1. Introduction
Ecklonia cava is an edible brown alga that inhabits the coastal waters of Korea and Japan. It contains many unique polyphenols, especially phlorotannins [
1]. Extracts of
Ecklonia cava have been reported to have various biological activities, such as antioxidant, anticancer, antidiabetes, and antiallergy effects [
2,
3]. Due to its bioactivities, it has been commercialized and is available as a therapeutic, Seapolynol (SN). The constituents of SN are known to include eckol, dieckol, and bieckol derivatives [
1]: dieckol (8.2%), 8,8′-bieckol (2.8%), 2-
O-(2,4,6-trihydroxyphenyl)-6,6′-bieckol (2.1%), 6,6′-bieckol (1.5%), phlorofurofucoeckol-A (1.4%), eckol (0.6%), 2-phloroeckol (0.4%), 7-phloroeckol (0.4%), and phlorotannin A (0.4%). SN, a mixture of these marine polyphenols, may be a promising edible agent for treating various pathological conditions.
Obesity is the result of excess fat storage, derived from an imbalance in energy. It is a constantly increasing metabolic disease worldwide, leading to atherosclerosis, diabetes mellitus, sleep apnea syndrome, and even some cancers [
4]. Treatment of obesity includes control of diets, exercise, and medications. Diet and exercise are important for the management of overweight subjects and the prevention of obesity, but the efficacy of such countermeasures is limited for treating morbid obesity, in particular, those with a body mass index (BMI) ≥ 30 kg/m
2. There is a continuing need for novel treatments for obesity, and many medications have been developed and marketed [
5]. However, current obesity drugs have adverse effects such as dizziness, insomnia, headache, rash, and mild gastrointestinal symptoms [
6]. Thus, the development of antiobesity drugs with fewer or no side effects is desirable.
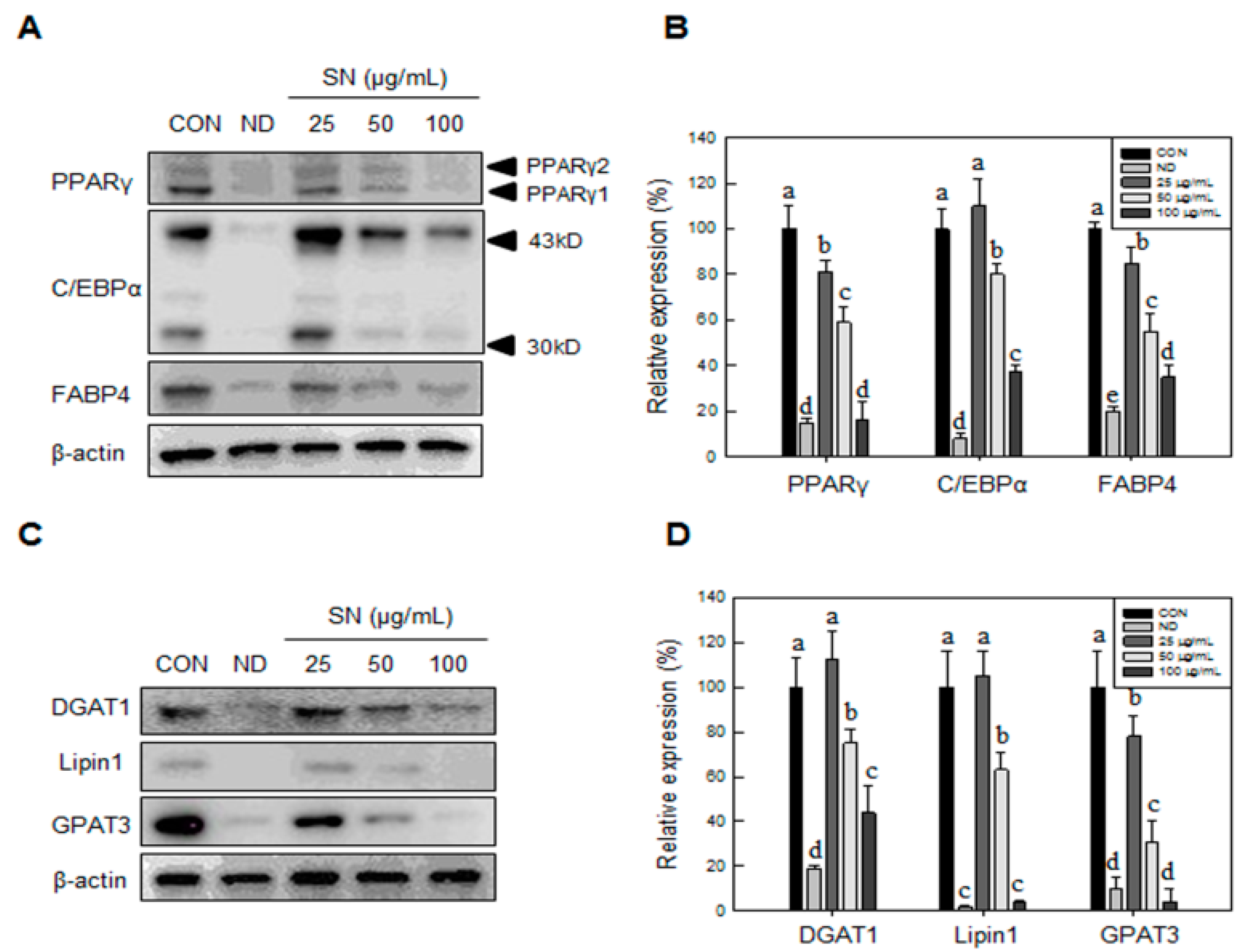
The accumulation of body fat depends on the differentiation status of adipocytes. In cell-based studies, adipocyte differentiation processes are well understood, with hormonal and nutritional regulation [
7,
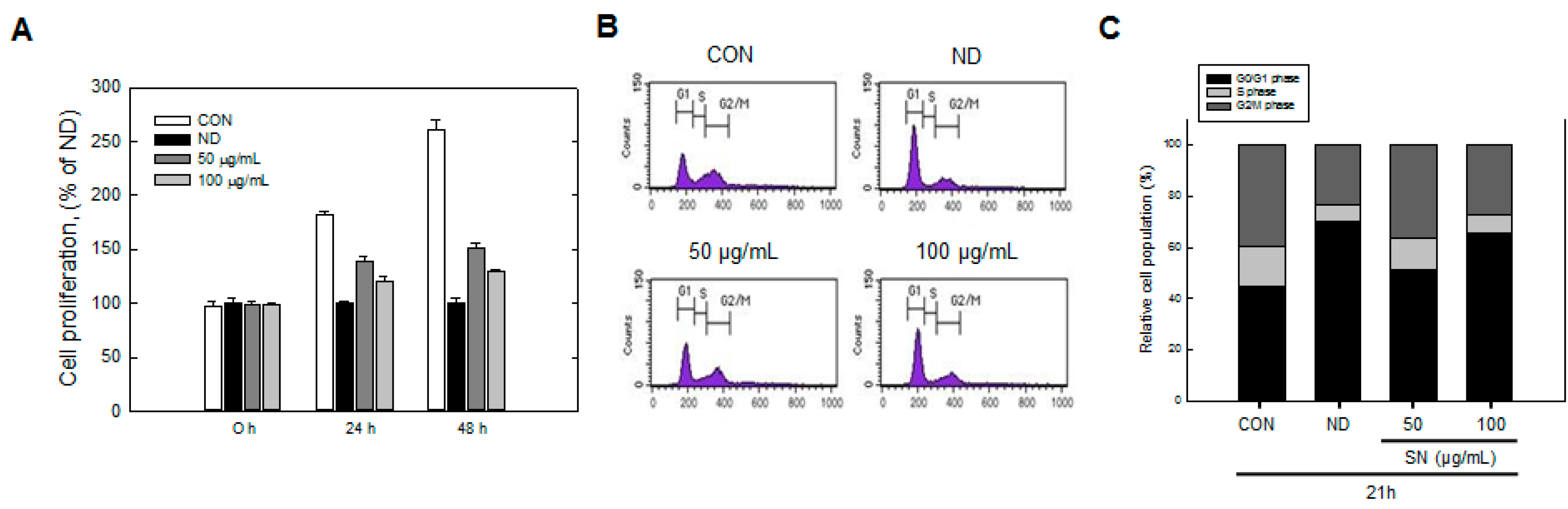
8]. The differentiation process begins from a growth-arrested confluent state of the cells, followed by the treatment of a hormone cocktail composed of insulin, dexamethasone, and 3-isobutyl-1-methylxanthine (IBMX). These differentiating cells then restart the cell cycle to increase cell numbers two- to four-fold by mitotic clonal expansion (MCE) [
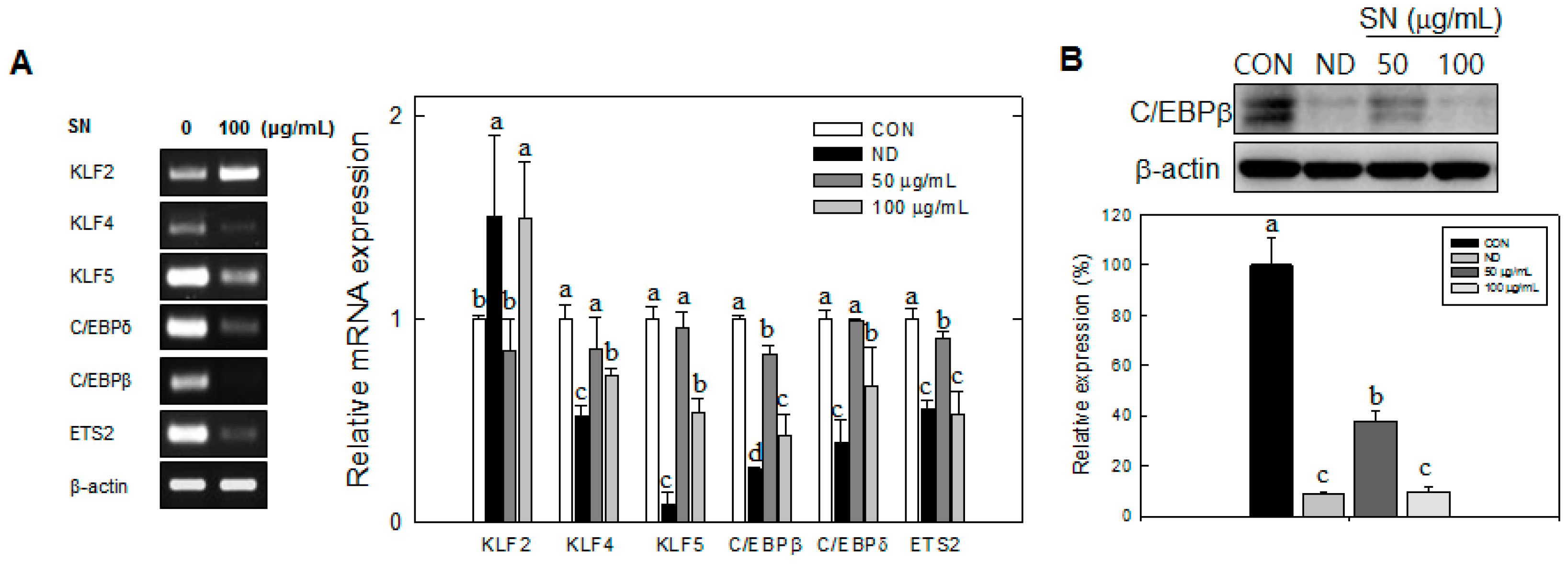
9]. This hyperplasia of adipocytes is accompanied by the expression of early adipogenic transcription factors, such as KLF4 and 5, C/EBPβ, C/EBPδ, and Protein C-ets-2 (ETS2), whereas KLF2 expression is negatively regulated during MCE [
10,
11,
12]. The regulation of early adipogenic factors is closely associated with the expression of late adipogenic factors, such as C/EBPα and peroxisome proliferator-activated receptor gamma (PPARγ). Activation of late adipogenic factors leads to the expression of enzymes involved in triglyceride synthesis, such as lipin1 and DGAT1, promoting lipid accumulation in the adipocyte [
13,
14,
15]. Adipogenic hyperplasia generally involves the activation of cell proliferative signaling pathways, such as the phosphoinositide 3-kinase/protein kinase B (PI3K/AKT) and extracellular signal-regulated kinase (ERK) pathways [
16,
17]. Moreover, AMPK signaling is involved in the lipid biosynthetic pathway, controlling energy metabolism in adipocytes [
18]. Activation of AMPK reduces fatty acid synthesis by deactivating acetyl-coA carboxylase (ACC), a major fatty acid synthetic enzyme [
19].
In recent years, the zebrafish has been identified as a useful animal model for the study of various human diseases. Benefits of the zebrafish model include its short generation time, abundant embryos, and lower costs compared with other animal models [
20]. Zebrafish also provide a transparent body for easier observation of some phenotypes. In particular, the programs for lipid accumulation of zebrafish are similar to those of mammals [
21]. Accordingly, research into obesity has been conducted using the zebrafish model [
22]. As the mouse is a well-established animal model of obesity, a comparative study of both models would enhance the reliability of any results.
Recently, SN, a mixture of edible marine polyphenols, has been reported to have antihyperlipidemic effects [
23]. However, the mechanistic understanding of the inhibitory effect of SN is limited. In this study, we investigated the inhibitory effects of SN on lipid accumulation using various models: 3T3-L1 cells, zebrafish, and mice. Additionally, the mechanistic relevance of cell cycle state and signaling in early adipogenesis was examined.
3. Discussion
Our study showed that SN inhibited lipid accumulation in 3T3-L1 cells and high-fat-diet-fed zebrafish and mice. Furthermore, we examined the underlying mechanisms of the SN-induced antiadipogenic effect including inhibition of MCE via cell cycle arrest, and activation of AMPKα signaling.
Ecklonia cava has been shown to inhibit adipogenesis in cell lines. Several polyphenols (dieckol, phloroeckol, dioxinodehydroeckol) derived from
Ecklonia cava were shown to have antiadipogenic effects in adipocytes [
1,
16]. A clinical study on
Ecklonia cava polyphenols was also performed in overweight subjects. In particular, the most abundant
Ecklonia cava polyphenol, dieckol [
3,
4], has been identified to have antiadipogenic effects in cells and animal models [
17]. SN, a standardized extract of
Ecklonia cava, includes active compounds including the polyphenols mentioned above, and is commercially available; SN contains dieckol, eckol, and bieckol derivatives [
1]. Recent studies have shown the biological effects of this extract.
Ecklonia cava extract or
Ecklonia cava-derived polyphenols have been reported to have antioxidant effects [
2,
25]. Kang
et al., showed the potential of
Ecklonia cava polyphenols as chemopreventive agents [
25], and the antioxidant and anti-inflammatory effects of
Ecklonia cava in osteoarthritis were evaluated.
Ecklonia cava extract was shown to have antibacterial effects on antibiotic-resistant food-borne pathogens [
26]. Additionally, Heo
et al., showed protective effects of
Ecklonia cava phlorotannins against photo-oxidative stress from UV-B radiation [
27]. Shibata
et al., reported the inhibitory effect of
Ecklonia cava phlorotannins on pro-inflammatory responses [
28]. In particular, an antiadipogenic effect of SN has been reported recently [
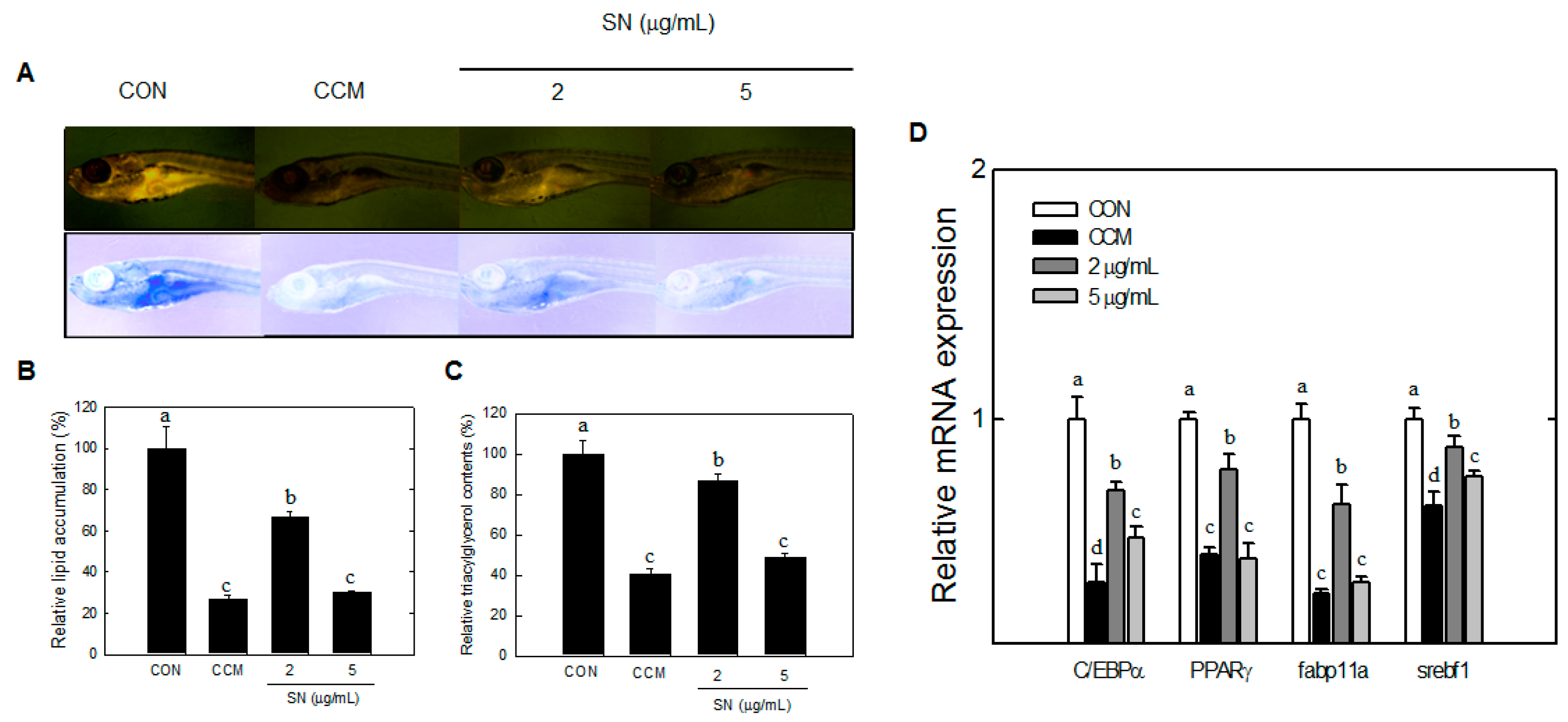
23]. However, the mechanisms underlying the antiadipogenic effects of SN should be investigated. The current study is the first to show the antiadipogenic effect of SN in two animal models with early adipogenic and signaling mechanisms, in which zebrafish was used to show the effects of SN in early adipogenesis. Lipid accumulation in zebrafish is known to be detectable from 15 dpf [
29]. Zebrafish were grown for 17–20 dpf to show early fat accumulation by Nile-red staining; SN inhibited fat accumulation in zebrafish from an early stage.
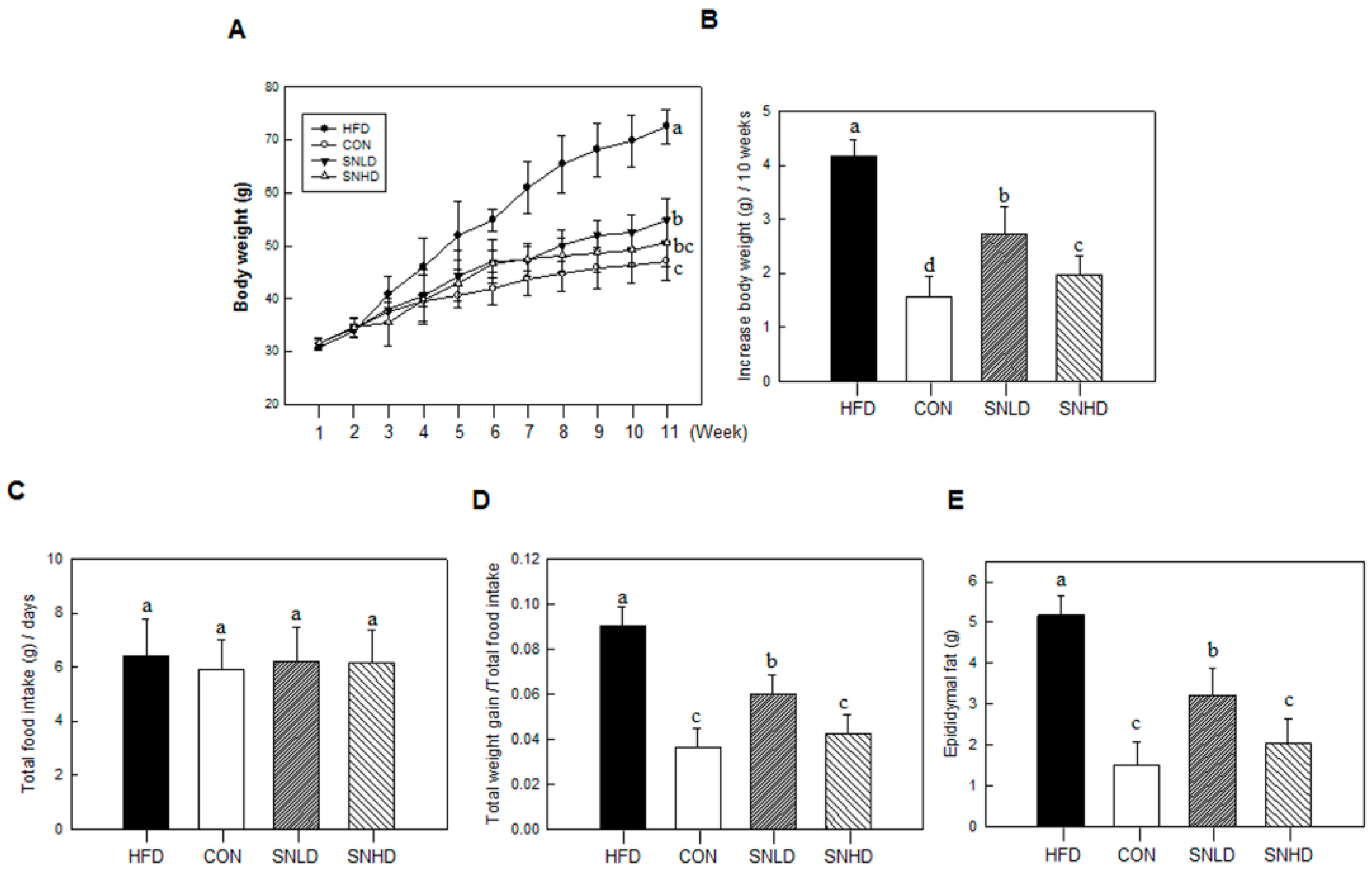
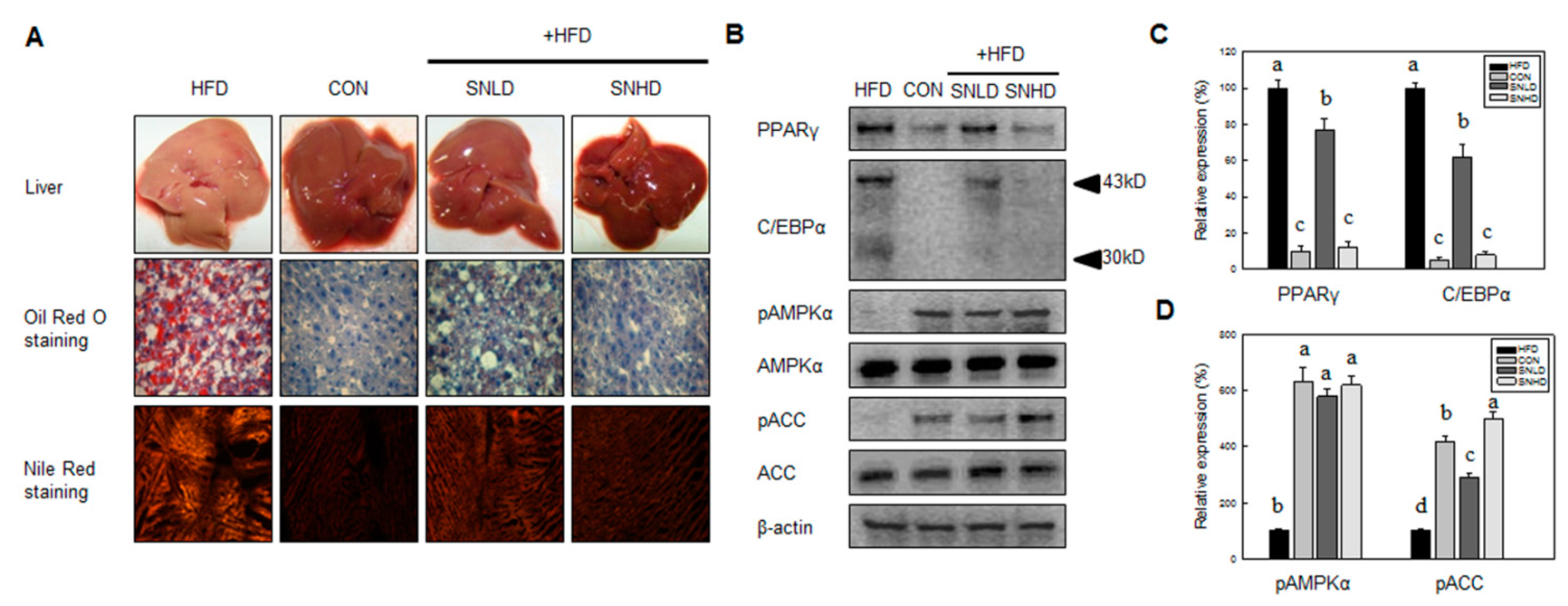
In addition, the diet induced obese mice has been used here were 5 weeks old, fed on HFD for 10 weeks. The HFD mice caused a marked increase the lipid accumulation in the liver or TG and total cholesterol in the blood compared to control diet mice, while SN dramatically repressed the lipid accumulation in the liver or TG and total cholesterol in the blood. In addition, SN significantly decreased HFD-induced abnormal expression of adipogenic and fatty liver associated genes. On the other hand, the high levels of glucose and insulin concentration are generally occurring in HFD-induced obese mice [
30]. However, we did not observed the sign of hyperglycemia and hyperinsulinemia in HFD-induced obese mice compared to the control diet group in this study.
The afore-mentioned evidence prompt us to speculate that some of the factors such as mouse strain, ages, diet composition or duration of HFD may affect to develop the intermediate stage obesity between obesity and type 2 diabetes. This model may occur the some of genetic changes in the tissues due to the HFD consumption in mice, but it has not yet appeared the hyperglycemia and hyperinsulinemia. However, additional animal study should be analyzed in further studies.
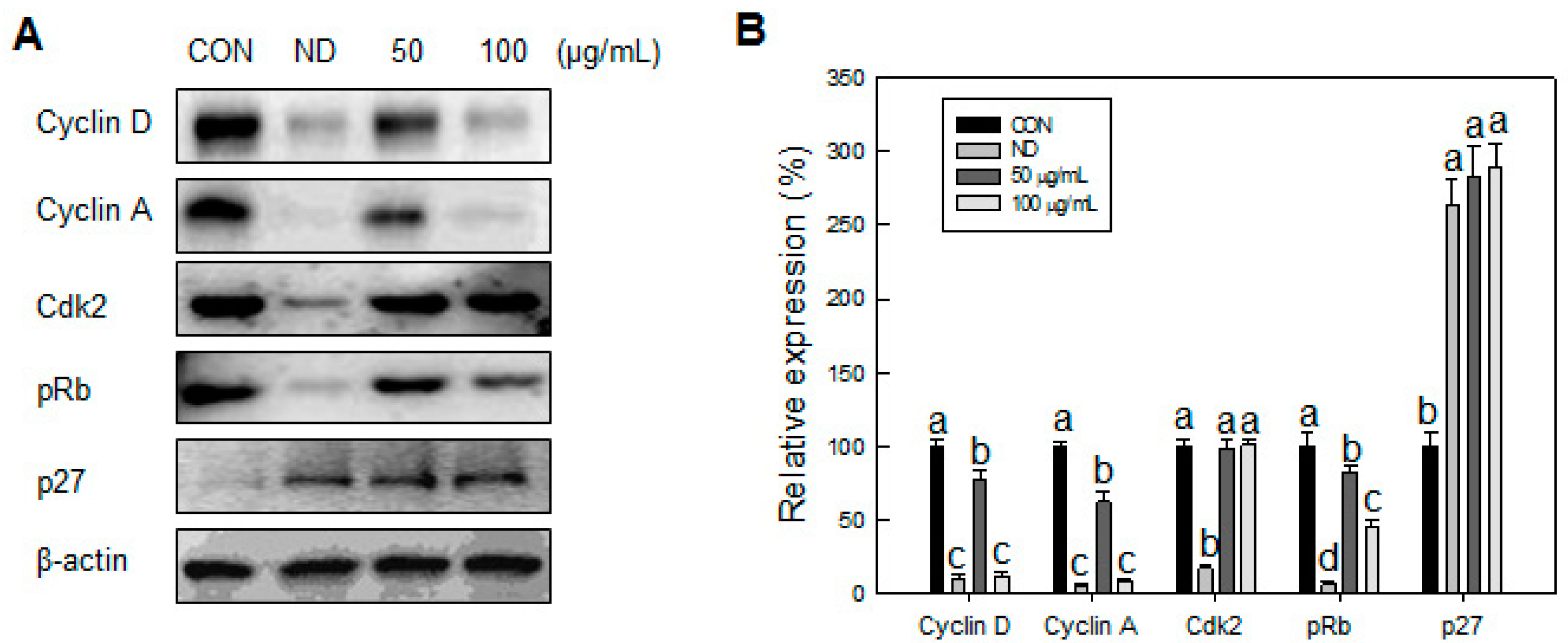
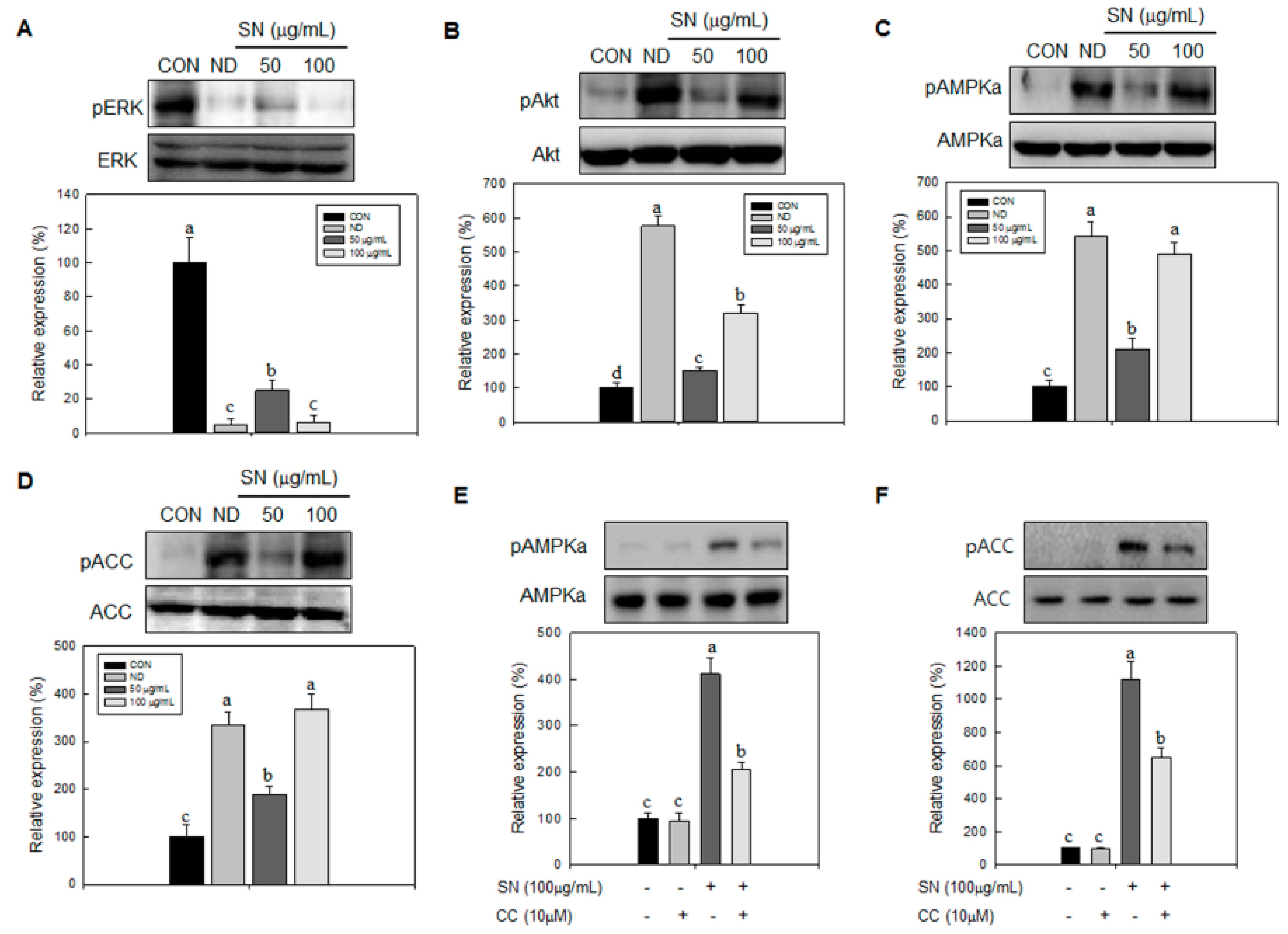
Our data showed that SN induced inhibition in early adipogenesis via regulating early adipogenic factors, such as KLF2, KLF4, and C/EBPβ. SN-mediated inhibition of early adipogenesis was associated with the inhibition of MCE via cell cycle arrest, and the inhibition of ERK and Akt signaling was underlined as a reason for the SN-induced inhibition of early adipogenesis. Recent studies have shown that several phytochemicals induce inhibition of MCE via cell cycle arrest as one of the major mechanisms of an antiadipogenic effect in 3T3-L1 cells [
31]. In particular, our data showed that SN activates AMPKα, a sensor in energy metabolism, and ACC, a major enzyme in fatty acid synthesis, in both adipocytes and a mouse model. Such AMPKα activation was also shown previously in a study of dieckol [
32]. Accordingly, the SN-induced activation of AMPKα is likely due to dieckol, a major component of SN. However, the other compounds in SN should also be investigated. The SN-mediated repression of ACC could contribute to the suppression of TG accumulation in adipocytes and mice models. Our inhibitor study showed SN-induced repression of ACC was mediated via activation of AMPKα signaling.
Our study showed that SN supplementation effectively improved the plasma lipid status of mice fed HFD, as was seen with dieckol [
32]. However, SN was superior to dieckol in terms of the suppression of lipid accumulation and cholesterol levels. SN decreased triglyceride levels by 40%—while dieckol reduced them by only ~10%—in mice fed, an HFD. LDL cholesterol levels were reduced by 66% in SN-supplemented HFD-fed mice, while dieckol supplementation showed a 54% reduction in LDL cholesterol [
32]. This result was supported by the study of Yeo
et al., [
23] which showed SN is more effective than dieckol on inhibition of lipid accumulation and weight increase. Given that dieckol comprises 8% of SN [
1], other SN-derived compounds likely contribute to the difference in effects on lipid accumulation and cholesterol levels between SN and dieckol. SN showed significant effects in decreasing triglyceride and cholesterol levels in HFD-fed mice.
The reductions in triglycerides and lipid accumulation by SN are supported by its suppression of adipogenic factors. However, the SN-induced reduction of (LDL) cholesterol level was not fully addressed in this study. In particular, downstream targets of AMPKα include various factors—including HMGCoA and SREBP1, which are responsible for cholesterol metabolism [
33]. Although SREBF-1 was shown to be regulated by SN in zebrafish, other cholesterol-related factors should be examined. AMPKα also regulates carbohydrate metabolism via factors such as ChREBP, but glucose and insulin levels were unaffected by SN, suggesting that SN does not greatly affect carbohydrate metabolism.
AMPKα is known to be associated with factors related to the energy expenditure, such as the NAD-dependent protein deacetylase sirtuin 1 (SIRT1) and peroxisome proliferator-activated receptor-γ coactivator 1α (PGC1α) [
29]. These signaling processes are known to activate thermogenic responses, such as heat production with upregulation of uncoupling proteins [
29]. A recent study showed that a phytochemical, indole-3-cabinol, directly activated SIRT1 via binding in 3T3-L1 cells [
34]. Thus, the roles of SN and SN-derived compounds should be investigated in thermogenic signaling in future studies.
We also used the zebrafish model, and showed SN-mediated inhibition in early adipogenesis and lipid accumulation. Zebrafish provides various advantages for in vivo research but also has several limitations. Full sets of antibodies for lipid metabolism studies have not yet been established. Further studies should include data for the antiadipogenic effects of SN in protein levels. Finally, our data demonstrated that SN effectively suppressed HFD-induced lipid accumulation in cell line, zebrafish, and mouse models.
Ecklonia cava contains various polyphenols, known as phlorotannins. Phloroeckol, dioxinodehydroeckol, and dieckol have been shown to have antiadipogenic activities in adipocytes and animals [
23], and dieckol, a major compound of SN, could give an important contribution to antiadipogenic effects of SN. However, the effect may be derived from the synergistic or combinational effects of constituents in SN. Since other
Ecklonia cava-derived polyphenols, which may also have antiadipogenic effects, have yet to be fully investigated, further study should be performed on SN-derived compounds, additionally, comparative and combinational studies on SN-derived compounds would be executed in the future.
4. Experimental Section
4.1. Materials
Seapolynol was obtained from Botamedi (Jeju, Korea). Dulbecco’s modified Eagle’s medium (DMEM), bovine calf serum (BCS), fetal bovine serum (FBS), penicillin-streptomycin (P/S), phosphate-buffered saline (PBS), and trypsin-EDTA were obtained from Gibco (Gaithersburg, MD, USA). Dexamethasone (DEX), IBMX, insulin, Oil red O, and nitrobluetetrazolium (NBT) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Antibodies against pRB, Akt, pAkt, ERK, pERK, C/EBPβ, AMPKα, pAMPKα, ACC, pACC, and β‑actin were purchased from Cell Signaling Technology (Danvers, MA, USA). Antibodies against PPARγ, C/EBPα, FABP4, GPAT3, Lipin1, DGAT1, Cyclin A, Cyclin D, Cdk2, and P27 were purchased from Santa Cruz Biotechnology, Inc. (Santa Cruz, CA, USA). Cell lysis buffer and phosphatase inhibitor cocktails I and II were purchased from Sigma. Dorsomorphin dihydrochloride (compound C) was purchased from Santa Cruz Biotechnology (Dallas, TX, USA). All other chemicals were purchased from Sigma.
4.2. Cell Culture
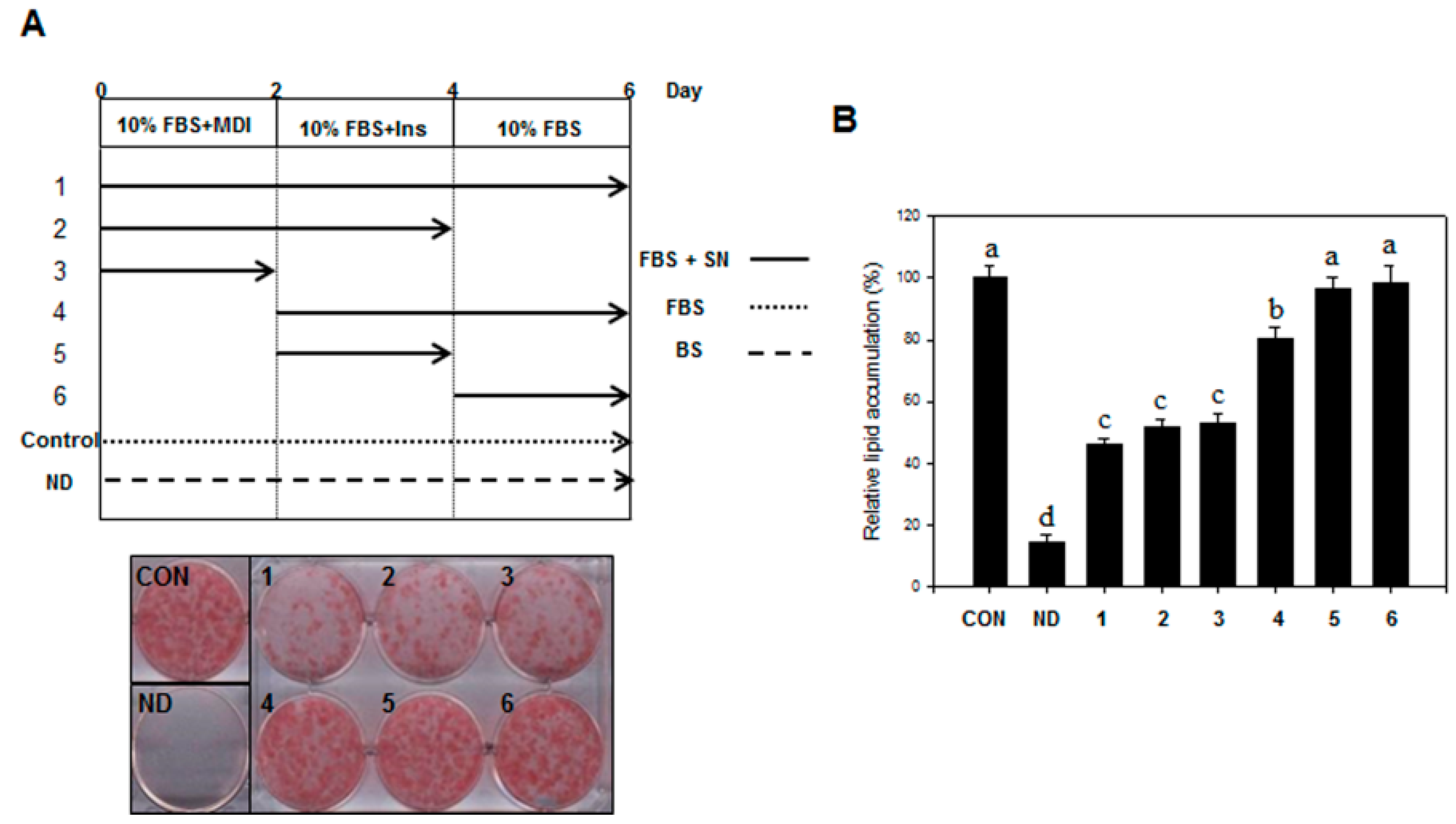
3T3-L1 preadipocytes were obtained from the American Type Culture Collection (ATCC, CL-173, Manassas, VA, USA). The preadipocytes were plated and cultured in DMEM medium containing 3.7 g/L sodium bicarbonate, 1% P/S, and 10% BCS at 37 °C in 5% CO2. Adipocyte differentiation was induced by treating two-day post-confluent cells with 10% FBS and a hormone mixture (MDI), consisting of 0.5 mM IBMX, 1.0 μM DEX, and 1.67 μM insulin (day 0). The medium was replaced with DMEM containing 1.67 μM insulin and 10% FBS for two additional days (day 2). This medium was then replenished with DMEM containing 10% FBS every other day until the indicated time (days 6 to 12). SN (25, 50, and 100 µg/mL) was dissolved in DMSO, the final concentration of DMSO was set to 0.25% in all experiments.
4.3. Cell Viability
3T3-L1 preadipocytes or confluent cells were treated with SN (0–400 μg/mL), and incubated for 48 h. XTT (2,3-Bis(2-methoxy-4-nitro-5-sulfophenyl)-2Htetrazolium-5-carbox-anilide) reagent and PMS (N-methyl dibenzopyrazine methyl sulfate) were added to the culture and incubated for 2 h at 37 °C. Produced soluble formazan salt in media was measured at 450 nm against 690 nm. Cell viability (%) was expressed as the ratio of viable cells compared to the non-treated cells.
4.4. Trypan Blue Assay
Post-confluent 3T3-L1 cells were induced to differentiate with MDI cocktail for two days in the presence or absence of SN. Cells were trypsinized after two washes with PBS. Trypan blue dye (0.5%, Sigma-Aldrich) was added to the cells for 3 min. Stained viable cells were counted under a microscope.
4.5. Oil Red O Staining and Lipid Quantification
Differentiated 3T3-L1 cells or liver tissue samples were fixed with 4% formaldehyde at 4 °C for 1 h after washing with PBS. After washing fixed cells with phosphate-buffered saline, cells were stained with 0.5% Oil red O in 60:40 (v/v) isopropanol/H2O for 2 h at room temperature, and then washed four times with distilled water. The extent of lipid storage was observed microscopically and photographed. Isopropanol (100%) was used to elute Oil red O dye for determination of optical density at 490 nm. Accumulated triglycerides were measured using a Total Triglyceride Assay Kit (Zen-Bio, Inc., Research Triangle Park, NC, USA).
4.6. Western Blotting
Proteins extracted from differentiated or undifferentiated and mouse liver tissues were resolved by SDS-PAGE in 8%–12% polyacrylamide gels before they transferred onto PVDF (polyvinylidene fluoride) membrane. An immunoblot analysis was conducted with the commercial antibodies indicated. Secondary antibodies conjugated with horseradish peroxidase (1:1000) were applied for 1 h. Bands were visualized by enhanced chemiluminescence, and detected using the LAS-4000 imaging software (Fuji, New York, NY, USA).
4.7. Semi-Quantitative RT-PCR or Real-Time RT-PCR (qRT-PCR)
Total RNA was isolated from 3T3-L1 cells using the TRIzol reagent (Invitrogen, Carlsbad, CA, USA. RNA samples with an OD260/OD280 ratio higher than 2.0 were used for semi-quantitative RT-PCR. One microgram of total RNA was used to produce cDNA with a RT-PCR system.
For the qRT-PCR, cells, mouse tissues and zebrafishes were washed with phosphate-buffered saline. Then, total RNA was extracted from differentiated (day 6 or 8) or undifferentiated adipocytes, zebrafish, and mouse liver tissues using the TRIzol reagent (Invitrogen, Carlsbad, CA, USA), according to the manufacturer’s protocol. cDNA was produced from 1 µg of total RNA using a Maxime RT PreMix Kit (iNtRON Biotechnology, Inc., Seoul, Korea). cDNA was amplified using SYBR Green 2× master mix kit (M-Biotech. Inc., Salt Lake City, UT, USA) and the specific primers listed in
Table 2. The reaction was conducted with a CFX96 Real-Time system and c1000 thermal cycler (Bio Rad, Inc., Hercules, CA, USA) and subjected to PCR analysis at 40 cycles. Quantification analysis was generated using the CFX Manager software (Bio Rad, Inc.).
Table 2.
Primer sequences.
Table 2.
Primer sequences.
| Name | Forward (5′ to 3′) | Reverse (5′ to 3′) |
|---|
| PPARγ | CTGTGAGACCAACAGCCTGA | AATGCGAGTGGTCTTCCATC |
| C/EBPα | TGAAGGAACTTGAAGCACAA | TCAGAGCAAAACCAAAACAA |
| aP2 | TCACCTGGAAGACAGCTCCT | AATCCCCATTTACGCTGATG |
| C/EBPβ | CAAGCTGAGCGACGAGTACA | AGCTGCTCCACCTTCTTCTG |
| C/EBPδ | AGAAGCTGGTGGAGTTGTCG | CGCAGGTCCCAAAGAAACTA |
| Krox20 | AGTTGGGTCTCCAGGTTGTG | GGAGATCCAGGGGTCTCTTC |
| KLF4 | CTGAACAGCAGGGACTGTCA | GTGTGGGTGGCTGTTCTTTT |
| KLF5 | ACGTACACCATGCCAAGTCA | GTGGGAGAGTTGGCGAATTA |
| KLF2 | GCCTGTGGGTTCGCTATAAA | AAGGAATGGTCAGCCACATC |
| ETS2 | CGCCCCAAGATATTCTGTGT | TGGAAGATCCCTCCTGATTG |
| β-actin | AGCCATGTACGTAGCCATCC | CTCTCAGCTGTGGTGGTGAA |
| PPARγ (D) | CAGTTTGCAGAGAACAGCGT | GGCTCTTCTTGTGTATGCGG |
| C/EBPα (D) | ATCAGCGCCTACATTGATCC | TTGCTTGGCTGTCGTAGATG |
| aP2 (D) | GCAAACTTGTGCAGAAACA | GAACTGAGCCTGGCATCTTC |
| β-actin (D) | CTCTTCCAGCCTTCCTTCCT | CTTCTGCATACGGTCAGCAA |
4.8. Analysis of Cell Cycle Progression
Cell cycle progression of cultured cells was determined by flow cytometry. Post-confluent preadipocytes were treated with MDI in the presence or absence of SN (50 or 100 µg/mL) for 24 h. Collected cells were fixed with 70% ethanol for 2 h at 4 °C, washed with PBS, and centrifuged (1000× g) to remove ethanol. For staining the DNA, the resulting pellet was resuspended in 40 µg/mL propidium iodine solution containing 1 mg/mL RNase A at 37 °C for 30 min. The cell cycle progression of samples (10,000 cells per experiment) was analyzed using a BD FACSCalibur flow cytometer (Becton Dickinson, CA, USA), according to the manufacturer’s instructions.
4.9. Animal Care and Experimental Protocol
All experimental mice were housed in a specific pathogen-free facility at CHA University, Seongnam, Korea. The project was approved by the Institutional Animal Care and Use Committee of CHA University (IACUC140001). In total, 40 male Imprinting Control Region (ICR) mice (5 weeks old) were purchased from Joong-Ang Experimental Animal Co. (Seoul, Korea) and were housed in a pathogen-free facility under the following conditions: 21 ± 2.0 °C, 50% ± 5% relative humidity, and 12/12-h light/dark cycle. All the mice were fed rodent chow and tap water ad libitum for one week prior to their division into the following experimental groups (n = 10 per group): normal diet, HFD, and SN-supplemented diets (SNLD; 30 mg/kgBW/day and SNHD; 75 mg/kgBW/day). The normal diet was a purified diet based on the Purina Laboratory Rodent Diet 38057 (Dyets Inc., Bethlehem, PA, USA). The HFD was identical to the ND but supplemented with 350 g fat/kg (300-g lard plus 50-g corn oil) and 1% cholesterol (Research Diets Inc., Bethlehem, PA, USA).
After 10 weeks of high fat diet feeding, mice are sacrificed to collect their blood and internal organs, following a 12-h fast. To analyze plasma lipid levels, blood was drawn into EDTA-coated tubes via cardiac puncture. Plasma was isolated by centrifuging the blood at 2000× g for 15 min at 4 °C. The epididymal and visceral fat pads were removed, rinsed with PBS, and weighed. The liver was weighed, photographed, and stored at 80 °C.
All zebrafish experiments were approved by the internal Animal Ethics Committee at CHA University. Embryos and larvae of zebrafish (Danio rerio) were initially obtained from Chungnam National University (Daejeon, Korea).
Larvae were maintained in a 100-mm plate at a density of ~20 larvae per 100 mL, and fed ad libitum with hardboiled egg yolk as a high-fat diet (HFD) once per day, in the presence or absence of SN for 12–15 days (17–20 dpf). SN and vehicle (dimethyl sulfoxide, DMSO) were treated at a concentration of 0.1% (v/v) in each group plate. Two concentrations of SN (2 and 5 µM) were examined, and CCM (curcumin, 2.5 µM) was used as a positive control. Zebrafish larvae used for imaging analysis were starved for 24 h prior to Nile red staining, to ensure that their digestive tracts were empty.
4.10. Quantification of Triglycerides
Triglyceride was evaluated using a triglyceride assay kit (Zen-Bio). Zebrafish was washed with PBS to remove residual medium, and the zebrafish was lysed with RIPA buffer. Triglyceride was digested with Reagent B for 2 h to release hydrolyzed glycerols into the buffer. Diluted hydrolysates were incubated with Reagent A containing peroxidase to produce quinoneimine dye, which shows a absorbance maximum in spectrophotometric detection at 540 nm.
4.11. Nile Red Staining, Fluorescence Imaging, and Quantification
For staining of zebrafish, stock solution (1.25 mg/mL, Invitrogen N-1142) of Nile red (Invitrogen N-1142) was prepared in acetone, and store in dark at −20 °C. Briefly, Nile red stock solution was supplemented with water to a final concentration of 50 ng/mL. The fish were incubated in this water for 5–10 min at 28 °C in the dark, after which they were washed with distilled water three times. A few drops of a tricaine (Sigma) stock solution (4 mg/mL, pH 7) were added to induce anesthesia. The fish were mounted in 3% methylcellulose, and the Nile red-stained area was imaged under a fluorescence dissecting microscope (TE300, Nikon, Tokyo, Japan) equipped with a green fluorescent protein (GFP) long-pass filter. Fluorescence images were acquired using a Pixera Penguin 600CL digital camera and the InStudio software (Pixera, Santa Clara, CA, USA).
4.12. Biochemical Analysis
Plasma concentrations of total cholesterol, high-density lipoprotein (HDL)-cholesterol, low-density lipoprotein (LDL)-cholesterol, triglyceride, albumin, glucose (Asan Pharmaceutical, Gyeonggi, Korea), and insulin (Shibayagi, Gunma, Japan) were determined enzymatically using commercial kits.
4.13. Statistical Analysis
All data are expressed as means ± standard error of the mean of triplicate determinants. All statistical analyses were performed using the SAS (version 9.1, Statistical Analysis Software, Cary, NC, USA). One-way analysis of variance (ANOVA) was used for comparisons among groups. Significant differences between the mean values were assessed using Duncan’s test. p values < 0.05 were considered to indicate statistical significance.