Polyphenols from Bee Pollen: Structure, Absorption, Metabolism and Biological Activity
Abstract
:1. Introduction
2. Bee Pollen
2.1. Polyphenols
2.1.1. Phenolic Acids
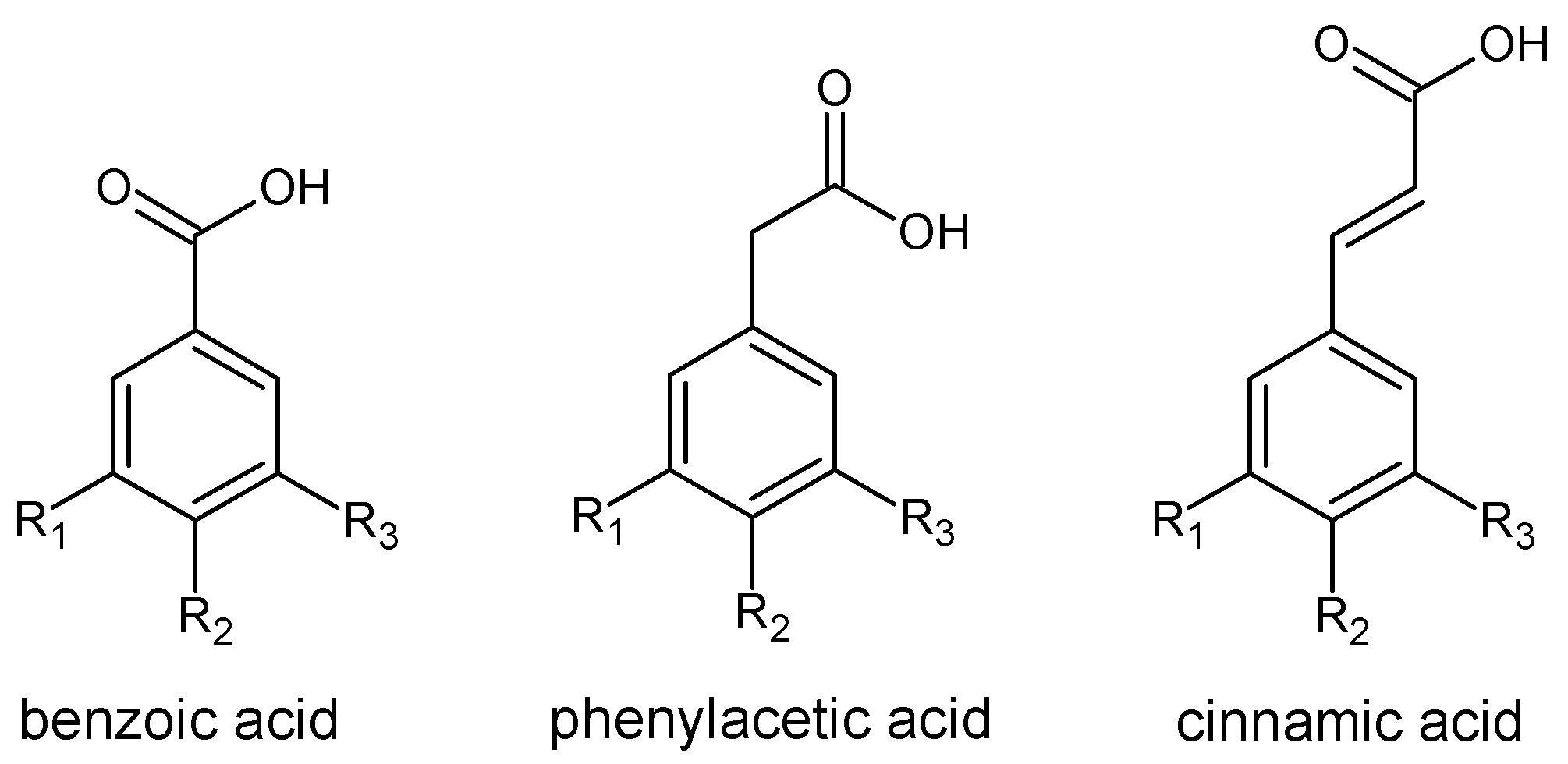

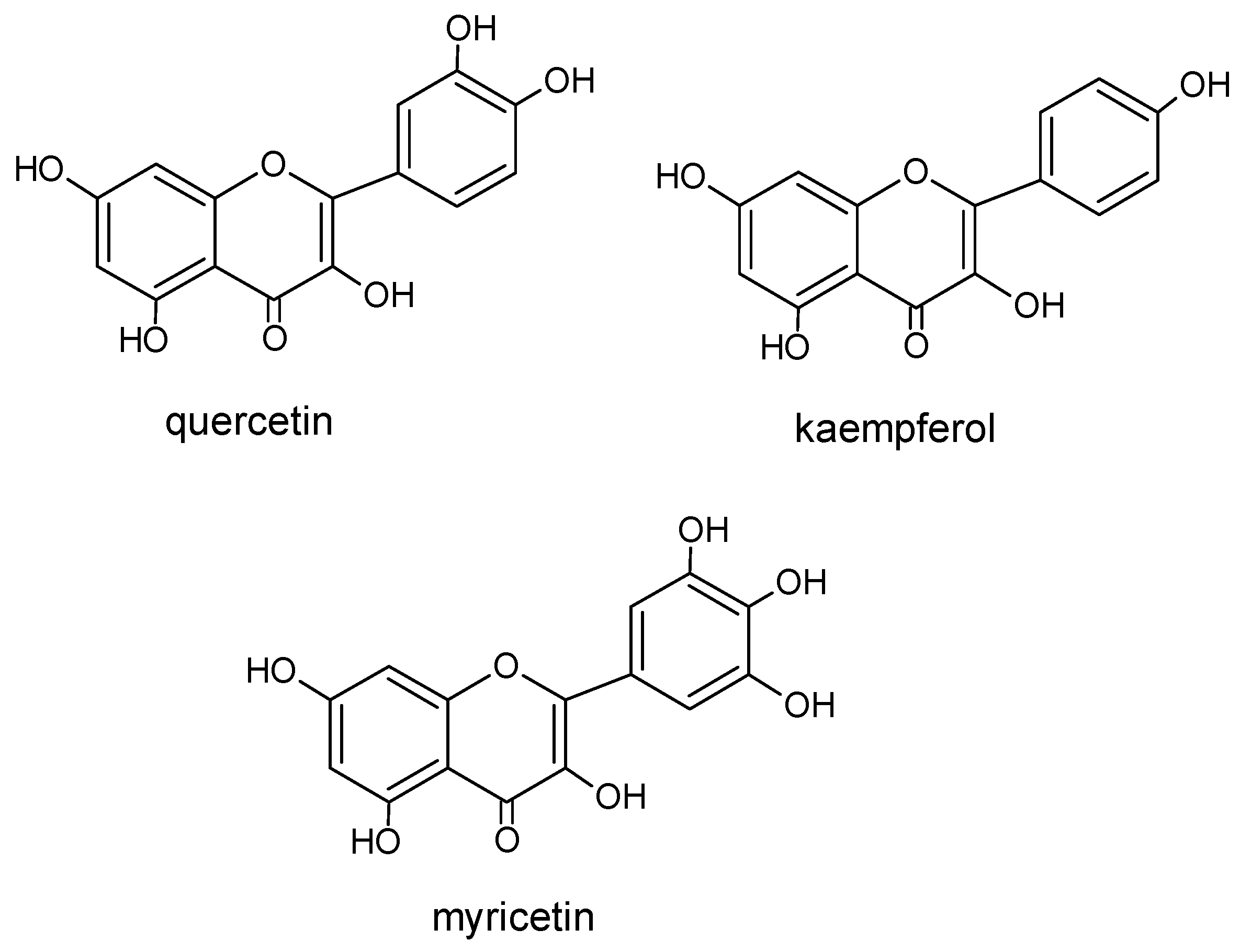
2.1.2. Flavonoids

| MAIN POLYPHENOLIC COMPOUNDS OF BEE POLLEN | |||
|---|---|---|---|
| Bee Pollen Compound and the Structures of Major Classes | Free Hydroxyl Groups Position [3,36] | TEAC a (mM) [3] | |
| 1. PHENOLIC ACIDS | |||
| HYDROXYBENZOIC ACIDS: | |||
| Gallic acid |  | 3,4,5 | 3,0 |
| Protocatechuic acid | 3,4 | 1,2 | |
| HYDROXYCINNAMIC ACIDS: | |||
| Caffeic acid |  | 3,4 | 1,3 |
| Ferulic acid | 4 | 1,9 | |
| Chlorogenic acid | 3,4 | 1,3 | |
| para-Coumaric acid | 4 | 2,2 | |
| ortho-Coumaric acid | 2 | 1,0 | |
| 2. FLAVONOIDS | |||
| FLAVONES: | |||
| Luteolin |  | 5,7,3′,4′ | 2,1 |
| Apigenin | 5,7,4′ | 1,5 | |
| Chrysin | 5,7 | 1,4 | |
| FLAVONOLS: | |||
| Quercetin |  | 3,5,7,3′,4′ | 4,7 |
| Rutin (Q 3-o-rutoside) | 5,7,3′,4′ | 2,4 | |
| Kaempherol | 3,5,7,4′ | 1,3 | |
| Myricetin | 3,5,7,3′,4′,5′ | 3,1 | |
| Galangin | 3,5,7 | N/D | |
| FLAVANONES: | |||
| Naringenin |  | 5,7,4′ | 1,5 |
| Pinocembrin | 5,7 | N/D | |
| ISOFLAVONES: | |||
| Genistein |  | 5,7,4′ | N/D |
3. Absorption and Metabolism of Phenolic Acids and Flavonoids
4. Properties of Bee Pollen
| Main Effect | Mechanism of the Biological Activity |
|---|---|
| Nutritive | Source of proteins (23.9% a; vital role), exogenous amino acids (8.6% a; vital role, malnutrition), carbohydrates (13%–55% a), lipids (0.3%–20% a; hypolipidemic) including unsaturated fatty acids (2.7% a; anti-inflammatory, anticancerogenic), phospholipids (1.5% a; cardioprotective, anti-inflammatory), phytosterols (1.1% a; antiartherosclerotic) vitamins (0.7% a), bioelements (1.6% a) [22,31,59,60] |
| Antioxidative | Scavenging hydroxyl radicals; complexing metals [7,12,13,14,17,18,19,20,21] |
| Cardioprotective | Inhibition of ACE activity; inhibition of blood platelets aggregation [61,62,63] |
| Hepatoprotective | Reduction of lipofuscin; detoxifying activity in industrial poisoning [64] |
| Anti-inflammatory | Inhibition of COX-2; inhibition of NO production [25,65,66,67,68,69,70,71] |
| Antibacterial | Disruption of bacteria metabolism, especially in: Staphyllococcus aureus, Staphylococcus epidermidis, Bacillus cereus, Bacillus subtilis, Pseudomonas aeruginosa, Salmonella enterica, Listeria monocytogenes, Escherichia coli [72,73,74] |
| Anticarcinogenic | Brassica camperstris L. bee pollen extract increases the activity of caspase-3 enzyme, and decreases the expression of anti-apoptic proteins Bcl-2; Cistus incanus L. and Salix alba L. bee pollens inhibit 17β-estradiol activity [75,76] |
| Antianaemic | Decrease of the number of blood platelets; increase in haemoglobin level [77,78] |
| Effects on bone tissue | Cystus ladaniferus L. bee pollen inhibits the resorption of the femur and formation of osteoclastic cells, and increases the level of alkaline phosphatase [79,80,81] |
4.1. Nutritive Properties
4.2. Antioxidant Activity
4.3. Cardioprotective Effects
4.4. Hepatoprotective Effects
4.5. Anti-Inflammatory Properties
4.6. Antibacterial Effects
4.7. Anticarcinogenic Properties
4.8. Immunostimulatory Activity
4.9. Antianaemic Effects
4.10. Effects on Osseous Tissue
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Luthria, D.L. Significance of sample preparation in developing analytical methodologies for accurate estimation of bioactive compounds in functional foods. J. Sci. Food Agric. 2006, 86, 2266–2272. [Google Scholar] [CrossRef]
- Kroyer, G.; Hegedus, N. Evaluation of bioactive properties of pollen extracts as functional dietary food supplement. Innov. Food Sci. Emerg. Technol. 2001, 2, 171–174. [Google Scholar] [CrossRef]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free Radic. Biol. Med. 1996, 20, 933–956. [Google Scholar] [CrossRef]
- Iriti, M. Introduction to Polyphenols, Plant Chemicals for Human Health. Mini Rev. Med. Chem. 2011, 11, 1183–1185. [Google Scholar] [PubMed]
- Gómez-Caravaca, A.M.; Gómez-Romero, M.; Arráez-Román, D.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Advances in the analysis of phenolic compounds in products derived from bees. J. Pharm. Biomed. Anal. 2006, 41, 1220–1234. [Google Scholar] [CrossRef] [PubMed]
- Tyszka-Czochara, M.; Knapik-Czajka, M.; Goździalska, A.; Francik, R.; Jaśkiewicz, J. Polyphenols in a diet. Some aspects of metabolism and bioavailability of phenolic compounds. Farm. Polska 2003, 59, 589–597. [Google Scholar]
- Leja, M.; Mareczek, A.; Wyżgolik, G.; Klepacz-Baniak, J.; Czekońska, K. Antioxidative properties of bee pollen in selected plant species. Food Chem. 2007, 100, 237–240. [Google Scholar] [CrossRef]
- Grajek, W. Antioxidants in Food, 1st ed.; WNT: Warsaw, Poland, 2007; pp. 258–259. [Google Scholar]
- Bartosz, G. The Second Face of Oxygen, 2nd ed.; PWN: Warsaw, Poland, 2003; pp. 30–57. [Google Scholar]
- Grygiel-Górniak, B. Oxidative stress as a cardio-diabetologic risk factor-facts and myths. Bromatol. Chem. Toksykol. 2010, 43, 109–117. [Google Scholar]
- Budryn, G.; Nebesny, E. Phenolic acids-their properties, occurance in plant materials, absorption and metabolism. Bromatol. Chem. Toksykol. 2006, 39, 103–110. [Google Scholar]
- Carpes, S.T.; Mourão, G.B.; Alencar, S.M.; Masson, M.L. Chemical composition and free radical scavenging activity of Apis mellifera bee pollen from Southern Brazil. Braz. J. Food Technol. 2009, 12, 220–229. [Google Scholar] [CrossRef]
- Nagai, T.; Inoue, R.; Suzuki, N.; Myoda, T.; Nagashima, T. Antioxidative ability in a linoleic acid oxidation system and scavenging abilities against active oxygen species of enzymatic hydrolysates from pollen Cistus ladaniferus. Int. J. Mol. Med. 2005, 15, 259–263. [Google Scholar] [CrossRef] [PubMed]
- Graikou, K.; Kapeta, S.; Aligiannis, N.; Sotiroudis, G.; Chondrogianni, N.; Gonos, E.; Chinou, I. Chemical analysis of Greek pollen—Antioxidant, antimicrobial, and proteasome activation properties. Chem. Cent. J. 2011, 5. [Google Scholar] [CrossRef] [PubMed]
- Gulcin, I. Antioxidant activity of food constituents: An overview. Arch. Toxicol. 2012, 86, 345–391. [Google Scholar] [CrossRef] [PubMed]
- Almaraz-Abarca, N.; da Graça Campos, M.; Ávila-Reyes, J.A.; Naranjo-Jiménez, N.; Herrera-Corral, J.; Gonzáles-Valdez, L.S. Variability of antioxidant activity among honeybee-collected pollen of different botanical origin. Interciencia-Caracas 2004, 29, 574–578. [Google Scholar]
- Almaraz-Abarca, N.; da Graça Campos, M.; Ávila-Reyes, J.A.; Naranjo-Jiménez, N.; Carrol, H.J.; González-Valdez, L.S. Antioxidant activity of polyphenolic extract of monofloral honeybee-collected pollen from mesquite (Prosopis juliflora, Leguminosae). J. Food Compos. Anal. 2007, 20, 119–124. [Google Scholar] [CrossRef]
- Morais, M.; Moreira, L.; Feás, X.; Estevinho, L.M. Honeybee-collected pollen from five Portuguese natural parks: Palynological origin, phenolic content, antioxidant properties and antimicrobial activity. Food Chem. Toxicol. 2011, 49, 1096–1101. [Google Scholar] [CrossRef] [PubMed]
- Izuta, H.; Narahara, Y.; Shimazawa, M.; Mishima, S.; Kondo, S.; Hara, H. 1,1-Diphenyl-2-picrylhydrazyl radical scavenging activity of bee products and their constituents determined by ESR. Biol. Pharm. Bull. 2009, 32, 1947–1951. [Google Scholar] [CrossRef] [PubMed]
- Marghitas, L.A.; Stanciu, O.G.; Dezmirean, D.S.; Bobis, O.; Popescu, O.; Bogdanov, S.; Campos, M.G. In vitro antioxidant capacity of honeybee-collected pollen of selected floral origin harvested from Romania. Food Chem. 2009, 115, 878–883. [Google Scholar] [CrossRef]
- Eraslan, G.; Kanbur, M.; Silici, S.; Liman, B.C.; Altinordulu, S.; Sarica, Z.S. Evaluation of protective effect of bee pollen against propoxur toxicity in rat. Ecotoxicol. Environ. Saf. 2009, 72, 931–937. [Google Scholar] [CrossRef] [PubMed]
- Komosińska-Vassev, K.; Olczyk, P.; Kaźmierczak, J.; Mencner, Ł.; Olczyk, K. Bee pollen: Chemical composition and therapeutic application. Evid. Based Complement. Altern. Med. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, C.; Iglesias, A.; Feás, X.; Estevinho, L.M. Commercial bee pollen with different geographical origins: A comprehensive approach. Int. J. Mol. Sci. 2012, 13, 11173–11187. [Google Scholar] [CrossRef] [PubMed]
- Rzepecka-Stojko, A.; Maciejewska-Paszek, I.; Stec, M.; Kurzeja, E.; Kęska, A.; Pawłowska-Góral, K. The influence of extraction method on obtaining polyphenolic compounds from bee pollen. Farm. Prz. Naukowy 2010, 1, 38–41. [Google Scholar]
- Maruyama, H.; Sakamoto, T.; Araki, Y.; Hara, H. Anti-inflammatory effect of bee pollen ethanol extract from Cistus sp. of Spanish on carrageenan-induced rat hind paw edema. BMC Complement. Altern. Med. 2010, 10. [Google Scholar] [CrossRef] [PubMed]
- Almeida-Muradian, L.B.; Pamplona, L.C.; Coimbra, S.; Barth, O.M. Chemical composition and botanical evaluation of dried bee pollen pellets. J. Food Compos. Anal. 2005, 18, 105–111. [Google Scholar] [CrossRef]
- Campos, M.G.; Bogdanov, S.; Almeida-Muradian, L.B.; Szczesna, T.; Mancebo, Y.; Frigerio, C.; Ferreira, F. Pollen composition and standardization of analytical methods. J. Apic. Res. 2008, 47, 154–161. [Google Scholar] [CrossRef]
- Arráez-Román, D.; Zurek, G.; Bässmann, C.; Almaraz-Abarca, N.; Quirantes, R.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Identification of phenolic compounds from pollen extracts using capillary electrophoresis-electrospray time-of-flight mass spectrometry. Anal. Bioanal. Chem. 2007, 389, 1909–1917. [Google Scholar] [CrossRef] [PubMed]
- Castellucio, C.; Paganga, G.; Melikan, N.; Bowell, G.P.; Pridham, J.; Sampson, J.; Rice-Evans, C. Antioxidant potential of intermediates in phenylopropanoid metabolism in higher plants. FEBS Lett. 1995, 368, 188–192. [Google Scholar] [CrossRef]
- Gawlik-Dziki, U. Phenolic acids as bioactive compounds in food products. Żywność Nauka Technol. Jakość 2004, 4, 29–40. [Google Scholar]
- Kędzia, B. Chemical composition and adaptogenic activity of honeybee-collected pollen. Part one. Chemical composition. Postep. Fitoter. 2008, 1, 47–58. [Google Scholar]
- Šarić, A.; Balog, T.; Sobočanec, S.; Kušić, B.; Šverko, V.; Rusak, G.; Likić, S.; Bubalo, D.; Pinto, B.; Reali, D.; et al. Antioxidant effects of flavonoid from Croatian Cystus incanus L. rich bee pollen. Food Chem. Toxicol. 2009, 47, 547–554. [Google Scholar] [CrossRef] [PubMed]
- Serra Bonvehi, J.; Soliva Torrentó, M.; Centelles Lorente, E. Evaluation of polyphenolic compounds in honeybee-collected pollen produced in Spain. J. Agric. Food Chem. 2001, 49, 1848–1853. [Google Scholar] [CrossRef] [PubMed]
- Chu, Q.; Tian, X.; Jiang, L.; Ye, J. Application of capillary electrophoresis to study phenolic profiles of honeybee-collected pollen. J. Agric. Food Chem. 2007, 31, 8864–8869. [Google Scholar] [CrossRef] [PubMed]
- Robak, J. Flavonoids as drugs inhibiting aggregation of blood platelets. Farm. Polska 1993, 49, 1–4. [Google Scholar]
- Cholbi, M.R.; Paya, M.; Alcaraz, M.J. Inhibitory effects of phenolic compounds on CCl4-induced microsomal lipid peroxidation. Experientia 1991, 47, 195–199. [Google Scholar] [CrossRef] [PubMed]
- Cook, N.C.; Samman, S. Flavonoids—Chemistry, metabolism, cardioprotective effects and dietary sources. J. Nutr. Biochem. 1996, 7, 66–76. [Google Scholar] [CrossRef]
- Silva, T.M.S.; Camara, C.A.; da Silva Lins, A.C.; Barbosa-Filho, J.M.; da Silva, E.M.S.; Freitas, B.M.; dos Santos, F.D.A.R. Chemical composition and free radical scavenging activity of pollen loads from stingless bee Melipona subnitida Ducke. J. Food Compos. Anal. 2006, 19, 507–511. [Google Scholar] [CrossRef]
- Čeksteryte, V.; Kazlauskas, S. Composition of flavonoids in Lithuanian honey and beebread. Biologija 2006, 2, 28–33. [Google Scholar]
- Campos, M.G.; Webby, R.F.; Markham, K.R.; Mitchell, K.A.; da Cuhna, A.P. Age-induced diminution of free radical scavenging capacity in bee pollens and the contribution of constituent flavonoids. J. Agric. Food Chem. 2003, 51, 742–745. [Google Scholar] [CrossRef] [PubMed]
- Markham, K.R.; Campos, M. 7- and 8-O-methylherbacetin-3-O-sophorosides from bee pollens and some structure/activity observations. Phytochemistry 1996, 43, 763–767. [Google Scholar] [CrossRef]
- Dauguet, J.C.; Bert, M.; Dolley, J.; Bekaerta, A.; Lewin, G. 8-Methoxykaempferol 3-neohesperidoside and other flavonoids from bee pollen of Crataegus monogyna. Phytochemistry 1993, 33, 1503–1505. [Google Scholar] [CrossRef]
- Puzanowska-Tarasiewicz, H.; Kuźmicka, L.; Tarasiewicz, M. Antioxidants and reactive oxygen species. Bromatol. Chem. Toksykol. 2010, 43, 9–14. [Google Scholar]
- Sikorski, Z.E. Nutritional and health properties of food compounds. In Food Chemistry, 1st ed.; Sikorski, Z., Ed.; WNT: Warsaw, Poland, 2007; Volume 3, pp. 204–220. [Google Scholar]
- Kędzia, B.; Hołderna-Kędzia, E. Bee Products in Medicine, 1st ed.; Apis: Lublin, Poland, 2007; pp. 45–61. [Google Scholar]
- Bogdanov, S. Quality and standards of pollen and beeswax. Apiacta 2004, 38, 334–341. [Google Scholar]
- Konishi, Y.; Hitomi, Y.; Yoshioka, E. Intestinal absorption of p-coumaric and gallic acids in rats after oral administration. J. Agric. Food Chem. 2004, 52, 2527–2532. [Google Scholar] [CrossRef] [PubMed]
- Donovan, J.L.; Crespy, V.; Manach, C.; Morand, C.; Besson, C.; Scalbert, A.; Rémésy, C. Catechin is metabolized by both the small intestine and liver of rats. J. Nutr. 2001, 131, 1753–1757. [Google Scholar] [PubMed]
- Hollman, P.C.; van Het Hof, K.H.; Tijbutg, L.B.; Katan, M.B. Addidion of milk does not affect the absorption of flavonols from tea in man. Free Radic. Res. 2001, 34, 297–300. [Google Scholar] [CrossRef] [PubMed]
- Hollman, P.C. Absorption, bioavailability and metabolism of flavonoids. Pharm. Biol. 2004, 42, 74–83. [Google Scholar] [CrossRef]
- Cermak, R.; Vujicic, Z.; Scharrer, E.; Wolfram, S. The impact of different flavonoid classes on colonic Cl- secretion in rats. Biochem. Pharmacol. 2001, 62, 1145–1151. [Google Scholar] [CrossRef]
- Aherne, S.A.; O’Brien, N.M. Dietary flavonols: Chemistry, food content and metabolism. Nutrition 2002, 18, 75–81. [Google Scholar] [CrossRef]
- Hollman, P.C.; Katan, M.B. Absorption, metabolism and health effects of dietary flavonoids in man. Biomed. Pharmacother. 1997, 51, 305–310. [Google Scholar] [CrossRef]
- Olthof, M.R.; Hollman, P.C.; Buijsman, M.N.; van Amelsvoort, J.M.; Katan, M.B. Chlorogenic acid, quercetin-3-rutinoside and black tea phenols are extensively metabolized in humans. J. Nutr. 2003, 133, 1806–1814. [Google Scholar] [PubMed]
- Walle, T. Absorption and metabolism of flavonoids. Free Radic. Biol. Med. 2004, 36, 829–837. [Google Scholar] [CrossRef] [PubMed]
- King, R.A.; Broadbent, L.J.; Head, R.J. Absorption and excretion of the soy isoflavone genistenin in rats. J. Nutr. 1996, 126, 176–182. [Google Scholar] [PubMed]
- Bravo, L. Polyphenols: Chemistry, dietary sources, metabolism and nutritional significance. Nutr. Rev. 1998, 56, 317–333. [Google Scholar] [CrossRef] [PubMed]
- Fiorani, M.; de Sanctis, R.; de Bellis, R.; Dachà, M. Intracellular flavonoids as electron donors for extracellular ferricyanide reduction in human erythrocytes. Free Radic. Biol. Med. 2002, 32, 64–72. [Google Scholar] [CrossRef]
- Feás, X.; Vázquez-Tato, M.P.; Estevinho, L.; Seijas, J.A.; Iglesias, A. Organic bee pollen: Botanical origin, nutritional value, bioactive compounds, antioxidant activity and microbiological quality. Molecules 2012, 17, 8359–8377. [Google Scholar] [CrossRef] [PubMed]
- Szczęsna, T. Long chain fatty acids composition of honeybee-collected pollen. J. Apic. Sci. 2006, 50, 65–79. [Google Scholar]
- Nagai, T.; Inoue, R.; Suzuki, N.; Tanoue, Y.; Kai, N.; Nagashima, T. Antihypertensive activities of enzymatic hydrolysates from honeybee-collected pollen of Cistus ladaniferus. Int. J. Food Agric. Environ. 2007, 5, 86–89. [Google Scholar]
- Samochowiec, L.; Wójcicki, J. Influence of Cernitin extracts on serum and liver lipids in rats on a high-fat diet. Herba Pol. 1983, 29, 165–170. [Google Scholar]
- Wójcicki, J. Experimental studies and clinical of flower pollen extracts. Herba Pol. 1987, 33, 49–54. [Google Scholar]
- Liu, X.; Li, L. Morphological observation of effect of bee pollen on intercellular lipofuscin in NIH mice. China J. Chin. Mater. Med. 1990, 15, 561–563. [Google Scholar]
- Campos, M.G.; Frigerio, C.; Lopes, J.; Bogdanov, S. What is the future of Bee-Pollen? J. ApiProd. ApiMed. Sci. 2010, 2, 131–144. [Google Scholar] [CrossRef]
- Ulbricht, C.; Conquer, J.; Giese, N.; Khalsa, K.P.S.; Sklar, J.; Weissner, W.; Woods, J. An evidence-based systematic review of bee pollen by the Natural Standard Research Collaboration. J. Diet. Suppl. 2009, 6, 290–312. [Google Scholar] [CrossRef] [PubMed]
- Yasumoto, R.; Kawanishi, H.; Tsujino, T.; Tsujita, M.; Nishisaka, N.; Horii, A.; Kishimoto, T. Clinical evaluation of long-term treatment using cernitin pollen extract in patients with benign prostatic hyperplasia. Clin. Ther. 1995, 17, 82–87. [Google Scholar] [CrossRef]
- Elist, J. Effects of pollen extract preparation Prostat/Poltit on lower urinary tract symptoms in patients with chronic nonbacterial prostatitis/chronic pelvic pain syndrome: A randomized, double-blind, placebo-controlled study. Urology 2006, 67, 60–63. [Google Scholar] [CrossRef] [PubMed]
- Buck, A.C.; Cox, R.; Rees, R.W.M.; Ebeling, L.; John, A. Treatment of outflow tract obstruction due to benign prostatic hyperplasia with the pollen extract, cernilton. A double-blind, placebo-controlled study. Br. J. Urol. 1990, 66, 398–404. [Google Scholar] [CrossRef] [PubMed]
- Choi, E.M. Antinociceptive and anti-inflammatory activities of pine (Pinus densiflora) pollen extract. Phytother. Res. 2007, 21, 471–475. [Google Scholar] [CrossRef] [PubMed]
- Wilt, T.J.; Macdonald, R.; Ishani, A.; Rutks, I.; Stark, G. Cernilton for benign prostatic hyperplasia. Cochrane Database Syst. Rev. 2011, 5. [Google Scholar] [CrossRef]
- Baltrusaityte, V.; Venskutonis, P.R.; Čeksteryte, V. Antibacterial activity of honey and Bee bread of different origin against S. aureus and S. epidermidis. Food Technol. Biotechnol. 2007, 45, 201–208. [Google Scholar]
- Knazovicka, V.; Melich, M.; Kacaniova, M.; Fikselova, M.; Hascik, P.; Chlebo, R. Antimicrobial activity of selected bee products. Acta Fytotech. Zootech. 2009, 12, 280–285. [Google Scholar]
- Basim, E.; Basim, H.S.; Özcan, M. Antibacterial activities of Turkish pollen and propolis extracts against plant bacterial pathogens. J. Food Eng. 2006, 77, 992–996. [Google Scholar] [CrossRef]
- Wu, Y.D.; Lou, Y.J. A steroid fraction of chloroform extract from bee pollen of Brassica campestris induces apoptosis in human prostate cancer PC-3 cells. Phytother. Res. 2007, 21, 1087–1091. [Google Scholar] [CrossRef] [PubMed]
- Pinto, B.; Caciagli, F.; Riccio, E.; Reali, D.; Šarić, A.; Balog, T.; Likić, S.; Scarpato, R. Antiestrogenic and antigenotoxic activity of bee pollen from Cystus incanus and Salix alba as evaluated by the yeast estrogen screen and the micronucleus assay in human lymphocytes. Eur. J. Med. Chem. 2010, 45, 4122–4128. [Google Scholar] [CrossRef] [PubMed]
- Haro, A.; López-Aliaga, I.; Lisbona, F.; Barrionuevo, M.; Alférez, M.J.; Campos, M.S. Beneficial effect of pollen and/or propolis on the metabolism of iron, calcium, phosphorus, and magnesium in rats with nutritional ferropenic anemia. J. Agric. Food Chem. 2000, 48, 5715–5722. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.S.; Fan, H.F.; Xu, H.J. Effects of bee pollen on blood and hemopoietic system in mice and rats. Chin. Tradit. Herb. Drugs 1993, 24, 588–591. [Google Scholar]
- Hamamoto, R.; Ishiyama, K.; Yamaguchi, M. Inhibitory effects of bee pollen Cistus ladaniferus extract on bone resorption in femoral tissues and osteoclast-like cell formation in bone marrow cells in vitro. J. Health Sci. 2006, 52, 268–275. [Google Scholar] [CrossRef]
- Yamaguchi, M.; Hamamoto, R.; Uchiyama, S.; Ishiyama, K.; Hashimoto, K. Anabolic effects of bee pollen Cistus ladaniferus extract on bone components in the femoral-diaphyseal and -metaphyseal tissues of rats in vitro and in vivo. J. Health Sci. 2006, 52, 43–49. [Google Scholar] [CrossRef]
- Yamaguchi, M.; Hamamoto, R.; Uchiyama, S.; Ishiyama, K.; Hashimoto, K. Preventive effects of bee pollen Cistus ladaniferus extract on bone loss in streptozotocin-diabetic rats in vivo. J. Health Sci. 2007, 53, 190–195. [Google Scholar] [CrossRef]
- Martins, M.C.T.; Morgano, M.A.; Vicente, E.; Baggio, S.R.; Rodriguez-Amaya, D.B. Physicochemical composition of bee pollen from eleven Brazilian states. J. Apic. Sci. 2011, 55, 107–116. [Google Scholar]
- Szczęsna, T. Study on the sugar composition of honeybee-collected pollen. J. Apic. Sci. 2007, 51, 15–22. [Google Scholar]
- Szczęsna, T.; Rybak-Chmielewska, H. Comparative research of the composition of bee pollen from different countries (Poland, South Corea, China). In Proceedings of the 35th Scientific Apiarian Conference, Puławy, Poland, 11–12 March 1998; pp. 76–77.
- Margaoan, R.; Mãrghitaş, L.; Dezmirean, D.; Mihai, C.M.; Bobis, O. Bee collected pollen—General aspects and chemical composition. Anim. Sci. Biotechnol. 2010, 67, 254–259. [Google Scholar]
- Szczęsna, T. Concentration of selected elements in honeybee-collected pollen. J. Apic. Sci. 2007, 51, 5–13. [Google Scholar]
- Tichonow, A.I.; Sodzawiczny, K.; Tichonowa, S.A.; Jarnych, T.G.; Bondarczuk, L.I.; Kolenko, A.M. Flower Pollen Bee Pollen in Pharmacy and Medicine. Theory, Technology, Application, Treatment, 1st ed.; Apipol Farma: Kraków, Poland, 2008; pp. 12–40, 79–85. [Google Scholar]
- Modro, A.F.H.; Message, D.; da Luz, C.F.P.; Meira Neto, J.A.A. Composição e qualidade de pólen apícola coletado em Minas Gerais. Pesqui. Agropecu. Bras. 2007, 42, 1057–1065. [Google Scholar] [CrossRef]
- Qian, W.L.; Khan, Z.; Watson, D.G.; Fearnley, J. Analysis of sugars in bee pollen and propolis by ligand exchange chromatography in combination with pulsed amperometric detection and mass spectrometry. J. Food Compos. Anal. 2008, 21, 78–83. [Google Scholar] [CrossRef]
- Kałużny, E. Apifitotherapy in nutrition, prevention and treatment. Part II. Wiad Ziel 2001, 43, 5–6. [Google Scholar]
- Czajka, A. Oxygen free radicals and body’s defense mechanism. Now Lekar 2006, 75, 582–586. [Google Scholar]
- Zabłocka, A.; Janusz, M. The two faces of reactive oxygen species. Postep. Hig. Med. Doświadczalnej 2008, 62, 118–124. [Google Scholar]
- Kuźnicki, D. Antioxidants and cholesterol’s level lowering agents in antiatherogenic plant materials. Postep. Fitoter. 2006, 4, 206–212. [Google Scholar]
- Bogdanov, S. Pollen: Production, Nutrition and Health: A Review. Bee-Hexagon.net. Available online: http://www.bee-hexagon.net/files/file/fileE/Health/PollenBook2Review.pdf (accessed on 17 June 2015).
- Markiewicz-Żukowska, R.; Naliwajko, S.K.; Bartosiuk, E.; Moskwa, J.; Isidorov, V.; Soroczyńska, J.; Borawska, M.H. Chemical composition and antioxidant activity of beebread, and its influence on the glioblastoma cell line (U87MG). J. Apic. Sci. 2013, 57, 147–157. [Google Scholar] [CrossRef]
- Kas’ianenko, V.I.; Komisarenko, I.A.; Dubtsova, E.A. Correction of atherogenic dyslipidemia with honey, pollen and bee bread in patients with different body mass. Terapevticheskii Arkhiv 2010, 83, 58–62. [Google Scholar]
- Uzbekova, D.G.; Makarova, V.; Khvoynitskaya, L.G.; Slepnev, A.A. Evaluation of bee-collected pollen influence on lipid peroxidation, antioxidant system and liver function in old animals. J. Hepatol. 2003, 38. [Google Scholar] [CrossRef]
- Anan’eva, T.V.; Dvoretskiĭ, A.I. Effect of beta-carotene oil and bee pollen on ion transport in rat brain slices following radiation-chemical exposure. Radiat. Biol. Radioecol. 1998, 39, 341–344. [Google Scholar]
- Kędzia, B.; Hołderna-Kędzia, E. Antihepatotoxic activity of bee pollen. Postep. Fitoter. 2011, 3, 202–207. [Google Scholar]
- Kędzia, B.; Hołderna-Kędzia, E. The possibility of application of bee products and herbal plants in the case of ethanol intoxication. Postep. Fitoter. 2009, 4, 248–255. [Google Scholar]
- Murakami, M.; Tsukada, O.; Okihara, K.; Hashimoto, K.; Yamada, H.; Yamaguchi, H. Beneficial effect of honeybee-collected pollen lump extract on benign prostatic hyperplasia BPH—A double-blind, placebo-controlled clinical trial. Food Sci. Technol. Res. 2008, 14, 306–310. [Google Scholar] [CrossRef]
- Lin, X.L.; Zhu, L.Q.; Yuan, Y.Y.; Li, L.M. Morphological changes in aged canine prostatic hyperplasia treated with bee pollen. Chin. Tradit. Herb. Drugs 1990, 21, 164–166. [Google Scholar]
- Campos, M.; Cunha, A.; Markham, K. Inhibition of Virulence of Pseudomonas Aeruginosa Cultures, by Flavonoids Isolated from Bee-Pollen: Possible Structure-Activity Relationships. In Polyphenol Communications 98, Proceedings of the XIX International Conference on Polyphenols, Lille, France, 1–4 September 1998; Groupe polyphenols: Bordeaux, France.
- Tichy, J.; Novak, J. Detection of antimicrobials in bee products with activity against viridans streptococci. J. Altern. Complement. Med. 2000, 6, 383–389. [Google Scholar] [CrossRef] [PubMed]
- Carpes, S.T.; Begnini, R.; Alencar, S.M.D.; Masson, M.L. Study of preparations of bee pollen extracts, antioxidant and antibacterial activity. Ciênc. Agrotecnol. 2007, 31, 1818–1825. [Google Scholar] [CrossRef]
- Kacániová, M.; Vuković, N.; Chlebo, R.; Haščík, P.; Rovná, K.; Cubon, J.; Dżugan, M.; Pasternakiewicz, A. The antimicrobial activity of honey, bee pollen loads and beeswax from Slovakia. Arch. Biol. Sci. 2012, 64, 927–934. [Google Scholar] [CrossRef]
- Izuta, H.; Shimazawa, M.; Tsuruma, K.; Araki, Y.; Mishima, S.; Hara, H. Bee products prevent VEGF-induced angiogenesis in human umbilical vein endothelial cells. BMC Complement. Altern. Med. 2009, 9. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Yuan, Q.; Rashid, F. Isolation, purification and immunobiological activity of a new water-soluble bee pollen polysaccharide from Crataegus pinnatifida Bge. Carbohydr. Polym. 2009, 78, 80–88. [Google Scholar] [CrossRef]
- Ishikawa, Y.; Tokura, T.; Nakano, N.; Hara, M.; Niyonsaba, F.; Ushio, H.; Yamamoto, Y.; Tadokoro, T.; Okumura, K.; Ogawa, H. Inhibitory Effect of Honeybee-Collected Pollen on Mast Cell Degranulation in Vivo and in Vitro. J. Med. Food 2008, 11, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, M.; Uchiyama, S.; Nakagawa, T. Anabolic effect of bee pollen Cistus ladaniferus extract in osteoblastic MC3T3-E1 cells in vitro. J. Health Sci. 2007, 53, 625–629. [Google Scholar] [CrossRef] [Green Version]
- Sample Availability: No samples are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rzepecka-Stojko, A.; Stojko, J.; Kurek-Górecka, A.; Górecki, M.; Kabała-Dzik, A.; Kubina, R.; Moździerz, A.; Buszman, E. Polyphenols from Bee Pollen: Structure, Absorption, Metabolism and Biological Activity. Molecules 2015, 20, 21732-21749. https://doi.org/10.3390/molecules201219800
Rzepecka-Stojko A, Stojko J, Kurek-Górecka A, Górecki M, Kabała-Dzik A, Kubina R, Moździerz A, Buszman E. Polyphenols from Bee Pollen: Structure, Absorption, Metabolism and Biological Activity. Molecules. 2015; 20(12):21732-21749. https://doi.org/10.3390/molecules201219800
Chicago/Turabian StyleRzepecka-Stojko, Anna, Jerzy Stojko, Anna Kurek-Górecka, Michał Górecki, Agata Kabała-Dzik, Robert Kubina, Aleksandra Moździerz, and Ewa Buszman. 2015. "Polyphenols from Bee Pollen: Structure, Absorption, Metabolism and Biological Activity" Molecules 20, no. 12: 21732-21749. https://doi.org/10.3390/molecules201219800
APA StyleRzepecka-Stojko, A., Stojko, J., Kurek-Górecka, A., Górecki, M., Kabała-Dzik, A., Kubina, R., Moździerz, A., & Buszman, E. (2015). Polyphenols from Bee Pollen: Structure, Absorption, Metabolism and Biological Activity. Molecules, 20(12), 21732-21749. https://doi.org/10.3390/molecules201219800










