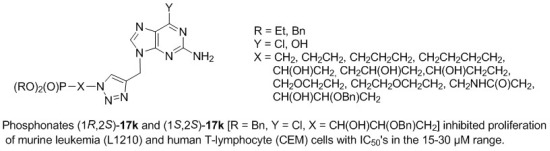
Phosphonylated Acyclic Guanosine Analogues with the 1,2,3-Triazole Linker
Abstract
:1. Introduction
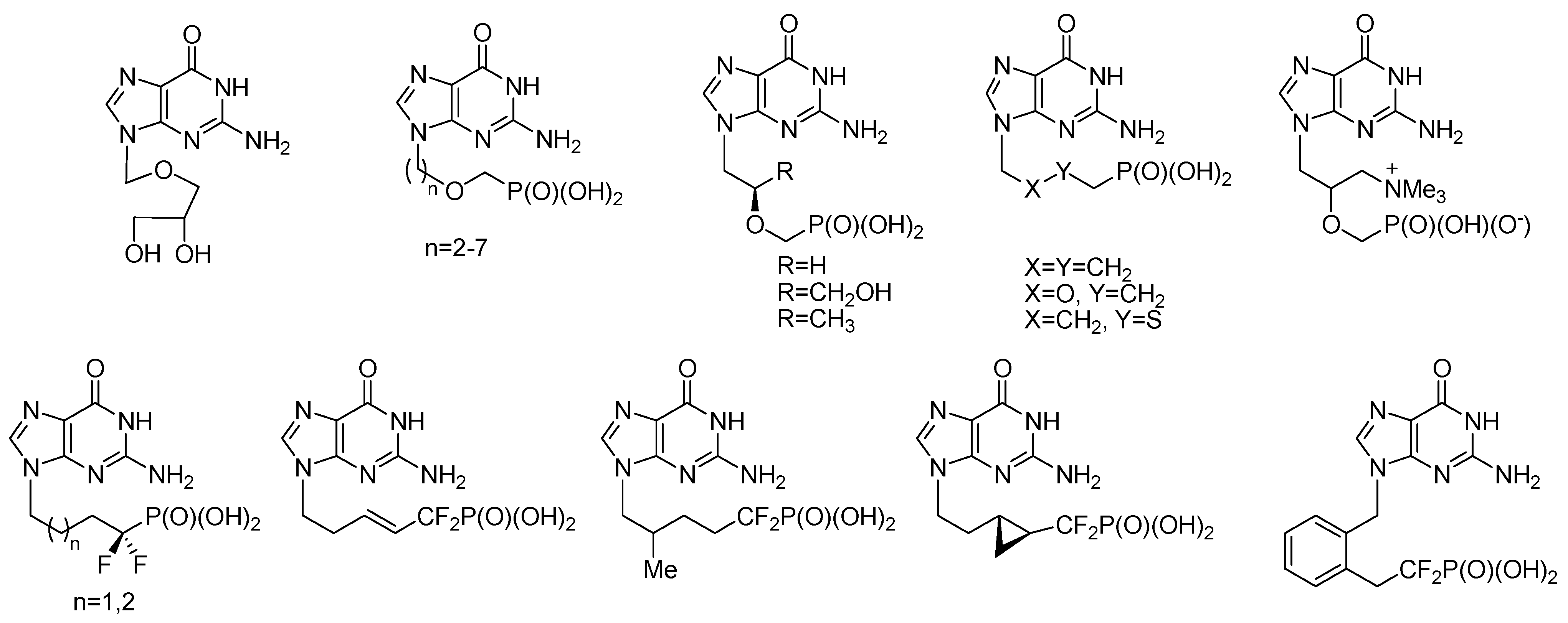
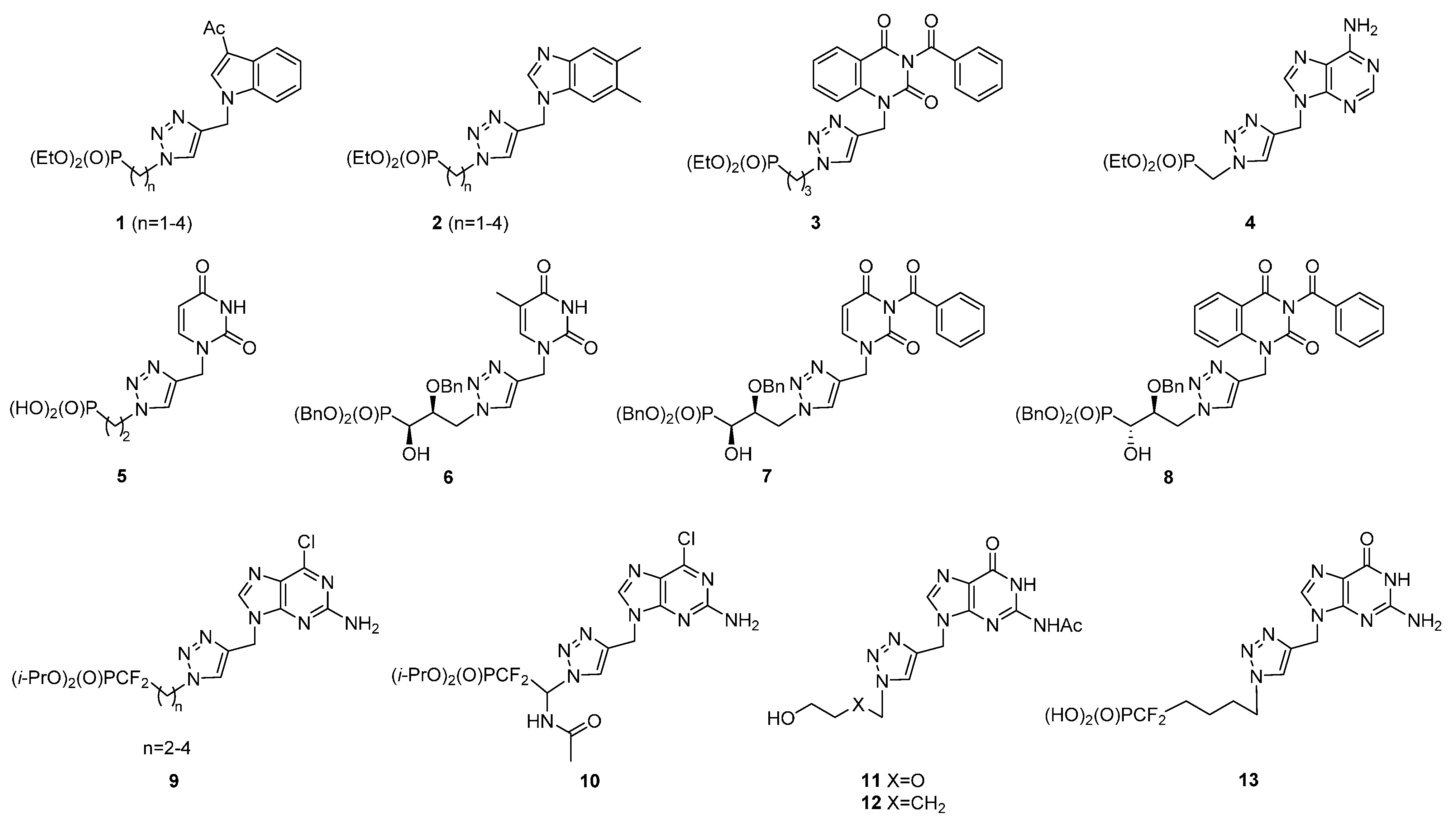
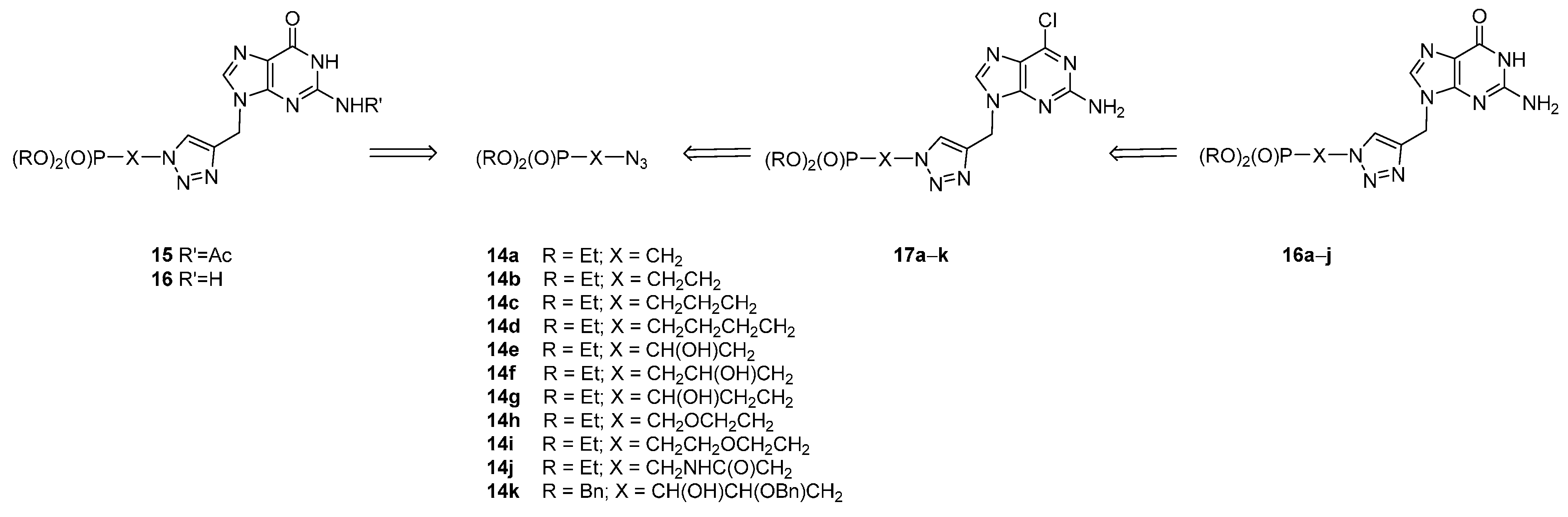
2. Results and Discussion
2.1. Chemistry


2.2. Antiviral Activity and Cytostatic/Cytotoxic Evaluation
| Compounds 16a–k and 17a–k | IC50a (µM) | ||
|---|---|---|---|
| L1210 | CEM | HeLa | |
| 17a | 283 ± 17 | ≥250 | ≥250 |
| 17b | ≥250 | >250 | 227 ± 32 |
| 17c | 227 ± 32 | >250 | ≥250 |
| 17d | >250 | >250 | ≥250 |
| 17e | >250 | >250 | >250 |
| 17f | >250 | >250 | >250 |
| 17g | >250 | >250 | >250 |
| 17h | >250 | ≥250 | >250 |
| 17i | >250 | >250 | >250 |
| 17j | >250 | >250 | >250 |
| (1R,2S)-17k | 21 ± 2 | 26 ± 8 | 90 ± 33 |
| (1S,2S)-17k | 16 ± 6 | 30 ± 16 | 84 ± 12 |
| 16a | >250 | >250 | >250 |
| 16b | >250 | >250 | >250 |
| 16c | >250 | >250 | 138 ± 52 |
| 16d | >250 | >250 | 148 ± 25 |
| 16e | >250 | >250 | 206 ± 49 |
| 16f | >250 | >250 | 195 ± 78 |
| 16g | >250 | >250 | 185 ± 35 |
| 16h | >250 | >250 | ≥250 |
| 16i | >250 | >250 | 210 ± 13 |
| 16j | >250 | >250 | 212 ± 54 |
| 5-fluorouracil | 0.33 ± 0.17 | 18 ± 5 | 0.54 ± 0.12 |
3. Experimental Section
3.1. General
3.2. General Procedure for the Synthesis of 17a–k
3.3. General Procedure for Transformation 17 into 16
3.4. Antiviral Activity Assays
3.5. Cytostatic Activity Assays
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Herdewijn, P. Modified Nucleosides: In Biochemistry, Biotechnology and Medicine; John Wiley & Sons: Weinheim, Germany, 2008. [Google Scholar]
- Chu, C.K. Antiviral Nucleosides: Chiral Synthesis and Chemotherapy; Elsevier: Amsterdam, The Netherlands, 2003. [Google Scholar]
- Dando, T.; Plosker, G. Adefovir dipivoxil: A review of its use in chronic hepatitis B. Drugs 2003, 63, 2215–2234. [Google Scholar] [CrossRef] [PubMed]
- Danta, M.; Dusheiko, G. Adefovir dipivoxil: Review of a novel acyclic nucleoside analogue. Int. J. Clin. Pract. 2004, 58, 877–886. [Google Scholar] [CrossRef] [PubMed]
- Cundy, K.C. Clinical pharmacokinetics of the antiviral nucleotide analogues cidofovir and adefovir. Clin. Pharmacokinet. 1999, 36, 127–143. [Google Scholar] [CrossRef] [PubMed]
- Srinivas, R.V.; Fridland, A. Antiviral activities of 9-R-2-phosphonomethoxypropyl adenine (PMPA) and bis(isopropyloxymethylcarbonyl)PMPA against various drug-resistant human immunodeficiency virus strains. Antimicrob. Agents Chemother. 1998, 42, 1484–1487. [Google Scholar]
- Ayoub, W.S. Tenofovir: A new option for treatment of chronic hepatitis B. Virus Adapt. Treat. 2009, 1, 5–10. [Google Scholar]
- Faulds, D.; Heel, R.C. Ganciclovir. Drugs 1990, 39, 597–638. [Google Scholar] [CrossRef] [PubMed]
- McGavin, J.K.; Goa, K.L. Ganciclovir: An update of its use in the prevention of cytomegalovirus infection and disease in transplant recipients. Drugs 2001, 61, 1153–1183. [Google Scholar] [CrossRef] [PubMed]
- Komatsu, T.E.; Pikis, A.; Naeger, L.K.; Harringhton, P.R. Resistance of human cytomegalovirus to ganciclovir/valganciclovir: A comprehensive review of putative resistance pathways. Antivir. Res. 2014, 101, 12–25. [Google Scholar] [CrossRef] [PubMed]
- Cvetković, R.S.; Wellington, K. Valganciclovir: A review of its use in the management of CMV infection and disease in immunocompromised patients. Drugs 2005, 65, 859–878. [Google Scholar] [CrossRef] [PubMed]
- Len, O.; Gavaldá, J.; Aguado, J.M.; Borrell, N.; Cervera, C.; Cisneros, J.M.; Cuervas-Mons, V.; Gurugi, M.; Martin-Dávila, P.; Montejo, M.; et al. Valganciclovir as treatment for cytomegalovirus disease in solid organ transplant recipients. Clin. Infect. Dis. 2008, 46, 20–27. [Google Scholar] [PubMed]
- Safrin, S.; Cherrinton, J.; Jaffe, H.S. Cidofovir: Review of current and potential clinical uses. Adv. Exp. Med. Biol. 1999, 458, 111–120. [Google Scholar] [PubMed]
- De Clercq, E. Towards an effective chemotherapy of virus infections: Therapeutic potential of cidofovir [(S)-1-[3-hydroxy-2-(phosphonomethoxy)propyl] cytosine, HPMPC] for the treatment of DNA virus infections. Collect. Czechoslv. Chem. Comunn. 1998, 63, 480–506. [Google Scholar] [CrossRef]
- Halazy, S.; Ehrhard, A.; Danzin, C. 9-(Difluorophosphonoalkyl)guanines as a new class of multisubstrate analog inhibitors of purine nucleoside phosphorylase. J. Am. Chem. Soc. 1991, 113, 315–317. [Google Scholar] [CrossRef]
- Yokomatsu, T.; Abe, H.; Sato, M.; Suemune, K.; Kihara, T.; Soeda, S.; Shimeno, H.; Shibuya, S. Synthesis of 1,1-difluoro-5-(1H-9-purinyl)-2-pentenylphosphonic acids and the related methano analogues. Remarkable effect of the nucleobases and thecyclopropane rings on inhibitory activity toward purine nucleoside phosphorylase. Bioorg. Med. Chem. 1998, 6, 2495–2505. [Google Scholar] [PubMed]
- Yokomatsu, T.; Hayakawa, Y.; Kihara, T.; Soeda, S.; Shimeno, H.; Shibuya, S. Synthesis and biological evaluation of 1,1-difluoro-2-(tetrahydro-3-furanyl)ethylphosphonic acids possessing a N9-purinylmethyl functional group at the ring. A new class of inhibitors for purine nucleoside phosphorylases. Bioorg. Med. Chem. Lett. 1999, 9, 2833–2836. [Google Scholar] [CrossRef]
- Kim, C.U.; Luh, B.Y.; Misco, P.F.; Bronson, J.J.; Hitchcock, M.J.M.; Ghazzouli, I.; Martin, J.C. Acyclic purine phosphonate analogs as antiviral agents. Synthesis and structure-activity relationships. J. Med. Chem. 1990, 33, 1207–1213. [Google Scholar] [PubMed]
- Dvořáková, H.; Masojídková, M.; Holý, A.; Balzarini, J.; Andrei, G.; Snoeck, R.; de Clerq, E. Synthesis of 2′-aminomethyl derivatives of N-(2-(phosphonomethoxy)ethyl) nucleotide analogues as potential antiviral agents. J. Med. Chem. 1996, 39, 3263–3268. [Google Scholar] [CrossRef] [PubMed]
- Holý, A.; Günter, J.; Dvořáková, H.; Masojídková, M.; Andrei, G.; Snoeck, R.; Balzarini, J.; de Clerq, E. Structure—Antiviral activity relationship in the series of pyrimidine and purine N-[2-(2-phosphonomethoxy)ethyl] nucleotide analogues. 1. Derivatives substituted at the carbon atoms of the base. J. Med. Chem. 1999, 42, 2064–2086. [Google Scholar] [PubMed]
- Yokomatsu, T.; Hayakawa, Y.; Kihara, T.; Koyanagi, S.; Soeda, S.; Shimeno, H.; Shibuya, S. Synthesis and evaluation of multisubstrate analogue inhibitors of purine nucleoside phosphorylases. Bioorg. Med. Chem. 2000, 8, 2571–2579. [Google Scholar] [CrossRef]
- Radi, S.; Lazrek, H.B. Synthesis of certain new 1,2,3-triazole acyclonucleosides via 1,3-dipolar cycloaddition. Bull. Korean. Chem. Soc. 2002, 23, 437–440. [Google Scholar] [CrossRef]
- Iwanow, M.; Magnowska, I.; Yokomatsu, T.; Shibuya, S.; Bzowska, A. Interactions of potent multisubstrate analogue inhibitors with purine nucleoside phosphorylase from calf spleen—kinetic and spectrofluorimetric studies. Nucleosides Nucleotides Nucleic Acids 2003, 22, 1567–1570. [Google Scholar] [CrossRef] [PubMed]
- Zhou, D.; Lagoja, I.M.; Van Aerschot, A.; Herdewijn, P. Synthesis of aminopropyl phosphonate nucleosides with purine and pyrimidine bases. Collect. Czechoslv. Chem. Commun. 2006, 71, 15–34. [Google Scholar] [CrossRef]
- Hikishima, S.; Isobe, M.; Koyanagi, S.; Soeda, S.; Shimeno, H.; Shibuya, S.; Yokomatsu, T. Synthesis and biological evaluation of 9-(5′,5′-difluoro-5′-phosphonopentyl)guanine derivatives for PNP-inhibitors. Bioorg. Med. Chem. 2006, 14, 1660–1670. [Google Scholar] [CrossRef] [PubMed]
- Keough, D.T.; Hocková, D.; Holý, A.; Naesens, L.; Skinner-Adams, T.S.; de Jersey, J.; Guddat, L. Inhibition of hypoxanthine-guanine phosphoribosyltransferase by acyclic nucleoside phosphonates: A new class of antimalarial therapeutics. J. Med. Chem. 2009, 52, 4391–4399. [Google Scholar] [CrossRef] [PubMed]
- Vertuani, S.; Baldisserotto, A.; Varani, K.; Borea, P.A.; Cruz, B.M.M.; Ferraro, L.; Manfredini, S.; Dalpiaz, A. Synthesis and in vitro stability of nucleoside 5′-phosphonate derivatives. Eur. J. Med. Chem. 2012, 54, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Tichý, T.; Andrei, G.; Snoeck, R.; Balzarini, J.; Dračínský, M.; Krečmerová, M. Synthesis and antiviral activities of hexadecyloxypropyl prodrugs of acyclic nucleoside phosphonates containing guanine or hypoxanthine and a (S)-HPMP or PEE acyclic moiety. Eur. J. Med. Chem. 2012, 55, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Kramer, R.A.; Bleicher, K.H.; Wennemers, H. Design and synthesis of nucleoproline amino acids for the straightforward preparation of chiral and conformationally constrained nucleopeptides. Helv. Chim. Acta 2012, 95, 2621–2633. [Google Scholar] [CrossRef]
- Keough, D.T.; Špaček, P.; Hocková, D.; Tichý, T.; Vrbková, S.; Slavětínská, L.; Janeba, Z.; Naesens, L.; Edstein, M.D.; Chavchich, M.; et al. Acyclic nucleoside phosphonates containing a second phosphonate group are potent inhibitors of 6-oxopurine phosphoribosyltransferases and have antimalarial activity. J. Med. Chem. 2013, 56, 2513–2526. [Google Scholar] [PubMed]
- Keough, D.T.; Špaček, P.; Hocková, D.; Tichý, T.; Vrbková, S.; Slavětínská, L.; Janeba, Z.; Naesens, L.; Edstein, M.D.; Chavchich, M.; et al. Plasmodium vivax hypoxanthine-guanine phosphoribosyltransferase: A target for anti-malarial chemotherap. Mol. Biochem. Parasitol. 2010, 173, 165–169. [Google Scholar]
- De Clercq, E. Strategies in the design of antiviral drugs. Nat. Rev. Drug Discov. 2002, 1, 13–25. [Google Scholar] [CrossRef] [PubMed]
- De Clercq, E.; Holý, A. Acyclic nucleoside phosphonates: A key class of antiviral drugs. Nat. Rev. Drug Discov. 2005, 4, 928–940. [Google Scholar] [CrossRef] [PubMed]
- De Clercq, E. The acyclic nucleoside phosphonates (ANPs): Antonín Holý’s legacy. Med. Res. Rev. 2013, 33, 1278–1303. [Google Scholar] [CrossRef] [PubMed]
- Holý, A. Phosphonomethoxyalkyl analogs of nucleotides. Curr. Pharm. Des. 2003, 9, 2567–2592. [Google Scholar] [CrossRef] [PubMed]
- Diab, S.A.; Hienzch, A.; Lebargy, C.; Guillarme, S.; Pfund, E.; Lequeux, T. Synthesis of fluorophosphonylated acyclic nucleotide analogues via copper(I)-catalyzed Huisgen 1-3 dipolar cycloaddition. Org. Biomol. Chem. 2009, 7, 4481–4490. [Google Scholar] [CrossRef] [PubMed]
- Elayadi, H.; Smietana, M.; Pannecouque, Ch.; Leyssen, P.; Neyts, J.; Vasseur, J.-J.; Lazrek, H.B. Straightforward synthesis of triazoloacyclonucleotide phosphonates as potential HCV inhibitors. Bioorg. Med. Chem. Lett. 2010, 20, 7365–7368. [Google Scholar] [CrossRef] [PubMed]
- Głowacka, I.E.; Balzarini, J.; Wróblewski, A.E. The synthesis, antiviral, cytostatic and cytotoxic evaluation of a new series of acyclonucleotide analogues with a 1,2,3-triazole linker. Eur. J. Med. Chem. 2013, 70C, 703–722. [Google Scholar] [CrossRef] [PubMed]
- Głowacka, I.E.; Balzarini, J.; Piotrowska, D.G. Design, synthesis, antiviral and cytostatic activity of ω-(1H-1,2,3-triazol-1-yl)(polyhydroxy)alkylphosphonates as acyclic nucleotide analogues. Bioorg. Med. Chem. 2014, 22, 3629–3641. [Google Scholar] [CrossRef] [PubMed]
- Diab, S.A.; de Schutter, C.; Muzard, M.; Plantier-Royon, R.; Pfund, E.; Lequeux, T. Fluorophosphonylated nucleoside derivatives as new series of thymidine phosphorylase multisubstrate inhibitors. J. Med. Chem. 2012, 55, 2758–2768. [Google Scholar] [CrossRef] [PubMed]
- Lazrek, H.B.; Taourirte, M.; Oulih, T.; Lebtoumi, M.; Barascut, J.L.; Imbach, J.L. Synthesis of new 1,2,3-triazole acyclonucleoside analogs of ACV and HBG. Nucleosides Nucleotides 1997, 16, 1115–1118. [Google Scholar] [CrossRef]
- Lazrek, H.B.; Taourirte, M.; Oulih, T.; Barascut, J.L.; Imbach, J.L.; Pannecouque, C.; Witrouw, M.; de Clercq, E. Synthesis and anti-HIV activity of new modified 1,2,3-triazole acyclonucleosides. Nucleosides Nucleotides Nucleic Acids 2001, 20, 1949–1960. [Google Scholar] [CrossRef] [PubMed]
- Krim, J.; Sillahi, B.; Taourirte, M.; Rakib, E.M.; Engels, J.W. Microwave-assisted click chemistry: Synthesis of mono and bis-1,2,3-triazole acyclonucleoside analogues of acyclovir via copper(I)-catalyzed cycloaddition. Arkivoc 2009, 142, 142–152. [Google Scholar]
- Luo, L.; He, X.P.; Shen, Q.; Li, J.-Y.; Shi, X.-X.; Xie, J.; Li, J.; Chen, G.-R. Synthesis of (glycopyranosyl-triazolyl)-purines and their inhibitory activities against protein tyrosine phosphatase 1B (PTP1B). Chem. Biodivers. 2011, 8, 2035–2043. [Google Scholar] [CrossRef] [PubMed]
- Nagapradeep, N.; Verma, S. Characterization of an unprecedented organomercury adduct via Hg(II)-mediated cyclization of N9-propargylguanine. Chem. Commun. 2011, 47, 1755–1757. [Google Scholar] [CrossRef] [PubMed]
- Hocková, D.; Keough, D.T.; Janeba, Z.; Naesens, L.; Wang, T.-H.; de Jersey, J.; Guddat, L.W. Synthesis of novel N-branched acyclic nucleoside phosphonates as potent and selective inhibitors of human, plasmodium falciparum and plasmodium vivax 6-oxopurine phosphoribosyltransferases. J. Med. Chem. 2012, 55, 6209–6223. [Google Scholar] [CrossRef] [PubMed]
- Krečmerová, M.; Dračinský, M.; Hocková, D.; Holý, A.; Keough, D.T.; Guddat, L.W. Synthesis of purine N9-[2-hydroxy-3-O-(phosphonomethoxy)propyl] derivatives and their side-chain modified analogues as potential antimalarial agents. Bioorg. Med. Chem. 2012, 20, 1222–1230. [Google Scholar] [CrossRef] [PubMed]
- Liao, S.; Fan, S-.Y.; Liu, Q.; Li, C.-K.; Chen, J.; Li, J.-L.; Zhang, Z.-W.; Zhang, Z.-Q.; Zhong, B.-H.; Xie, J.-W. In vitro evaluation of 9-(2-phosphonylmethoxyethyl)adenine ester analogues, a series of anti-HBV structures with improved plasma stability and liver release. Arch. Pharm. Res. 2014, 37, 1416–1425. [Google Scholar]
- Keith, K.A.; Hitchcock, M.J.M.; Lee, W.A.; Holý, A.; Kern, E.R. Evaluation of nucleoside phosphonates and their analogs and prodrugs for inhibition of orthopoxvirus replication. Antimicrob Agents Chemother. 2003, 47, 2193–2198. [Google Scholar] [CrossRef] [PubMed]
- Jansa, P.; Baszczyňski, O.; Dračínský, M.; Votruba, I.; Zídek, Z.; Bahador, G.; Stepan, G.; Cihlar, T.; Mackman, R.; Holý, A.; et al. A novel and efficient one-pot synthesis of symmetrical diamide (bis-amidate) prodrugs of acyclic nucleoside phosphonates and evaluation of their biological activities. Eur. J. Med. Chem. 2011, 46, 3748–3754. [Google Scholar] [PubMed]
- Pradère, U.; Roy, V.; Montagu, A.; Sari, O.; Hamada, M.; Balzarini, J.; Snoeck, R.; Andrei, G.; Agrofoglio, L.A. Synthesis and antiviral evaluation of bis(POM) prodrugs of (E)-[4′-phosphono-but-2′-en-1′-yl]purine nucleosides. Eur. J. Med. Chem. 2012, 57, 126–133. [Google Scholar] [CrossRef] [PubMed]
- De Clercq, E. Highlights in the discovery of antiviral drugs: A personal retrospective. J. Med. Chem. 2010, 53, 1438–1450. [Google Scholar] [CrossRef] [PubMed]
- Hecker, S.J.; Erion, M.D. Prodrugs of Phosphates and Phosphonates. J. Med. Chem. 2008, 51, 2328–2345. [Google Scholar] [CrossRef] [PubMed]
- Głowacka, I.E.; Balzarini, J.; Piotrowska, D.G. 1-Amino-3-(1H-1,2,3-triazol-1-yl)propylphosphonates as acyclic analogues of nucleotides. Arch. Pharm. Chem. Life Sci. 2014, 347, 496–505. [Google Scholar] [CrossRef] [PubMed]
- Lindsell, W.E.; Murray, Ch.; Preston, P.N.; Woodman, T.A.J. Synthesis of 1,3-diynes in the purine, pyrimidine, 1,3,5-triazine and acridine series. Tetrahedron 2000, 56, 1233–1245. [Google Scholar] [CrossRef]
- Vo, D.D.; Staedel, C.; Zehnacker, L.; Benhinda, R.; Darfeullie, F.; Duca, M. Targeting the production of oncogenic microRNAs with multimodal synthetic small molecules. ACS Chem. Biol. 2014, 9, 711–721. [Google Scholar] [CrossRef] [PubMed]
- Wróblewski, A.E.; Głowacka, I.E. Synthesis of (1S,2S)- and (1R,2S)-3-amino-1,2-dihydroxypropylphosphonic acids. Polish J. Chem. 2005, 79, 1895–1899. [Google Scholar]
- Wróblewski, A.E.; . Głowacka, I.E. Synthesis of four enantiomerically pure 4-(4-carbamoyl-1,2,3-triazol-1-yl)-2,3-dihydroxy-1-methoxybutylphosphonic acids. Tetrahedron 2005, 61, 11930–11938. [Google Scholar] [CrossRef]
- Głowacka, I.E.; Balzarini, J.; Wróblewski, A.E. Design, synthesis, antiviral, and cytotoxic evaluation of novel phosphonylated 1,2,3-triazoles as acyclic nucleotide analogues. Nucleosides Nucleotides Nucleic Acids 2012, 31, 293–318. [Google Scholar] [CrossRef] [PubMed]
- Głowacka, I.E.; Balzarini, J.; Wróblewski, A.E. Synthesis and biological evaluation of novel 1,2,3-triazolonucleotides. Arch. Pharm. Chem. Life Sci. 2013, 346, 278–291. [Google Scholar] [CrossRef] [PubMed]
- Głowacka, I.E.; Balzarini, J.; Wróblewski, A.E. Synthesis of a new series of phosphonylated 1,2,3-triazoles as acyclic analogs of ribavirin. Arch. Pharm. Chem. Life Sci. 2013, 346, 677–687. [Google Scholar] [CrossRef] [PubMed]
- Bankowska, E.; Balzarini, J.; Głowacka, I.E.; Wróblewski, A.E. Design, synthesis, antiviral and cytotoxic evaluation of novel acyclic phosphonate nucleotide analogues with a 5,6-dihydro-1H-[1,2,3]triazolo[4,5-d]pyridazine-4,7-dione system. Monatsh. Chem. 2013, 145, 663–673. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Not avaliable.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Głowacka, I.E.; Andrei, G.; Schols, D.; Snoeck, R.; Piotrowska, D.G. Phosphonylated Acyclic Guanosine Analogues with the 1,2,3-Triazole Linker. Molecules 2015, 20, 18789-18807. https://doi.org/10.3390/molecules201018789
Głowacka IE, Andrei G, Schols D, Snoeck R, Piotrowska DG. Phosphonylated Acyclic Guanosine Analogues with the 1,2,3-Triazole Linker. Molecules. 2015; 20(10):18789-18807. https://doi.org/10.3390/molecules201018789
Chicago/Turabian StyleGłowacka, Iwona E., Graciela Andrei, Dominique Schols, Robert Snoeck, and Dorota G. Piotrowska. 2015. "Phosphonylated Acyclic Guanosine Analogues with the 1,2,3-Triazole Linker" Molecules 20, no. 10: 18789-18807. https://doi.org/10.3390/molecules201018789