New Flavanol and Cycloartane Glucosides from Landoltia punctata
Abstract
:1. Introduction
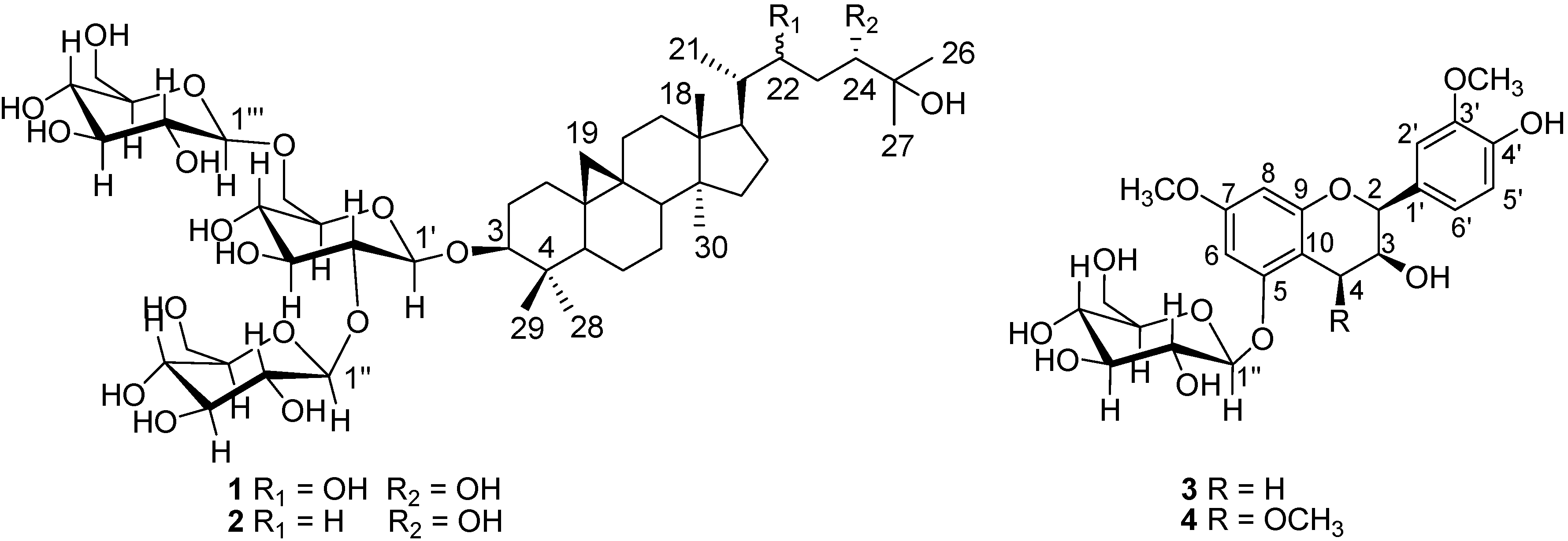
2. Results and Discussion
2.1. Structure Elucidation of Compounds 1–4
 + 51.2, which was determined by TLC analysis and optical rotation measurement [19,20]. Therefore, 1 was considered to be a cycloartane-type triterpene glucoside.
+ 51.2, which was determined by TLC analysis and optical rotation measurement [19,20]. Therefore, 1 was considered to be a cycloartane-type triterpene glucoside. 



2.2. Biological Activity Assay
3. Experimental
3.1. General Information
3.2. Material
3.3. Extraction and Isolation
3.4. Hydrolysis
3.5. Spectral Data
 −3.0 (c 0.1, MeOH). IR (KBr) νmax 3411, 2932, 1075 cm−1. 1H and 13C-NMR: See Table 1. HR-ESI-MS (positive mode) m/z 985.5371 [M+Na]+ (calc. for C48H82O19Na, 985.5343).
−3.0 (c 0.1, MeOH). IR (KBr) νmax 3411, 2932, 1075 cm−1. 1H and 13C-NMR: See Table 1. HR-ESI-MS (positive mode) m/z 985.5371 [M+Na]+ (calc. for C48H82O19Na, 985.5343). −4.0 (c 0.1, MeOH). IR (KBr) νmax 3418, 2891, 1043 cm−1. 1H and 13C-NMR: See Table 1. HR-ESI-MS (positive mode) m/z 969.5406 [M + Na]+ (calc. for C48H82O18Na, 969.5393).
−4.0 (c 0.1, MeOH). IR (KBr) νmax 3418, 2891, 1043 cm−1. 1H and 13C-NMR: See Table 1. HR-ESI-MS (positive mode) m/z 969.5406 [M + Na]+ (calc. for C48H82O18Na, 969.5393). −42.0 (c 0.1, MeOH). UV (MeOH) λmax (log ε) 218 (4.60), 227 (3.96), 282 (3.52) nm. IR (KBr) νmax 3434, 1623, 1080 cm−1. 1H and 13C-NMR: See Table 2. HR-ESI-MS (negative mode) m/z 479.1549 (calc. for C23H27O11, 479.1559).
−42.0 (c 0.1, MeOH). UV (MeOH) λmax (log ε) 218 (4.60), 227 (3.96), 282 (3.52) nm. IR (KBr) νmax 3434, 1623, 1080 cm−1. 1H and 13C-NMR: See Table 2. HR-ESI-MS (negative mode) m/z 479.1549 (calc. for C23H27O11, 479.1559).  −20.0 (c 0.1, MeOH). UV (MeOH) λmax (log ε) 226 (4.02), 280 (3.60) nm. IR (KBr) νmax 3420, 2926, 1597, 1424, 1079 cm−1. 1H and 13C-NMR: See Table 2. HR-ESI-MS (negative mode) m/z 509.1662 (calc. for C24H29O12, 509.1664).
−20.0 (c 0.1, MeOH). UV (MeOH) λmax (log ε) 226 (4.02), 280 (3.60) nm. IR (KBr) νmax 3420, 2926, 1597, 1424, 1079 cm−1. 1H and 13C-NMR: See Table 2. HR-ESI-MS (negative mode) m/z 509.1662 (calc. for C24H29O12, 509.1664).3.6. Biological Activity Assay
| No. | 1 | 2 | No. | 1 | 2 | ||||
|---|---|---|---|---|---|---|---|---|---|
| δC | δH | δC | δH | δC | δH | δC | δH | ||
| 1 | 33.3 | 1.65–1.67 (m), 1.32–1.34 (m) (m) | 30.3 | 2.50–2.53 (m), 1.25–1.26 (m) | 25 | 74.0 | 73.1 | ||
| 2 | 30.7 | 2.08–2.11 (m), 1.35–1.36 (m) | 29.3 | 1.81–1.82 (m), 1.95–1.97 (m) | 26 | 26.1 | 1.24 (s, 3H) | 26.5 | 1.56 (s, 3H) |
| 3 | 90.9 | 3.34–3.36 (m) | 89.1 | 3.45–3.48 (m) | 27 | 25.6 | 1.23 (s, 3H) | 26.2 | 1.54 (s, 3H) |
| 4 | 42.4 | 41.7 | 28 | 26.0 | 1.13 (s, 3H) | 26.0 | 1.29 (s, 3H) | ||
| 5 | 50.4 | 1.68–1.70 (m) | 47.8 | 1.20–1.22 (m) | 29 | 15.6 | 0.95 (s, 3H) | 15.7 | 1.19 (s, 3H) |
| 6 | 22.3 | 1.67–1.68 (m), 1.68–1.70 (m) | 27.2 | 1.87–1.88 (m), 1.37 (overlapped) | 30 | 20.1 | 1.01 (s, 3H) | 18.6 | 0.99 (s, 3H) |
| 7 | 27.4 | 1.39–1.40 (m), 1.36–1.37 (m) | 33.5 | 1.50–1.51 (m), 1.53 (overlapped) | 1' | 105.3 | 4.52 (d, J = 7.20, 1H) | 105.2 | 4.90 (d, J = 7.56, 1H) |
| 8 | 49.7 | 1.60–1.63 (m) | 53.3 | 1.64–1.65 (m) | 2' | 81.4 | 3.62–3.63 (m) | 83.5 | 4.15–4.17 (m) |
| 9 | 21.4 | 20.2 | 3' | 78.2 | 3.61–3.62 (m) | 77.3 | 4.10–4.11 (m) | ||
| 10 | 27.5 | 26.9 | 4' | 71.7 | 3.39–3.40 (m) | 72.0 | 4.20–4.21 (m) | ||
| 11 | 27.7 | 1.70–1.72 (m), 2.10–2.11 (m) | 21.5 | 1.45–1.46 (m), 0.70–0.72 (m) | 5' | 77.1 | 3.52–3.54 (m) | 77.2 | 4.03–4.04 (m) |
| 12 | 34.2 | 1.72, 1.76, overlapped | 32.6 | 1.64–1.65 (m), 1.24 (overlapped) | 6' | 70.2 | 3.85–3.87 (m), 4.16–4.18 (br. d) | 70.4 | 4.29–4.31 (m), 4.82 (br. d) |
| 13 | 46.9 | 45.9 | 1'' | 104.8 | 4.74 (d, J = 7.68, 1H) | 106.3 | 5.35 (d, J = 7.62, 1H) | ||
| 14 | 49.8 | 49.3 | 2'' | 76.5 | 3.28–3.29 (m) | 75.6 | 4.04–4.05 (m) | ||
| 15 | 37.0 | 1.40–1.41 (m), 1.42–1.43 (m) | 36.7 | 1.65–1.66 (m), 1.55 (overlapped) | 3'' | 78.4 | 3.60–3.61 (m) | 78.3 | 4.24–4.25 (m) |
| 16 | 28.4 | 1.42–1.43 (m), 2.03–2.05 (m) | 28.8 | 1.37–1.38 (m), 1.63–1.64 (m) | 4'' | 72.0 | 3.26–3,27 (m) | 71.8 | 4.28–4.29 (m) |
| 17 | 50.0 | 1.79–1.81 (m) | 48.4 | 1.43–1.44 (m) | 5'' | 78.1 | 3.30–3.31 (m) | 78.6 | 3.90–3.92 (m) |
| 18 | 18.7 | 1.11 (s, 3H) | 19.9 | 0.84 (s, 3H) | 6'' | 63.0 | 3.71–3.72 (m), 3.93–3.94 (m) | 63.0 | 4.45–4.46 (m), 4.52–4.53 (m) |
| 19 | 30.9 | 0.44 (d, J = 3.72) | 30.0 | 0.24 (d, J = 4.08) | 1''' | 105.0 | 4.48 (d, J = 7.74, 1H) | 105.7 | 5.13 ( d, J = 7.80, 1H) |
| 0.64 (d, J = 3.72) | 0.48 (d, J = 4.08) | 2''' | 75.4 | 3.24–3.26 (m) | 75.5 | 4.05–4.06 (m) | |||
| 20 | 44.0 | 1.78–1.79 (m) | 36.1 | 1.23–1.24 (m) | 3''' | 78.0 | 3.40–3.42 (m) | 78.8 | 3.94–3.96 (m) |
| 21 | 12.7 | 0.95 (s, 3H) | 18.9 | 1.01 (s, 3H) | 4''' | 71.9 | 3.31–3.32 (m) | 71.1 | 4.03–4.04 (m) |
| 22 | 71.3 | 4.01 (br. d) | 23.2 | 0.86–0.87 (m), 1.25 (overlapped) | 5''' | 78.1 | 3.29–3.30 (m) | 78.4 | 4.21–4.22 (m) |
| 23 | 32.9 | 1.40–1.41 (m), 1.53–1.57 (m) | 34.5 | 1.83–1.85 (m), 1.68 (overlapped) | 6''' | 63.3 | 3.70–3.71 (m), 3.90–3.92 (m) | 63.1 | 4.33–4.36 (m), 4.48–4.50 (m) |
| 24 | 76.2 | 3.58–3.60 (m) | 79.4 | 3.78–3.81 (m) | |||||
| No. | 3 | 4 | ||
|---|---|---|---|---|
| δC | δH | δC | δH | |
| 2 | 80.3 | 4.98 (br. s, 1H) | 78.6 | 5.05 (br. s, 1H) |
| 3 | 67.4 | 4.29 (br. s, 1H) | 69.2 | 4.04 (br. d, 1H) |
| 4 | 29.7 | 3.09 (d, J = 17.60, 1H)3.02(1H, dd) | 73.5 | 4.62 (d, J = 2.28, 1H) |
| 3.02 (dd, J = 17.60, 4.38, 1H) | ||||
| 5 | 158.6 | 157.9 | ||
| 6 | 97.1 | 6.49(d, J = 2.10) | 96.6 | 6.54 (d, J = 1.80) |
| 7 | 160.9 | 162.9 | ||
| 8 | 96.5 | 6.29 (d, J = 2.10) | 96.3 | 6.30 (d, J = 1.80) |
| 9 | 157.3 | 160.4 | ||
| 10 | 104.0 | 104.4 | ||
| 1' | 132.3 | 131.6 | ||
| 2' | 112.1 | 7.22 (d, J = 1.06) | 112.3 | 7.22 (d, J = 1.10) |
| 3' | 148.8 | 149.0 | ||
| 4' | 147.2 | 147.4 | ||
| 5' | 115.9 | 6.87 (d, J = 8.16) | 116.0 | 6.90 (d, J = 8.04) |
| 6' | 120.8 | 6.99 (dd, J = 1.06, 8.16) | 121.0 | 7.00 (dd, J = 1.10, 8.04) |
| 1'' | 102.8 | 4.95 (d, J = 7.20) | 102.6 | 4.99 (d, J = 7.50) |
| 2'' | 75.1 | 3.50–3.55 (overlapped) | 75.3 | 3.58–3.59 (overlapped) |
| 3'' | 78.4 | 3.50–3.55 (overlapped) | 78.5 | 3.53–3.56 (overlapped) |
| 4'' | 71.6 | 3.47–3.48 (m) | 71.6 | 3.46–3.48 (overlapped) |
| 5'' | 78.2 | 3.50–3.55 (overlapped) | 76.9 | 3.53–3.56 (overlapped) |
| 6'' | 62.7 | 3.79 (dd, J = 5.58, 12.10) | 62.8 | 3.79 (dd, J = 5.00, 12.20) |
| 3.98 (d, J = 12.10) | 4.00 (d, J = 12.20) | |||
| MeO-4 | 56.6 | 3.65 (s, 3H) | ||
| MeO-7 | 56.6 | 3.82 (s, 3H) | 56.0 | 3.84 (s, 3H) |
| MeO-3' | 56.0 | 3.95 (s, 3H ) | 57.8 | 3.96 (s, 3H) |
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ge, X.M.; Zhang, N.N.; Phillips, G.C.; Xu, J.F. Growing Lemna minor in agricultural wastewater and converting the duckweed biomass to ethanol. Bioresour. Technol. 2012, 124, 485–488. [Google Scholar] [CrossRef]
- Zhao, H.; Appenroth, K.; Landesman, L.; Salmeán, A.A.; Lam, E. Duckweed rising at Chengdu: Summary of the 1st International Conference on Duckweed Application and Research. Plant. Mol. Boil. 2012, 78, 627–632. [Google Scholar] [CrossRef]
- Cole, C.T.; Voskuil, M.I. Population genetic structure in duckweed (Lemna minor, Lemnaceae). Can. J. Bot. 1996, 74, 222–230. [Google Scholar] [CrossRef]
- Xu, J.; Zhao, H.; Stomp, A.M.; Cheng, J.J. The production of duckweed as a source of biofuels. Biofuels 2012, 3, 589–601. [Google Scholar] [CrossRef]
- Ozengin, N.; Elmaci, A. Performance of duckweed (Lemna minor L.) on different types of wastewater treatment. J. Environ. Biol. 2007, 28, 307–314. [Google Scholar]
- Mohedano, R.A.; Costa, R.H.; Tavares, F.A.; Filho, B.P. High nutrient removal rate from swine wastes and protein biomass production by full-scale duckweed ponds. Bioresour. Technol. 2012, 112, 98–104. [Google Scholar] [CrossRef]
- Cheng, J.J.; Stomp, A.M. Growing duckweed to recover nutrients from wastewaters and for production of fuel ethanol and animal feed. Clean 2009, 37, 17–26. [Google Scholar]
- Xu, J.; Cui, W.; Cheng, J.J.; Stomp, A.M. Production of high-starch duckweed and its conversion to bioethanol. Biosyst. Eng. 2011, 110, 67–72. [Google Scholar] [CrossRef]
- Xiao, Y.; Fang, Y.; Jin, Y.; Zhang, G.; Zhao, H. Culturing duckweed in the field for starch accumulation. Ind. Crop. Prod. 2013, 48, 183–190. [Google Scholar] [CrossRef]
- Jurd, L.; Geissman, T.A.; Seikel, M.K. The flavonoid constituents of Spirodela oligorrhiza II. The flavone constituents. Arch. Biochem. Biophys. 1957, 67, 284–297. [Google Scholar] [CrossRef]
- Lee, Y.H.; Lee, I.R.; Won, W.S.; Park, C.H. Flavonoids of Elscholtzia cristata. Arch. Pharm. Res. 1988, 11, 247–249. [Google Scholar] [CrossRef]
- Rayyan, S.; Fossen, T.; Nateland, H.S.; Andersen, Q.M. Isolation and identification of flavonoids, including flavone rotamers, from the herbal drug ‘crataegi folium cum flore’ (hawthorn). Phytochem. Anal. 2005, 16, 334–341. [Google Scholar] [CrossRef]
- Kawasaki, M.; Kanomata, T.; Yoshitama, K. Flavonoids in the leaves of twenty-eight polygonaceous plants. Bot. Mag. Tokyo 1986, 99, 63–74. [Google Scholar] [CrossRef]
- Dubois, M.A.; Zoll, A.; Markham, K.R.; Bouillant, M.L.; Dellamonica, G.; Chopin, J. 6-c-β-d-glucopyranosyl-8-c-β-d-galactopyranosylapigenin from Cerastium arvense. Phytochemistry 1984, 23, 706–707. [Google Scholar] [CrossRef]
- Kamboj, A.; Salujai, A.K. Isolation of stigmasterol and β-sitosterol from petroleum ether extract of aerial parts of Ageratum conyzoides (Asteraceae). Int. J. Pharm. Pharm. Sci. 2011, 3, 94–96. [Google Scholar]
- Cui, J.; Wang, H.; Huang, Y.; Xin, Y.; Zhou, A. Synthesis and cytotoxic analysis of some disodium 3β, 6β-dihydroxysterol disulfates. Steroids 2009, 74, 1057–1060. [Google Scholar] [CrossRef]
- Polat, E.; Caliskan-Alankus, O.; Perrone, A.; Piacente, S.; Bedir, E. Cycloartane-type glycosides from Astragalus amblolepis. Phytochemistry 2009, 70, 628–634. [Google Scholar] [CrossRef]
- Yokosuka, A.; Sato, K.; Yamori, T.; Mimaki, Y. Triterpene glycosides from Curculigo orchioides and their cytotoxic activity. J. Nat. Prod. 2010, 73, 1102–1106. [Google Scholar] [CrossRef]
- Chaturvedula, P.V.S.; Prakash, I. A new diterpene glycoside from Stevia rebaudiana. Molecules 2011, 16, 2937–2943. [Google Scholar] [CrossRef]
- Mohan, R.; Birari, R.; Karmase, A.; Jagtap, S.; Bhutani, K.K. Antioxidant activity of a new phenolic glycoside from Lagenaria siceraria Stand. fruits. Food Chem. 2012, 132, 244–251. [Google Scholar] [CrossRef]
- Bedir, E.; Calis, I.; Khan, I.A. Macrophyllosaponin E: A novel compound from the roots of Astragalus oleifolius. Chem. Pharm. Bull. 2000, 48, 1081–1083. [Google Scholar] [CrossRef]
- Hirotani, M.; Zhou, Y.; Rut aTsutomu Furuya, H. Cycloartane triterpene glycosides from the hairy root cultures of Astragalus. membranaceus. Phytochemistry 1994, 37, 1403–1407. [Google Scholar] [CrossRef]
- Thompson, R.S.; Jacques, D.; Haslam, E.; Tanner, R.J.N. Plant proanthocyanidins. Part I. Introduction; the isolation, structure, and distribution in nature of plant procyanidins. J. Chem. Soc. Perkin Trans. 1 1972, 1387–1399. [Google Scholar]
- Fletcher, A.C.; Porter, L.J.; Haslam, E.; Gupta, R.K. Plant proanthocyanidins. Part 3. Conformational and configurational studies of natural procyanidins. J. Chem. Soc. Perkin Trans. 1 1977, 1628–1637. [Google Scholar]
- Mateus, N.; Silva, A.M.; Santos-Buelga, C.; Rivas-Gonzalo, J.C.; de Freitas, V. Identification of anthocyanin-flavanol pigments in red wines by NMR and mass spectrometry. J. Agric. Food Chem. 2002, 50, 2110–2116. [Google Scholar] [CrossRef]
- Otsuka, H.; Hirata, E.; Shinzato, T.; Takeda, Y. Glochiflavanosides AD: Flavanol glucosides from the leaves of Glochidion zeylanicum (Gaertn) A. Juss. Chem. Pharm. Bull. 2001, 49, 921–923. [Google Scholar] [CrossRef]
- Li, S.Y.; Fuchino, H.; Kawahara, N.; Sekita, S.; Satake, M. New phenolic constituents from Smilax bracteata. J. Nat. Prod. 2002, 65, 262–266. [Google Scholar] [CrossRef]
- Li, F.; Zhan, Z.; Liu, F.; Yang, Y.; Li, L.; Feng, Z.; Jiang, J.S.; Zhang, P. Polyflavanostilbene A, a new flavanol-fused stilbene glycoside from Polygonum cuspidtum. Org. Lett. 2013, 15, 674–677. [Google Scholar]
- Agati, G.; Azzarello, E.; Pollastri, S.; Tattini, M. Flavonoids as antioxidants in plants: Location and functional significance. Plant. Sci. 2012, 196, 67–76. [Google Scholar] [CrossRef]
- Aron, P.M.; Kennedy, J.A. Flavan-3-ols: Nature, occurrence and biological activity. Mol. Nutr. Food Res. 2008, 52, 79–104. [Google Scholar] [CrossRef]
- Scheid, L.; Reusch, A.; Stehle, P.; Ellinger, S. Antioxidant effects of cocoa and cocoa products ex vivo and in vivo: Is there evidence from controlled intervention studies? Curr. Opin. Clin. Nutr. Metab. Care 2010, 13, 737–742. [Google Scholar] [CrossRef]
- Xu, G.B.; Yang, T.; Bao, J.K.; Fang, D.M.; Li, G.Y. Isochaetomium A2, a new bis (naphthodihydropyran-4-one) with antimicrobial and immunological activities from fungus Chaetomium microcephalum. Arch. Pharm. Res. 2013. [Google Scholar] [CrossRef]
- Tian, S.Z.; Pu, X.; Luo, G.Y.; Zhao, L.X.; Xu, L.H.; Li, W.J.; Luo, Y.G. Isolation and characterization of new p-terphenyls with antifungal, antibacterial, and antioxidant activities from halophilic actinomycete Nocardiopsis gilva YIM 90087. J. Agric. Food Chem. 2013, 61, 3006–3012. [Google Scholar] [CrossRef]
- Tao, X.; Fang, Y.; Xiao, Y.; Jin, Y.L.; Ma, X.R.; Zhao, Y.; He, K.Z.; Zhao, H.; Wang, H.Y. Comparative transcriptome analysis to investigate the high starch accumulation of duckweed (Landoltia punctata) under nutrient starvation. Biotechnol. Biofuels 2013, 6, 72–87. [Google Scholar] [CrossRef]
- Li, S.F.; Di, Y.T.; Luo, R.H.; Zheng, Y.T.; Wang, Y.H.; Fang, X.; Zhang, Y.; Li, L.; He, H.P.; Li, S.L. Cycloartane triterpenoids from Cassia occidentalis. Planta. Med. 2012, 78, 821–827. [Google Scholar] [CrossRef]
- Wang, X.Y.; Tang, G.H.; Yuan, C.M.; Zhang, Y.; Zou, T.; Yu, C.; Zhao, Q.; Hao, X.J.; He, H.P. Aphagrandinoids A–D, cycloartane triterpenoids with antibacterial activities from Aphanamixis grandifolia. Fitoterapia 2013, 85, 64–68. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 3, 6, 7–13 are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, N.; Xu, G.; Fang, Y.; Yang, T.; Zhao, H.; Li, G. New Flavanol and Cycloartane Glucosides from Landoltia punctata. Molecules 2014, 19, 6623-6634. https://doi.org/10.3390/molecules19056623
Wang N, Xu G, Fang Y, Yang T, Zhao H, Li G. New Flavanol and Cycloartane Glucosides from Landoltia punctata. Molecules. 2014; 19(5):6623-6634. https://doi.org/10.3390/molecules19056623
Chicago/Turabian StyleWang, Nini, Guobo Xu, Yang Fang, Tao Yang, Hai Zhao, and Guoyou Li. 2014. "New Flavanol and Cycloartane Glucosides from Landoltia punctata" Molecules 19, no. 5: 6623-6634. https://doi.org/10.3390/molecules19056623
APA StyleWang, N., Xu, G., Fang, Y., Yang, T., Zhao, H., & Li, G. (2014). New Flavanol and Cycloartane Glucosides from Landoltia punctata. Molecules, 19(5), 6623-6634. https://doi.org/10.3390/molecules19056623





