1. Introduction
Angelica Radix, the roots of the genus
Angelica (“Danggui” in Chinese), has been used for more than 2,000 years as a traditional medicine in China, Korea and Japan. Angelica Radix is often called “female ginseng” because it is traditionally applied to the treatment of gynecological disorders [
1]. The medical use of
Angelica root was first recorded in ~100 B.C in
Shen Nong Bencao Jing, which stated that
Angelica root is effective in replenishing the blood, reducing pain and moistening the intestine. Clinically, Angelica Radix can be applied to treat anemia [
2], to enhance the immune system [
3] and to relieve constipation [
4]. The Pharmacopoeia of People’s Republic of China (PRC) records more than 100 herbal formulas containing
Angelica roots in China and Japan [
5]. Nowadays,
Angelica root is not only commonly consumed in Asia, but also in the Western countries as a health food supplement. The Chinese Pharmacopoeia (2010) defines that
Angelica root in China is derived from the root of
Angelica sinensis (Oliv.) Diels. However,
Angelica acutiloba (Sieb.
et Zucc.) Kitag., mainly found in Japan, and
Angelica gigas Nakai, mainly found in Korea, are commonly used instead of
A. sinensis in Japan and Korea. The three commonly used
Angelica roots showed variation in their compositions, pharmacological properties and efficacy [
6]. In addition, the cultivated regions of
Angelica also have a significant difference in chemical quantities and biological response [
7] even when they are parts of a decoction [
8]. The study concluded that the source of herb should be considered when preparing decoctions in order to achieve the maximum biological efficacy and minimum toxicity [
8]. Thus, the authentication of different
Angelica species is an important task.
Angelica species were previously distinguished by sequencing of internal transcribed spacers, nuclear ribosomal DNA, 5S-rRNA spacer and 18S-rRNA [
6]. Although these DNA-based methods are effective, they can only be applied to the crude herb and not to the prepared product, such as ready-to-consume decoctions or tablets. Chemical marker identification is also applied on the quality control of
Angelica; ferulic acid and
Z-ligustilide were chosen as the markers for the quality control [
5,
9]. These two compounds however are not unique to
Angelica, and are not necessarily clinically relevant. It is also unclear if these markers are sufficient to distinguish the closely related
Angelica species.
Recently, there is an increasing interest in using metabolic profiling approaches to obtain comprehensive chemical signatures for quality control of herbal medicines [
10,
11,
12]. Kim
et al. have applied
1H-NMR and UPLC-MS for the quality control of
A. gigas of different geographical origins in Korea [
13]. By using methanol extraction with UPLC-MS, they successfully discriminate
A. gigas from 3 different regions in Korea. Kobayashi
et al. applied GC-MS metabolic profiling to discriminate
A. acutiloba and
A. sinensis [
11]. Here, we demonstrated that
1H-NMR metabolic profiling could be used to distinguish the hot water extracts of two different species of Angelica Radix from three geographically different cultivation regions. We established that mainly by measuring primary metabolites,
1H-NMR metabolic profiling can be used to distinguish these closely related herbal samples, even if a less potent extraction method is used. Importantly, by employing a more clinically relevant method of preparation of boiling in water, instead of methanol extraction used in previous studies, we showed that our method could be applied to commercial ready-to-consume herbal products.
2. Results and Discussion
The average spectra of
A. sinensis from China and
A. gigas from ROK and DPRK were obtained and are shown in
Figure 1. Visual inspection showed that all three spectra were indeed rather similar. The most intense peaks were found between 3.3–4 ppm, which corresponded to sugar compounds. Sucrose was the highest concentration among these peaks, in good agreement with the result of Kim
et al. [
13], who employed methanol extraction. In addition, we identified glucose and fructose, as well as other primary metabolites including a few amino acids and organic acids. However, we could not detect the specific secondary metabolites, such as decursin and other coumarin derivatives, as revealed by Kim
et al. [
13]. Decursin is considered as one of the active compounds in
Angelica, especially in
A. gigas, which is reported to have beneficial effects against cancer, oxidative stress, neurological disorders, fat accumulation and obesity-induced diabetes [
14]. Comparing our results to Kim
et al., we concluded that the typical method of preparing
Angelica roots for consumption (
i.e., boiling in water), which we mimicked in this study, did not produce detectable levels of these useful compounds.
Reduced-resolution spectra (~1,100 data points representing integrated regions of 0.01 ppm width) were exported to SIMCA-P for multivariate analysis. Principle component analysis (PCA) was applied to data analysis. PCA is the most basic and efficient method for analyzing complex data in metabonomics, which helps to extract and display systematic variations from the data, as well as detects groupings, trends and outliers if they are present in the data [
15].
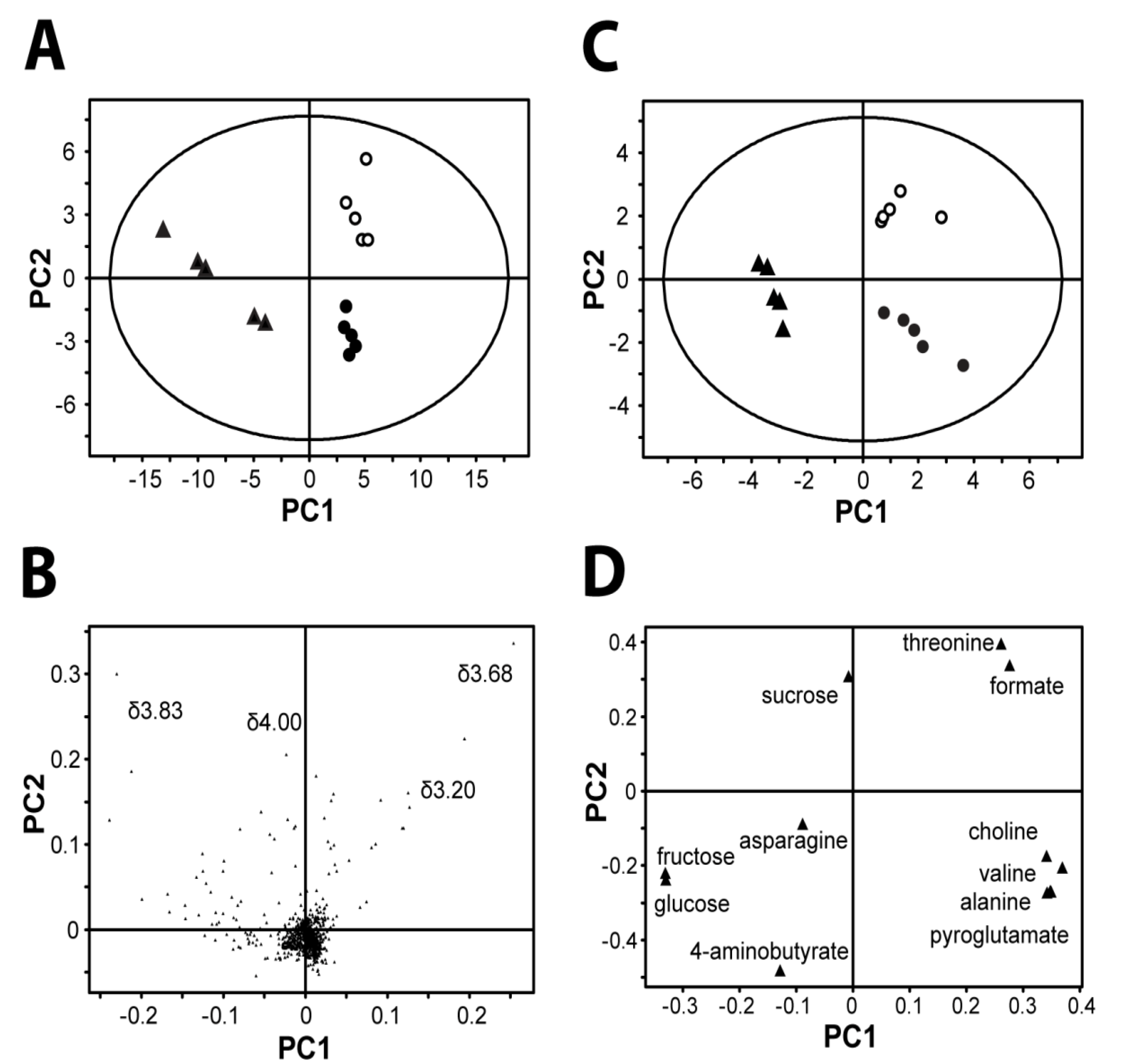
Figure 2A shows a PCA plot. Each point in the PCA score plot represented a single sample, and samples clustered together were considered to have similar characteristics—in this case, similar metabolic profiles. The three
Angelica roots,
A. sinensis root (China) and
A. gigas roots (ROK and DPRK) were successfully discriminated based on their water extracts alone. By studying the corresponding PCA loading plots, the discriminating features were clearly identified (
Figure 2B). The most discriminating peaks are found in the sugar region between 3.4–4 ppm. In order to understand how other metabolites also contribute to the discrimination, we then performed PCA on the integrated peak areas of the identified metabolites only (
Figure 2C). Clear separation of the three
Angelica samples was similarly obtained. The corresponding loading plot indicated that
A. sinesis from China showed a higher concentration of glucose, fructose and 4-aminobutyrate, while threonine, formate and succinate were found higher in
A. gigas from ROK. Choline, valine, acetate, alanine and pyroglutamate were found at higher levels in
A. gigas from DPRK (
Figure 2D).
Figure 1.
Average 1H-NMR Spectra of three Angelica extracts, A. sinensis from China (PRC) and A. gigas from ROK and DPRK. Spectra represented the mean of five replicates. By visual inspection, the average 1H-NMR spectra of the three herbs showed a very similar profile, but observable differences in the finer details. The most intense peaks were found between 3.3–4 ppm, which are associated with carbohydrates. A visible difference in peak pattern was also found between 5.5–8.5 ppm, associated with aromatic compounds.
Figure 1.
Average 1H-NMR Spectra of three Angelica extracts, A. sinensis from China (PRC) and A. gigas from ROK and DPRK. Spectra represented the mean of five replicates. By visual inspection, the average 1H-NMR spectra of the three herbs showed a very similar profile, but observable differences in the finer details. The most intense peaks were found between 3.3–4 ppm, which are associated with carbohydrates. A visible difference in peak pattern was also found between 5.5–8.5 ppm, associated with aromatic compounds.
All identified and integrated peaks were further evaluated with Student t-tests (
Figure 3 and
Table 1). Sucrose was found at the highest intensity in
Angelica roots, followed by fructose and glucose. One-way ANOVA determined that the two
A. gigas groups (ROK and DPRK) have significantly lower (
p < 0.001) levels of glucose and fructose than the
A. sinensis (PRC) group, and those sugars alone cannot distinguish the two
A. gigas groups significantly. Among the other metabolite examined, several amino acids, including threonine, asparagine and valine, can alone differentiate all three groups (
p < 0.01). Acetate, alanine, asparagines, choline, pyroglutamate and valine exhibit similar trends, with highest levels found in
A. gigas from DPRK, followed by
A. gigas from ROK (
Table 1 and
Figure 3). Levels of these metabolites are lowest in
A. sinensis.
Figure 2.
Chemometric analysis by principle component analysis (PCA). (A) and (B): Score and loading plots from pattern recognition (PCA) of whole 1H-NMR spectra are shown. The data sets are Pareto-scaled. Overall, discrimination of all three groups of Angelica (n = 5) was clearly shown in the score plot. The loading plot shows that 3.2–4 ppm, the sugar region, is the most discriminating feature. (PC1: R2 = 0.70, Q2 = 0.63; PC2: R2 = 0.86, Q2 = 0.78) (C) and (D): Scores and loading plots from pattern recognition (PCA) of a selection of targeted molecules. Data are unit-variance scaled. Loading plot shows which molecules contributed most to the discrimination. (PC1: R2 = 0.56, Q2 = 0.34; PC2: R2 = 0.74, Q2 = 0.36). Abbreviation: PRC, A. sinensis from China; DPRK, A. gigas from Democratic People’s Republic of Korea; ROK, A. gigas from Republic of Korea.
Figure 2.
Chemometric analysis by principle component analysis (PCA). (A) and (B): Score and loading plots from pattern recognition (PCA) of whole 1H-NMR spectra are shown. The data sets are Pareto-scaled. Overall, discrimination of all three groups of Angelica (n = 5) was clearly shown in the score plot. The loading plot shows that 3.2–4 ppm, the sugar region, is the most discriminating feature. (PC1: R2 = 0.70, Q2 = 0.63; PC2: R2 = 0.86, Q2 = 0.78) (C) and (D): Scores and loading plots from pattern recognition (PCA) of a selection of targeted molecules. Data are unit-variance scaled. Loading plot shows which molecules contributed most to the discrimination. (PC1: R2 = 0.56, Q2 = 0.34; PC2: R2 = 0.74, Q2 = 0.36). Abbreviation: PRC, A. sinensis from China; DPRK, A. gigas from Democratic People’s Republic of Korea; ROK, A. gigas from Republic of Korea.
▲ A. sinensis (PRC) ● A. gigas (DPRK) ○ A. gigas (ROK)
Figure 3.
Quantification of identified metabolites in the different
Angelica extracts. The peaks associated with identified metabolites (listed in
Table 1 with respective chemical shifts) in all of the
1H-NMR spectra were integrated to yield the concentrations in the extracts relative to the mean value for
A. sinensis (PRC) (basal level set at 1.0). The values are expressed as Means ± SEM (
n = 5). *
p ≤ 0.05, **
p ≤ 0.01, ***
p ≤ 0.001, by one-way ANOVA followed by post-hoc multiple comparison tests. Abbreviations: PRC,
A. sinensis from China; DPRK,
A. gigas from Democratic People’s Republic of Korea; ROK,
A. gigas from Republic of Korea.
Figure 3.
Quantification of identified metabolites in the different
Angelica extracts. The peaks associated with identified metabolites (listed in
Table 1 with respective chemical shifts) in all of the
1H-NMR spectra were integrated to yield the concentrations in the extracts relative to the mean value for
A. sinensis (PRC) (basal level set at 1.0). The values are expressed as Means ± SEM (
n = 5). *
p ≤ 0.05, **
p ≤ 0.01, ***
p ≤ 0.001, by one-way ANOVA followed by post-hoc multiple comparison tests. Abbreviations: PRC,
A. sinensis from China; DPRK,
A. gigas from Democratic People’s Republic of Korea; ROK,
A. gigas from Republic of Korea.
Table 1.
Chemical shifts of metabolites identified in 1H-NMR Spectra of different sources of Angelica roots used for quantification.
Table 1.
Chemical shifts of metabolites identified in 1H-NMR Spectra of different sources of Angelica roots used for quantification.
| Metabolite | Chemical Shift (ppm) |
|---|
| Acetate | 1.9(s) |
| Alanine | 1.5(d) |
| Asparagine | 2.93 (m) |
| 4-aminobutyrate | 2.3(t) |
| Choline | 3.2(s) |
| Formate | 8.4 (s) |
| Fructose | 4.6(d) |
| Glucose | 5.2(d) |
| Pyroglutamate | 2.0(m) |
| Succinate | 2.4(s) |
| Sucrose | 5.4(d) |
| Threonine | 1.3(d) |
| Valine | 1.1(d) |
| Ferulic Acid | 6.3(d) |
Ferulic acid (δ 6.3) and
Z-ligustilide are often considered as important biomarkers in
Angelica. Ferulic acid inhibits platelet aggregation and serotonin release while
Z-ligustilide has anti-asthmatic and spasmolytic activity [
1]. By simple solubility tests using pure standards, we found that
Z-lingustilide is water insoluble, while ferulic acid has a low water solubility (~6 mg/mL). Kim
et al. did not show that they could detect Z-ligustilide or ferulic acid by methanol extraction with
1H-NMR and UPLC-MS [
13]. Similarly, Kobayashi
et al. did not report observing these two markers by GC-MS metabolic profiling on
A. acutiloba and
A. sinensis [
11]. Here,
Z-ligustilide was not detected in the
1H-NMR spectra, but ferulic acid was observed at 6.3 ppm. Our result suggests that
A. sinensis from China has a higher average ferulic acid concentration than
A. gigas from Korea but the difference is not statistically significant. This result partially agreed with Zhao
et al. [
6], who also detected higher ferulic acid concentrations in
A. sinensis than in
A. gigas. However, we did not observe as large as difference as that was reported by Zhao
et al. [
6]. The level of ferulic acid in the DPRK
A. gigas sample was not detected, because the peak of interest at 6.3 ppm was overlapped with another substantial peak of unknown identity. Unresolved peaks in our analyses could potentially be identified and quantified by using 2D-NMR or hyphenated techniques, such as LC-NMR, in the future.
We have applied NMR metabolic profiling as our tool for the quality control of Angelica roots. We detected many common amino acids and sugars, instead of the presumed active ingredients of Angelica. This could be due to the water extraction that we used here instead of methanol extraction used in previous studies. These active compounds might be too hydrophobic to be extracted efficiently by boiling water, resulting in a concentration lower than the detection limit of our 1H-NMR method (approximately 10 μM). Although these compounds were not detected, we still managed to distinguish the different sources of Angelica roots, based on their primary metabolites.
NMR profiling is a promising method for quality control of commercial herbal medicine products, with its quick analysis time (about 5 min per sample), and its simple sample preparation. It is also unbiased (all compounds with protons are profiled) and non-destructive. Although
1H-NMR spectra of mixtures may exhibit overlapping peaks, which poses challenges in peak identification and integration, this does not interfere with the reproducibility of
1H-NMR for the purpose of sample differentiation and quality control. In comparison, LC-MS metabolic profiling has a higher sensitivity than
1H-NMR (10
−9–10
−11 mol in comparison with 10
−19 mol), and which can detect lower-abundance metabolites [
16]. However, absolute quantification is impossible with LC-MS, unless pure standards are available, and the experimental and data analysis procedure of LC-MS is usually more time-consuming.