Crataegus pinnatifida: Chemical Constituents, Pharmacology, and Potential Applications
Abstract
:1. Introduction
2. Chemical Composition
| Classification | No. | Chemical component | Part of Plant | Reference |
|---|---|---|---|---|
| Flavonoids | 1 | Apigenin | Leaves | [6] |
| 2 | Luteolin | Leaves | [7] | |
| 3 | Orientin | Leaves | [8] | |
| 4 | Isoorientin | Leaves | [8] | |
| 5 | Vitexin | Flower | [9] | |
| 6 | Vitexin rhamnoside | Flower | [9] | |
| 7 | Isovitexin | Leaves | [10] | |
| 8 | Hyperoside | Leaves | [11] | |
| 9 | Pinnatifinoside A | Leaves | [12] | |
| 10 | Pinnatifinoside B | Leaves | [12] | |
| 11 | Pinnatifinoside C | Leaves | [12] | |
| 12 | Pinnatifinoside D | Leaves | [12] | |
| 13 | Pinnatifinoside I | Leaves | [12] | |
| 14 | 3′′′, 4′′′-di-O-Acetyl-2′′-O-α-rhamuosylvitexin | Leaves | [13] | |
| 15 | Schaftoside | Leaves | [14] | |
| 16 | Isoschaftoside | Leaves | [14] | |
| 17 | Neoschaftoside | Leaves | [14] | |
| 18 | Neoisoschaftoside | Leaves | [14] | |
| 19 | Cratenacin | Leaves | [15] | |
| 20 | Acetylvitexin | Flower | [16] | |
| 21 | Crataequinone B | Leaves | [17] | |
| 22 | Kaempferol | Leaves | [18] | |
| 23 | Quercetin | Leaves | [11] | |
| 24 | Bioquercetin | Leaves | [9] | |
| 25 | Herbacetin | Leaves | [19] | |
| 26 | Santin | Leaves | [19] | |
| 27 | 5-Hydroxyauranetin | Leaves | [19] | |
| 28 | Rutin | Leaves | [17] | |
| 29 | 8-Methoxykaempferol | Flower | [9] | |
| 30 | Pinnatifidin | Flower | [20] | |
| 31 | Kaempferol 3-neohesperidoside | Leaves,Fruit | [21] | |
| 32 | 8-Methoxykaempferol 3-neohesperidoside | Leaves,Fruit | [21] | |
| 33 | Naringenin-5,7-di-glucoside | Leaves | [22] | |
| 34 | Eriodictyol-5,3′-di-glucoside | Leaves | [22] | |
| 35 | (+)-Taxifolin | Leaves | [23] | |
| 36 | (+)-Taxifolin 3-O-arabinopyranoside 3-O-arabinopyranoside | Leaves | [23] | |
| 37 | (+)-Taxifolin 3-O-xylopyranoside | Leaves | [23] | |
| 38 | Crateside | Leaves | [24] | |
| 39 | (+)-Catechin | Leaves | [13] | |
| 40 | (−) E-picatechin | Leaves | [13] | |
| 41 | Leucocyanidin | Fruit | [25] | |
| 42 | Proanthocyanidin A2 | Leaves, Flower | [26] | |
| 43 | Procyanidin B2 | Leaves, Flower | [26] | |
| 44 | Procyanidin B4 | Leaves, Flower | [26] | |
| 45 | Procyanidin B5 | Leaves, Flower | [26] | |
| 46 | Procyanidin C1 | Leaves, Flower | [26] | |
| 47 | Procyanidin D1 | Leaves, Flower | [26] | |
| 48 | Epicatechin-(4β→6)-Epicatechin-(4β→8)- epicatechin | Leaves, Flower | [26] | |
| 49 | Epicatechin-(4β→8)- epicatechin-(4β→6)-epicatechin | Leaves, Flower | [26] | |
| 50 | Procyanidin E1 | Leaves, Flower | [26] | |
| Triterpenoids & Steroids | 51 | Ursolic acid | Fruit | [27] |
| 52 | 2α,3β,19α-trihydroxyl ursolic acid | Leaves | [28] | |
| 53 | Corosolic acid | Fruit | [29] | |
| 54 | Cuneataol | Fruit | [30] | |
| 55 | Cycloartenol | Stem, Leaves | [31] | |
| 56 | Uvaol | Fruit | [27] | |
| 57 | Oleanolic acid | Seeds | [32] | |
| 58 | Maslinic acid | Fruit | [29] | |
| 59 | Butyrospermol | Stem, Leaves | [31] | |
| 60 | 24-Methylene-24-dihydrolanosterol | Stem, Leaves | [31] | |
| 61 | Betulin | Fruit | [27] | |
| 62 | 18,19-seco,2α,3β-Dihydroxy-19-oxo-urs-11,13(18)-dien-28-oic acid | Leaves | [33] | |
| 63 | β-Sitosterol | Fruit | [34] | |
| 64 | β-Daucosterol | Fruit | [34] | |
| 65 | Stigmosterol | Fruit | [34] | |
| 66 | 24-Methylene-24-dihydrolanosterol | Stem, Leaves | [31] | |
| Monoterpenes & sesquiterpenes | 67 | 3,9-Dihydroxymegastigma-5-ene | Leaves | [33] |
| 68 | (3S,5R,6R,7E)-Megatsigmane-7-ene-3-hydroxy-5, 6-epoxy-9-O-β-d-glucopyranoside | Leaves | [33] | |
| 69 | (3R,5S,6S,7E,9S)-Megastigman-7-ene-3,5,6,9-tetrol 9-O-β-d-glucopyranoside | Leaves | [35] | |
| 70 | (6S,7E,9R)-6,9-Dihydroxy-4,7-megastigmadien-3-one 9-O-[β-d-xylopyranosyl-(1′′→6′)-β-d-glucopyranoside] | Leaves | [35] | |
| 71 | Linarionoside C | Leaves | [36] | |
| 72 | Linarionoside A | Leaves | [36] | |
| 73 | Linarionoside B | Leaves | [36] | |
| 74 | 3β-d-Glucopyranosyloxy-β-ionone | Leaves | [36] | |
| 75 | Icariside B6 | Leaves | [36] | |
| 76 | Pisumionoside | Leaves | [36] | |
| 77 | (3S,5R,6R,7E,9R)-3,6-Epoxy-7-megastigmen-5,9-diol-9-O-β-d-glucopyranoside | Leaves | [36] | |
| 78 | (6S,7E,9R)-Roseoside | Leaves | [36] | |
| 79 | (6R,9R)-3-Oxo-α-ionol-9-O-β-d-glucopyranoside | Leaves | [36] | |
| 80 | 4-[4β-O-β-d-Xylopyranosyl-(1′′→6′)-β-d-glucopyranosyl-2,6,6-trimethyl-1-cyclohexen-1-yl]-butan-2-one | Leaves | [35] | |
| 81 | (3S,9R)-3,9-Dihydroxy-megastigman-5-ene 3-O-primeveroside | Leaves | [35] | |
| 82 | (3R,5S,6S,7E,9S)-Megastiman-7-ene-3,5,6,9-tetrol | Leaves | [35] | |
| 83 | 1β,9α-Dihydroxyeudesm-3-en-5β,6α,7α,11α H-12,6-olide | Fruit | [37] | |
| 84 | (5Z)-6-[5-(2-Hydroxypropan-2-yl)-2-methyltetrahydrofuran-2-yl] -3-methylhexa-1,5-dien-3-ol | Leaves | [35] | |
| 85 | (5Z)-6-[5-(2-O-β-d-Glucopyranosyl-propan-2-yl)-2-methyl tetrahydrofur-an-2-yl]-3-methylhexa-1,5-dien-3-ol | Leaves | [35] | |
| 86 | 5-Ethenyl-2-[2-O-β-d-glucopyranosyl-(1′′→6′)-β-d-glucopyranosyl-propan-2-yl]-5-methyltetrahydrofuran-2-ol | Leaves | [35] | |
| 87 | Gibberellic acid | Fruit | [38] | |
| Lignans | 88 | (2,3-Dihydro-2-(4-O-β-d-glueopyranosyl-3-methoxy-Phenyl)-3-hydroxymethyl-5-(3-hydroxypropyl)-7-methoxybenzofuran) | Leaves | [39] |
| 89 | Shanyenoside A | Leaves | [40] | |
| 90 | (7S,8R)-Urolignoside | Leaves | [36] | |
| 91 | (−)-2a-O-(β-d-Glucopyranosyl)- lyoniresinol | Leaves | [36] | |
| 92 | Tortoside A | Leaves | [36] | |
| 93 | Verbascoside | Leaves | [36] | |
| 94 | Acernikol-4′′-O-β- d-glucopyranoside | Leaves | [36] | |
| 95 | erythro-1-(4-O-β-d-Glucopyranosyl-3-methoxyphenyl)-2-[4-(3-hydroxypropyl)-2,6-dimethoxyphenoxy]-1,3-propanediol | Leaves | [36] | |
| 96 | (7S, 8R)-5-Methoxydihydrodehydrodiconiferyl alcohol 4-O-β- d-glucopyranoside | Leaves | [36] | |
| 97 | Pinnatifidanin C I | Seeds | [41] | |
| 98 | Pinnatifidanin C II | Seeds | [41] | |
| 99 | Pinnatifidanin C III | Seeds | [41] | |
| 100 | Pinnatifidanin C IV | Seeds | [41] | |
| 101 | Pinnatifidanin C V | Seeds | [41] | |
| 102 | Pinnatifidanin C VI | Seeds | [40] | |
| 103 | Pinnatifidanin C VII | Seeds | [41] | |
| 104 | Pinnatifidanin C VIII | Seeds | [41] | |
| 105 | Pinnatifidanin B I | Seeds | [42] | |
| 106 | Pinnatifidanin B II | Seeds | [42] | |
| 107 | Pinnatifidanin B III | Seeds | [42] | |
| 108 | Pinnatifidanin B IV | Seeds | [42] | |
| 109 | Pinnatifidanin B V | Seeds | [42] | |
| 110 | Pinnatifidanin B VI | Seeds | [42] | |
| 111 | Pinnatifidanin B VII | Seeds | [42] | |
| 112 | Pinnatifidanin B VIII | Seeds | [42] | |
| 113 | Pinnatifidanin B IX | Seeds | [42] | |
| Hydroxycinnamic acids | 114 | Chlorogenic acid | Leaves | [19] |
| 115 | β-Coumaric acid | Fruit | [38] | |
| 116 | Caffeic acid | Fruit | [38] | |
| 117 | Ferulic acid | Fruit | [38 | |
| Organic acids | 118 | Benzoic acid | Leaves | [33] |
| 119 | (p-Hydroxyphenyl) benzoic acid | Seed | [43] | |
| 120 | Gallic acid | Seed | [43] | |
| 121 | Protocatechuic acid | Seed | [43] | |
| 122 | Anisic acid | Fruit | [38] | |
| 123 | Vanillic acid | Fruit | [38] | |
| 124 | Syringic acid | Fruit | [38] | |
| 125 | Gentisic acid | Fruit | [38] | |
| 126 | Malic acid | Fruit | [44] | |
| 127 | Citric acid | Fruit | [44] | |
| 128 | Quinic acid | Fruit | [44] | |
| 129 | Pyruvic acid | Fruit | [44] | |
| 130 | Tartaric acid | Fruit | [44] | |
| 131 | Succinic acid | Fruit | [34] | |
| 132 | Fumaric acid | Seed | [32] | |
| 133 | Ascorbic acid | Shoot | [45] | |
| 134 | 2-(4-Hydroxy-2-benzyl) malic acid | Seed | [32] | |
| 135 | Palmitic acid | Fruit | [46] | |
| 136 | Stearic acid | Fruit | [46] | |
| 137 | Oleic acid | Fruit | [46] | |
| 138 | Linoleic acid | Fruit | [46] | |
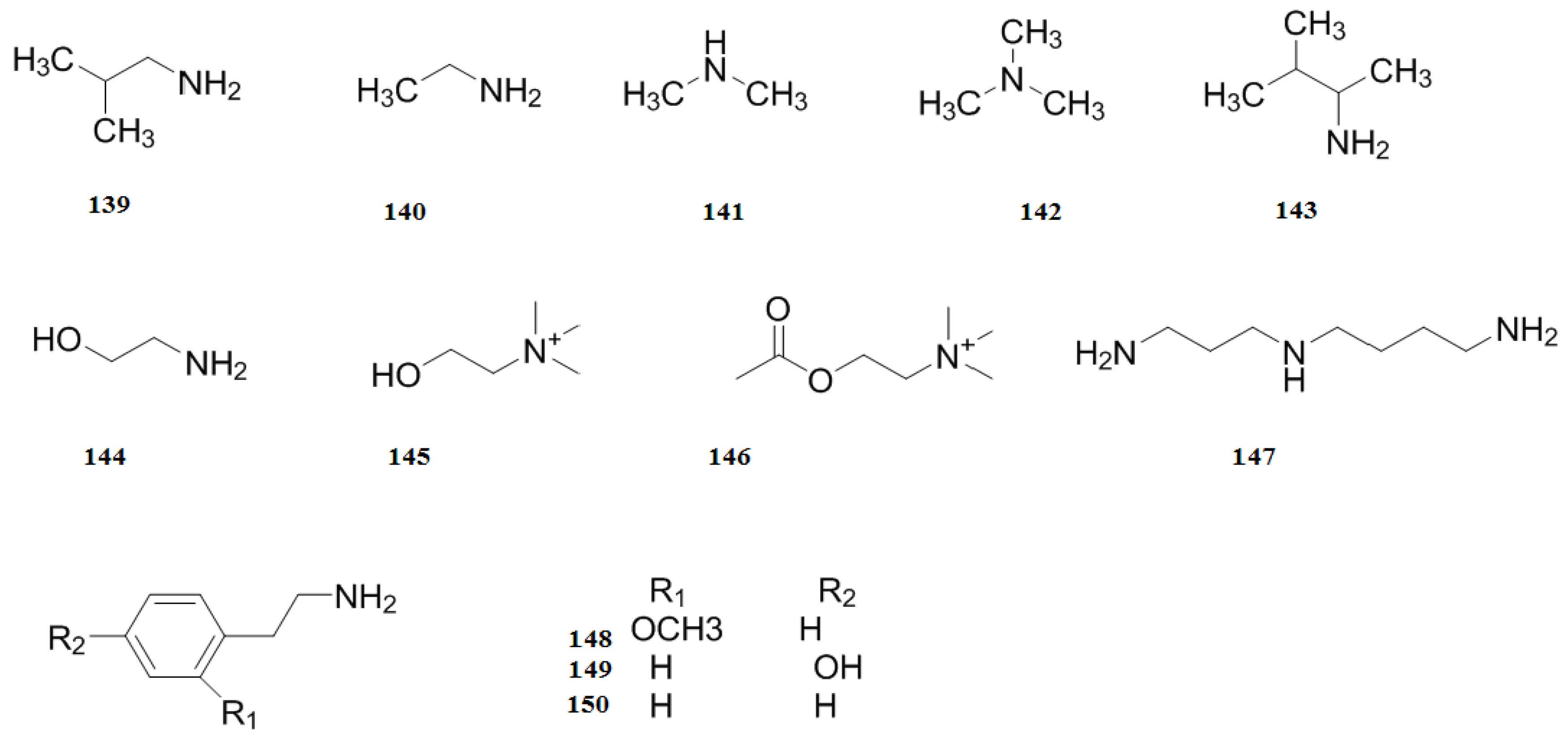
| Nitrogenous compounds | 139 | Isobutylamine | Leaves | [47] |
| 140 | Ethylamine | Leaves | [47] | |
| 141 | Dimethylamine | Leaves | [47] | |
| 142 | Trimethylamine | Leaves | [47] | |
| 143 | Isoamyl amine | Leaves | [47] | |
| 144 | Ethanolamine | Leaves | [47] | |
| 145 | Choline | Leaves | [47] | |
| 146 | Acetylcholine | Leaves | [47] | |
| 147 | Spermindine | Leaves | [47] | |
| 148 | O-Methoxyphenethylamine | Leaves | [47] | |
| 149 | Tyramine | Leaves | [47] | |
| 150 | Phenylethylamine | Leaves | [47] | |
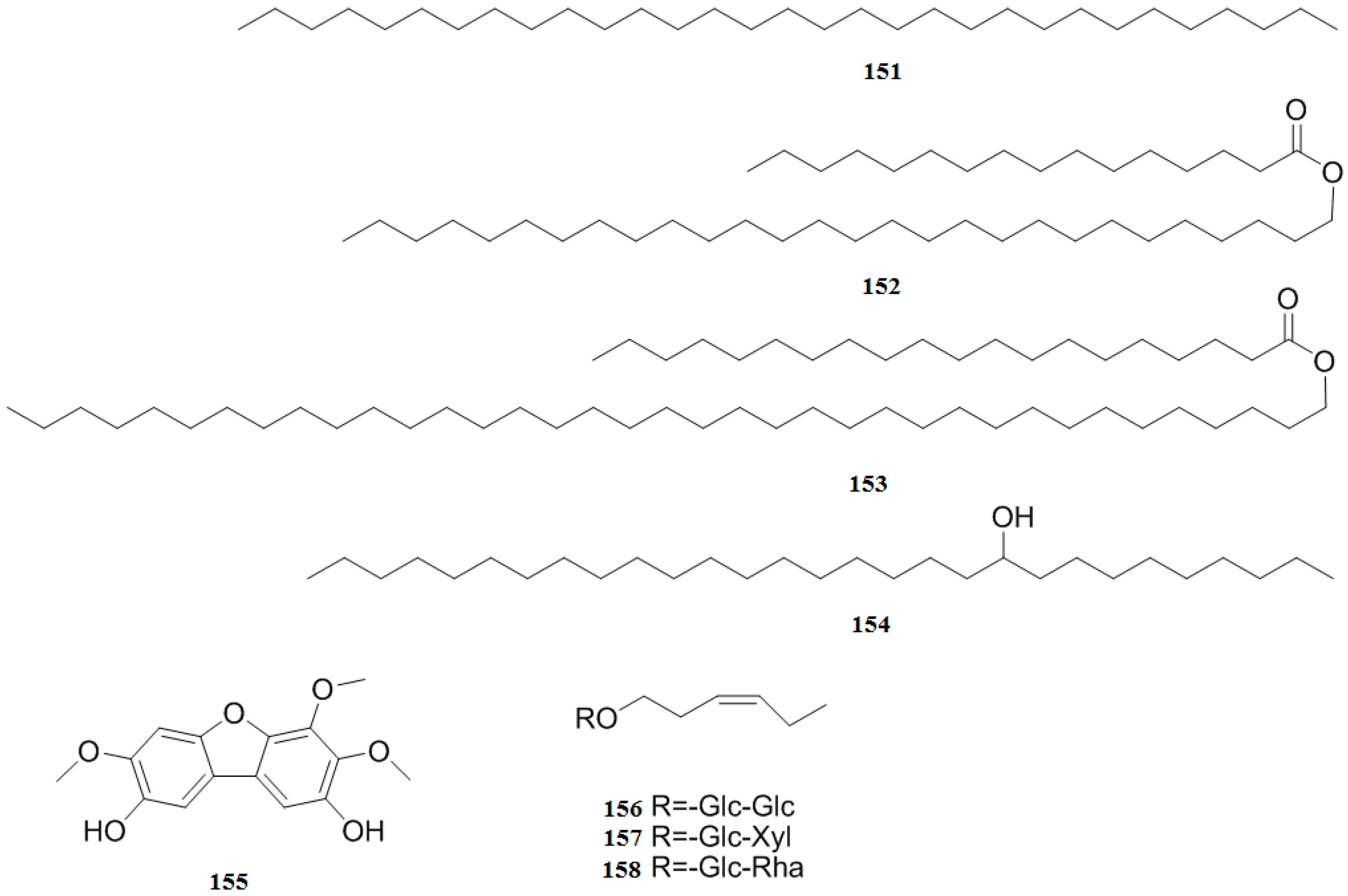
| Other compounds | 151 | Hentriacontane | Fruit | [27] |
| 152 | Hexadecanoic acid, octaconsyl ester | Fruit | [27] | |
| 153 | Eicosanoic acid, octatriacontyl ester | Fruit | [27] | |
| 154 | Nonacosan-10-ol | Fruit | [27] | |
| 155 | 2,8-Dihydroxy-3,4,7-trimethoxydibenzofuran | Bark, sapwood | [48] | |
| 156 | (Z)-3-hexenyl-O-β-d-glucopyranosyl-(1′′→6′)-β-d-glucopyranoside | Leaves | [35] | |
| 157 | (Z)-3-Hexenyl-O-β-d-xylopyranosyl-(1′′→6′)-β-d-glucopyranoside | Leaves | [35] | |
| 158 | (Z)-3-Hexenyl-O-β-d-rhamnopyranosyl-(1′′→6′)-β-d-glucopyranoside | Leaves | [35] |
2.1. Flavonoids
2.1.1. Flavones
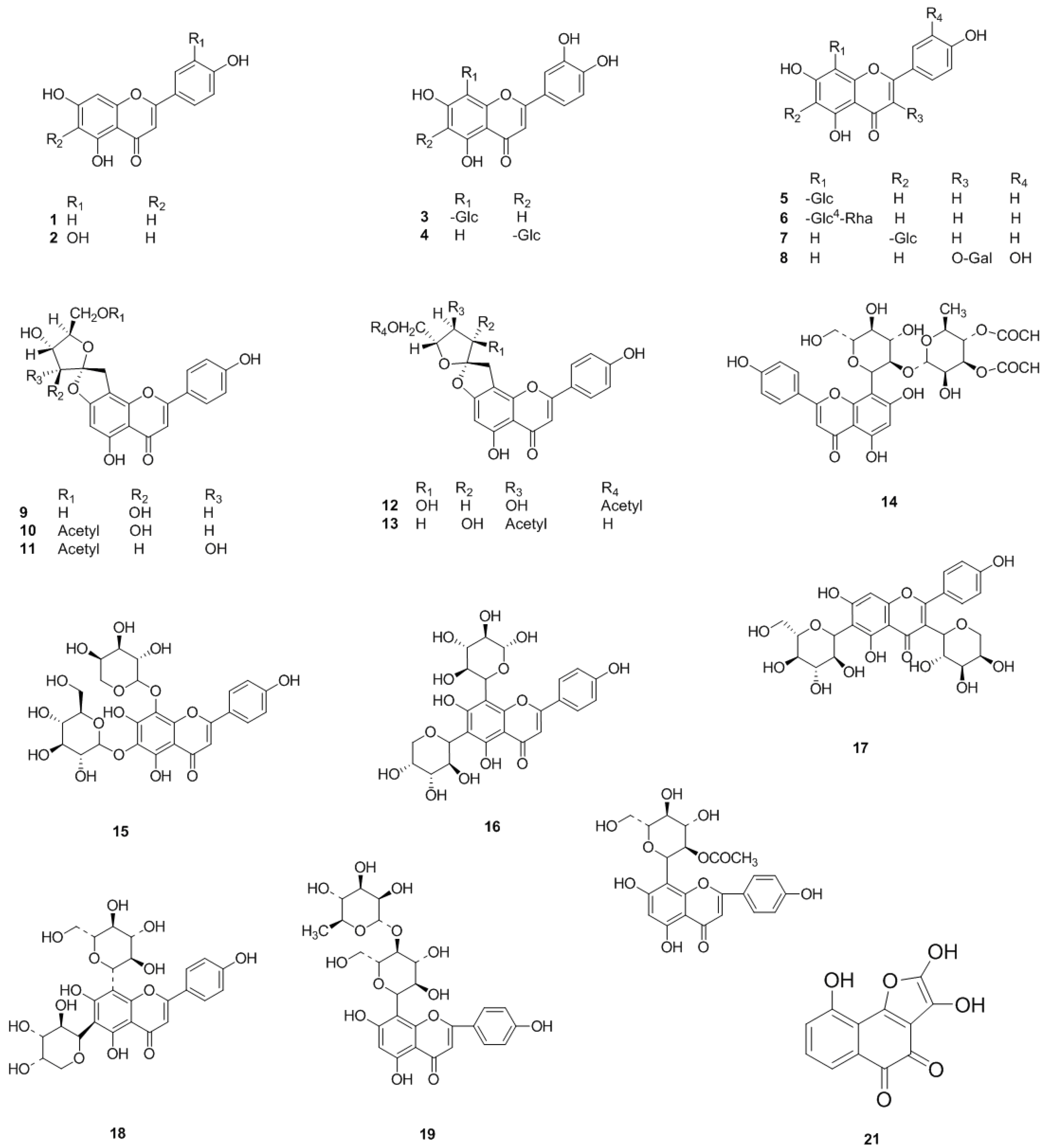
2.1.2. Flavonols

2.1.3. Flavanones and Flavanonols
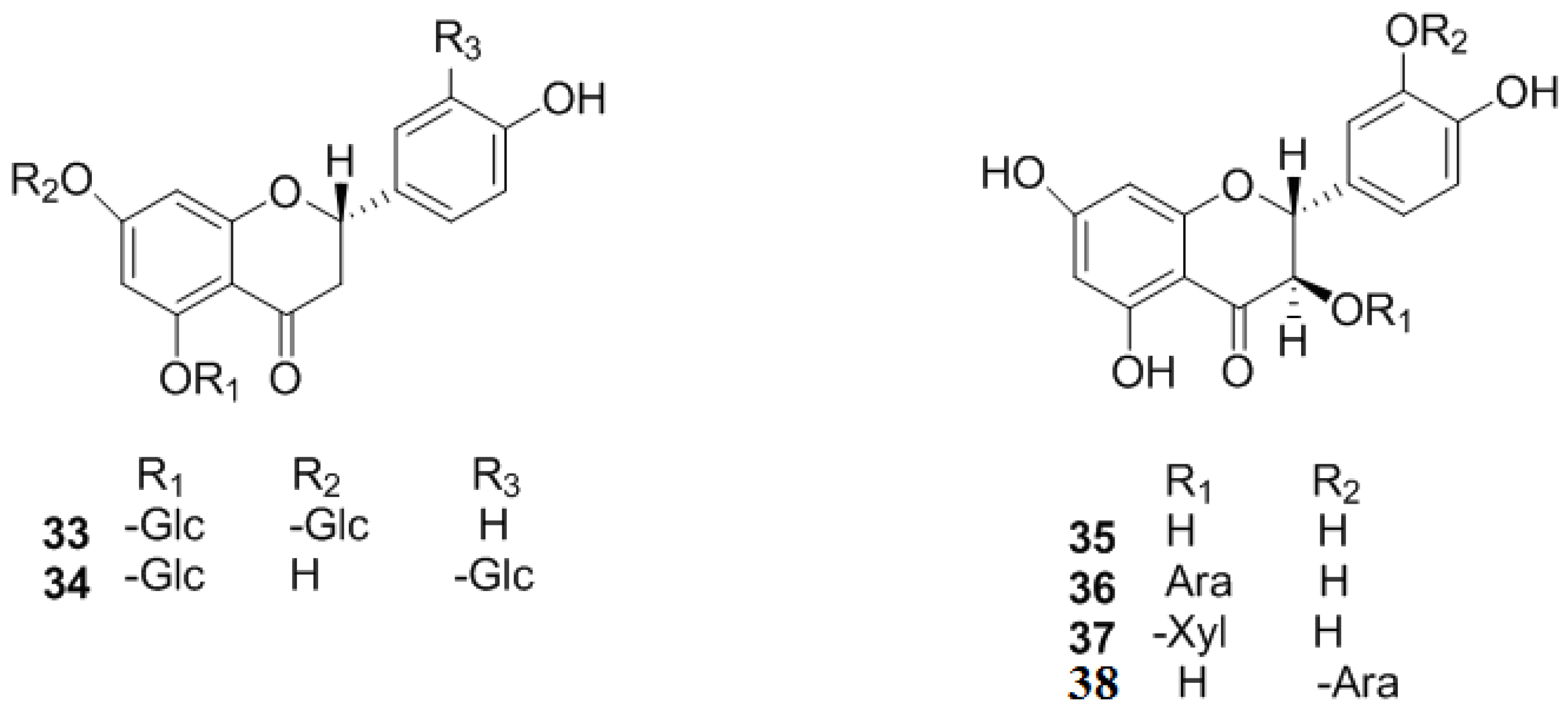
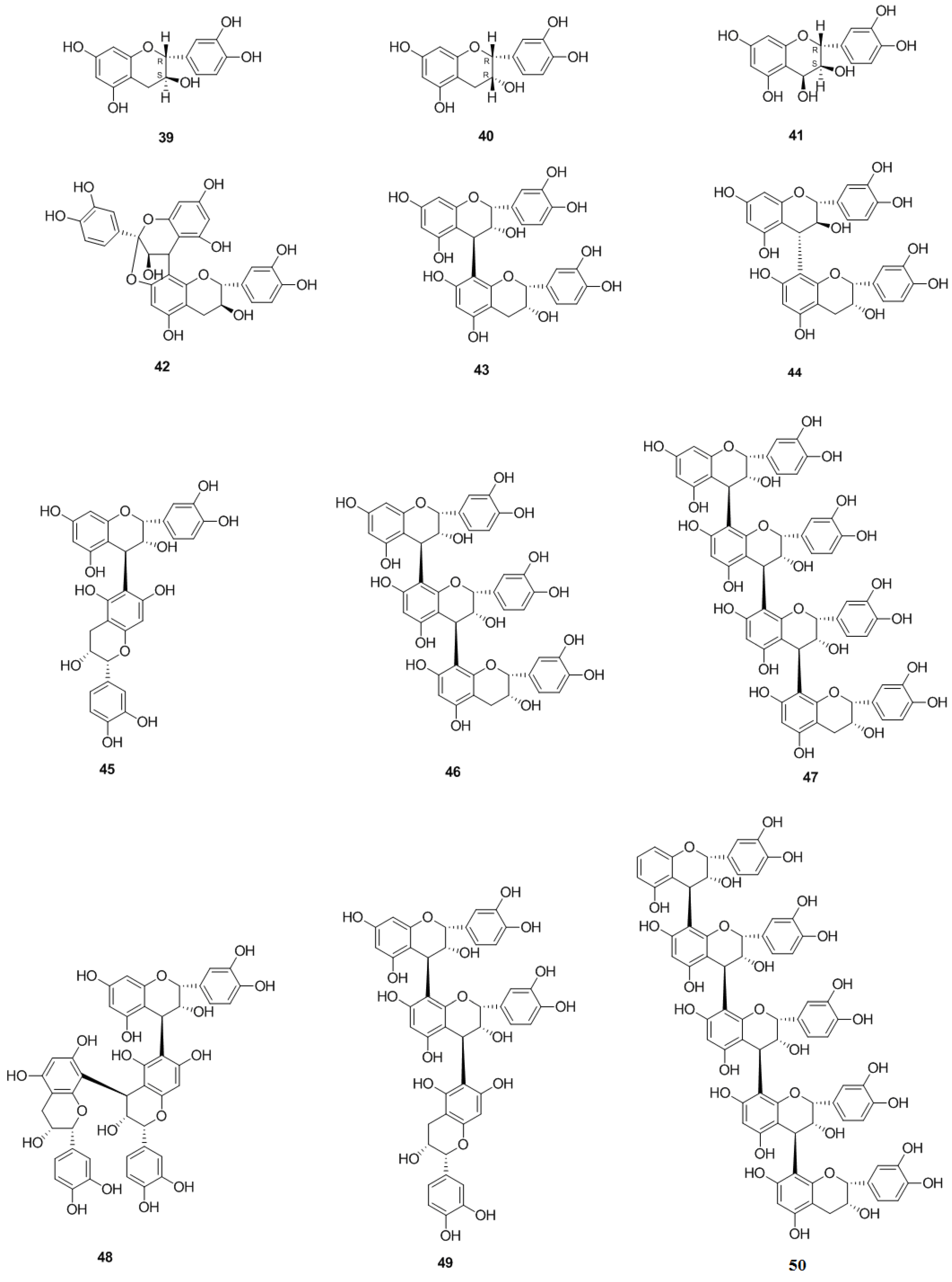
2.1.4. Flavanols and the Polymers of Flavanols
2.2. Triterpenoids and Steroids
2.2.1. Triterpenoids
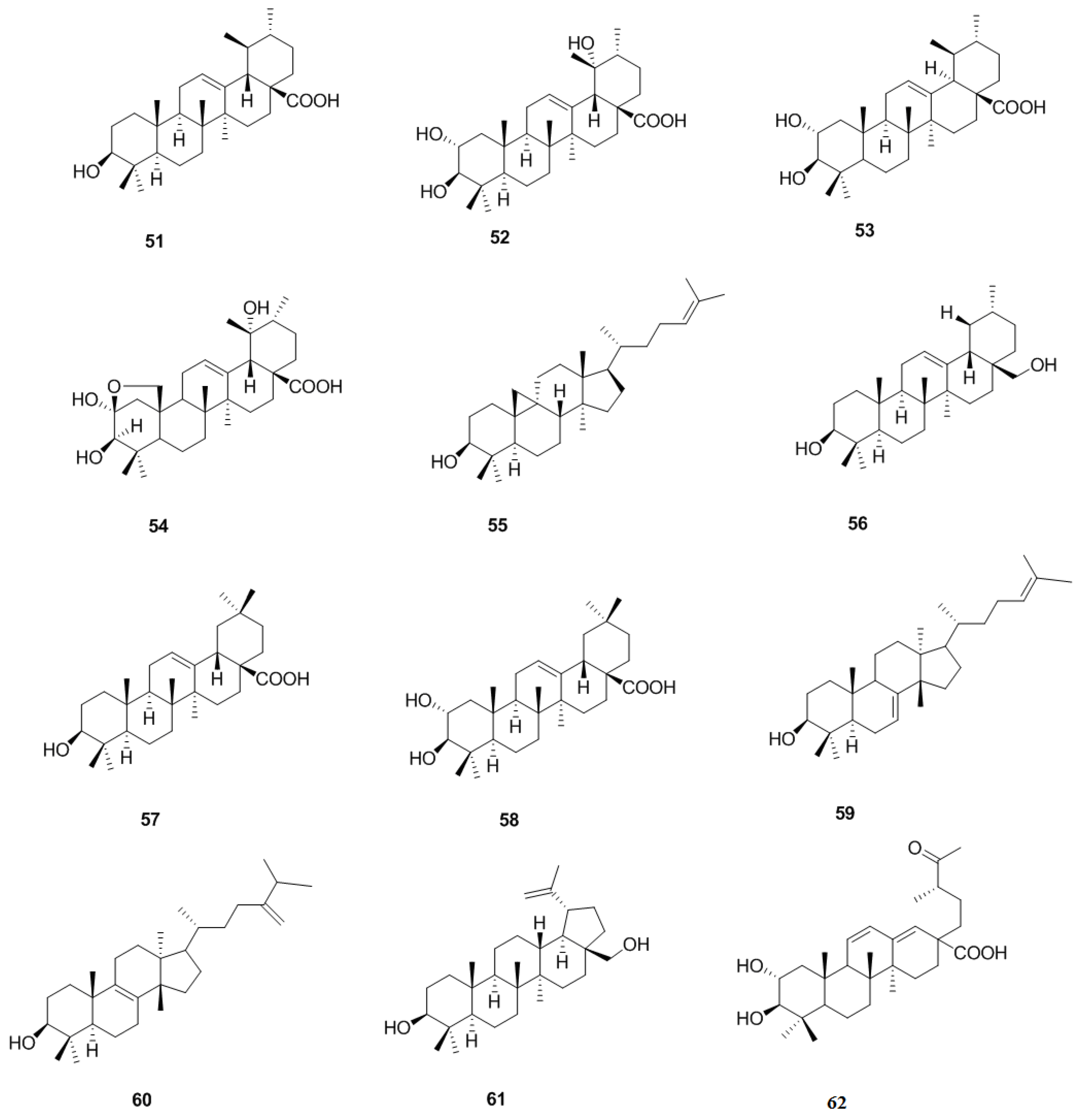
2.2.2. Steroids
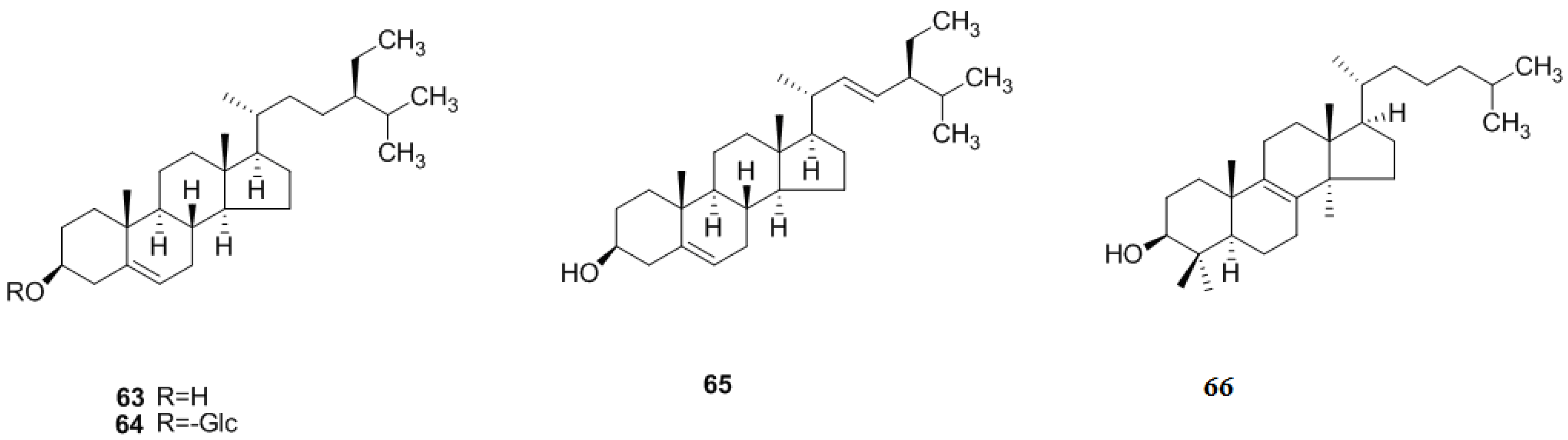
2.3. Monoterpenoids and Sesquiterpenoids

2.4. Lignans
2.5. Hydroxycinnamic Acids
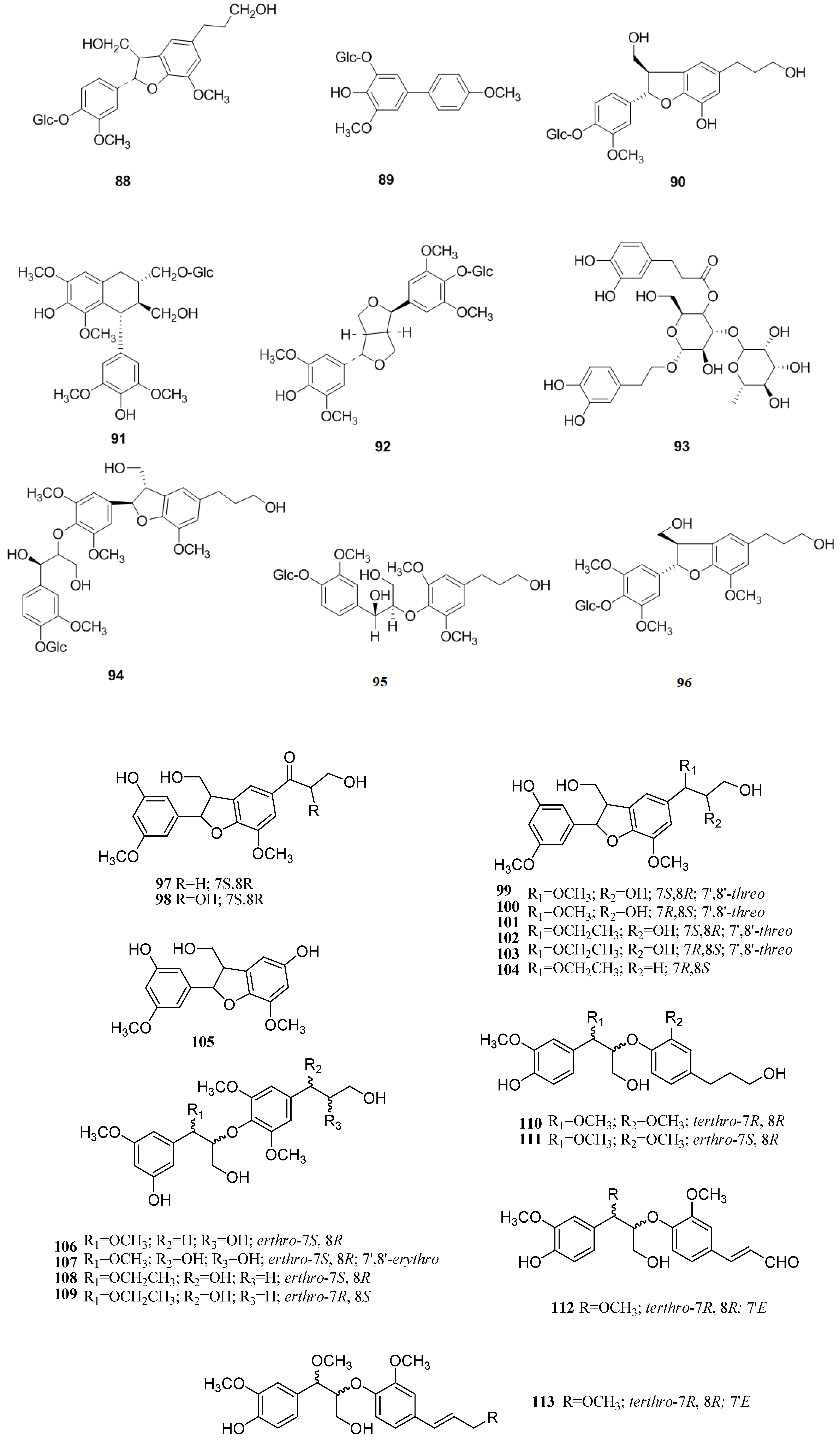
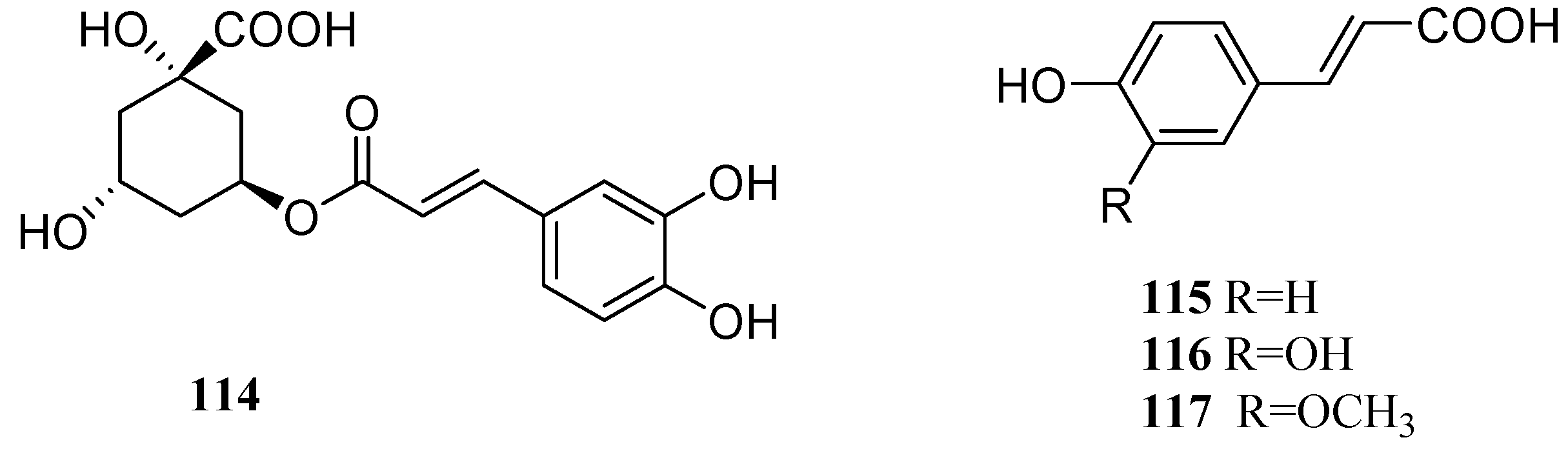
2.6. Organic Acids
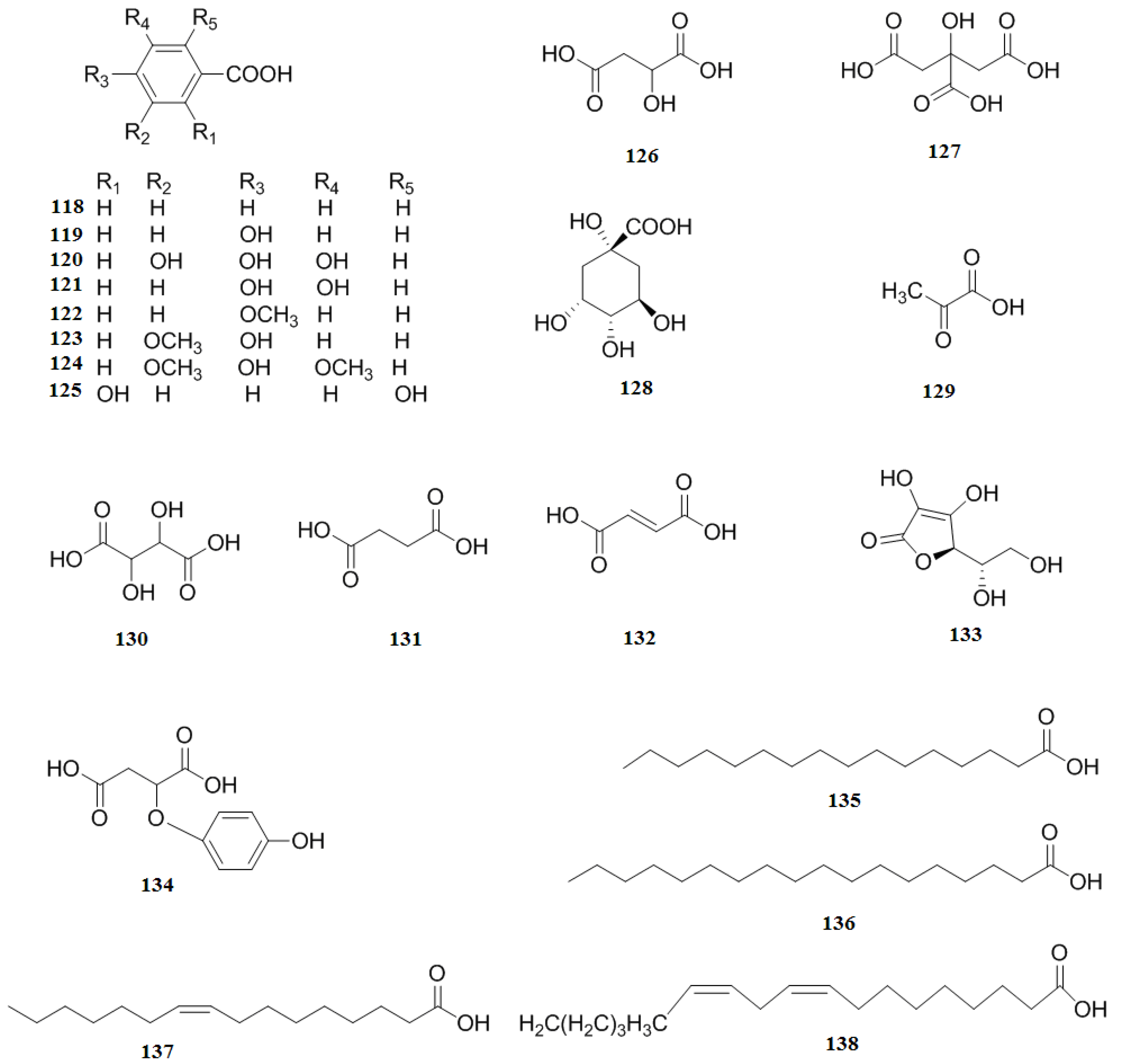
2.7. Nitrogen-containing Compounds
2.8. Others
3. Biological Properties
3.1. Cardiovascular System Effects
3.1.1. Lipid Regulating and Anti-atherosclerosis Effects
3.1.2. Resistance to Chronic Heart Failure
3.1.3. Antihypertensive Effects
3.1.4. Anti-myocardial Ischemia and Reperfusion Injury Effect
3.2. Digestive System Effects
3.2.1. Gastrointestinal Function Regulating Effect
3.2.2. Digestive Enzyme Promotion Effects
3.3. Effects on Pathogenic Microorganisms
3.3.1. Antibacterial Effects
3.3.2. Synergistic Antibacterial Effects
3.4. Effects on Tumors and the Immune System
3.4.1. Anticancer and Sperm Distortion Inhibiting Effects
3.4.2. Immunoregulating Effects
3.5. Endocrine System Effects
3.6. Coagulation System Effects
3.7. Other Effects
3.7.1. Antiinflammatory Effects
3.7.2. Antioxidant Effects
3.7.3. Osteoporosis Inhibiting Effects
3.7.4. Retina Protecting Effects
4. Toxicology
5. Future Perspectives and Conclusions
Conflicts of Interest
References
- Gao, G.Y.; Feng, Y.X. The literatures of traditional Chinese medicine hawthorn. Zhongguo Zhong Yao Za Zhi 1994, 5, 259–260. [Google Scholar]
- Wang, C.L.; Lu, B.Z.; Hou, G.L. Chemical constituent, pharmacological effects and clinical application of Crataegus pinnatifida. Strait Pharm. J. 2010, 3, 75–78. [Google Scholar]
- Wu, S.J.; Li, Q.J.; Xiao, X.F.; Li, M.; Yang, X.R.; Lv, T. The research of chemical constituent and pharmacological effects of Crataegus pinnatifida. Drug Eval. Res. 2010, 4, 316–319. [Google Scholar]
- Editorial Committee of Dictionary of Chinese Materia Medica. Dictionary of Chinese Materia Medica, 2001, ed.; People’s Medical Publishing House: Beijing, China, 2001. [Google Scholar]
- Yang, B.S.; Li, Y.L.; Niu, L.Y.; Feng, H.G.; Li, W.L. Experimental study on genotoxicity of oral corn pollen haw liquor. J. Xinxiang Med. Coll. 1998, 1, 6–8. [Google Scholar]
- Melikoglu, G.; Mericli, F.; Mericli, A.H. Flavonoids of Crataegus orientalis. Boll. Chim. Farm. 1999, 7, 351–352. [Google Scholar]
- Titova, A.A.; Batyuk, V.S. C-glycosides of crataegus curvisepala. Chem. Nat. Comp. 1986, 22, 348. [Google Scholar] [CrossRef]
- Nikolov, N.; Batyuk, V.S.; Kovalev, I.P.; Ivanov, V. Glycoflavonoids of crataegus monogyna and C. pentagyna. Chem. Nat. Compd. 1973, 9, 110. [Google Scholar]
- Bykov, V.I.; Glyzin, V.I.; Ban’kovskii, A.I. Flavonoids of the genus crataeugs. The structure of bioquercetin. Chem. Nat. Compd. 1972, 8, 657. [Google Scholar]
- Nikolov, N. New flavone c-biosides from Crataegus monogyna and Cr. Pentagyna. Chem. Nat. Compd. 1975, 11, 434–435. [Google Scholar] [CrossRef]
- Batyuk, V.S.; Prokopenko, A.P.; Kolesnikov, D.G. New flavonoids from the leaves of Crataegus curvisepala lindm. Chem. Nat. Compd. 1965, 1, 225–226. [Google Scholar]
- Zhang, P.C.; Xu, S.X. Flavonoid ketohexosefuranosides from the leaves of Crataegus pinnatifida Bge.var.major N.E.Br. Phytochemistry 2001, 57, 1249–1253. [Google Scholar] [CrossRef]
- El-Mousallamy, A.M.D. Chemical investigation of the constitutive flavonoid glycosides of the leaves of Crataegus sinaica. Nat. Prod. Sci. 1998, 4, 53–57. [Google Scholar]
- Nikolov, N.; Dellamonica, G.; Chopin, J. Di-c-glycosyl flavones from Crataegus monogyna. Phytochemistry 1981, 20, 2780–2781. [Google Scholar] [CrossRef]
- Batyuk, V.S.; Chernobrovaya, N.V.; Prokopenko, A.P. Cratenacin-A new flavone glycoside from Crataegus curvisepala. Chem. Nat. Compd. 1966, 2, 90–93. [Google Scholar]
- Kashnikova, M.V. Flavonoids of the flowers of Crataegus sanguinea. Chem. Nat. Compd. 1984, 20, 105–106. [Google Scholar] [CrossRef]
- Sözer, U.; Dönmez, A.A.; Mericli, A.H. Constituents from the leaves of Crataegus davisii Browicz. Sci. Pharm. 2006, 74, 203–208. [Google Scholar] [CrossRef]
- Zhang, P.C.; Xu, S.X. Chamical constituents from the leaves of Rataegus pinnatifida Bge. Var. Major N.E.Br. Yao Xue Xue Bao 2001, 10, 754–757. [Google Scholar]
- Mericli, A.H.; Ergezen, K. Flavonoids of Crataegus tanacetifolia (Rosaceae), an endemic species from Turkey. Sci. Pharm. 1994, 3, 277–281. [Google Scholar]
- Bykov, V.I.; Glyzin, V.I.; Ban’kovakii, A.I. Pinnatifidin-a new flavonol glycoside from Crataegus pinnatifida. Chem. Nat. Compd. 1972, 8, 699–701. [Google Scholar]
- Zhao, Y.P.; Wang, C.X.; Du, L.X. Study on chemical constituents in fruits and leaves of Crataegus L. Beverage Ind. 2002, 6, 8–12. [Google Scholar]
- Kowalewski, Z.; Mrugasiewicz, K. New falvanone glycosides in Crataegus phaenopyeum. Planta Med. 1971, 19, 311–317. [Google Scholar] [CrossRef]
- Shahat, A.A.; Ismail, S.I.; Hammouda, F.M.; Azzam, A.A.; Lemiere, G.; de-Bruyne, T.; de-Swaef, S.; Pieters, L.; Vlietinck, A. Anti-HIV activity of flavonoids and proanthocyanidins from Crataegus sinaica. Phytomedicine 1998, 2, 133–136. [Google Scholar]
- Nikolov, N.; Batyuk, V.S.; Ivanov, V. Crateside—A new flavonol glycoside from Crataegus monogyna and C. pentagyna. Chem Nat Comp 1973, 9, 150–151. [Google Scholar] [CrossRef]
- Petrova, V.P. Catechins and leuconathocyanins of Crataegus fruit. Ukr. Bot. Zh. 1972, 2, 144–147. [Google Scholar]
- Svedström, U.; Vuorela, H.; Kostiainen, R.; Tuominen, J.; Kokkonen, J.; Rauha, J.P.; Laakso, I.; Hiltunen, R. Isolation and identification of oligomeric procyanidins from Crataegus leaves and flowers. Phytochemistry 2002, 60, 821–825. [Google Scholar] [CrossRef]
- Shi, Y.P.; Ding, X.B. Studies on chemical constituents from fruit of Crataegus pinnatifida. Chin. Tradit. Herbal Drugs 2000, 3, 173–175. [Google Scholar]
- Song, S.J.; Chen, J.; Kou, X.; Song, Y.H.; Xu, S.X. Chemical constituents from leaves of Crataegus pinnatifida Bge (I). J. Shenyang Pharm. Univ. 2006, 23, 88–90. [Google Scholar]
- Chen, L.S.; Lv, L.; Xu, S.W.; Xin, Y. Study on the Triterpene Acids in Fruit of Crataegus pinnatifida. Lishizhen Med. Mater. Med. Res. 2008, 19, 2909–2910. [Google Scholar]
- Ikeda, T.; Ogawa, Y.; Nohara, T. A new triterpenoid from Crataegus cuneata. Chem. Pharm. Bull. 1999, 10, 1487–1488. [Google Scholar] [CrossRef]
- García, M.D.; Sáenz, M.T.; Ahumada, M.C. A.Cert.Isolation of three triterpenes and several aliphatic alcohols from Crataegus monogyna Jacq. J. Chromatogr. A 1997, 767, 340–342. [Google Scholar] [CrossRef]
- Sun, X.F.; Yao, Q.Y. Chemical constituents from seed of Crataegus pinnatifida Bge. Chin. Tradit. Herbal Drugs 1987, 18, 441–454. [Google Scholar]
- Huang, X.X.; Niu, C.; Gao, P.Y.; Li, L.Z.; Ming, M.; Song, S.J. Chemical constituents from the leaves of the Crataegus pinnatifida Bge (III). J. Shenyang Pharm. Univ. 2010, 27, 615–618. [Google Scholar]
- Zhang, P.C.; Xu, S.X.; Guo, H. Study on chemical component in the fruit of Crataegus pinnatifida. J. Shenyang Inst. Chem. Technol. 1999, 16, 87–89. [Google Scholar]
- Song, S.J.; Li, L.Z.; Gao, P.Y.; Peng, Y.; Yang, J.Y.; Wu, C.F. Terpenoids and hexenes from the leaves of Crataegus pinnatifida. Food Chem. 2011, 129, 933–939. [Google Scholar] [CrossRef]
- Gao, P.Y.; Li, L.Z.; Peng, Y.; Li, F.F.; Niu, C.; Huang, X.X.; Ming, M.; Song, S.J. Monoterpene and lignin glycosides in the leaves of Crataegus pinnatifida. Biochem. Syst. Ecol. 2010, 38, 988–992. [Google Scholar] [CrossRef]
- Ahmed, A.A.; Khattab, A.M.; Grace, M.H.; Sahl, M.M. A new eudesmanolide from Crataegus flava fruits. Fitoterapia 2001, 72, 759. [Google Scholar]
- Schrall, R.; Becker, H. Production of catechins and oligomeric proanthocyanidins in callus and suspension cultures of Crataegus monogyna, C. oxyacantha and Ginkgo biloba. Planta Med. 1977, 4, 297–318. [Google Scholar] [CrossRef]
- Hao, D.F.; Yang, R.P.; Zhou, Y.Z.; Chen, H.; Li, Z.F.; Pei, Y.H. Chemical constituents from the leaves of the Crataegus pinnatifida Bge (II). J. Shenyang Pharm. Univ. 2009, 26, 282–284. [Google Scholar]
- Chen, J.; Song, S.J.; He, J.; Xu, S.X. A new biphenyl glucoside from the leaves of Crataegus pinnatifida. J. Shenyang Pharma. Univ. 2006, 23, 430–431. [Google Scholar]
- Huang, X.X.; Zhou, C.C.; Li, L.Z.; Peng, Y.; Lou, L.L.; Liu, S.; Li, D.M.; Ikejima, T.; Song, S.J. Cytotoxic and antioxidant dihydrobenzofuran neolignans from the seeds of Crataegus pinnatifida. Fitoterapia 2013, 91, 217–223. [Google Scholar] [CrossRef]
- Huang, X.X.; Zhou, C.C.; Li, L.Z.; Li, F.F.; Lou, L.L.; Li, D.M.; Ikejima, T.; Peng, Y.; Song, S.J. The cytotoxicity of 8-O-4′ neolignans from the seeds of Crataegus pinnatifida. Bioorg. Med. Chem. Lett. 2013, 23, 5599–5604. [Google Scholar]
- Wang, X.S.; Che, Q.M.; Li, Y.M.; He, Y.Q. A study on chemical constituents in seeds of Crataegus pinnatifida Bge.var. Major N. E. Br. Zhongguo Zhong Yao Za Zhi 1999, 17, 739–740. [Google Scholar]
- Chapman, G.W., Jr.; Horvat, R.J.; Payne, J.A. The nonvolatile acid and suger composition of mayhaw fruit. Food Qual. 1991, 14, 435–439. [Google Scholar] [CrossRef]
- Roddewig, C.; Hensel, H. Reaction of local myocardial blood flow in non-anesthetized dogs and anesthetized cats to the oral and parenteral administration of a Crateagus fraction (Oligomere procyanidines). Arzneim.-Forsch. 1977, 27, 1407–1410. [Google Scholar]
- Evdokimova, O.V.; Samylina, I.A.; Nesterova, O.V. Examination of a lipophilic Crataegus fruit fraction. Farmatsiya 1992, 41, 60–61. [Google Scholar]
- Hobbs, S.; Foster, S. Hawthom: a literature review. Herbal Gram. 1990, 22, 19–33. [Google Scholar]
- Kokubun, T.; Harborne, J.B.; Eagles, J.; Waterman, P.G. Dibenzofuran phytoalexins from the sapwood tissue of Photinia, Pyracantha and Crataegus species. Phytochemistry 1995, 5, 1033–1037. [Google Scholar]
- Jennifer, E.; Edwards, J.E.; Brown, P.N.; Talent, N.; Dickinson, T.N.; Shipley, P.R. A review of the chemistry of the genus Crataegus. Phytochemistry 2012, 79, 5–26. [Google Scholar] [CrossRef]
- Liu, Q.L.; Yang, Z.L. Comparison of total flavonoids of different purities from Folium crataegi in effect of antihyperlipidemia. Strait Pharm. J. 2008, 20, 32–52. [Google Scholar]
- Liu, X.Y.; Zhou, L.; Liang, R.Y. Study on lipid regulation mechanism of total flavonoids from Folium crataegi by 3T3-L1 cells. Chin. Arch. Tradit. Chin. Med. 2009, 27, 1066–1068. [Google Scholar]
- Xie, W.H.; Sun, C.; Liu, S.M. Effect of hawthorn flavanone on blood-fat and expression of lipogenesis and lipolysis genes of hyperlipoidemia model mouse. Zhongguo Zhong Yao Za Zhi 2009, 34, 224–229. [Google Scholar]
- Yang, R.M.; Chen, H.M.; Gao, N.N.; Song, X.; Li, J.L.; Cai, D.Y. Mechanism of early atherosclerosis in guinea pig: abnormal metabolism of LDL-C. Acta Lab. Anim. Sci. Sin. 2011, 23, 237–241. [Google Scholar]
- Liu, J.; Sun, H.B.; Duan, W.G.; Mu, D.Y.; Zhang, L.Y. Maslinic acid reduces blood glucose in KK-Ay mice. Biol. Pharm. Bull. 2007, 30, 2075–2078. [Google Scholar] [CrossRef]
- Li, G.H.; Sun, J.Y.; Zhang, X.L. Experimental studies on antihyperlipidemia effects of two compositions from hawthorn in mice. Chin. Tradit. Herbal Drugs 2002, 33, 50–52. [Google Scholar]
- Yang, Y.J.; Lin, J.; Wang, C.M. Effect of FHL on intervening early hyperlipoidemia model rat. Chin. Tradit. Herbal Drugs 2008, 39, 1848–1850. [Google Scholar]
- Ye, X.Y.; Zhang, L.; Shen, J. Effect of hawthorn leaf flavonoids on metabolism of glucose and lipids in diabetic mice. Chin. Tradit. Herbal Drugs 2005, 36, 1683–1686. [Google Scholar]
- Yang, Y.J.; Wang, C.M.; Dang, X.W. Protection of flavonoids in hawthorn leaf against vascular disfunction of hyperlipoidemic rats. Chin. Tradit. Herbal Drugs 2007, 38, 1687–1690. [Google Scholar]
- Degenring, F.H.; Suter, A.; Weber, M.; Saller, R. A randomised double blind placebo controlled clinical trial of a standardised extract of fresh Crataegus berries (Crataegisan®) in the treatment of patients with congestive heart failure NYHA II. Phytomedicine 2003, 11, 363–369. [Google Scholar] [CrossRef]
- Schmidt, U.; Kuhn, U.; Ploch, M.; Hübner, W.-D. Efficacy of the hawthorn (Crataegus) preparation LI 132 in 78 patients with chronic congestive heart failure defined as NYHA functional class II. Phytomedicine 1994, 1, 17–24. [Google Scholar] [CrossRef]
- Zapfe, G. Clinical efficacy of Crataegus extract WS® 1442 in congestive heart failure NYHA class II. Phytomedicine 2001, 49, 262–266. [Google Scholar] [CrossRef]
- Tauchert, M. Efficacy and safety of Crataegus extract WS 1442 in comparison with placebo in patients with chronic stable New York Heart Association class-III heart failure. Am. Heart J. 2002, 155, 910–915. [Google Scholar] [CrossRef]
- Ju, X.Y.; Fang, T.H.; Zhang, W.T. Effect of freezedrying powder of total flavonoids in chinese hawthorn leaf on myocardial infarction caused by coronary ligation of anesthetic dogs. J. Nanjing Univ. Tradit. Chin. Med. 2005, 19, 381–383. [Google Scholar]
- Editorial Committee of Modern Clinical Chinese Materia Medica. Modern Clinical Chinese Materia Medica, 1998 ed.; China Press of Traditional Chinese Medicine: Beijing, China, 1998. [Google Scholar]
- Yang, X.P. Medicinal value of Crataegus pinnatifida. Jilin Med. J. 1998, 19, 41. [Google Scholar]
- Yuan, Y.; Zhao, J.; Gao, H.J.; Wang, J.H. Experimental study on effect of hawthorn on compounding hypertension and hyperlipoidemia rats. J. Xinjiang Med. Univ. 2013, 35, 52–57. [Google Scholar]
- Yu, B.; Li, H.Y.; Zhang, L. Protective effect of total flavonoids from hawthorn leaf on ligation-induced acute myocardial ischemia in anesthetized dogs. Tradit. Chin. Drug Res. Clin. Pharmacol. 2008, 19, 461–464. [Google Scholar]
- Huang, S.S.; Lin, Y.; Diao, Y.P. Effects of charred fructus Crataegi alcohol extract on contractility of isolated rat gastric and intestine muscle strips. Prog. Mod. Biomed. 2009, 9, 612–614. [Google Scholar]
- Deng, S.; Lin, Y.; Diao, Y.P. Effects of hawthorn alcohol extract on the contractility of isolated rat gastric and intestine muscle strips. Prog. Mod. Biomed. 2009, 9, 1262–1264. [Google Scholar]
- Wen, X.P.; Deng, S.; Lin, Y.; Diao, Y.P.; Huang, S.S.; Zhang, H.L. Effects of Folium crataegi water extract on the contractility of isolated gastric and intestinal muscle strips in rats. China Pharm. 2010, 21, 978–980. [Google Scholar]
- Yang, S.X.; Wang, X.C.; Wang, Z.X.; Han, J.; Liu, D.L.; Zhang, Y. Discussion of Haw foods in the reduction of azithromycin’s side-effect. Chin. Med. Herald 2010, 7, 175–176. [Google Scholar]
- Editorial Committee of Pharmacology of Chinese Materia Medica. Pharmacology of Chinese Materia Medica, 2000 ed.; People’s Medical Publishing House: Beijing, China, 2000. [Google Scholar]
- Wu, J.H.; Sun, J.Y. Effect of Crataegus pinnatifida organic acids on the gastrointestinal motility. Shanxi J. Tradit. Chin. Med. 2009, 25, 1402–1403. [Google Scholar]
- Lin, L.; Chen, Y.J.; Li, L.; Cao, Y.; Sun, Q.X. Experimental observation on germicidal efficacy of hawthorn liquid and its influencing factors. Chin. J. Disinfect. 2000, 17, 85–88. [Google Scholar]
- Li, C.Q.; Wu, W.; Tong, Y. Study on germicidal efficacy of extract of hawthorn fruit pit and its influencing factors. Chin. J. Disinfect. 2007, 24, 50–52. [Google Scholar]
- Jiang, K.; Qin, R.X.; Zhu, T.F.; Zhou, H.; Zheng, J. Antibacterial activity of active component of Crataegus pinnatifida combined with β-lactam antibiotics against methicillin-resistant Staphylococcus aureus in vitro. Acta Acad. Med. Milit. Tert. 2009, 19, 1887–1889. [Google Scholar]
- Qin, R.X.; Xiao, K.K.; Li, B.; Jiang, W.W.; Peng, W.; Zheng, J.; Zhou, H. The combination of Catechin and Epicatechin Gallate from Fructus crataegi potentiates β-Lactam antibiotics against Methicillin-resistant Staphylococcus aureus (MRSA) in vitro and in vivo. Int. J. Mol. Sci. 2013, 14, 1802–1821. [Google Scholar] [CrossRef]
- Wang, B.; Qiu, W.W.; Yu, Y.Y.; Yang, F.; Tang, J. Progress of study on maslinic acid. Chin. Bull. Life Sci. 2009, 21, 264–269. [Google Scholar]
- Xu, H.X.; Zeng, F.Q.; Wan, M.; Sim, K.Y. Anti-HIV triterpene acids from Geum japonicum. J. Nat. Prod. 1996, 59, 643–645. [Google Scholar] [CrossRef]
- Numata, A.; Yang, P.; Takahashi, C.; Fujiki, R.; Nabae, M.; Fujita, E. Cytotoxic triterpenes from a Chinese medicine, goreishi. Chem. Pharm. Bul1. 1989, 37, 648–651. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, H.W.; Sun, J.P. Extraction-Separation of total flavone from the fruit of Crataegus pinnatifida and anticancer activities in vitro. Chin. Tradit. Herbal Drugs 2004, 35, 787–789. [Google Scholar]
- Cui, T.C.; Liu, X.Q.; Xu, H.Q.; Wu, G.J.; Zhang, Z.B. Effect of the extracts of C. pinnatifida on inhibiting sperm distortion of mice induced by cyclophosphamide. Chin. Public Health 2002, 3, 266–267. [Google Scholar]
- Chang, J.; Jin, Z.P.; Gao, G. Effect of decoctum of Crataegus pinnatifida Bge on the cellular immune function of mice. J. Baotou Med. Coll. 1996, 12, 10. [Google Scholar]
- Jin, Z.C.; Bai, L.H. Effect of hawthorn injection on the immunity of rabbit. Chin. Tradit. Herbal Drugs 1992, 23, 592–593. [Google Scholar]
- Dong, H.; Zhang, T.P.; Peng, S.M.; Li, J.; Zhang, H.Y. Extraction of sitosterol from hawthorn fruits and effect of sitosterol on immunological function and serum lipid. Nat. Prod. Res. Dev. 2009, 21, 60–63. [Google Scholar]
- Yan, Q.G. Effect of hawthorn polysaccharides on immunity of mice. J. China Tradit. Chin. Med. Inf. 2009, 29, 134–135. [Google Scholar]
- Oikonomakos, N.G. Glycogen phosphorylase as a moleculartarget for type II diabetes therapy. Curr. Protein Pept. Sci. 2002, 3, 561–586. [Google Scholar] [CrossRef]
- Somsák, L.; Nagya, V.; Hadady, Z.; Docsa, T.; Gergely, P. Glucose analog inhibitors of glycogen phosphorylases as potential antidiabetic agents: Recent developments. Curr. Pharm. Des. 2003, 15, 1177–1189. [Google Scholar]
- Wen, X.A.; Zhang, P.; Liu, J.; Zhang, L.Y.; Wu, X.M.; Ni, P.Z.; Sun, H.B. Pentacyclic triterpenes. Part 2: Synthesis and biological evaluation of maslinic acid derivatives as glycogen phosphorylase inhibitors. Bioorg. Med. Chem. Lett. 2006, 16, 722–726. [Google Scholar] [CrossRef]
- Liu, B.L.; Dong, J.S.; Ni, X.H. In vivo study on flavonoids extraction from hawthorn fruits and effects of hawthorn leaves flavonoids. Food Sci. 2007, 28, 324–327. [Google Scholar]
- Yang, L.P.; Wang, C.L.; Wang, Y.L.; Li, Y.S.; Fu, S.X. The effects of extracts from the leaves of Crataegus pinnatifida on platelet aggregation of rabbits and myocardial ischemia of rats. Chin. Tradit. Herbal Drugs 1993, 24, 482–483. [Google Scholar]
- Shi, J.; Wang, Z.X.; Lu, X.H. Synergetic Antiplatelet effects of Fructus crataegi and Rhizoma alismatis. Chin. Tradit. Herbal Drugs 1996, 27, 350–352. [Google Scholar]
- Jiang, K.Y.; Gu, Z.L.; Ruan, C.G. Effect of WH505 on experimental thrombosis and its mechanism. Chin. Tradit. Patent Med. 2000, 20, 203–205. [Google Scholar]
- Jiao, L.P. Effect of flavone mixture of crataegus leaves on hemorheology index of acute blood stasis rat model. Chin. Remedies Clin. 2008, 8, 49–50. [Google Scholar]
- Yang, Y.J.; Dong, X.Q.; Guo, J.J. Experimental research of blood-quickening stasis-transforming actions of hawthorn leaf flavonoids. Hebei Med. 2009, 31, 22–23. [Google Scholar]
- Shatoor, A.S.; Soliman, H.; Al-Hashem, F.; El-Gamal, B.; Othman, A.; El-Menshawy, N. Effect of hawthorn (Crataegus aronia syn. Azarolus (l)) on platelet function in albino wistar rats. Thrombosis Res. 2012, 130, 75–80. [Google Scholar] [CrossRef]
- Quan, Y.C.; Guan, L.P. Anti-inflammation, analgesic effects of extract of hawthorn leaf. Lishizhen Med. Mater. Med. Res. 2006, 17, 556–557. [Google Scholar]
- Wang, Y.; Sun, G.H.; Zhang, R.F.; Zhang, M.; Wang, L.J. Anti-inflammation, analgesic effects of extracts of leaves of Crataegus pinnatifida. Acta Chin. Med. Pharmacol. 2012, 40, 38–39. [Google Scholar]
- Ji, Y.S.; Li, H.; Yang, S.J. Protective effect and its molecular mechanism of FMCL on PC 12 cells apoptosis induced by H2O2. Chin. Pharmacol. Bull. 2006, 22, 760–762. [Google Scholar]
- Li, L.; Lv, H.; Pang, H. Anti-aging effect of total flavone of hawthorn leaf. Lishizhen Med. Mater. Med. Res. 2007, 9, 2143–2144. [Google Scholar]
- Chen, Z.Y.; Yan, M.X.; He, B.H. The Change and the immpact of IFHL on oxidative stress in the formation of NASH in rats. J. Med. Res. 2007, 36, 33–36. [Google Scholar]
- Chen, X.; Cheng, X.W.; Xu, M.R. Protective effect of total flavones of hawthorn leaf on renal ischemia/reperfusion injury in rats. J. Appl. Clin. Pediatr. 2007, 22, 359–360. [Google Scholar]
- Li, C.H.; Yang, Z.F.; Li, Z.X.; Ma, Y.; Zhang, L.P.; Zheng, C.B.; Qiu, W.W.; Wu, X.; Wang, X.; Li, H.; et al. Maslinic acid suppresses osteoclastogenesis and prevents ovariectomy-induced bone loss by regulating RANKL-mediated NF-κB and MAPK signaling pathways. J. Bone Mine. Res. 2011, 26, 644–656. [Google Scholar]
- Tian, Q.F.; Guo, X.R.; Yin, W.J.; Yin, P.Z.; Dong, Y.L.; Hu, Z.M.; Li, J.X. Histological study of haw drink compound in protecting against experimental carbon disulfide toxic damage of relina. Chin. J. Ocular Fundus Dis. 1999, 15, 249–252. [Google Scholar]
- Wei, J.Z.; Feng, L.; Bai, J.P.; Zhang, H.F.; Yang, L.Z.; Ma, C.G. The toxicology experiments of “semen cassiae hawthorn oat capsules”. J. Shanxi Datong Univ. (Nat. Sci. Ed.) 2011, 25, 41–44. [Google Scholar]
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wu, J.; Peng, W.; Qin, R.; Zhou, H. Crataegus pinnatifida: Chemical Constituents, Pharmacology, and Potential Applications. Molecules 2014, 19, 1685-1712. https://doi.org/10.3390/molecules19021685
Wu J, Peng W, Qin R, Zhou H. Crataegus pinnatifida: Chemical Constituents, Pharmacology, and Potential Applications. Molecules. 2014; 19(2):1685-1712. https://doi.org/10.3390/molecules19021685
Chicago/Turabian StyleWu, Jiaqi, Wei Peng, Rongxin Qin, and Hong Zhou. 2014. "Crataegus pinnatifida: Chemical Constituents, Pharmacology, and Potential Applications" Molecules 19, no. 2: 1685-1712. https://doi.org/10.3390/molecules19021685
APA StyleWu, J., Peng, W., Qin, R., & Zhou, H. (2014). Crataegus pinnatifida: Chemical Constituents, Pharmacology, and Potential Applications. Molecules, 19(2), 1685-1712. https://doi.org/10.3390/molecules19021685




