A New Dicoumarinyl Ether from the Roots of Stellera chamaejasme L
Abstract
:1. Introduction
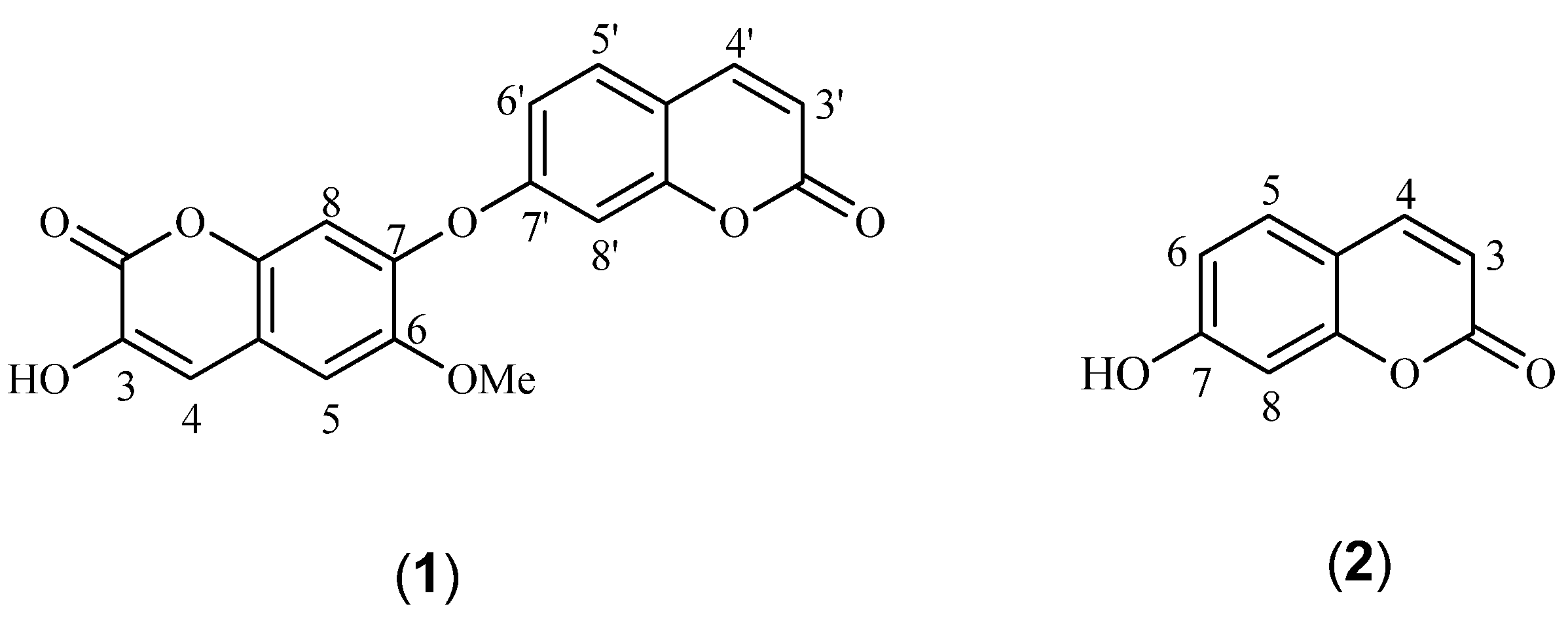
2. Results and Discussion

| NO. | δH Mult (J = Hz) | δC | HMBC | NO. | δH Mult (J = Hz) | δC | HMBC |
|---|---|---|---|---|---|---|---|
| 2 | 157.8 s | 3' | 6.31 d (9.6) | 115.0 d | 2', 10' | ||
| 3 | 137.5 s | 4' | 7.96 d (9.6) | 144.4 d | 2', 5', 9' | ||
| 4 | 7.80 s | 131.5 d | 2, 3, 5, 9 | 5' | 7.68 d (8.6) | 130.6 d | 4', 7', 9' |
| 5 | 7.24 s | 109.9 d | 4, 6, 7, 9 | 6' | 7.10 dd (8.6, 2.0) | 114.3 d | 8', 10' |
| 6 | 146.6 s | 7' | 161.1 s | ||||
| 7 | 151.2 s | 8' | 7.06 d (2.0) | 104.9 d | 6', 7' | ||
| 8 | 6.90 s | 103.7 d | 6, 7, 9, 10 | 9' | 156.5 s | 10' | |
| 9 | 149.0 s | 10' | 115.5 s | ||||
| 10 | 111.6 s | 6-OCH3 | 56.7 q | ||||
| 2' | 160.7 s | 3.92 s | 6 |
3. Experimental
3.1. General
3.2. Plant Material
3.3. Extraction and Isolation
4. Conclusions
Supplementary Materials
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Xu, Z.H.; Qin, G.W.; Li, X.Y.; Xu, R.S. New biflavanones and bioactive compounds from Stellera chamaejasme L. Acta Pharm. Sin. 2001, 36, 668–671. [Google Scholar]
- Zhang, C.; Zhou, S.S.; Feng, L.Y.; Zhang, D.Y.; Lin, N.M.; Zhang, L.H.; Pan, J.P.; Wang, J.B.; Li, J. In vitro anti-cancer activity of chamaejasmenin B and neochamaejasmin C isolated from the root of Stellera chamaejasme L. Acta Pharmacol. Sin. 2013, 34, 262–270. [Google Scholar] [CrossRef]
- Niwa, M.; Takamizawa, H.; Tatematsu, H.; Hirata, Y. Piscicidal constituents of Stellera. chamaejasme L. Chem. Pharm. Bull. 1982, 30, 4518–4520. [Google Scholar] [CrossRef]
- Niwa, M.; Tatematsu, S.; Liu, G.Q.; Hirata, Y. Isolation and structures of two new C-3/C-3'-biflavanones, neochamaejasmin A and neochamaejasmin B. Chem. Lett. 1984, 539–542. [Google Scholar]
- Niwa, M.; Liu, G.Q.; Tatematsu, S.; Hirata, Y. Chamaechromone, a novel rearranged biflavonoid from Stellera. chamaejasme L. Tetrahedron Lett. 1984, 25, 3735–3738. [Google Scholar] [CrossRef]
- Tatematsu, H.; Kurokawa, M.; Niwa, M.; Hirata, Y. Piscicidal constituents of Stellera. chamaejasme L. II. Chem. Pharm. Bull. 1984, 32, 1612–1613. [Google Scholar] [CrossRef]
- Liu, G.Q.; Tatematsu, H.; Kurokawa, M.; Niwa, M.; Hirata, Y. Novel C-3/C-3''-biflavanones from stellera chamaejasme L. Chem. Pharm. Bull. 1984, 32, 362–365. [Google Scholar] [CrossRef]
- Niwa, M.; Otsuji, S.; Tatematsu, S.; Liu, G.Q.; Chen, S.F.; Hirata, Y. Stereostructures of two biflavanones from Stellera. chamaejasme L. Chem. Pharm. Bull. 1986, 34, 3249–3251. [Google Scholar] [CrossRef]
- Jiang, Z.H.; Tanaka, T.; Sakamoto, T.; Kouno, I.; Duan, J.A.; Zhou, R.H. Biflavanones, diterpenes, and coumarins from the roots of Stellera. chamaejasme L. Chem. Pharm. Bull. 2002, 50, 137–139. [Google Scholar] [CrossRef]
- Yang, G.H.; Liao, Z.X.; Xu, Z.Y.; Zhang, H.P.; Chen, D.F. Antimitotic and antifungal C-3/C-3'-biflavanones from Stellera. chamaejasme. Chem. Pharm. Bull. 2005, 53, 776–779. [Google Scholar] [CrossRef]
- Asada, Y.; Sukemori, A.; Watanabe, T.; Malla, K.J.; Yoshikawa, T.; Li, W.; Kolike, K.; Chen, C.H.; Akiyama, T.; Qian, K.; et al. Stelleralides A-C, novel potent anti-HIV daphnane-type diterpenoids from Stellera. chamaejasme L. Org. Lett. 2011, 13, 2904–2907. [Google Scholar] [CrossRef]
- Li, J.; Zhao, W.; Hu, J.L.; Cao, X.; Yang, J.; Li, X.R. A new C-3/C-3''-biflavanone from the roots of Stellera. chamaejasme L. Molecules 2011, 16, 6465–6469. [Google Scholar]
- Feng, B.M.; Pei, Y.H.; Han, B. Research on chemical constituents of Stellera. chamaejasme L. J. Shenyang Pharm. Univ. 2000, 17, 258–259. [Google Scholar]
- Sample Availability: Available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, J.; Shen, Q.; Bao, C.-H.; Chen, L.-T.; Li, X.-R. A New Dicoumarinyl Ether from the Roots of Stellera chamaejasme L. Molecules 2014, 19, 1603-1607. https://doi.org/10.3390/molecules19021603
Li J, Shen Q, Bao C-H, Chen L-T, Li X-R. A New Dicoumarinyl Ether from the Roots of Stellera chamaejasme L. Molecules. 2014; 19(2):1603-1607. https://doi.org/10.3390/molecules19021603
Chicago/Turabian StyleLi, Jie, Qian Shen, Chen-Hao Bao, Li-Ting Chen, and Xiang-Rong Li. 2014. "A New Dicoumarinyl Ether from the Roots of Stellera chamaejasme L" Molecules 19, no. 2: 1603-1607. https://doi.org/10.3390/molecules19021603
APA StyleLi, J., Shen, Q., Bao, C.-H., Chen, L.-T., & Li, X.-R. (2014). A New Dicoumarinyl Ether from the Roots of Stellera chamaejasme L. Molecules, 19(2), 1603-1607. https://doi.org/10.3390/molecules19021603




