3.3. General Method for Palladium-Catalyzed Macrocyclizations
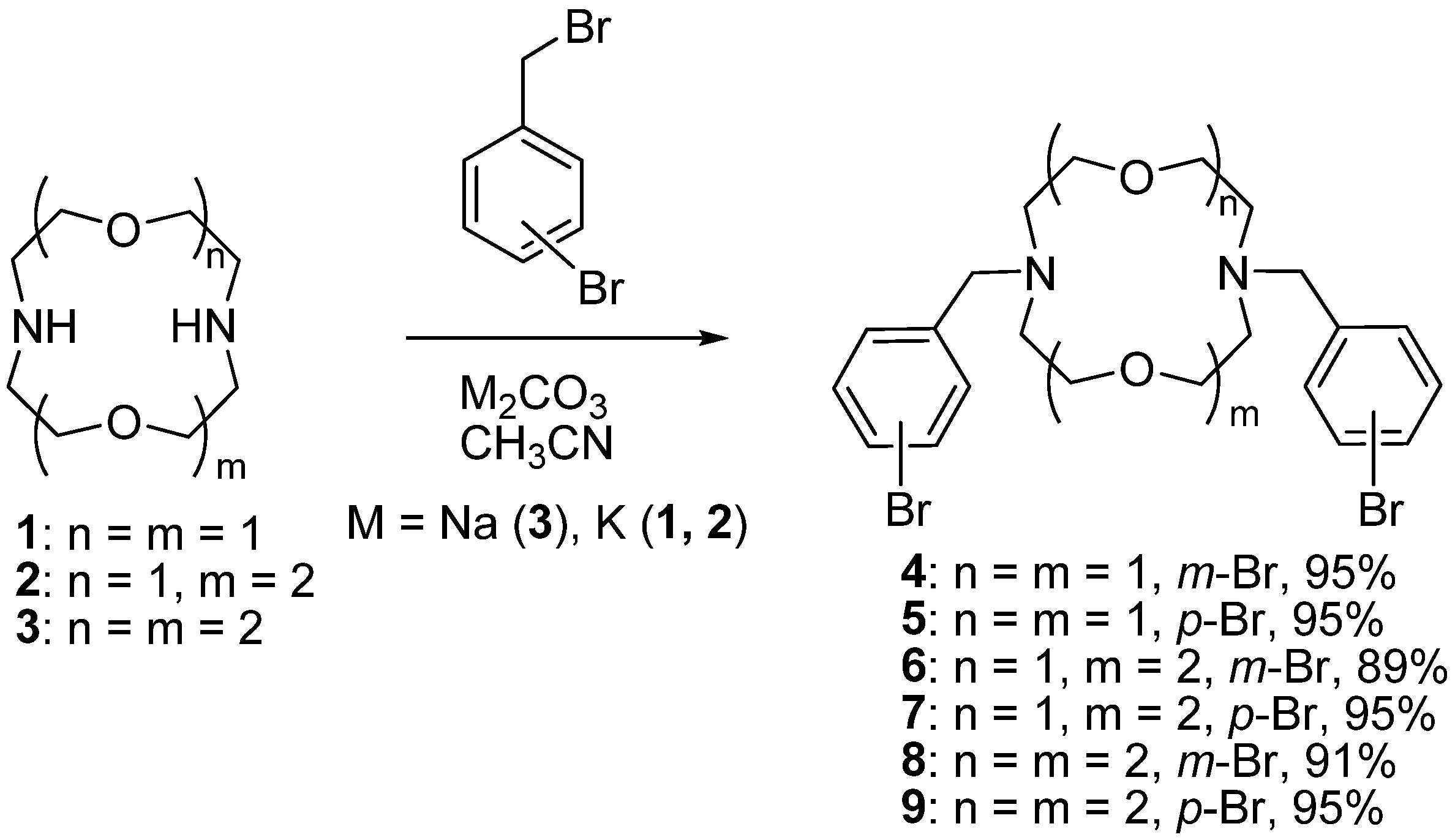
A two-neck flask equipped with a magnetic stirrer and reflux condenser, flushed with dry argon, was charged with diazacrown derivative 4–9 (0.2–0.25 mmol), Pd(dba)2 (8–16 mol%), BINAP or DavePhos ligand (9–18 mol%), absolute dioxane (10–12 mL), the reaction mixture was stirred for several minutes, then the corresponding polyamine (0.2–0.25 mmol) and NaOt-Bu (0.6–0.75 mmol) were added, and the reaction mixture was stirred at reflux for 24 h. After cooling down to room temperature the residue was filtered off, washed with CH2Cl2 (5–10 mL), the combined organic fractions were evaporated in vacuo, the residue was dissolved in CH2Cl2 (10 mL), washed with distilled water (3 × 10 mL), dried over 4Å molecular sieves, and the solvent was evaporated in vacuo. The solid residue was chromatographed on silica gel (40–60 μm) using a sequence of eluents: CH2Cl2, CH2Cl2–MeOH 100:1–3:1, CH2Cl2–MeOH–NH3(aq) 100:20:1–10:4:1.
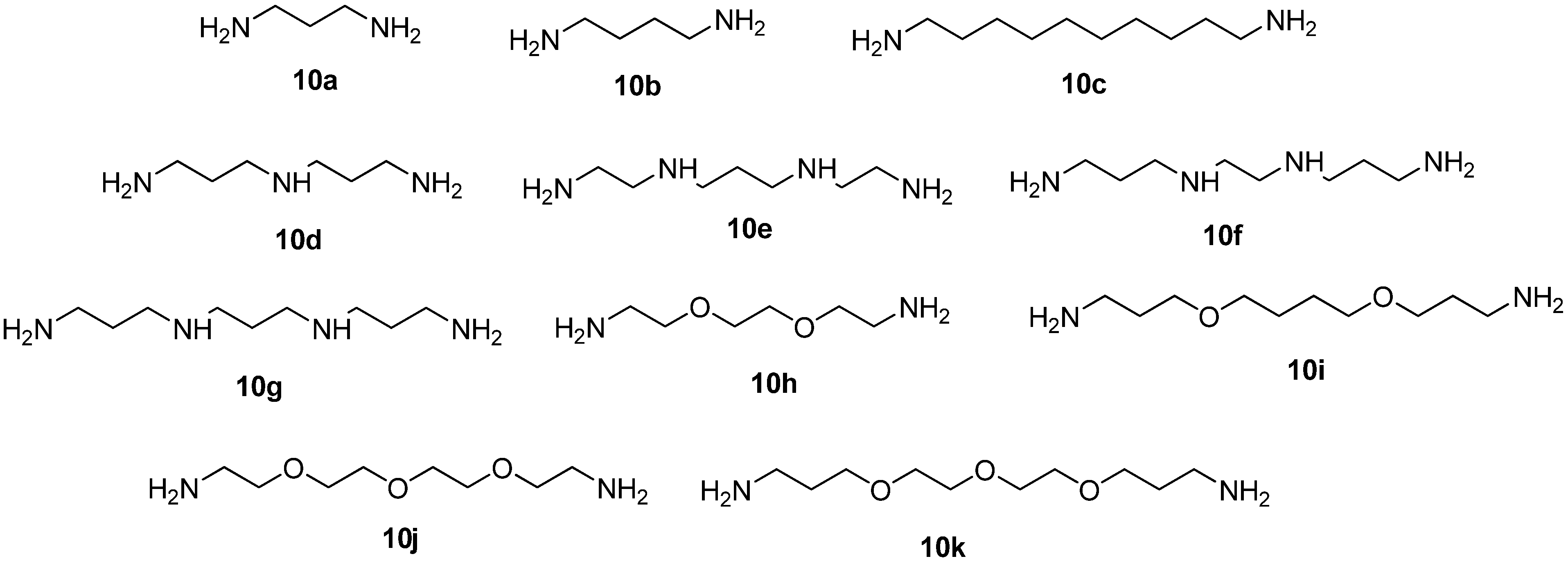
11,14,27,32-Tetraoxa-1,8,17,24-tetraazatetracyclo[22.5.5.13,7.118,22]hexatriaconta-3(36),4,6,18(35),19,21-hexaene (11h). Obtained from compound 4 (0.2 mmol, 102 mg), dioxadiamine 10h (0.2 mmol, 30 mg) in the presence of Pd(dba)2 (18 mg, 16 mol%), BINAP (22 mg, 18 mol%), NaOt-Bu (0.6 mmol, 57 mg) in abs. dioxane (10 mL). Eluent CH2Cl2–MeOH–NH3(aq) = 100:20:2. Yield 13 mg (13%), of a yellowish viscous oil. 1H-NMR (CDCl3) δ 2.76 (br.s, 8H, CH2N), 3.32 (t, 3J = 4.9 Hz, 4H, CH2NPh), 3.58 (br.s, 12H, CH2O, PhCH2N), 3.66 (s, 4H, CH2O), 3.72 (t, 3J = 4.9 Hz, 4H, CH2O), 4.17 (br.s, 2H, NH), 6.46–6.54 (m, 4H, H4(Ph), H6(Ph)), 6.98 (s, 2H, H2(Ph), 7.06 (t, 3J = 7.7 Hz, 2H, H5(Ph)). 13C-NMR (CDCl3) δ 43.8 (2C, CH2NPh), 55.3 (4C, CH2N), 62.0 (2C, PhCH2N), 69.5 (4C, CH2O), 69.7 (2C, CH2O), 70.4 (2C, CH2O), 111.3 (2C, CH(Ph)), 114.5 (2C, CH(Ph)), 118.2 (2C, CH(Ph)), 128.8 (2C, C5(Ph)), 139.8 (2C, C1(Ph)), 148.7 (2C, C3(Ph)). HRMS (MALDI-TOF): C28H43N4O4 (M+H)+ calcd.; 499.3284 observed; 499.3251.
11,14,17,30,35-Pentaoxa-1,8,20,27-tetraazatetracyclo[25.5.5.13,7.121,25]nonatriaconta-3(39),4,6,21 (38),22,24-hexaene (11j). Obtained from compound 4 (0.2 mmol, 102 mg), trioxadiamine 10j (0.2 mmol, 38 mg) in the presence of Pd(dba)2 (9 mg, 8 mol%), BINAP (11 mg, 9 mol%), NaOt-Bu (0.6 mmol, 57 mg) in abs. dioxane (10 mL). Eluent CH2Cl2–MeOH–NH3(aq) = 100:20:1. Yield 34 mg (31%), yellowish viscous oil. 1H-NMR (CDCl3) δ 2.86 (br.s, 8H, CH2N), 3.32 (t, 3J = 3.9 Hz, 4H, CH2NPh), 3.61 (br.s, 8H, CH2O), 3.66 (s, 8H, CH2O), 3.70 (t, 3J = 3.9 Hz, 4H, CH2O), 6.48–6.56 (m, 4H, H4(Ph), H6(Ph)), 7.07 (t, 3J = 7.6 Hz, 2H, H5(Ph)), 7.12 (br.s, 2H, H2(Ph)), two NH protons were not assigned. 13C-NMR (CDCl3) δ 43.7 (2C, CH2NPh), 54.6 (4C, CH2N), 60.4 (2C, PhCH2N), 68.7 (4C, CH2O), 69.5 (2C, CH2O), 70.3 (2C, CH2O), 70.8 (2C, CH2O), 111.6 (2C, CH(Ph)), 114.6 (2C, CH(Ph)), 117.8 (2C, CH(Ph)), 128.9 (2C, C5(Ph)), 149.2 (2C, C3(Ph)), two quaternary carbon atoms C1(Ph) were not assigned. HRMS (MALDI-TOF): C30H47N4O5 (M+H)+ calcd.; 543.3546 observed; 543.3598.
12,15,18,32,37-Pentaoxa-1,8,22,29-tetraazatetracyclo[27.5.5.13,7.123,27]hentetraconta-3(41),4,6,23 (40),24,26-hexaene (11k). Obtained from compound 4 (0.2 mmol, 102 mg), trioxadiamine 10k (0.2 mmol, 44 mg) in the presence of Pd(dba)2 (9 mg, 8 mol%), BINAP (11 mg, 9 mol%), NaOt-Bu (0.6 mmol, 57 mg) in abs. dioxane (10 mL). Eluent CH2Cl2–MeOH–NH3(aq) = 100:20:2. Yield 22 mg (19%), yellowish viscous oil. 1H-NMR (CDCl3) δ 1.84 (quintet, 3J = 6.0 Hz, 4H, CH2CH2CH2), 2.74 (br.s, 8H, CH2N), 3.23 (t, 3J = 6.4 Hz, 4H, CH2NPh), 3.51–3.63 (m, 16H, CH2O, PhCH2N), 3.64–3.69 (m, 4H, CH2O), 6.48 (d, 3J = 8.1 Hz, 2H, H4(Ph) or H6(Ph)), 6.58 (d, 3J = 7.2 Hz, 2H, H6(Ph) or H4(Ph)), 6.90 (s, 2H, H2(Ph)), 7.07 (t, 3J = 7.6 Hz, 2H, H5(Ph)), two NH protons were not assigned. 13C-NMR (CDCl3) δ 29.0 (2C, CH2CH2CH2), 41.7 (2C, CH2NPh), 54.9 (4C, CH2N), 61.1 (2C, PhCH2N), 69.6 (4C, CH2O), 69.7 (2C, CH2O), 70.2 (2C, CH2O), 70.6 (2C, CH2O), 111.0 (2C, CH(Ph), 113.5 (2C, CH(Ph)), 117.3 (2C, CH(Ph)), 128.7 (2C, C5(Ph)), 140.5 (2C, C1(Ph)), 148.9 (2C, C3(Ph)). HRMS (MALDI-TOF): C32H51N4O5 (M+H)+ calcd.; 571.3859 observed; 571.3832.
Cyclodimer 13k. Obtained as the second product in the synthesis of macrobicycle 11k. Eluent CH2Cl2–MeOH–NH3(aq) = 100:20:2. Yield 27 mg (23%) of a yellowish glassy compound. 1H-NMR (CDCl3) δ 1.84 (quintet, 3J = 6.0 Hz, 8H, CH2CH2CH2), 2.73 (br.s, 16H, CH2N), 3.20 (t, 3J = 6.3 Hz, 8H, CH2NPh), 3.50–3.70 (m, 48H, CH2O, PhCH2N), 6.43–6.46 (m, 8H, H4(Ph), H6(Ph)), 6.63 (s, 4H, H2(Ph)), 7.07 (t, 3J = 7.6 Hz, 4H, H5(Ph)), four NH protons were not assigned. 13C-NMR (CDCl3) δ 29.1 (4C, CH2CH2CH2), 41.6 (4C, CH2NPh), 57.7 (8C, CH2N), 61.2 (4C, PhCH2N), 69.4 (4C, CH2O), 69.6 (8C, CH2O), 70.2 (4C, CH2O), 70.6 (4C, CH2O), 111.1 (4C, CH(Ph)), 113.5 (4C, CH(Ph)), 117.8 (4C, CH(Ph)), 128.9 (4C, C5(Ph)), 140.3 (4C, C1(Ph)), 148.5 (4C, C3(Ph)). MS (MALDI-TOF): C64H101N8O10 (M+H)+ calcd.; 1141.76 observed; 1141.74.
10,13,25,30-Tetraoxa-1,7,16,22-tetraazatetracyclo[20.5.5.23,6.217,20]hexatriaconta-3,5,17,19,33,35-hexaene (12h). Obtained from compound 5 (0.2 mmol, 102 mg), dioxadiamine 10h (0.2 mmol, 30 mg) in the presence of Pd(dba)2 (18 mg, 16 mol%), BINAP (22 mg, 18 mol%), NaOt-Bu (0.6 mmol, 57 mg) in abs. dioxane (10 mL). Eluent CH2Cl2–MeOH–NH3(aq) = 100:20:2. Yield 30 mg (30%), of a yellowish viscous oil. 1H-NMR (CDCl3) δ 2.73 (br.s, 8H, CH2N), 3.30 (t, 3J = 5.1 Hz, 4H, CH2NPh), 3.52 (s, 4H, PhCH2N), 3.58 (br.s, 8H, CH2O), 3.66 (s, 4H, CH2O), 3.73 (t, 3J = 5.1 Hz, 4H, CH2O), 4.07 (br.s, 2H, NH), 6.58 (d, 3Jobs = 8.3 Hz, 4H, H3(Ph)), 7.24 (d, 3Jobs = 8.3 Hz, 4H, H2(Ph)). 13C-NMR (CDCl3) δ 43.9 (2C, CH2NPh), 55.3 (4C, CH2N), 60.1 (2C, PhCH2N), 69.3 (2C, CH2O), 70.1 (6C, CH2O), 113.1 (4C, C3(Ph)), 129.7 (4C, C2(Ph)), 132.1 (2C, C1(Ph)), 147.2 (2C, C4(Ph)). HRMS (MALDI-TOF): C28H43N4O4 (M+H)+ calcd.; 499.3284 observed; 499.3318.
Cyclodimer 14h. Obtained as the second product in the synthesis of macrobicycle 12h. Eluent CH2Cl2–MeOH–NH3(aq) = 100:20:2. Yield 27 mg (27%) of a yellowish glassy compound. 1H-NMR (CDCl3) δ 2.71 (br.s, 16H, CH2N), 3.28 (t, 3J = 4.4 Hz, 8H, CH2NPh), 3.51–3.61 (m, 24H, CH2O, PhCH2N), 3.64 (s, 8H, CH2O), 3.70 (t, 3J = 4.4 Hz, 8H, CH2O), 4.06 (br.s, 4H, NH), 6.56 (d, 3Jobs = 8.2 Hz, 8H, H3(Ph)), 7.16 (d, 3Jobs = 8.2 Hz, 8H, H2(Ph)). 13C-NMR (CDCl3) δ 43.5 (4C, CH2NPh), 54.7 (8C, CH2N), 60.5 (4C, PhCH2N), 69.3 (8C, CH2O), 69.7 (4C, CH2O), 70.2 (4C, CH2O), 112.8 (8C, C3 (Ph)), 130.1 (8C, C2(Ph)), 131.9 (4C, C1(Ph)), 147.1 (4C, C4(Ph)). MS (MALDI-TOF): C56H85N8O8 (M+H)+ calcd.; 997.65 observed; 997.66.
10,13,16,28,33-Pentaoxa-1,7,19,25-tetraazatetracyclo[23.5.5.23,6.220,23]nonatriaconta-3,5,20,22,36, 38-hexaene (12j). Obtained from compound 5 (0.2 mmol, 102 mg), trioxadiamine 10j (0.2 mmol, 38 mg) in the presence of Pd(dba)2 (9 mg, 8 mol%), BINAP (11 mg, 9 mol%), NaOt-Bu (0.6 mmol, 57 mg) in abs. dioxane (10 mL). Eluent CH2Cl2–MeOH–NH3(aq) = 100:20:2. Yield 26 mg (20%), of a yellowish viscous oil. 1H-NMR (CDCl3) δ 2.71 (t, 3J = 4.0 Hz, 8H, CH2N), 3.31 (t, 3J = 5.1 Hz, 4H, CH2NPh), 3.50 (s, 4H, PhCH2N), 3.60 (t, 3J = 4.0 Hz, 8H, CH2O), 3.67 (s, 8H, CH2O), 3.72 (t, 3J = 5.1 Hz, 4H, CH2O), 6.63 (d, 3Jobs = 8.3 Hz, 4H, H3(Ph)), 7.30 (d, 3Jobs = 8.3 Hz, 4H, H2(Ph)), two NH protons were not assigned. 13C-NMR (CDCl3) δ 43.7 (2C, CH2NPh), 55.2 (4C, CH2N), 60.0 (2C, PhCH2N), 69.6 (2C, CH2O), 70.1 (4C, CH2O), 70.4 (2C, CH2O), 70.8 (2C, CH2O), 112.9 (4C, C3(Ph)), 129.0 (2C, C1(Ph)), 129.6 (4C, C2(Ph)), 147.1 (2C, C4(Ph)). HRMS (MALDI-TOF): C30H47N4O5 (M+H)+ calcd.; 543.3546 observed; 543.3511.
11,14,17,30,35-Pentaoxa-1,7,21,27-tetraazatetracyclo[25.5.5.23,6.222,25]hentetraconta-3,5,22,24,38, 40-hexaene (12k). Obtained from compound 5 (0.2 mmol, 102 mg), trioxadiamine 10k (0.2 mmol, 44 mg) in the presence of Pd(dba)2 (9 mg, 8 mol%), BINAP (11 mg, 9 mol%), NaOt-Bu (0.6 mmol, 57 mg) in abs. dioxane (10 mL). Eluent CH2Cl2–MeOH–NH3(aq) = 100:20:2. Yield 18 mg (15%), of a yellowish viscous oil. 1H-NMR (CDCl3) δ 1.89 (quintet, 3J = 5.9 Hz, 4H, CH2CH2CH2), 2.71 (t, 3J = 4.0 Hz, 8H, CH2N), 3.25 (t, 3J = 6.3 Hz, 4H, CH2NPh), 3.51 (s, 4H, PhCH2N), 3.57 (t, 3J = 4.0 Hz, 8H, CH2O), 3.60–3.65 (m, 4H, CH2O), 3.62 (t, 3J = 5.2 Hz, 4H, CH2O), 3.68–3.72 (m, 4H, CH2O), 4.24 (br.s, 2H, NH), 6.57 (d, 3Jobs = 8.3 Hz, 4H, H3(Ph)), 7.24 (d, 3Jobs = 8.3 Hz, 4H, H2(Ph)). 13C-NMR (CDCl3) δ 28.9 (2C, CH2CH2CH2), 42.3 (2C, CH2NPh), 55.1 (4C, CH2N), 60.5 (2C, PhCH2N), 69.8 (4C, CH2O), 70.1 (2C, CH2O), 70.3 (2C, CH2O), 70.6 (2C, CH2O), 112.4 (4C, C3(Ph)), 130.0 (4C, C2(Ph)), 132.0 (2C, C1(Ph)), 147.6 (2C, C4(Ph)). HRMS (MALDI-TOF): C32H51N4O5 (M+H)+ calcd.; 571.3859 observed; 571.3890.
Cyclodimer 14k. Obtained as the second product in the synthesis of macrobicycle 12k. Eluent CH2Cl2–MeOH–NH3(aq) = 100:20:3. Yield 20 mg (17%), of a yellowish glassy compound. 1H-NMR (CDCl3) δ 1.86 (quintet, 3J = 5.3 Hz, 8H, CH2CH2CH2), 2.70 (br.s, 16H, CH2N), 3.19 (t, 3J = 5.7 Hz, 8H, CH2NPh), 3.50–3.61 (m, 40H, CH2O, PhCH2N), 3.63–3.67 (m, 8H, CH2O), 3.85 (br.s, 4H, NH), 6.53 (d, 3Jobs = 8.3 Hz, 8H, H3(Ph)), 7.11 (d, 3Jobs = 8.3 Hz, 8H, H2(Ph)). 13C-NMR (CDCl3) δ 29.1 (4C, CH2CH2CH2), 41.8 (4C, CH2NPh), 54.4 (8C, CH2N), 60.6 (4C, PhCH2N), 69.4 (8C, CH2O), 69.7 (4C, CH2O), 70.2 (4C, CH2O), 70.6 (4C, CH2O), 112.4 (8C, C3(Ph)), 130.2 (8C, C2(Ph)), 130.4 (4C, C1(Ph)), 147.5 (4C, C4(Ph)). MS (MALDI-TOF): C64H101N8O10 (M+H)+ calcd.; 1141.76 observed; 1141.78.
22,25,30-Trioxa-1,8,12,19-tetraazatetracyclo[17.8.5.13,7.113,17]tetratriaconta-3(34),4,6,13(33),14,16-hexaene (15а). Obtained from compound 6 (0.25 mmol, 139 mg), diamine 10a (0.25 mmol, 19 mg) in the presence of Pd(dba)2 (12 mg, 8 mol%), BINAP (14 mg, 9 mol%), tBuONa (0.75 mmol, 72 mg) in abs. dioxane (12 mL). Eluent CH2Cl2–MeOH = 3:1. Yield 22 mg (19%), of a yellowish glassy compound. 1H-NMR (CDCl3) δ 1.75 (br.s, 1Н, CH2CH2CH2), 2.06 (br.s, 1Н, CH2CH2CH2), 2.17 (d, 2J = 13.3 Hz, 2H), 2.38 (dd, 2J = 12.1 Hz, 2H), 2.72–2.96 (m, 4Н), 3.18–3.25 (m, 2Н), 3.26–3.32 (m, 4Н), 3.35-3.41 (m, 2Н), 3.47–3.78 (m, 8Н), 3.91 (d, 3J = 11.6 Hz, 2H), 4.02 (t, 3J = 9.0 Hz, 2H), 4.10 (br.s, 2Н, NH), 6.35 (d, 3Jobs = 6.9 Hz, 2H, H4(Ph) or H6(Ph)), 6.47 (d, 3J = 7.8 Hz, 2H, H6(Ph) or H4(Ph)), 7.01 (t, 3J = 7.8 Hz, 2H, H5(Ph)), 7.43 (br.s, 2Н, H2(Ph)). 13C-NMR (CDCl3) δ 28.9 (1C, CH2CH2CH2), 41.7 (2C, CH2NPh), 53.2 (2C, CH2N), 54.7 (2C, CH2N), 60.7 (2C, PhCH2N), 70.2 (2C, CH2O), 111.8 br (2C, CH(Ph)), 113.9 br (2C, CH(Ph)), 118.8 br (2C, CH(Ph)), 129.1 (2С, C5(Ph)), 148.7 (2C, C3(Ph)), two quaternary atoms С1(Ph) were not assigned due to a broad signal line; four СН2О carbon atoms give a very broad signal in the region 68–70 ppm). HRMS (MALDI-TOF): C27H41N4O3 (M+H)+ calcd.; 469.3178 observed; 469.3143.
1,8,12,19,28,35,39,46-Octaazaheptacyclo[44.8.5.519,28.13,7.113,17.130,34.140,44]octahexaconta-3(68),4,6, 13(67),14,16,30(61),31,33,40(60),41,43-dodecaene (19а). Obtained as the second product in the synthesis of macrobicycle 15a. Eluent CH2Cl2–MeOH = 3:1. Yield 22 mg (19%) of a yellowish glassy compound. 1H-NMR (CDCl3) δ 1.84 (br.s, 4Н ССН2С), 2.71–2.96 (m, 16Н, CH2N), 3.29 (t, 3J = 5.4 Hz, 8H, CH2NPh), 3.48–3.79 (m, 32Н, CH2O, PhCH2N), 6.49 (br.s, 4H, H4(Ph) or H6(Ph)), 6.55 (br.s, 4Н, H6(Ph) or H4(Ph)), 7.02–7.08 (m, 8Н, H2(Ph), H5(Ph)), NH protons were not assigned. 13C-NMR (CDCl3) δ 27.3 (2C, CCH2C), 42.9 (4C, CH2NPh), 53.2 (4C, CH2N), 54.7 (4C, CH2N), 61.9 (4C, PhCH2N), 67.3 (4C, CH2O), 67.7 (4C, CH2O), 69.4 (4C, CH2O), 110.7 (4C, CH(Ph)), 115.6 (4C, CH(Ph)), 118.2 (4C, CH(Ph)), 128.5 (4С, C5(Ph)), 137.7 (4C, C1(Ph)), 149.6 (4C, C3(Ph)). HRMS (MALDI-TOF): C54H81N8O8 (M+H)+ calcd.; 937.6279 observed; 937.6385.
23,26,3,-Trioxa-1,8,13,20-tetraazatricyclo[18.8.5.13,7.114,18]pentatriaconta-3(35),4,6,14(34),15,17-hexaene (15b). Obtained from compound 6 (0.25 mmol, 139 mg), diamine 10b (0.25 mmol, 22 mg) in the presence of Pd(dba)2 (12 mg, 8 mol%), BINAP (14 mg, 9 mol%), NaOt-Bu (0.75 mmol, 72 mg) in abs. dioxane (12 mL). Eluent CH2Cl2–MeOH = 3:1. Yield 14 mg (12%) of a yellowish glassy compound. 1H-NMR (CDCl3) δ 1.83 (br.s, 4Н, CCH2CH2C), 2.85–3.08 (m, 4Н, CH2N), 3.15–3.22 (m, 4Н, CH2N), 3.40 (br.s, 4Н, CH2NPh), 3.53–3.88 (m, 16Н, CH2O, PhCH2N), 3.97 (br.s, 2Н, NH), 6.41 (br.s, 2Н, H4(Ph) or H6(Ph)), 6.49 (br.s, 2H, H6(Ph) pr H4(Ph)), 7.03 (t, 3J = 7.6 Hz, 2H, H5(Ph)), 7.39 (br.s, 2Н, H2(Ph)). 13C-NMR (CDCl3) δ 26.6 (2C, CCH2CH2C), 43.5 br (2C, CH2NAr), 53.2 (2C, CH2N), 54.2 (2C, CH2N), 61.3 (2C, PhCH2N), 69.0 (2C, CH2O), 70.0 (4C, CH2O), 111.1 br (2C, CH(Ph)), 115.8 br (2C, CH(Ph)), 118.1 br (2C, CH(Ph)), 128.6 (2С, C5(Ph)), 148.8 (2C, C3(Ph)), two quaternary atoms С1(Ph) were not assigned due to a broad signal line. HRMS (MALDI-TOF): C28H43N4O3 (M+H)+ calcd.; 483.3335 observed; 483.3275.
22,25,50,53,58,65-Hexaoxa-1,7,12,19,28,35,40,47-octaazaheptacyclo-[45.8.5.519,28.23,6.113,17.130,34.141,45]octahexaconta-3,5,13(68),14,16,30(62),31,33,41(61),42,44,69-dodecaene (19b). Obtained as the second product in the synthesis of macrobicycle 15b. Eluent CH2Cl2–MeOH = 3:1. Yield 25 mg (21%) of a yellowish glassy compound. 1H-NMR (CDCl3) δ 1.71 (br.s, 8Н ССН2С), 2.45–3.08 (m, 16Н CH2N), 3.18 (t, 3J = 5.5 Hz, 8H, CH2NPh), 3.52–3.82 (m, 32Н, CH2O, PhCH2N), 4.65 (br.s, 4Н, NH), 6.48 (d, 3Jobs = 7.2 Hz, 4H, H(Ph) or Н6(Ph)), 6.56 (br.s, 4Н, Н6(Ph) or H4(Ph)), 7.01 (br.s, 4Н, Н2(Ph)), 7.03 (t, 3J = 7.6 Hz, 4H, H5(Ph)). 13C-NMR (CDCl3) δ 26.6 (4C, CCH2CH2C), 43.1 (4C, CH2NPh), 53.9 (4C, CH2N), 54.9 (4C, CH2N), 60.3 (4C, PhCH2N), 67.2 (4C, CH2O), 67.4 (4C, CH2O), 67.9 (4C, CH2O), 112.6 (4C, CH(Ph)), 112.9 (4C, CH(Ph)), 118.1 (4C, CH(Ph)), 128.9 (4С, C5(Ph)), 137.5 (4C, C1(Ph)), 148.6 (4C, C3(Ph)). HRMS (MALDI-TOF): C56H85N8O6 (M+H)+ calcd.; 965.6592 observed; 965.6511.
29,32,37-Trioxa-1,8,19,26-tetraazatetracyclo[24.8.5.13,7.120,24]hentetraconta-3(41),4,6,20(40),21,23-hexaene (15c). Obtained from compound 6 (0.25 mmol, 139 mg), diamine 10c (0.25 mmol, 43 mg) in the presence of Pd(dba)2 (12 mg, 8 mol%), BINAP (14 mg, 9 mol%), NaOt-Bu (0.75 mmol, 72 mg) in abs. dioxane (12 mL). Eluent CH2Cl2–MeOH = 10:1. Yield 38 mg (27%) of a yellowish glassy compound. 1H-NMR (CDCl3) δ 1.10–1.37 (m, 12Н, CCH2C), 1.45–1.55 (m, 4H, CH2CNPh) 2.60–3.21 (m, 12Н, CH2N, CH2NPh), 3.58–3.83 (m, 16Н, CH2O, PhCH2N), 4.22 (br.s, 2Н, NH), 6.48 (d, 3Jobs = 6.7 Hz, 2H, H4(Ph)), 6.59 (d, 3Jobs = 5.7 Hz, 2H, H6(Ph)), 6.84 (br.s 2H, H2(Ph)), 7.05 (t, 3J = 7.7 Hz, 2H, H5(Ph)). 13C-NMR (CDCl3) δ 26.1 (2C, CCH2C), 28.1 (2C, CCH2C), 28.2 (2C, CCH2C), 28.7 (2C, СCCH2C), 43.5 (2C, CH2NPh), 53.9 (2C, CH2N), 54.3 (2C, CH2N), 60.2 (2C, PhCH2N), 67.3 (2C, CH2O), 67.5 (2C, CH2O), 70.0 (2C, CH2O), 112.0 (2C, CH(Ph)), 114.0 (2C, CH(Ph)), 117.8 (2C, CH(Ph)), 129.2 (2С, C5(Ph)), 137.4 (2C, C1(Ph)), 148.9 (2C, C3(Ph)). HRMS (MALDI-TOF): C34H55N4O3 (M+H)+ calcd.; 567.4274 observed; 567.4225.
29,32,63,66,71,78-Hexaoxa-1,8,19,26,35,42,53,60-octaazaheptacyclo-[58.8.5.526,35.13,7.120,24.137,41.154,58]dooctaconta-3(82),4,6,20(81),21,23,37(75),38,40,54(74),55,57-dodecaene (19с). Obtained as the second product in the synthesis of macrobicycle 15c. Eluent CH2Cl2–MeOH = 3:1. Yield 21 mg (15%) of a yellowish glassy compound. 1H-NMR (CDCl3) δ 1.15–1.37 (m, 24Н, CCH2C), 1.50–1.59 (m, 8H, CH2CNAr), 2.67–2.92 (m, 16Н, CH2N), 3.02 (br.s, 8Н, CH2NPh), 3.50–3.75 (m, 32Н, CH2O, PhCH2N), 4.38 (br.s, 4Н, NH), 6.46 (d, 3J = 8.1 Hz, 4H, H4(Ph)), 6.56 (br.s, 4H, H6(Ph)), 6.72 (br.s, 4Н, H2(Ph)), 7.05 (t, 3J = 7.7 Hz, 4H, H5(Ph)). 13C-NMR (CDCl3) δ 27.1 (4C, CCH2C), 29.4 (12C, CCH2C), 43.9 (4C, CH2NPh), 53.3 (4C, CH2N), 53.9 (4C, CH2N), 60.2 (4C, PhCH2N), 67.0–70.0 (m, 12C, CH2O), 111.1 (4C, CH(Ph)), 114.2 (4C, CH(Ph)), 118.0 (4C, CH(Ph)), 129.0 (4С, C5(Ph)), 148.8 (4C, C3(Ph)), four quaternary C1(Ph) atoms were not assigned. HRMS (MALDI-TOF): C68H109N8O6 (M+H)+ calcd.; 1133.8470 observed; 1133.8562.
26,29,34-Trioxa-1,8,12,16,23-pentaazatetracyclo[21.8.5.13,7.117,21]octatriaconta-3(38),4,6,17(37), 18,20-hexaene (15d). Obtained from compound 6 (0.25 mmol, 139 mg), triamine 10d (0.25 mmol, 33 mg) in the presence of Pd(dba)2 (12 mg, 8 mol%), BINAP (14 mg, 9 mol%), NaOt-Bu (0.75 mmol, 72 mg) in abs. dioxane (12 mL). Eluent CH2Cl2–MeOH–NH3(aq) = 100:20:3. Yield 47 mg (36%) of a yellow glassy compound. 1H-NMR (CDCl3) δ 1.92 (br.s, 4Н, ССН2С), 2.65–2.75 (m, 8Н, CH2N), 2.88 (br.s, 4Н СН2NHСН2), 3.21 (t, 3J = 5.4 Hz, 4H, CH2NPh), 3.53 (s, 4Н, PhCH2N), 3.55–3.65 (m, 12H, CH2O), 5.10 (br.s, 2Н, PhNH), 6.43 (br.s, 2Н, H4(Ph) or H6(Ph)), 6.50 (d, 3Jobs = 6.7 Hz, 2H, H6(Ph) or H4(Ph)), 6.90 (br.s, 2Н, H2(Ph)), 7.03 (t, 3J = 7.7 Hz, 2H, Н5(Ph)), one NH proton was not assigned. 13C-NMR (CDCl3) δ 26.4 (2C, CCH2C), 41.6 (2C, CH2NPh), 46.3 (2C, CH2NHCH2), 54.2 (2C, CH2N), 54.9 (2C, CH2N), 60.4 (2C, PhCH2N), 69.2 (4C, CH2O), 70.0 (2C, CH2O), 110.7 (2C, CH(Ph)), 113.6 (2C, CH(Ph)), 117.5 (2C, CH(Ph)), 128.9 (2С, C5(Ph)), 140.4 (2C, C1(Ph)), 148.2 (2C, C3(Ph)). HRMS (MALDI-TOF): C30H48N5O3 (M+H)+ calcd.; 526.3757 observed; 526.3798.
26,29,57,60,65,72-Hexaoxa-1,8,12,16,23,32,39,43,47,54-decaazaheptacyclo-[52.8.5.523,32.13,7.117,21.134,38.148,52]-hexaheptaconta-3(76),4,6,17(75),18,20,34(69),35,37,48(68),49,51-dodecaene (19d). Obtained as the second product in the synthesis of macrobicycle 15d. Eluent CH2Cl2–MeOH–NH3(aq) = 100:25:5. Yield 12 mg (9%) of a yellowish glassy compound. 1H-NMR (CDCl3) δ 1.80 (br.s, 8Н, ССН2С), 2.60–2.82 (m, 24Н, CH2N), 3.14 (br.s, 8Н, CH2NPh), 3.48–3.68 (m, 32H, CH2O, PhCH2N), 6.44 (br.s, 4Н, H4(Ph) or H6(Ph)), 6.55 (br.s 4H, H6(Ph) or H4(Ph)), 6.79 (br.s, 4Н, H2(Ph)), 7.05 (br.s, 4Н, Н5(Ph)), NH protons were not assigned. MS (MALDI-TOF): C60H95N10O6 (M+H)+ calcd.; 1051.74 observed; 1051.72.
28,31,36-Trioxa-1,8,11,15,18,25-hexatetracyclo[23.8.5.13,7.119,23]tetraconta-3(40),4,6,19(39),20,22-hexaene (15е). Obtained from compound 6 (0.25 mmol, 139 mg), tetraamine 10e (0.25 mmol, 40 mg) in the presence of Pd(dba)2 (12 mg, 8 mol%), BINAP (14 mg, 9 mol%), NaOt-Bu (0.75 mmol, 72 mg) in abs. dioxane (12 mL). Eluent CH2Cl2–MeOH–NH3(aq) = 100:25:5. Yield 39 mg (28%) of a yellow glassy compound. 1H-NMR (CDCl3) δ 1.78 (quintet, 3J = 5.5 Hz, 2H, ССН2С), 2.70 (t, 3J = 5.5 Hz, 8H, СН2NHСН2), 2.82 (t, 3J = 5.7 Hz, 4H, СН2N), 2.87 (t, 3J = 5.2 Hz, 4H, CH2N), 3.31 (t, 3J = 4.7 Hz, 4H, CH2NPh), 3.53 (s, 4H, PhCH2N), 3.56–3.66 (m, 12H, CH2O), 4.79 (br.s, 2Н, PhNH), 6.51 (d, 3J = 7.7 Hz, 2H, H4(Ph) or H6(Ph)), 6.54 (dd, 3J = 7.8 Hz, 4J = 1.5 Hz, 2H, H6(Ph) or H4(Ph)), 6.96 (br.s, 2Н, H2(Ph)), 7.03 (t, 3J = 7.8 Hz, 2H, Н5(Ph)), two NH protons were not assigned. 13C-NMR (CDCl3) δ 25.9 (1C, CCH2C), 42.5 (2C, CH2NPh), 47.7 (2C, CH2NHCH2), 48.8 (2C, CH2NHCH2), 54.9 (2C, CH2N), 55.3 (2C, CH2N), 60.5 (2C, PhCH2N), 69.3 (2C, CH2O), 69.6 (2C, CH2O), 70.1 (2C, CH2O), 110.8 (2C, CH(Ph)), 113.7 (2C, CH(Ph)), 117.6 (2C, CH(Ph)), 128.8 (2С, C5(Ph)), 140.9 (2C, C1(Ph)), 148.3 (2C, C3(Ph)). HRMS (MALDI-TOF): C31H51N6O3 (M+H)+ calcd.; 555.4022 observed; 555.3979.
29,32,37-Trioxa-1,8,12,15,19,26-hexaazatetracyclo[24.8.5.13,7.120,24]hentetraconta-3(41),4,6,20(40), 21,23-hexaene (15f). Obtained from compound 6 (0.25 mmol, 139 mg), tetraamine 10f (0.25 mmol, 44 mg) in the presence of Pd(dba)2 (12 mg, 8 mol%), BINAP (14 mg, 9 mol%), NaOt-Bu (0.75 mmol, 72 mg) in abs. dioxane (12 mL). Eluent CH2Cl2–MeOH–NH3(aq) = 100:25:5. Yield 47mg (33%) of a yellow glassy compound. 1H-NMR (CDCl3) δ 1.74 (quintet, 3J = 6.3 Hz, 4H, ССН2С), 2.55–2.81 (m, 16Н, CH2N), 3.15 (t, 3J = 6.3 Hz, 4H, CH2NPh), 3.51–3.66 (m, 16H, CH2O, PhCH2N), 6.43 (d, 3J = 7.8 Hz, 2H, H4(Ph) or H6(Ph)), 6.54 (d, 3J = 7.3 Hz, 2H, H6(Ph) or H4(Ph)), 6.90 (br.s, 2Н, H2(Ph)), 7.04 (t, 3J = 7.7 Hz, 2H, Н5(Ph)) NH protons were not assigned. 13C-NMR (CDCl3) δ 28.8 (2C, CCH2C), 42.5 (2C, CH2NPh), 47.7 (2C, CH2NHCH2), 48.3 (2C, CH2NHCH2), 54.5 (2C, CH2N), 54.9 (2C, CH2N), 60.4 (2C, PhCH2N), 64.9 (4C, CH2O), 70.1 (2C, CH2O), 110.4 (2C, CH(Ph)), 113.9 (2C, CH(Ph)), 117.3 (2C, CH(Ph)), 128.8 (2С, C5(Ph)), 140.5 (2C, C1(Ph)), 148.7 (2C, C3(Ph)). HRMS (MALDI-TOF): C32H53N6O3 (M+H)+ calcd.; 569.4179 observed; 569.4142.
30,33,38-Trioxa-1,8,12,16,20,27-hexaazatetracyclo[25.8.5.13,7.121,25]dodecatetraconta-3(42),4,6,21 (41),22,24-hexaene (15g). Obtained from compound 6 (0.25 mmol, 139 mg), tetraamine 10g (0.25 mmol, 47 mg) in the presence of Pd(dba)2 (12 mg, 8 mol%), BINAP (14 mg, 9 mol%), NaOt-Bu (0.75 mmol, 72 mg) in abs. dioxane (12 mL). Eluent CH2Cl2–MeOH–NH3(aq) = 100:25:5. Yield 34 mg (24%) of a yellow glassy compound. 1H-NMR (CDCl3) δ 1.74 (quintet, 3J = 7.3 Hz, 2H, ССН2С), 1.78 (quintet, 3J = 5.3 Hz, 4H, ССН2С), 2.67–2.76 (m, 12Н, CH2N, CH2NHCH2), 2.79 (t, 3J = 5.6 Hz, 4H, CH2N), 3.12 (t, 3J = 6.3 Hz, 4H, CH2NPh), 3.53–3.65 (m, 16H, CH2O, PhCH2N), 4.27 (br.s, 2Н, PhNH), 6.46 (dd, 3J = 7.7 Hz, 4J = 1.8 Hz, 2H, H4(Ph) or H6(Ph)), 6.58 (d, 3J = 7.7 Hz, 2H, H6(Ph) or H4(Ph)), 6.89 (br.s, 2Н, H2(Ph)), 7.05 (t, 3J = 7.7 Hz, 2H, Н5(Ph), two NH protons were not assigned. 13C-NMR (CDCl3) δ 27.5 (1C, CCH2C), 28.4 (2C, CCH2C), 42.4 (2C, CH2NPh), 47.9 (2C, CH2NHCH2), 49.2 (2C, CH2NHCH2), 54.7 (2C, CH2N), 55.0 (2C, CH2N), 60.5 (2C, PhCH2N), 69.5 (4C, CH2O), 70.2 (2C, CH2O), 110.7 (2C, CH(Ph)), 113.6 (2C, CH(Ph)), 117.5 (2C, CH(Ph)), 128.8 (2С, C5(Ph)), 140.9 (2C, C1(Ph)), 148.7 (2C, C3(Ph)). HRMS (MALDI-TOF): C33H55N6O3 (M+H)+ calcd.; 583.4335 observed; 583.4390.
11,14,27,30,35-Pentaoxa-1,8,17,24-tetraazatetracyclo[22.8.5.13,7.118,22]nonatriaconta-3(39),4,6,18 (38),19,21-hexaene (15h). Obtained from compound 6 (0.25 mmol, 139 mg), dioxadiamine 10h (0.25 mmol, 37 mg) in the presence of Pd(dba)2 (12 mg, 8 mol%), BINAP (14 mg, 9 mol%), NaOt-Bu (0.75 mmol, 72 mg) in abs. dioxane (12 mL). Eluent CH2Cl2–MeOH = 10:1. Yield 27 mg (20%) of a yellowish glassy compound. 1H-NMR (CDCl3) δ 2.61–3.15 (m, 8Н, CH2N), 3.30 (br.s, 4Н, CH2NPh), 3.50–3.75 (m, 24Н, CH2O, PhCH2N), 6.53 (dd, 3J = 8.1 Hz, 4J = 1.6 Hz, 2H, H4(Ph) or H6(Ph)), 6.58 (br.s, 2H, H6(Ph) or H4(Ph)), 7.06 (t, 3J = 7.8 Hz, 2H, H5(Ph)), 7.19 (br.s, 2H, H2(Ph)), NH protons were not assigned. 13C-NMR (CDCl3) δ 43.9 (2C CH2NPh), 53.4 (2C, CH2N), 54.7 (2C, CH2N), 61.0 (2C, PhCH2N), 67.4 (2C, CH2O), 67.8 (2C, CH2O), 69.3 (4C, CH2O), 70.2 (2C, CH2O), 111.1 (2C, CH(Ph)), 116.3 (2C, CH(Ph)), 118.9 (2C, CH(Ph)), 128.9 (2С, C5(Ph)), 137.8 (2C, C1(Ph)), 149.1 (2C, C3(Ph)). HRMS (MALDI-TOF): C30H47N4O5 (M+H)+ calcd.; 543.3543 observed; 543.3588.
10,13,26,29,42,45,58,61,66,73-Decaoxa-1,7,16,23,32,39,48,55-octaazaheptacyclo-[53.8.5.5.5.32,32.23,6.117,21.134,38.149,53]-ocatheptaconta-3,5,17(76),18,20,34(70),35,37,49(69),50,52,77-dodecaene (19h). Obtained as the second product in the synthesis of macrobicycle 15h. Eluent CH2Cl2–MeOH = 3:1. Yield 14 mg (10%) of a yellowish glassy compound. 1H-NMR (DMSO-d6, 363K) δ 2.84 (br.s, 16Н, CH2N), 3.21 (t, 3J = 5.6 Hz, 8H, CH2NPh), 3.52-3.68 (m, 48Н, CH2O, PhCH2N), 6.50 (d, 3J = 8.1 Hz, 4H, H4(Ph) or H6(Ph)), 6.52 (d, 3J = 8.3 Hz, 4H, H6(Ph) or H4(Ph)), 6.72 (br.s, 4Н, H2(Ph)), 6.99 (t, 3J = 7.8 Hz, 4H, H5(Ph)), NH protons were not assigned. 13C-NMR (DMSO-d6, 363K) δ 42.7 (4C, CH2NPh), 53.5 (4C, CH2N), 53.9 (4C, CH2N), 59.6 (4C, PhCH2N), 67.5 (4C, CH2O), 68.0 (4C, CH2O), 68.9 (4C, CH2O), 69.5 (8C, CH2O), 111.7 (4C, CH(Ph)), 112.2 (4C, CH(Ph)), 116.5 (4C, CH(Ph)), 128.1 (4C, C5(Ph)), 148.3 (4С, C3(Ph)), four quaternary C1(Ph) atoms were not assigned. HRMS (MALDI-TOF): C60H93N8O10 (M+H)+ calcd.; 1085.7014 observed; 1085.7086.
11,16,29,32,37-Pentaoxa-1,8,19,26-tetraazatetracyclo[24.8.5.13,7.120,24]hentetraconta-3(41),4,6,20 (40),21,23-hexaene (15i). Obtained from compound 6 (0.25 mmol, 139 mg), dioxadiamine 10i (0.25 mmol, 51 mg) in the presence of Pd(dba)2 (12 mg, 8 mol%), BINAP (14 mg, 9 mol%), NaOt-Bu (0.75 mmol, 72 mg) in abs. dioxane (12 mL). Eluent CH2Cl2–MeOH = 10:1. Yield 56 mg (37%) as a yellowish glassy compound. 1H-NMR (CDCl3) δ 1.57–1.67 (m, 4H, ССH2CH2C), 1.80 (quintet, 3J = 5.3 Hz, 4H, NCCH2CN), 2.57–3.16 (m, 12Н, CH2N), 3.36-3.43 (m, 4Н, CH2O), 3.47 (t, 3J = 4.7 Hz, 4H, CH2O), 3.54–3.75 (m, 16Н, CH2O, PhCH2N), 6.47 (d, 3Jobs = 7.1 Hz, 4H, H4(Ph), H6(Ph)), 6.96 (br.s, 2Н, H2(Ph)), 7.05 (t, 3J = 7.7 Hz, 2H, H5(Ph)), NH protons were not assigned. 13C-NMR (CDCl3) δ 26.4 (2C, ССH2CH2C), 29.1 (2C, NССH2N), 41.7 (2C, CH2NPh), 53.0 (2C, CH2N), 54.3 (2C, CH2N), 60.2 (2C, PhCH2N), 67.2 (2C, CH2O), 67.5 (2C, CH2O), 69.0 (2C, CH2O), 69.2 (2C, CH2O), 70.5 (2C, CH2O), 110.1 (2C, CH(Ph)), 115.3 (2C, CH(Ph)), 118.2 (2C, CH(Ph)), 128.9 (2С, C5(Ph)), 137.4 (2C, C1(Ph)), 149.3 (2C, C3(Ph)). HRMS (MALDI-TOF): C34H55N4O5 (M+H)+ calcd.; 599.4172 observed; 599.4130.
12,15,18,32,35,40-Hexaoxa-1,8,22,29-tetraazatetracyclo[27.8.5.13,7.123,27]tetratetraconta-3(44),4,6,23 (43),24,26-hexaene (15k). Obtained from compound 6 (0.25 mmol, 139 mg), trioxadiamine 10k (0.25 mmol, 51 mg) in the presence of Pd(dba)2 (12 mg, 8 mol%), BINAP (14 mg, 9 mol%), NaOt-Bu (0.75 mmol, 72 mg) in abs. dioxane (12 mL). Eluent CH2Cl2–MeOH = 10:1. Yield 58 mg (38%) of a yellowish glassy compound. 1H-NMR (CDCl3) δ 1.82 (quintet, 3J = 6.1 Hz, 4H, ССН2С), 2.74 (t, 3J = 4.7 Hz, 4H, CH2N), 2.77 (t, 3J = 4.7 Hz, 4H, СН2N), 3.18 (t, 3J = 6.3 Hz, 4H, CH2NPh), 3.57 (t, 3J = 5.8 Hz, 4H, CH2O), 3.57–3.67 (m, 24H, CH2O, PhCH2N), 4.10 (br.s, 2Н, NH), 6.45 (d, 3J = 7.7 Hz, 2H, H4(Ph) or H6(Ph)), 6.55 (d, 3J = 7.3 Hz, 2H, H6(Ph) or H4(Ph)), 6.83 (br.s, 2Н, H2(Ph)), 7.04 (t, 3J = 7.8 Hz, 2H, Н5(Ph)). 13C-NMR (CDCl3) δ 28.9 (2C, CCH2C), 41.5 (2C, CH2NPh), 53.0 (2C, CH2N), 54.4 (2C, CH2N), 60.3 (2C, PhCH2N), 67.2 (2C, CH2O), 67.6 (2C, CH2O), 69.0 (2C, CH2O), 69.6 (2C, CH2O), 70.0 (2C, CH2O), 70.4 (2C, CH2O), 110.4 (2C, CH(Ph)), 115.2 (2C, CH(Ph)), 118.3 (2C, CH(Ph)), 128.8 (2С, C5(Ph)), 137.3 (2C, C1(Ph)), 149.4 (2C, C3(Ph)). HRMS (MALDI-TOF): C34H55N4O6 (M+H)+ calcd.; 615.4121 observed; 615.4157.
12,15,18,32,35,49,52,55,69,72,77,84-Dodecaoxa-1,8,22,29,38,45,59,66-octaazaheptacyclo-[64.8.5.529,38.13,7.123,27.140,44.160,64]octaoctaconta-3(88),4,6,23(87),24,26,40(81),41,43,60(80),61,63-dodeca-ene (19k). Obtained as the second product in the synthesis of macrobicycle 15k. Eluent CH2Cl2–MeOH = 3:1. Yield 37 mg (24%) of a yellowish glassy compound. 1H-NMR (CDCl3) δ 1.84 (quintet, 3J = 5.8 Hz, 8H, ССН2С), 2.75 (t, 3J = 4.8 Hz, 8H, CH2N), 2.78 (t, 3J = 4.8 Hz, 8H, СН2N), 3.18 (t, 3J = 5.9 Hz, 8H, CH2NPh), 3.53-3.68 (m, 56H, CH2O, PhCH2N), 4.06 (br.s, 4Н, NH), 6.44 (d, 3J = 7.6 Hz, 4H, H4(Ph) or H6(Ph)), 6.59 (d, 3J = 7.2 Hz, 4H, H6(Ph) or H4(Ph)), 6.69 (br.s, 4Н, H2(Ph)), 7.05 (t, 3J = 7.7 Hz, 4H, Н5(Ph)). 13C-NMR (CDCl3) δ 29.0 (4C, CCH2C), 41.4 (4C, CH2NPh), 54.2 (8C, CH2N), 60.5 (4C, PhCH2N), 69.3 (4C, CH2O), 69.4 (4C, CH2O), 69.9 (4C, CH2O), 70.0 (4C, CH2O), 70.2 (4C, CH2O), 70.4 (4C, CH2O), 110.6 (4C, CH(Ph)), 113.2 (4C, CH(Ph)), 117.2 (4C, CH(Ph)), 128.7 (4С, C5(Ph)), 140.3 (4C, C1(Ph)), 148.5 (4C, C3(Ph)). MS (MALDI-TOF): C68H109N8O6 (M+H)+ calcd.; 1229.82 observed; 1229.84.
10,13,25,28,33-Pentaoxa-1,7,16,22-tetraazatetracyclo[20.8.5.23,6.217,20]nonatriaconta-3,5,17,19,36, 38-hexaene (16h). Obtained from compound 7 (0.5 mmol, 278 mg), dioxadiamine 10h (0.5 mmol, 74 mg) in the presence of Pd(dba)2 (46 mg, 16 mol%), BINAP (56 mg, 18 mol%), NaOt-Bu (1.5 mmol, 144 mg) in abs. Dioxane (25 ml). Eluent CH2Cl2–MeOH = 5:1, CH2Cl2–MeOH–NH3(aq) = 100:20:1. Yield 49 mg (18%) as a yellow glassy compound. 1H-NMR (CDCl3) δ 2.69–2.86 (m, 8H, CH2N), 3.27 (t, 3J = 4.5 Hz, 4H, CH2NPh), 3.51–3.70 (m, 24H, CH2O, PhCH2N), 6.54 (d, 3Jobs = 7.0 Hz, 4H, H3(Ph), H3'(Ph)), 7.09 (d, 3Jobs = 7.5 Hz, 4H, H2(Ph), H2'(Ph)), NH protons were not assigned. 13C-NMR (CDCl3) δ 43.4 (2C, CH2NPh), 52.8 (2C, CH2N), 54.8 (2C, CH2N), 60.1 (2C, PhCH2N), 67.0 (2C, CH2O), 69.0–69.9 (m, 8C, CH2O), 113.0 (4C, C3(Ph), C3'(Ph)), 126.0 (2C, C1(Ph)), 131.4 (4C, C2(Ph), C2'(Ph)), 148.3 (2C, C4(Ph)). HRMS (MALDI-TOF): C30H47N4O5 (M+H)+ calcd.; 543.3546 observed; 543.3577.
10,13,25,40,43,55,58,63,72-Decaoxa-1,7,16,22,31,37,46,52-octaazaheptacyclo-[50.8.5.522,31.23,6.217,20.233,36.247,50]octaheptaconta-3,5,17,19,33,35,47,49,66,68,75,77-dodecaene (20h). Obtained as the second product in the synthesis of macrobicycle 16h. Eluent CH2Cl2–MeOH–NH3(aq) = 100:20:1–100:20:3. Yield 85 mg (31%) of a yellow glassy compound. 1H-NMR (CDCl3) δ 2.59 (t, 3J = 5.0 Hz, 8H, CH2N), 2.67 (t, 3J = 4.6 Hz, 8H, CH2N), 3.27 (t, 3J = 4.5 Hz, 8H, CH2NPh), 3.47 (br.s, 8H, CH2O), 3.51–3.64 (m, 16H, CH2O), 3.66 (s, 8H, CH2O or PhCH2N), 3.68 (s, 8H, PhCH2N or CH2O), 3.74 (t, 3J = 5.0 Hz, 8H, CH2O), 4.04 (br.s, 4H, NH), 6.51 (d, 3Jobs = 8.1 Hz, 8H, H3(Ph), H3'(Ph)), 7.16 (d, 3Jobs = 8.1 Hz, 8H, H2(Ph), H2'(Ph)). 13C-NMR (CDCl3) δ 43.6 (4C, CH2NPh), 52.2 (4C, CH2N), 55.3 (4C, CH2N), 61.2 (4C, PhCH2N), 67.0–69.9 (m, 20C), 113.3 (8C, C3(Ph), C3'(Ph)), 125.0 (4C, C1(Ph)), 131.4 (8C, C2(Ph), C2'(Ph)), 147.6 (4C, C4(Ph)). HRMS (MALDI-TOF): C60H93N8O10 (M+H)+ calcd.; 1085.7014 observed; 1085.6952.
11,16,29,32,37-Pentaoxa-1,7,20,26-tetraazatetracyclo[24.8.5.23,6.221,24]tritetraconta-3,5,21,23,42-hexaene (16i). Obtained from compound 7 (0.25 mmol, 139 mg), dioxadiamine 10i (0.25 mmol, 51 mg) in the presence of Pd(dba)2 (23 mg, 16 mol%), BINAP (28 mg, 18 mol%), NaOt-Bu (0.75 mmol, 72 mg) in abs. dioxane (12 mL). Eluent CH2Cl2–MeOH = 5:1. Yield 15 mg (10%) as a yellow glassy compound. 1H-NMR (CDCl3) δ 1.65 (br.s, 4H, CCH2CH2C), 1.86 (quintet, 3J = 5.7 Hz, 4H, NCCH2CN), 2.58–3.10 (m, 8H, CH2N), 3.20 (t, 3J = 5.7 Hz, 4H, CH2NPh), 3.37–3.45 (m, 4H, CH2O), 3.47–3.72 (m, 16H, CH2O, PhCH2N), 3.78-3.93 (m, 4H, CH2O), 6.51 (d, 3Jobs = 8.1 Hz, 4H, H3(Ph), H3'(Ph)), 7.21 (d, 3Jobs = 8.1 Hz, 4H, H2(Ph), H2'(Ph)), NH protons were not assigned. 13C-NMR (CDCl3) δ 26.5 (2C, CCH2CH2C), 29.3 (2C, CCH2C), 42.6 (2C, CH2NPh), 53.2 (2C, CH2N), 54.0 (2C, CH2N), 60.1 (2C, PhCH2N), 67.6 (2C, CH2O), 69.5 (2C, CH2O), 70.7 (2C, CH2O), 71.0 (4C, CH2O), 112.3 (4C, C3(Ph), C3'(Ph)), 127.5 (2C, C1(Ph)), 131.6 (4C, C2(Ph), C2'(Ph)), 148.8 (2C, C4(Ph)). HRMS (MALDI-TOF): C34H55N4O5 (M+H)+ calcd.; 599.4172 observed; 599.4131.
11,16,29,32,45,50,63,66,71,80-Decaoxa-1,7,20,26,35,41,54,60-ocatazaheptacyclo-[58.8.5.526,35.23,6.221,24.237,40.155,58]hexaoctaconta-3,5,21,23,37,39,55,57,74,76,83,85-dodecaene (20i). Obtained as the second product in the synthesis of macrobicycle 16i. Eluent CH2Cl2–MeOH = 5:1. Yield 15 mg (10%) of a yellow glassy compound. 1H-NMR (CDCl3) δ 1.66 (br.s, 8H, CCH2CH2C), 1.86 (quintet, 3J = 5.7 Hz, 8H, NCCH2CN), 2.47 (br.s, 8H, CH2N), 3.06 (br.s, 8H, CH2N), 3.19 (t, 3J = 5.3 Hz, 8H, CH2NPh), 3.37-3.45 (m, 8H, CH2O), 3.47-3.72 (m, 32H, CH2O, PhCH2N), 3.88 (br.s, 8H, CH2O), 4.45 (br.s, 4H, NH), 6.57 (d, 3Jobs = 8.0 Hz, 8H, H3(Ph), H3'(Ph)), 7.23 (d, 3Jobs = 8.0 Hz, 8H, H2(Ph), H2'(Ph)). 13C-NMR (CDCl3) δ 26.7 (4C, CCH2CH2C), 29.6 (4C, CCH2C), 42.3 (4C, CH2NPh), 51.9 (4C, CH2N), 54.9 (4C, CH2N), 60.1 (4C, PhCH2N), 67.2 (8C, CH2O), 69.1 (4C, CH2O), 69.4 (4C, CH2O), 70.0 (4C, CH2O), 112.5 (8C, C3(Ph), C3'(Ph)), 125.2 (4C, C1(Ph)), 131.4 (8C, C2(Ph), C2'(Ph)), 148.1 (4C, C4(Ph)). HRMS (MALDI-TOF): C68H109N8O10 (M+H)+ calcd.; 1197.8266 observed; 1197.8215.
11,14,17,30,33,38-Hexaoxa-1,7,21,27-tetraazatetracyclo[25.8.5.23,6.222,25]tetratetraconta-3,5,22,24, 41,43-hexaene (16k). Obtained from compound 7 (0.25 mmol, 139 mg), trioxadiamine 10k (0.25 mmol, 55 mg) in the presence of Pd(dba)2 (23 mg, 16 mol%), BINAP (28 mg, 18 mol%), NaOt-Bu (0.75 mmol, 72 mg) in abs. dioxane (12 mL). Eluent CH2Cl2–MeOH = 5:1. Yield 15 mg (10%) of a yellow glassy compound. 1H-NMR (CDCl3) δ 1.85 (quintet, 3J = 5.3 Hz, 4H, CCH2C), 2.90–3.12 (m, 8H, CH2N), 3.21 (t, 3J = 5.5 Hz, 4H, CH2NPh), 3.52–3.73 (m, 24H, CH2O, PhCH2N), 3.82–3.94 (m, 4H, CH2O), 6.46 (d, 3Jobs = 7.9 Hz, 4H, H3(Ph), H3'(Ph)), 7.16 (d, 3Jobs = 7.9 Hz, 4H, H2(Ph), H2'(Ph)), NH protons were not assigned. 13C-NMR (CDCl3) δ 28.6 (2C, CCH2C), 41.7 (2C, CH2NPh), 52.8 (2C, CH2N), 54.1 (2C, CH2N), 59.9 (2C, PhCH2N), 69.7 (2C, CH2O), 69.9 (2C, CH2O), 70.2 (4C, CH2O), 70.6 (4C, CH2O), 112.3 (4C, C3(Ph), C3'(Ph)), 127.5 (2C, C1(Ph)), 131.8 (4C, C2(Ph), C2'(Ph)), 148.8 (2C, C4(Ph)). HRMS (MALDI-TOF): C34H55N4O6 (M+H)+ calcd.; 615.4121 observed; 615.4063.
11,14,17,30,33,46,49,52,66,69,74,82-Dodecaoxa-1,7,21,27,36,42,56,63-octaazaheptacyclo-[61.8.5. 527,36.23,6.222,25.238,41.157,61]octaoctaconta-3,5,22,24,38,40,57(77),58,60,78,85,87-dodecaene (20k). Obtained as the second product in the synthesis of macrobicycle 16k. Eluent CH2Cl2–MeOH = 5:1. Yield 29 mg (19%) of a yellow glassy compound. 1H-NMR (CDCl3) δ 1.86 (quintet, 3J = 5.8 Hz, 8H, CCH2C), 2.45 (br.s, 8H, CH2N), 3.04 (br.s, 8H, CH2N), 3.18 (t, 3J = 6.3 Hz, 8H, CH2NPh), 3.38–3.72 (m, 48H, CH2O, PhCH2N), 3.89 (br.s, 8H, CH2O), 4.49 (br.s, 4H, NH), 6.56 (d, 3Jobs = 7.8 Hz, 8H, H3(Ph), H3'(Ph)), 7.21 (d, 3Jobs = 7.8 Hz, 8H, H2(Ph), H2'(Ph)). 13C-NMR (CDCl3) δ 29.0 (4C, CCH2C), 42.1 (4C, CH2NPh), 52.0 (4C, CH2N), 54.9 (4C, CH2N), 60.2 (4C, PhCH2N), 67.2 (4C, CH2O), 67.6 (4C, CH2O), 69.0 (4C, CH2O), 70.0 (4C, CH2O), 70.1 (4C, CH2O), 70.4 (4C, CH2O), 112.5 (8C, C3(Ph), C3'(Ph)), 125.1 (4C, C1(Ph)), 131.4 (8C, C2(Ph), C2'(Ph)), 148.0 (4C, C4(Ph)). MS (MALDI-TOF): C68H109N8O12 (M+H)+ calcd.; 1229.82 observed; 1229.83.
26,29,34,37-Tetraoxa-1,8,12,16,23-pentaazatetracyclo[21.8.8.13,7.117,21]hentetraconta-3(41),4,6,17 (40),18,20-hexaene (17d). Obtained from compound 8 (0.25 mmol, 150 mg), triamine 10d (0.25 mmol, 33 mg) in the presence of Pd(dba)2 (12 mg, 8 mol%), BINAP (14 mg, 9 mol%), NaOt-Bu (0.75 mmol, 72 mg) in abs. dioxane (12 mL). Eluent CH2Cl2–MeOH–NH3(aq) = 100:25:5. Yield 35 mg (25%) of a yellow glassy compound. 1H-NMR (CDCl3) δ 1.78 (quintet, 3J = 6.3 Hz, 4H, CCH2C), 2.75 (t, 3J = 6.4 Hz, 4H, CH2NHCH2), 2.76 (t, 3J = 5.6 Hz, 8H, CH2N), 3.21 (t, 3J = 6.4 Hz, 4H, CH2NPh), 3.56–3.62 (m, 20H, CH2O, PhCH2N), 6.43 (dd, 3J = 7.8 Hz, 4J = 1.9 Hz, 2H, H6(Ph) or H4(Ph)), 6.51 (d, 3J = 7.3 Hz, 2H, H4(Ph) or H6(Ph)), 6.88 (br.s, 2H, H2(Ph)), 7.06 (t, 3J = 7.7 Hz, 2H, H5(Ph)), NH protons were not assigned. 13C-NMR (CDCl3) δ 24.2 (2C, CCH2C), 42.9 (2C, CH2NPh), 48.2 (2C, CH2NHCH2), 54.6 (4C, CH2N), 60.0 (2C, PhCH2N), 70.0 (4C, CH2O), 70.6 (4C, CH2O), 110.2 (2C, CH(Ph)), 113.7 (2C, CH(Ph)), 117.1 (2C, CH(Ph)), 128.8 (2C, C5(Ph)), 140.9 (2C, C1(Ph)), 148.7 (2C, C3(Ph)). HRMS (MALDI-TOF): C32H52N5O4 (M+H)+ calcd.; 570.4019 observed; 530.3976.
26,29,57,60,65,68,75,78-Octaoxa-1,8,12,16,23,32,39,43,47,54-decaazaheptacyclo-[52.8.8.832,32.13,7.117,21.134,38.148,52]dooctaconta-3(82),4,6,17(81),18,20,34(72),35,37,48(71),49,51-dodecaene (21d). Obtained as the second product in the synthesis of macrobicycle 17d Eluent CH2Cl2–MeOH–NH3(aq) = 100:35:6. Yield 9 mg (6%) of a yellow glassy compound. 1H-NMR (CDCl3) δ 1.79 (br.s, 8H, CCH2C), 2.73 (t, 3J = 6.3 Hz, 8H, CH2NHCH2), 2.76–2.82 (m, 16H, CH2N), 3.16 (t, 3J = 6.1 Hz, 8H, CH2NPh), 3.54–3.62 (m, 40H, CH2O, PhCH2N), 6.45 (d, 3J = 7.8 Hz, 4H, H6(Ph) or H4(Ph)), 6.60 (d, 3Jobs = 6.3 Hz, 4H, H4(Ph) or H6(Ph)), 6.67 (br.s, 4H, H2(Ph)), 7.05 (t, 3J = 7.6 Hz, 4H, H5(Ph)), NH protons were not assigned. 13C-NMR (CDCl3) δ 29.6 (4C, CCH2C), 42.7 (4C, CH2NPh), 48.3 (4C, CH2NHCH2), 53.8 (8C, CH2N), 60.2 (4C, PhCH2N), 70.1 (8C, CH2O), 70.7 (8C, CH2O), 111.1 (4C, CH(Ph)), 113.3 (4C, CH(Ph)), 117.8 (4C, CH(Ph)), 129.0 (4C, C5(Ph)), 140.7 (4C, C1(Ph)), 148.6 (4C, C3(Ph)). MS (MALDI-TOF): C64H103N10O8 (M+H)+ calcd.; 1139.80 observed; 1139.79.
29,32,37,40-Tetraoxa-1,8,12,15,19,26-hexaazatetracyclo[24.8.8.13,7.120,24]tetratetraconta-3(44),4,6,20 (43),21,23-hexaene (17f). Obtained from compound 8 (0.25 mmol, 150 mg), tetraamine 10f (0.25 mmol, 44 mg) in the presence of Pd(dba)2 (12 mg, 8 mol%), BINAP (14 mg, 9 mol%), NaOt-Bu (0.75 mmol, 72 mg) in abs. dioxane (12 mL). Eluent CH2Cl2–MeOH–NH3(aq) = 100:25:5–100:35:6. Yield 15 mg (10%) of a yellow glassy compound. 1H-NMR (CDCl3) δ 1.77 (quintet, 3J = 5.7 Hz, 4H, CCH2C), 2.58–2.85 (m, 16H, CH2N), 3.15 (t, 3J = 5.9 Hz, 4H, CH2NPh), 3.45-3.70 (m, 20H, CH2O, PhCH2N), 6.44 (d, 3J = 7.9 Hz, 2H, H6(Ph) or H4(Ph)), 6.56 (d, 3J = 7.2 Hz, 2H, H4(Ph) or H6(Ph)), 6.82 (br.s, 2H, H2(Ph)), 7.05 (t, 3J = 7.7 Hz, 2H, H5(Ph)), NH protons were not assigned. 13C-NMR (CDCl3) δ 28.8 (2C, CCH2C), 42.7 (2C, CH2NPh), 47.9 (2C, CH2NHCH2), 48.2 (2C, CH2NHCH2), 54.5 (4C, CH2N), 60.0 (2C, PhCH2N), 69.8 (4C, CH2O), 70.6 (4C, CH2O), 110.5 (2C, CH(Ph)), 113.7 (2C, CH(Ph)), 117.4 (2C, CH(Ph)), 128.9 (2C, C5(Ph)), 139.5 (2C, C1(Ph)), 145.8 (2C, C3(Ph)). HRMS (MALDI-TOF): C34H57N6O4 (M+H)+ calcd.; 613.4441 observed; 613.4412.
29,32,63,66,71,74,81,84-Octaoxa-1,8,12,15,19,26,35,42,46,49,53,60-dodecaazaheptacyclo-[58.8.8.826,35.13,7.120,24.137,41.154,58]octaoctaconta-3(88),4,6,20(87),21,23,37(78),38,40,54(77),55,57-dodecaene (21f). Obtained as the second product in the synthesis of macrobicycle 17d. Eluent CH2Cl2–MeOH–NH3(aq) = 100:35:6. Yield 8 mg (5%) of a yellow glassy compound. 1H-NMR (CDCl3) δ 1.76 (br.s, 8H, CCH2C), 2.68–2.81 (m, 32H, CH2N), 3.15 (br.s, 8H, CH2NPh), 3.50–3.63 (m, 40H, CH2O, PhCH2N), 6.43 (d, 3Jobs = 7.2 Hz, 4H, H6(Ph) or H4(Ph)), 6.60 (br.s, 4H, H2(Ph)), 6.62 (d, 3J = 8.0 Hz, 4H, H4(Ph) or H6(Ph)), 7.05 (t, 3J = 7.5 Hz, 4H, H5(Ph)), NH protons were not assigned. MS (MALDI-TOF): C68H113N12O8 (M+H)+ calcd.; 1225.88 observed; 1125.90.
11,14,17,31,34,40-Hexaoxa-1,8,21,28-tetraazatetracyclo[26.8.6.13,7.122,26]tetratetraconta-3(44),4,6,22 (43),23,25-hexaene (17h). Obtained from compound 8 (0.25 mmol, 150 mg), dioxadiamine 10h (0.25 mmol, 37 mg) in the presence of Pd(dba)2 (12 mg, 8 mol%), BINAP (14 mg, 9 mol%), NaOt-Bu (0.75 mmol, 72 mg) in abs. dioxane (12 mL). Eluent CH2Cl2–MeOH = 10:1–3:1. Yield 83 mg (57%) of a yellowish glassy compound. 1H-NMR (CDCl3) δ 2.73 (br.s, 8H, CH2N), 3.23 (t, 3J = 5.1 Hz, 4H, CH2NPh), 3.34 (br.s, 4H, CH2O), 3.51 (br.s, 8H, CH2O), 3.58 (t, 3J = 4.9 Hz, 8H, CH2O), 3.62 (s, 4H, PhCH2N), 3.68 (t, 3J = 5.1 Hz, 4H, CH2O), 6.45 (br.s, 4H, H(Ph)), 6.47 (d, 3J = 8.0 Hz, 2H, H(Ph)), 7.07 (t, 3J = 7.8 Hz, 2H, H5(Ph)), NH protons were not assigned. 13C-NMR (CDCl3) δ 43.6 (2C, CH2NPh), 52.8 (4C, CH2N), 59.6 (2C, PhCH2N), 67.3 (4C, CH2O), 68.3 (4C, CH2O), 69.4 (2C, CH2O), 70.2 (2C, CH2O), 110.9 (2C, CH(Ph)), 115.8 (2C, CH(Ph)), 118.0 (2C, CH(Ph)), 129.3 (2C, C5(Ph)), 138.1 (2C, C1(Ph)), 149.0 (2C, C3(Ph)). HRMS (ESI-TOF): C32H51N4O6 (M+H)+ calcd.; 587.3809 observed; 587.3815.
12,17,31,34,39,42-Hexaoxa-1,8,21,28-tetraazatetracyclo[26.8.8.13,7.122,26]hexatetraconta-3(46),4,6,22 (45),23,25-hexaene (17i). Obtained from compound 8 (0.25 mmol, 150 mg), dioxadiamine 10i (0.25 mmol, 51 mg) in the presence of Pd(dba)2 (12 mg, 8 mol%), BINAP (14 mg, 9 mol%), NaOt-Bu (0.75 mmol, 72 mg) in abs. dioxane (12 mL). Eluent CH2Cl2–MeOH = 3:1, CH2Cl2–MeOH–NH3(aq) = 100:20:1. Yield 45 mg (28%) as a yellowish glassy compound. 1H-NMR (CDCl3) δ 1.65–1.71 (m, 4H, CCH2CH2C), 1.87 (quintet, 3J = 6.0 Hz, 4H, CCH2C), 2.77 (t, 3J = 5.2 Hz, 8H, CH2N), 3.21 (t, 3J = 6.3 Hz, 4H, CH2NPh), 3.42–3.48 (m, 4H, CH2O), 3.54 (t, 3J = 5.8 Hz, 4H, CH2O), 3.56-3.64 (m, 20H, CH2O, PhCH2N), 6.44 (dd, 3J = 8.0 Hz, 4J = 1.8 Hz, 2H, H6(Ph) or H4(Ph)), 6.56 (d, 3Jobs = 7.0 Hz, 2H, H4(Ph) or H6(Ph)), 6.78 (br.s, 2H, H2(Ph)), 7.06 (t, 3J = 7.7 Hz, 2H, H5(Ph)), NH protons were not assigned. 13C-NMR (CDCl3) δ 26.7 (2C, CCH2CH2C), 29.3 (2C, CCH2C), 42.0 (2C, CH2NPh), 54.4 (4C, CH2N), 60.1 (2C, PhCH2N), 69.5 (2C, CH2O), 70.0 (4C, CH2O), 70.7 (2C, CH2O), 70.8 (4C, CH2O), 110.3 (2C, CH(Ph)), 113.6 (2C, CH(Ph)), 117.2 (2C, CH(Ph)), 128.8 (2C, C5(Ph)), 140.9 (2C, C1(Ph)), 148.8 (2C, C3(Ph)). HRMS (MALDI-TOF): C36H59N4O6 (M+H)+ calcd.; 643.4435 observed; 643.4479.
12,15,18,32,35,40,43-Heptaoxa-1,8,22,29-tetraazatetracyclo[27.8.8.13,7.123,27]heptatetraconta-3(47), 4,6,23(46),24,26-hexaene (17k). Obtained from compound 8 (0.25 mmol, 150 mg), trioxadiamine 10k (0.25 mmol, 55 mg) in the presence of Pd(dba)2 (12 mg, 8 mol%), BINAP (14 mg, 9 mol%), NaOt-Bu (0.75 mmol, 72 mg) in abs. dioxane (12 mL). Eluent CH2Cl2–MeOH = 10:1. Yield 57 mg (35%) of a yellowish glassy compound. 1H-NMR (CDCl3) δ 1.82 (quintet, 3J = 6.0 Hz, 4H, CCH2C), 2.92 (br.s, 8H, CH2N), 3.18 (t, 3J = 6.4 Hz, 4H, CH2NPh), 3.54–3.60 (m, 12H, CH2O), PhCH2N), 3.62–3.67 (m, 12H, CH2O), 3.76 (br.s, 4H, CH2O), 4.18 (br.s, 2H, NH), 6.48 (d, 3Jobs = 7.2 Hz, 2H, H4(Ph) or H6(Ph)), 6.61 (d, 3Jobs = 7.2 Hz, 2H, H6(Ph) or H4(Ph)), 6.75 (br.s, 2H, H2(Ph)), 7.05 (t, 3J = 7.7 Hz, 2H, H5(Ph)). 13C-NMR (CDCl3) δ 29.0 (2C, CCH2C), 41.4 (2C, CH2NPh), 52.9 (2C, CH2N), 54.0 (2C, CH2N), 59.8 (2C, PhCH2N), 67.3 (4C, CH2O), 68.5 (4C, CH2O), 69.5 (2C, CH2O), 70.1 (2C, CH2O), 70.5 (2C, CH2O), 110.8 (2C, CH(Ph)), 115.1 (2C, CH(Ph)), 117.4 (2C, CH(Ph)), 129.3 (2C, C5(Ph)), 138.2 (2C, C1(Ph)), 149.0 (2C, C3(Ph)). HRMS (ESI-TOF): C36H59N4O7 (M+H)+ calcd.; 659.4384 observed; 659.4389.
12,15,18,32,35,49,52,55,69,72,77,80,87,90-Tetradecaoxa-1,8,22,29,38,45,59,66-octaazaheptacyclo-[64.8.8.829,38.13,7.123,27.140,44.160,64]tetranonaconta-3(94),4,6,23(93),24,26,40(84),41,43,60(83),61,63-dodecaene (21k). Obtained as the second product in the synthesis of macrobicycle 17k. Eluent CH2Cl2–MeOH = 10:3. Yield 28 mg (17%) of a yellow glassy compound. 1H-NMR (CDCl3) δ 1.83 (quintet, 3J = 5.7 Hz, 8H, CCH2C), 2.81 (br.s, 16H, CH2N), 3.18 (t, 3J = 6.3 Hz, 8H, CH2NPh), 3.50–3.68 (m, 64H, CH2O, PhCH2N), 4.56 (br.s, 4H, NH), 6.46 (d, 3J = 7.8 Hz, 4H, H4(Ph) or H6(Ph)), 6.55 (d, 3Jobs = 7.0 Hz, 4H, H6(Ph) or H4(Ph)), 6.73 (s, 4H, H2(Ph)), 7.05 (t, 3Jobs = 7.5 Hz, 4H, H5(Ph)). MS (MALDI-TOF): C72H117N8O14 (M+H)+ calcd.; 1317.87 observed; 1317.84.
10,13,25,28,33,36-Hexaoxa-1,7,16,22-tetraazatetracyclo[20.8.8.23,6.217,20]dotetraconta-3,5,17,19,39,41-hexaene (18h). Obtained from compound 9 (0.25 mmol, 150 mg), dioxadiamine 10h (0.25 mmol, 37 mg) in the presence of Pd(dba)2 (24 mg, 16 mol%), BINAP (28 mg, 18 mol%), NaOt-Bu (0.75 mmol, 72 mg) in abs. dioxane (12 mL). Eluent CH2Cl2–MeOH = 3:1, CH2Cl2–MeOH–NH3(aq) = 100:20:1–100:20:3. Yield 37 mg (25%) of a yellowish glassy compound. 1H-NMR (CDCl3) δ 2.68 (t, 3J = 5.4 Hz, 8H, CH2N), 3.27 (t, 3J = 4.8 Hz, 4H, CH2NPh), 3.53 (s, 4H, CH2O), 3.59 (t, 3J = 5.4 Hz, 8H, CH2O), 3.62 (s, 8H, CH2O), 3.65 (s, 4H, PhCH2N), 3.73 (t, 3J = 4.8 Hz, 4H, CH2O), 6.55 (d, 3Jobs = 8.5 Hz, 4H, H3(Ph), H3'(Ph)), 7.17 (d, 3Jobs = 8.5 Hz, 4H, H2(Ph), H2'(Ph)), NH protons were not assigned. 13C-NMR (CDCl3) δ 43.7 (2C, CH2NPh), 54.9 (4C, CH2N), 59.7 (2C, PhCH2N), 69.4 (2C, CH2O), 69.8 (4C, CH2O), 70.0 (2C, CH2O), 70.8 (4C, CH2O), 113.1 (4C, C3(Ph), C3'(Ph)), 128.8 (2C, C1(Ph)), 129.8 (4C, C2(Ph), C2'(Ph)), 147.2 (2C, C4(Ph)). HRMS (ESI-TOF): C32H51N4O6 (M+H)+ calcd.; 587.3809 observed; 587.3829.
10,13,25,28,40,43,55,58,63,66,75,78-dodecaoxa-1,7,16,22,31,37,46,52-octaazaheptacyclo-[50.8.8.822,31.23,6.217,20.233,36.247,50]tetraoctaconta-3,5,17,19,33,35,47,49,69,71,81,83-dodecaene (22h). Obtained as the second product in the synthesis of macrobicycle 18h. Eluent CH2Cl2–MeOH–NH3(aq) = 100:25:5. Yield 15 mg (10%) of a yellow glassy compound. 1H-NMR (CDCl3) δ 2.77 (br.s, 16H, CH2N), 3.26 (br.s, 8H, CH2NPh), 3.58 (br.s, 40H, CH2O), 3.63 (s, 8H, PhCH2N), 3.68 (br.s, 8H, CH2O), 3.95 (br.s, 4H, NH), 6.54 (d, 3Jobs = 8.2 Hz, 8H, H3(Ph), H3'(Ph)), 7.09 (d, 3Jobs = 8.2 Hz, 8H, H2(Ph), H2'(Ph)). HRMS (MALDI-TOF): C64H101N8O12 (M+H)+ calcd.; 1173.7538 observed; 1173.7472.
11,14,17,30,33,38,41-Heptaoxa-1,7,21,27-tetraazatetracyclo[25.8.8.23,6.222,25]heptatetraconta-3,5,22, 24,44,46-hexaene (18k). Obtained from compound 9 (0.25 mmol, 150 mg), trioxadiamine 10k (0.25 mmol, 55 mg) in the presence of Pd(dba)2 (24 mg, 16 mol%), BINAP (28 mg, 18 mol%), NaOt-Bu (0.75 mmol, 72 mg) in abs. dioxane (12 mL). Eluent CH2Cl2–MeOH = 10:1, 3:1. Yield 59 mg (36%) as a yellowish glassy compound. 1H-NMR (CDCl3) δ 1.80 (br.s, 4H, CCH2C), 2.70 (br.s, 8H, CH2N), 3.13 (br.s, 4H, CH2NPh), 3.34 (br.s, 4H, CH2O), 3.50 (br.s, 8H, CH2O), 3.58 (s, 8H, CH2O), 3.63–3.69 (m, 12H, CH2O, PhCH2N), 6.50 (d, 3Jobs = 8.1 Hz, 4H, H3(Ph), H3'(Ph)), 6.79 (d, 3Jobs = 8.1 Hz, 4H, H2(Ph), H2'(Ph)), NH protons were not assigned. 13C-NMR (CDCl3) δ 28.9 (2C, CCH2C), 41.8 (2C, CH2NPh), 52.2 (4C, CH2N), 58.6 (2C, PhCH2N), 66.8 (4C, CH2O), 68.5 (4C, CH2O), 69.7 (2C, CH2O), 70.2 (2C, CH2O), 70.4 (2C, CH2O), 112.7 (4C, C3(Ph), C3'(Ph)), 124.5 (2C, C1(Ph)), 130.5 (4C, C2(Ph), C2'(Ph)), 148.4 (2C, C4(Ph)). HRMS (ESI-TOF): C36H59N4O7 (M+H)+ calcd.; 659.4384 observed; 659.4375.
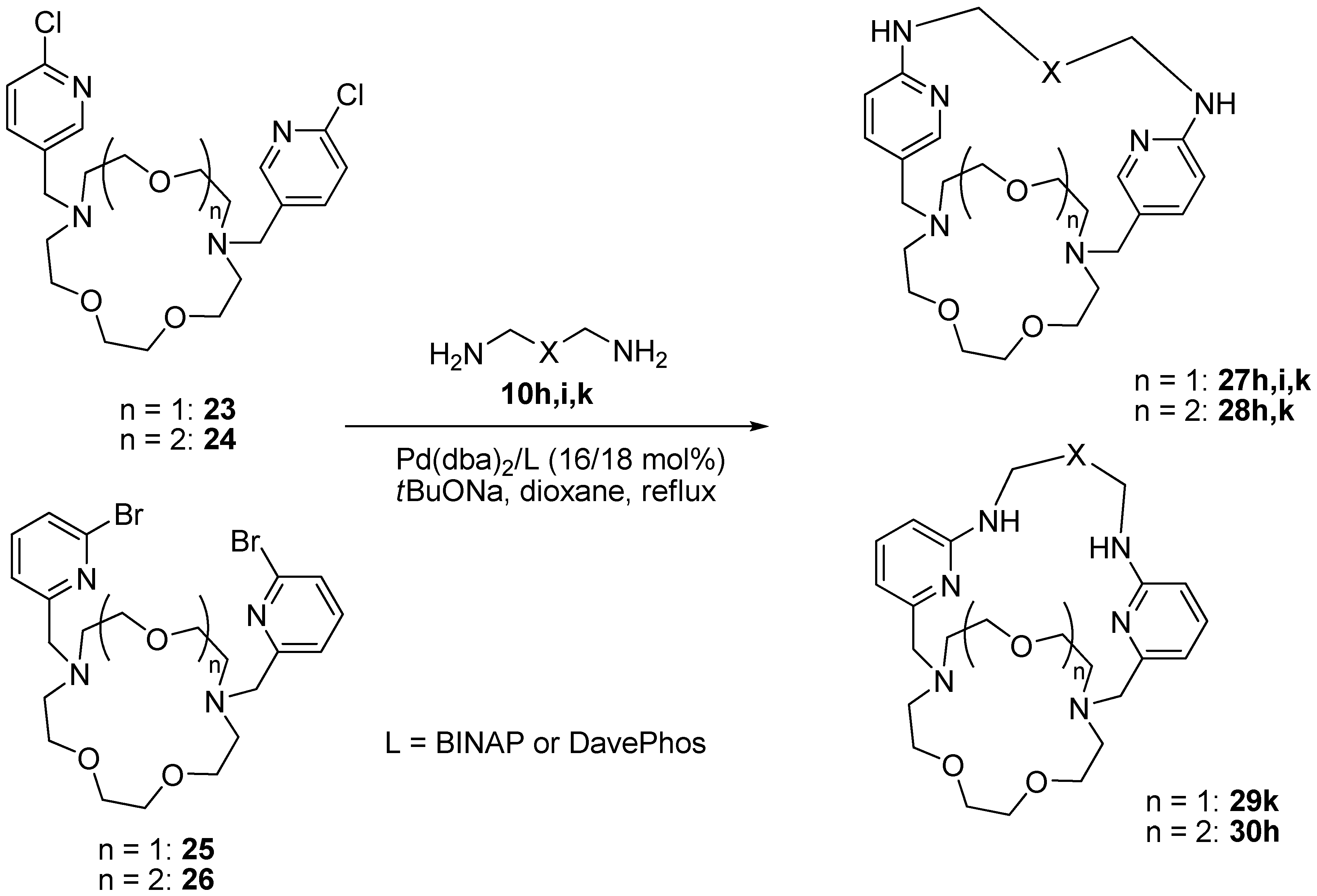
10,13,25,28,33-Pentaoxa-1,5,7,16,18,22-hexaazatetracyclo[20.8.5.23,6.217,20]nonatriaconta-3,5,17,19,36,38-hexaene (27h). Obtained from compound 23 (0.5 mmol, 235 mg), dioxadiamine 10h (0.5 mmol, 74 mg) in the presence of Pd(dba)2 (46 mg, 16 mol%), DavePhos (36 mg, 18 mol%), NaOt-Bu (1.5 mmol, 144 mg) in abs. Dioxane (25 mL). Eluent CH2Cl2–MeOH–NH3(aq) = 100:20:2. Yield 60 mg (22%) of a yellow glassy compound. 1H-NMR (CDCl3) δ 2.56 (t, 3J = 5.1 Hz, 4H, CH2N), 2.66 (t, 3J = 4.6 Hz, 4H, CH2N), 3.42 (br.s, 4H, CH2NPy), 3.52 (t, 3J = 5.1 Hz, 4H, CH2O), 3.55–3.62 (m, 8H, CH2O), 3.66 (s, 4H, CH2O or PyCH2N), 3.67 (s, 4H, PyCH2N or CH2O), 3.72 (t, 3J = 5.1 Hz, 4H, CH2O), 5.11 (br.s, NH), 6.30 (d, 3J = 8.5 Hz, 2H, H5(Py)), 7.47 (d, 3J = 8.5 Hz, 2H, H6(Py)), 7.94 (s, 2H, H2(Py)). 13C-NMR (CDCl3) δ 41.6 (2C, CH2NPy), 54.7 (2C, CH2N), 55.2 (2C, CH2N), 57.4 (2C, PyCH2N), 69.6 (4C, CH2O), 69.7 (2C, CH2O), 70.0 (2C, CH2O), 70.8 (2C, CH2O), 108.3 (2C, C5(Py)), 123.9 (2C, C1(Py)), 138.6 (2C, C6(Py)), 147.4 (2C, C2(Py)), 158.0 (2C, C4(Py)). HRMS (MALDI-TOF): C28H45N6O5 (M+H)+ calcd.; 545.3451 observed; 545.3480.
10,15,27,30,35-Pentaoxa-1,5,7,18,20,24-hexaazatetracyclo[22.8.5.23,6.219,22]hentetraconta-3,5,19,21, 38,40-hexaene (27i). Obtained from compound 23 (0.25 mmol, 117 mg), dioxadiamine 10i (0.25 mmol, 51 mg) in the presence of Pd(dba)2 (23 mg, 16 mol%), DavePhos (18 mg, 18 mol%), NaOt-Bu (0.75 mmol, 72 mg) in abs. dioxane (12 mL). Eluent CH2Cl2–MeOH–NH3(aq) = 100:20:1–100:20:3. Yield 17 mg (11%) of a yellow glassy compound. 1H-NMR (CDCl3) δ 1.63–1.70 (m, 4H, CCH2CH2C), 1.87 (quintet, 3J = 5.5 Hz, 4H, CCH2C), 2.71 (br.s, 8H, CH2N), 3.39 (t, 3J = 5.2 Hz, 4H, CH2NPy), 3.42–3.48 (m, 4H, CH2O), 3.50–3.64 (m, 20H, CH2O, PyCH2N), 5.26 (br.s, 2H, NH), 6.35 (d, 3J = 8.5 Hz, 2H, H5(Py)), 7.58 (d, 3J = 8.5 Hz, 2H, H6(Py)), 7.91 (br.s, 2H, H2(Py)). 13C-NMR (CDCl3) δ 26.8 (2C, CCH2CH2C), 29.3 (2C, CCH2C), 40.7 (2C, CH2NPy), 54.4 (2C, CH2N), 54.5 (2C, CH2N), 57.4 (2C, PyCH2N), 69.0 (2C, CH2O), 69.5 (4C, CH2O), 70.6 (2C, CH2O), 71.0 (2C, CH2O), 107.1 (2C, C5(Py)), 122.5 (2C, C1(Py)), 139.4 (2C, C6(Py)), 147.4 (2C, C2(Py)), 158.1 (2C, C4(Py)). HRMS (MALDI-TOF): C32H53N6O5 (M+H)+ calcd.; 601.4077 observed; 601.4043.
11,14,17,30,33,38-Hexaoxa-1,5,7,21,23,27-hexaazatetracyclo[25.8.5.23,6.222,25]tetratetraconta-3,5,22, 24,41,43-hexaene (27k). Obtained from compound 23 (0.25 mmol, 117 mg), trioxadiamine 10k (0.25 mmol, 55 mg) in the presence of Pd(dba)2 (23 mg, 16 mol%), DavePhos (18 mg, 18 mol%), NaOt-Bu (0.75 mmol, 72 mg) in abs. dioxane (12 mL). Eluent CH2Cl2–MeOH = 5:1, CH2Cl2–MeOH–NH3(aq) = 100:20:1–100:20:3. Yield 14 mg (9%) of a yellow glassy compound. 1H-NMR (CDCl3) δ 1.87 (quintet, 3J = 5.9 Hz, 4H, CCH2C), 2.71 (t, 3J = 5.3 Hz, 4H, CH2N), 2.77 (t, 3J = 4.7 Hz, 4H, CH2N), 3.39 (br.s, 4H, CH2NPy), 3.51-3.70 (m, 28H, CH2O, PyCH2N), 5.64 (br.s, 2H, NH), 6.32 (d, 3J = 8.6 Hz, 2H, H5(Py)), 7.54 (d, 3J = 8.6 Hz, 2H, H6(Py)), 7.90 (br.s, 2H, H2(Py)). 13C-NMR (CDCl3) δ 29.0 (2C, CCH2C), 40.0 (2C, CH2NPy), 54.1 (2C, CH2N), 54.3 (2C, CH2N), 57.2 (2C, PyCH2N), 68.7 (2C, CH2O), 69.2 (2C, CH2O), 69.4 (2C, CH2O), 70.1 (2C, CH2O), 70.5 (2C, CH2O), 70.6 (2C, CH2O), 107.5 (2C, C5(Py)), 123.0 (2C, C1(Py)), 139.9 (2C, C6(Py)), 146.6 (2C, C2(Py)), 157.7 (2C, C4(Py)). HRMS (MALDI-TOF): C32H53N6O6 (M+H)+ calcd.; 617.4027 observed; 617.3967.
10,13,25,28,33,36-Hexaoxa-1,5,7,16,18,22-hexaazatetracyclo[20.8.8.23,6.217,20]dotetraconta-3,5,17,19,39,41-hexaene (28h). Obtained from compound 24 (0.25 mmol, 128 mg), dioxadiamine 10h (0.25 mmol, 37 mg) in the presence of Pd(dba)2 (23 mg, 16 mol%), DavePhos (18 mg, 18 mol%), NaOt-Bu (0.75 mmol, 72 mg) in abs. dioxane (12 mL). Eluent CH2Cl2–MeOH–NH3(aq) = 100:20:3. Yield 7 mg (5%) as a yellow glassy compound. 1H-NMR (CDCl3) δ 2.70 (br.s, 8H, CH2N), 3.47–3.54 (m, 8H, CH2NPy, CH2O), 3.55–3.61 (m, 16H, CH2O), 3.67 (s, 4H, PyCH2N), 3.73 (t, 3J = 4.7 Hz, 4H, CH2O), 5.61 (br.s, 2H, NH), 6.37 (d, 3J = 8.2 Hz, 2H, H6(Py)), 7.52 (d, 3J = 8.2 Hz, 2H, H5(Py)), 7.97 (br.s, 2H, H2(Py)). HRMS (ESI-TOF): C30H49N6O6 (M+H)+ calcd.; 589.3713 observed; 589.3715.
11,14,17,30,33,38,41-Heptaoxa-1,5,7,21,23,27-hexaazatetracyclo[25.8.8.23,6.222,25]heptatetraconta-3,5,22,24,44,46-hexaene (28k). Obtained from compound 24 (0.25 mmol, 128 mg), trioxadiamine 10k (0.25 mmol, 55 mg) in the presence of Pd(dba)2 (23 mg, 16 mol%), DavePhos (18 mg, 18 mol%), NaOt-Bu (0.75 mmol, 72 mg) in abs. dioxane (12 mL). Eluent CH2Cl2–MeOH–NH3(aq) = 100:20:1–100:20:2. Yield 40 mg (24%) of a yellow glassy compound. 1H-NMR (CDCl3) δ 1.87 (quintet, 3J = 6.0 Hz, 4H, CCH2C), 2.69 (t, 3J = 5.5 Hz, 4H, CH2N), 2.79 (t, 3J = 4.6 Hz, 4H, CH2N), 3.38 (q, 3J = 6.1 Hz, 4H, CH2NPy), 3.52-3.69 (m, 32H, CH2O, PyCH2N), 5.20 (br.s, 2H, NH), 6.32 (d, 3J = 8.6 Hz, 2H, H5(Py)), 7.47 (dd, 3J = 8.6 Hz, 4J = 2.0 Hz, 2H, H6(Py)), 7.89 (d, 4J = 2.0 Hz, 2H, H2(Py)). 13C-NMR (CDCl3) δ 29.1 (2C, CCH2C), 39.8 (2C, CH2NPy), 54.1 (4C, CH2N), 56.8 (2C, PyCH2N), 69.4 (2C, CH2O), 69.9 (4C, CH2O), 70.2 (4C, CH2O), 70.5 (2C, CH2O), 70.8 (2C, CH2O), 107.1 (2C, C5(Py)), 123.1 (2C, C1(Py)), 138.8 (2C, C6(Py)), 147.7 (2C, C2(Py)), 158.2 (2C, C4(Py)). HRMS (ESI-TOF): C34H57N6O7 (M+H)+ calcd.; 661.4288 observed; 661.4241.
12,15,18,32,35,40-Hexaoxa-1,8,22,29,43,44-hexaazatetracyclo[27.8.5.13,7.123,27]tetratetraconta-3(44), 4,6,23(43),24,26-hexaene (29k). Obtained from compound 25 (0.25 mmol, 140 mg), trioxadiamine 10k (0.25 mmol, 55 mg) in the presence of Pd(dba)2 (23 mg, 16 mol%), BINAP (28 mg, 18 mol%), NaOt-Bu (0.75 mmol, 72 mg) in abs. dioxane (12 mL). Eluent CH2Cl2–MeOH = 3:1. Yield 21 mg (12%) of a yellow glassy compound. 1H-NMR (CDCl3) δ 2.02 (br.s, 4H, CCH2C), 2.60–2.88 (m, 8H, CH2N), 3.15 (q, 3J = 5.6 Hz, 4H, CH2NPy), 3.28 (t, 3J = 5.6 Hz, 4H, CH2O), 3.31–3.38 (m, 4H, CH2O), 3.40–3.65 (m, 20H, CH2O, PyCH2N), 5.82 (t, 3J = 4.8 Hz, 2H, NH), 6.41 (d, 3J = 8.5 Hz, 2H, H4(Py) or H6(Py)), 6.51 (d, 3J = 7.1 Hz, 2H, H6(Py) or H4(Py)), 7.45 (dd, 3J = 8.5 Hz, 3J = 7.1 Hz, 2H, H5(Py)). 13C-NMR (CDCl3) δ 28.8 (2C, CCH2C), 39.3 (2C, CH2NPy), 53.8 (2C, CH2N), 56.1 (2C, CH2N), 61.9 (2C, PyCH2N), 66.8 (2C, CH2O), 66.9 (2C, CH2O), 67.5 (2C, CH2O), 69.0 (2C, CH2O), 70.3 (2C, CH2O), 70.6 (2C, CH2O), 105.1 (2C, C4(Py) or C6(Py)), 112.5 (2C, C6(Py) or C4(Py)), 139.3 (2C, C5(Py)), 156.4 (2C, C1(Py)), 160.1 (2C, C3(Py)). HRMS (MALDI-TOF): C32H52N6NaO6 (M+Na)+ calcd.; 639.3846 observed; 639.3803.
11,14,27,30,35,38-Hexaoxa-1,8,17,24,41,42-hexaazatetracyclo[22.8.8.13,7.118,22]dotetraconta-3(42),4, 6,18(41),19,21-hexaene (30h). Obtained from compound 26 (0.227 mmol, 137 mg), dioxadiamine 10h (0.23 mmol, 34 mg) in the presence of Pd(dba)2 (21 mg, 16 mol%), DavePhos (16 mg, 18 mol%), NaOt-Bu (0.75 mmol, 72 mg) in abs. dioxane (12 mL). Eluent CH2Cl2–MeOH = 3:1. Yield 21 mg (16%) of a yellow glassy compound. 1H-NMR (CDCl3) δ 2.64 (br.s, 8H,CH2N), 3.03 (br.s, 4H, CH2NPy), 3.30 (s, 4H, CH2O), 3.33–3.38 (m, 8H, CH2O), 3.47 (t, 3J = 4.5 Hz, 4H, CH2O), 3.50–3.68 (m, 12H, CH2O, PyCH2N), 6.42 (d, 3J = 8.5 Hz, 2H, H4(Py) or H6(Py)), 6.51 (d, 3J = 7.2 Hz, 2H, H6(Py) or H4(Py)), 7.46 (t, 3J = 7.8 Hz, 2H, H5(Py)), NH protons were not assigned. 13C-NMR (CDCl3) δ 42.5 (2C, CH2NPy), 52.3 (4C, CH2N), 60.4 (2C, PyCH2N), 67.1 (4C, CH2O), 67.3 (4C, CH2O), 68.4 (2C, CH2O), 69.8 (2C, CH2O), 105.4 (2C, C4(Py) or C6(Py)), 112.8 (2C, C6(Py) or C4(Py)), 138.8 (2C, C5(Py)), 158.2 (2C, C1(Py)), 161.7 (2C, C3(Py)). HRMS (MALDI-TOF): C30H49N6O6 (M+H)+ calcd.; 589.3714 observed; 589.3675.

 10h
10h 10j
10j 10k
10k 10h
10h 10j
10j 10k
10k
 (10a)
(10a) (10b)
(10b) (10c)
(10c) (10d)
(10d) (10e)
(10e) (10f)
(10f) (10g)
(10g) (10h)
(10h) (10i)
(10i) (10k)
(10k) (10k)
(10k) (10k)
(10k) (10k)
(10k) (10i)
(10i) (10h)
(10h) (10d)
(10d) (10f)
(10f) (10h)
(10h) (10i)
(10i) (10k)
(10k) (10h)
(10h) (10k)
(10k)
 (10h)
(10h) (10i)
(10i) (10k)
(10k) (10h)
(10h) (10k)
(10k) (10k)
(10k) (10h)
(10h) (10h)
(10h)





