Study on the Mechanism of Intestinal Absorption of Epimedins A, B and C in the Caco-2 Cell Model
Abstract
:1. Introduction
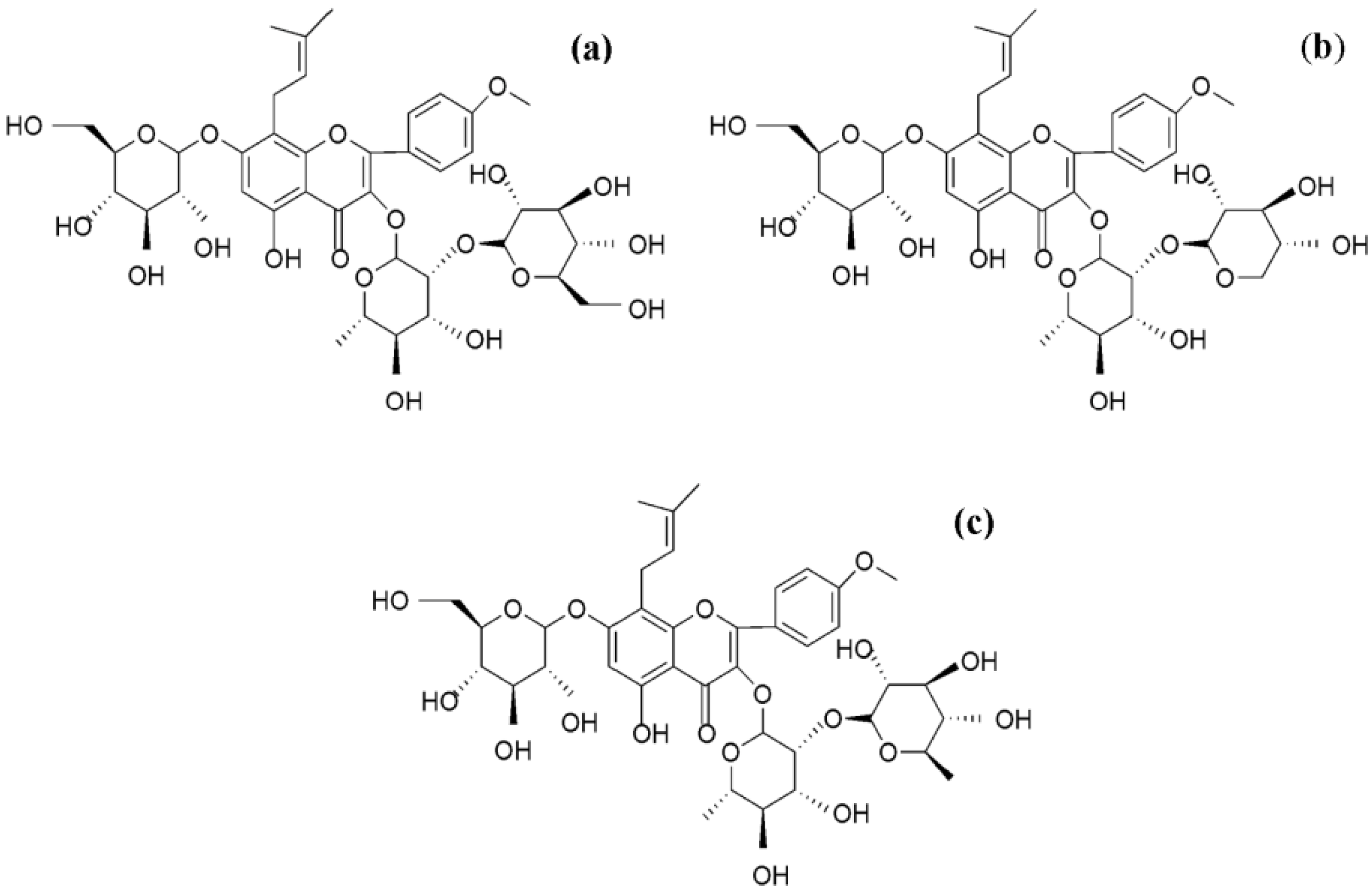
2. Results and Discussion
2.1. Cell Viability Assay
2.2. Transport of Epimedins A, B, and C at Different Concentrations


2.3. Effect of Inhibitors on the Absorption of Epimedins A, B, and C
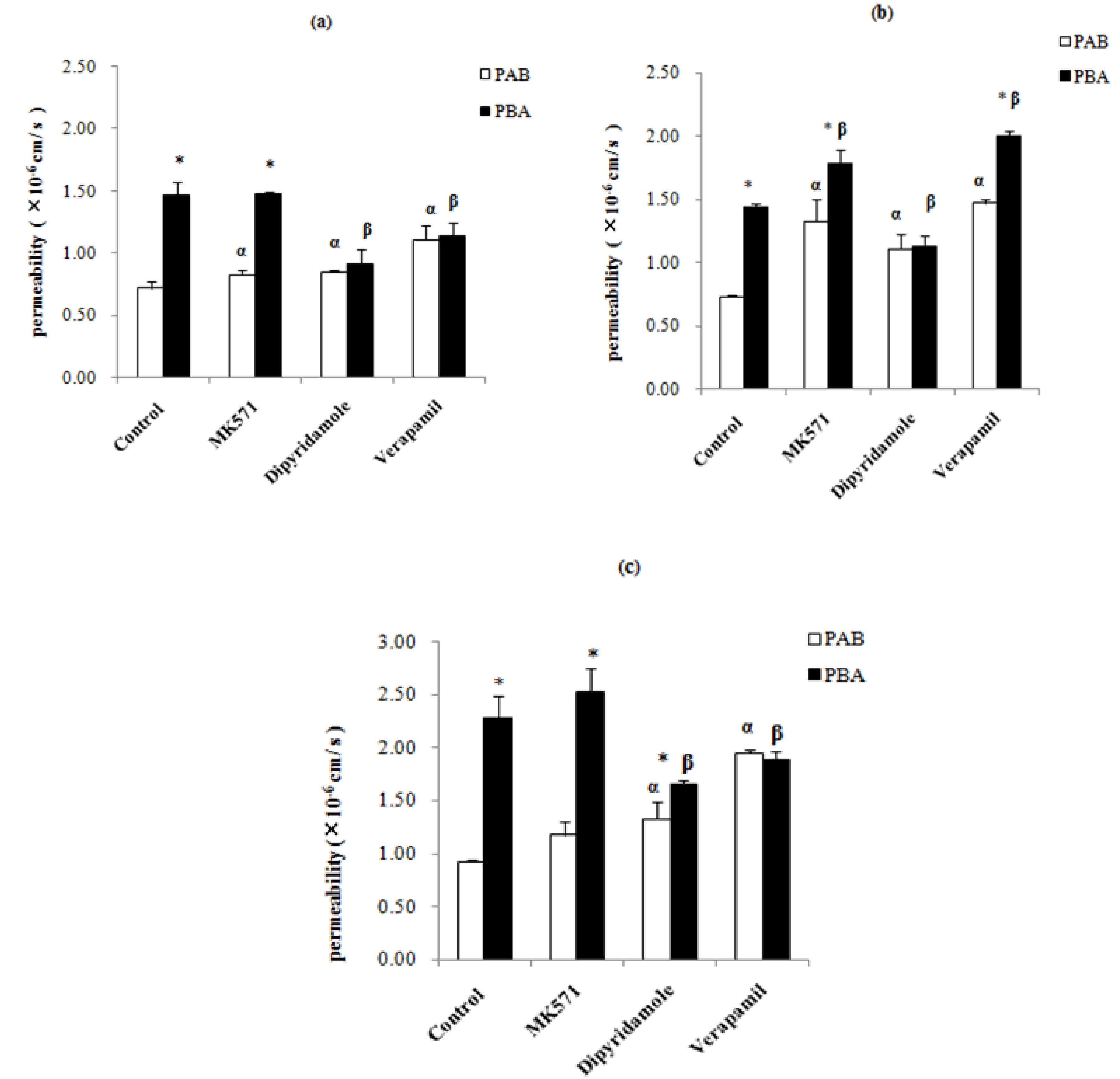
3. Experimental
3.1. Materials and Reagents
3.2. Cell viability Assay
3.3. Cell Culture
3.4. Sample Preparation
3.5. Transport Experiments through Caco-2 Cell Culture Model
3.6. UPLC Analysis of Transport Samples
3.7. Data Analysis
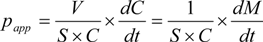
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wu, Y.T.; Lin, C.W.; Lin, L.C.; Chiu, A.W.; Chen, K.K.; Tsai, T.H. Analysis of biliary excretion of icariin in rats. J. Agric. Food Chem. 2010, 58, 9905–9911. [Google Scholar]
- Meng, F.H.; Li, Y.B.; Xiong, Z.L.; Jiang, Z.M.; Li, F.M. Osteoblastic proliferative activity of Epimedium brevicornum Maxim. Phytomedicine 2005, 12, 189–193. [Google Scholar]
- Wang, Y.K.; Huang, Z.Q. Protective effects of icariin on human umbilical vein endothelial cell injury induced by H2O2 in vitro. Pharmacol. Res. 2005, 52, 174–182. [Google Scholar]
- An, D.N.; Victoria, P.; Stuart, M.; Denis, D.K. Estrogenic activity of a polyphenolic extract of the leaves of epimedium brevicornum. Fitoterapia 2005, 76, 35–40. [Google Scholar]
- Zhang, H.F.; Yang, T.S.; Li, Z.Z.; Wang, Y. Simultaneous extraction of epimedin A, B, C and icariin from herba epimedii by ultrasonic technique. Ultrason. Sonochem. 2008, 15, 376–385. [Google Scholar]
- Lin, C.C.; Ng, L.T.; Hsu, F.F.; Shieh, D.E.; Chiang, L.C. Cytotoxic effects of Coptis chinensis and Epimedium sagittatum extracts and their major constituents (berberine, coptisine and icariin) on hepatoma and leukaemia cell growth. Clin. Exp. Pharmacol. Physiol. 2004, 31, 65–69. [Google Scholar] [CrossRef]
- Liu, T.Z.; Chen, C.Y.; Yiin, S.J.; Chen, C.H.; Cheng, J.T.; Shih, M.K.; Wang, Y.S.; Chern, C.L. Molecular mechanism of cell cycle blockage of hepatoma SK-Hep-1 cells by Epimedin C through suppression of mitogen-activated protein kinase activation and increased expression of CDK inhibitors p21(Cip1) and p27(Kip1). Food Chem. Toxicol. 2006, 44, 227–235. [Google Scholar] [CrossRef]
- Yap, S.P.; Shen, P.; Butler, M.S.; Gong, Y.; Loy, C.J.; Yong, E.L. New estrogenic prenylflavone from Epimedium brevicornum inhibits the growth of breast cancer cells. Planta Med. 2005, 71, 114–119. [Google Scholar] [CrossRef]
- Chen, Y.; Zhao, Y.H.; Jia, X.B.; Hu, M. Intestinal absorption mechanisms of prenylated flavonoids present in the heat-processed Epimedium. koreanum Nakai. (Yin Yanghuo). Pharm. Res. 2008, 25, 2190–2199. [Google Scholar] [CrossRef]
- Peterson, M.D.; Mooseker, M.S. Characterization of the enterocyte-like brush border cytoskeleton of the C2BBe clones of the human intestinal cell line, Caco-2. J. Cell Sci. 1992, 102, 581–600. [Google Scholar]
- Li, C.R.; Zhang, L.; Zhou, L.B.; Wo, S.K.; Lin, G.; Zuo, Z. Comparison of intestinal absorption and disposition of structurally similar bioactive flavones in Radix Scutellariae. AAPS J. 2012, 14, 23–33. [Google Scholar]
- Planas, J.M.; Alfaras, I.; Colom, H.M.; Juan, M.E. The bioavailability and distribution of trans-resveratrol are constrained by ABC transporters. J. Arch. Biochem. Biophys. 2012, 527, 67–73. [Google Scholar]
- Fricker, G.; Miller, D.S. Relevance of multidrug resistance proteins for intestinal drug absorption in vitro and in vivo. J. Pharmacol. Toxicol. 2002, 90, 5–13. [Google Scholar]
- Juan, M.E.; González-Pons, E.; Planas, J.M. Multidrug resistance proteins restrain the intestinal absorption of trans-resveratrol in rats. J. Nutr. 2010, 140, 489–495. [Google Scholar]
- Hellwig, M.; Geissler, S.; Matthes, R.; Peto, A.; Silow, C.; Brandsch, M.; Henle, T. Transport of free and peptide-bound glycated amino acids: Synthesis, transepithelial flux at Caco-2 cell monolayers, and interaction with apical membrane transport proteins. ChemBioChem 2011, 12, 1270–1279. [Google Scholar]
- Van de Wetering, K.; Burkon, A.; Feddema, W.; Bot, A.; de Jonge, H.; Somoza, V.; Borst, P. Intestinal breast cancer resistance protein (BCRP)/Bcrp1 and multidrug resistance protein 3 (MRP3)/Mrp3 are involved in the pharmacokinetics of resveratrol. Mol. Pharmacol. 2009, 75, 876–885. [Google Scholar]
- Mease, K.; Sane, R.; Podila, L.; Taub, M.E. Differential selectivity of efflux transporter inhibitors in Caco-2 and MDCK–MDR1 Monolayers: A strategy to assess the interaction of a new chemical entity with P-gp, BCRP, and MRP2. J. Pharm. Sci. 2012, 101, 1888–1897. [Google Scholar]
- Dahan, A.; Sabit, H.; Amidon, G.L. Multiple efflux pumps are involved in the transepithelial transport of colchicine: Combined effect of p-glycoprotein and multidrug resistance-associated protein 2 leads to decreased intestinal absorption throughout the entire small intestine. J. Drug Metab. Dispos. 2009, 37, 2028–2036. [Google Scholar]
- Brand, W.; van der Wel, P.A.; Rein, M.J.; Barron, D.; Williamson, G.; van Bladeren, P.J.; Rietiens, M. Metabolism and transport of the citrus flavonoid hesperetin in Caco-2 cell monolayers. Drug Metab. Dispos. 2008, 36, 1794–1802. [Google Scholar]
- Jeong, E.J.; Liu, Y.; Lin, H.; Hu, M. In situ single-pass perfused rat intestinal model for absorption and metabolism. In Methods in Pharmacology and Toxicology—Optimization in Drug Discovery: In Vitro Methods; Yan, Z., Caldwell, G.W., Eds.; Human Press: Totowa, NJ, USA, 2004; pp. 65–76. [Google Scholar]
- Liang, E.; Proudfoot, J.; Yazdanian, M. Mechanisms of transport and structure-permeability relationship of sulfasalazine and its analogs in Caco-2 cell monolyers. Pharm. Res. 2000, 17, 1168–1174. [Google Scholar]
- Xia, C.Q.; Liu, N.; Yang, D.; Miwa, G.; Gan, L.S. Expression, localization, and functional characteristics of breast cancer resistance protein in Caco-2 cells. Drug Metab. Dispos. 2005, 33, 637–643. [Google Scholar]
- Kobayashi, S.; Shinohara, M.; Nagai, T.; Konishi, Y. Transport mechanisms of soy isoflavones and their microbial metabolites dihydrogenistein and dihydrodaidzein across monolayers and membranes. Pharm. Anal. Acta 2013, 77, 2210–2217. [Google Scholar]
- Schutte, M.E.; Freidig, A.P.; van de Sandt, J.J.; Alink, G.M.; Rietjens, I.M.; Groten, J.P. An in vitro and in silico study on the flavonoid-mediated modulation of the transport of 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine (PhIP) through Caco-2 monolayers. Toxicol. Appl. Pharmacol. 2006, 217, 204–215. [Google Scholar]
- Xue, C.F.; Guo, J.M.; Qian, D.W.; Duan, J.A.; Shu, Y. Absorption of flavonoids from Abelmoschus manihot extract by in situ intestinal perfusion. Yao Xue Xue Bao 2011, 46, 454–459. [Google Scholar]
- Wolfgang, L.; Heidrun, P. Blood-brain barrier active efflux transporters: ATP-binding cassette gene family. NeuroRX 2005, 2, 86–98. [Google Scholar]
- Akao, T.; Hanada, M.; Sakashita, Y.; Sato, K.; Morita, M.; Imanaka, T. Efflux of baicalin, a flavone glucuronide of Scutellariae Radix, on Caco-2 cells through multidrug resistance-associated protein 2. J. Pharm. Pharmacol. 2007, 59, 87–93. [Google Scholar]
- Lin, J.B.; Dou, J.; Xu, J.L.; Haji, A.A. Chemical composition, antimicrobial and antitumor activities of the essential oils and crude extracts of Euphorbia macrorrhiza. Molecules 2012, 17, 5030–5039. [Google Scholar] [CrossRef]
- Hu, M.; Chen, J.; Zhu, Y.; Dantzig, A.H.; Stratford, R.E., Jr.; Kuhfeld, M.T. Mechanism and kinetics of transcellular transport of a new beta -lactam antibiotic loracarbef across an intestinal epithelial membrane model system (Caco-2). Pharm. Res. 1994, 11, 1405–1413. [Google Scholar] [CrossRef]
- Lv, L.Z.; Tong, C.Q.; Lv, Q.; Tang, X.J.; Li, L.M.; Fang, Q.X.; Yu, J.; Han, M.; Gao, J.Q. Enhanced absorption of hydroxysafflor yellow A using a self-double-emulsifying drug delivery system: In vitro and in vivo studies. Int. J. Nanomed. 2012, 7, 4099–4107. [Google Scholar]
- Guan, M.; Zhu, Q.L.; Liu, Y.; Bei, Y.Y.; Gu, Z.L.; Zhang, X.N.; Zhang, Q. Uptake and transport of a novel anticancer drug-delivery system: Lactosyl-norcantharidin-associated N-trimethyl chitosan nanoparticles across intestinal Caco-2 cell monolayers. Int. J. Nanomed. 2012, 7, 1921–1930. [Google Scholar]
- Karamustafa, F. Transport of Alendronate Through Human Intestinal Cell Line Caco-2. In Proceedings of the 33rd Annual Meeting & Exposition of the Controlled Release Society, Vienna, Austria, 22–26 June 2006.
- Lev, B.; Valery, A. Effects of polyether-modified poly(acrylic acid) microgels on doxorubicin transport in human intestinal epithelial Caco-2 cell. J. Control. Release 2003, 88, 11–22. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chen, Y.; Wang, Y.; Zhou, J.; Gao, X.; Qu, D.; Liu, C. Study on the Mechanism of Intestinal Absorption of Epimedins A, B and C in the Caco-2 Cell Model. Molecules 2014, 19, 686-698. https://doi.org/10.3390/molecules19010686
Chen Y, Wang Y, Zhou J, Gao X, Qu D, Liu C. Study on the Mechanism of Intestinal Absorption of Epimedins A, B and C in the Caco-2 Cell Model. Molecules. 2014; 19(1):686-698. https://doi.org/10.3390/molecules19010686
Chicago/Turabian StyleChen, Yan, Ying Wang, Jing Zhou, Xia Gao, Ding Qu, and Congyan Liu. 2014. "Study on the Mechanism of Intestinal Absorption of Epimedins A, B and C in the Caco-2 Cell Model" Molecules 19, no. 1: 686-698. https://doi.org/10.3390/molecules19010686



