Physicochemical Characteristics and Anti-Inflammatory Activities of Antrodan, a Novel Glycoprotein Isolated from Antrodia cinnamomea Mycelia
Abstract
:1. Introduction
2. Results and Discussion
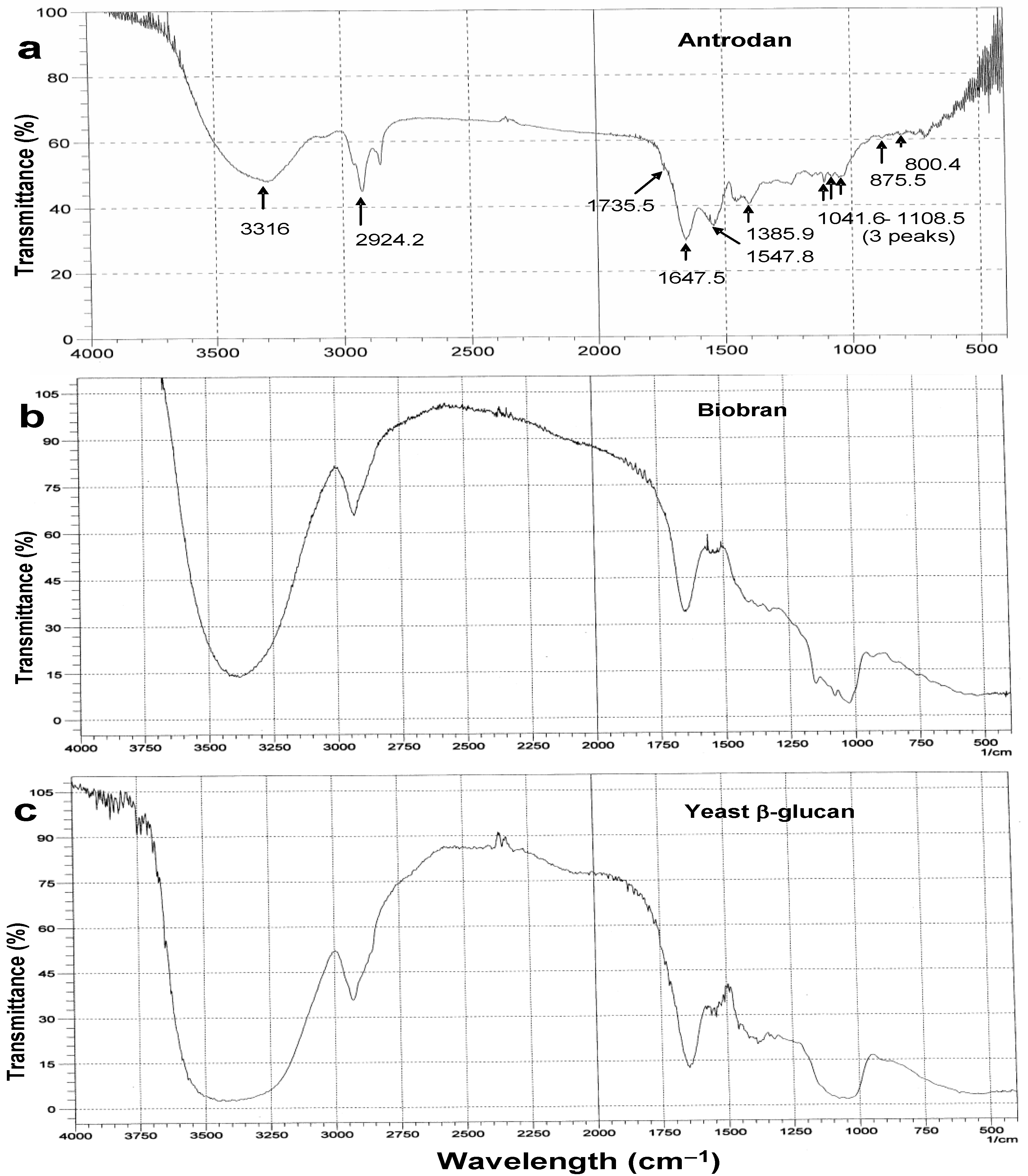
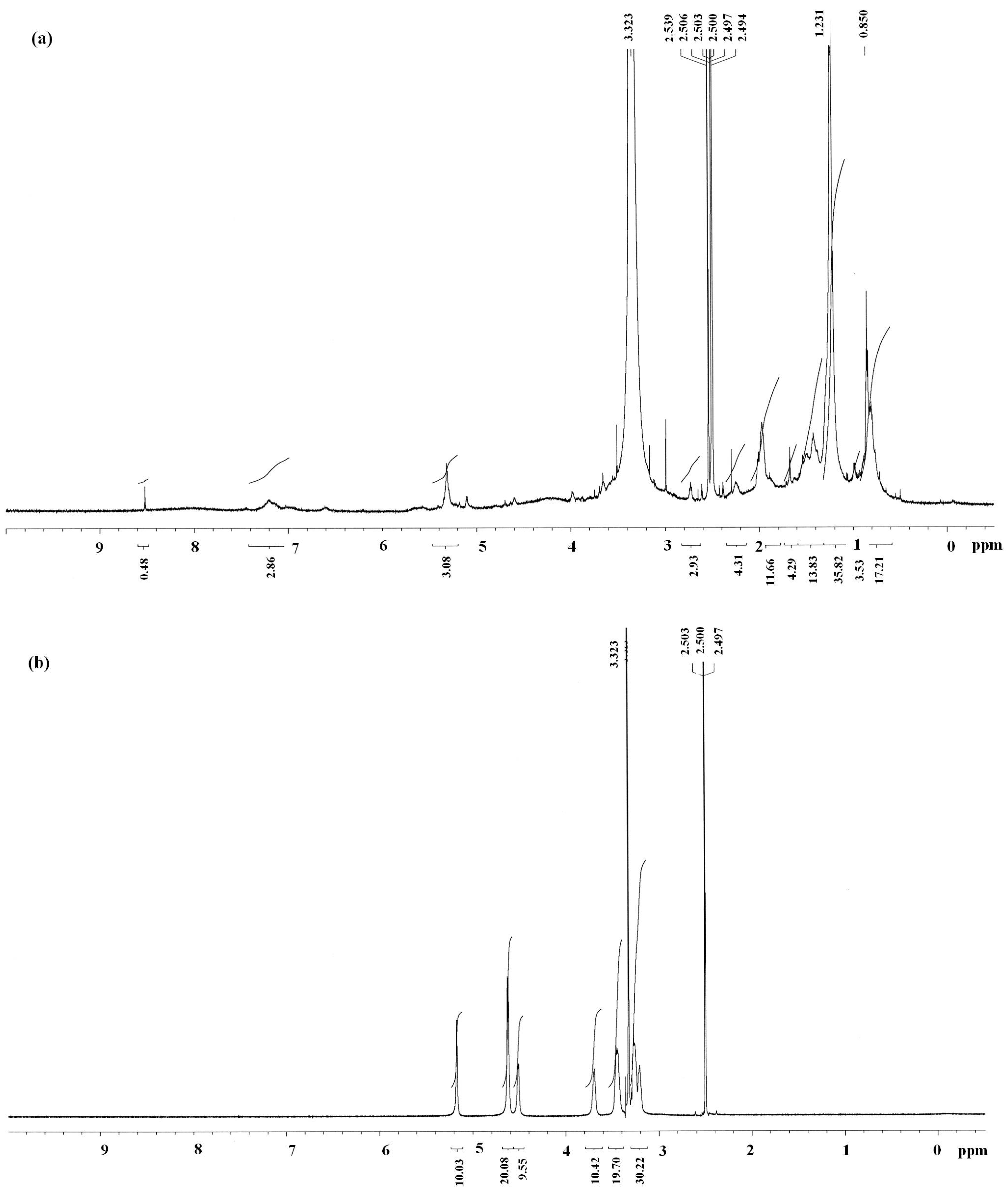
2.1. Characterization of Antrodan

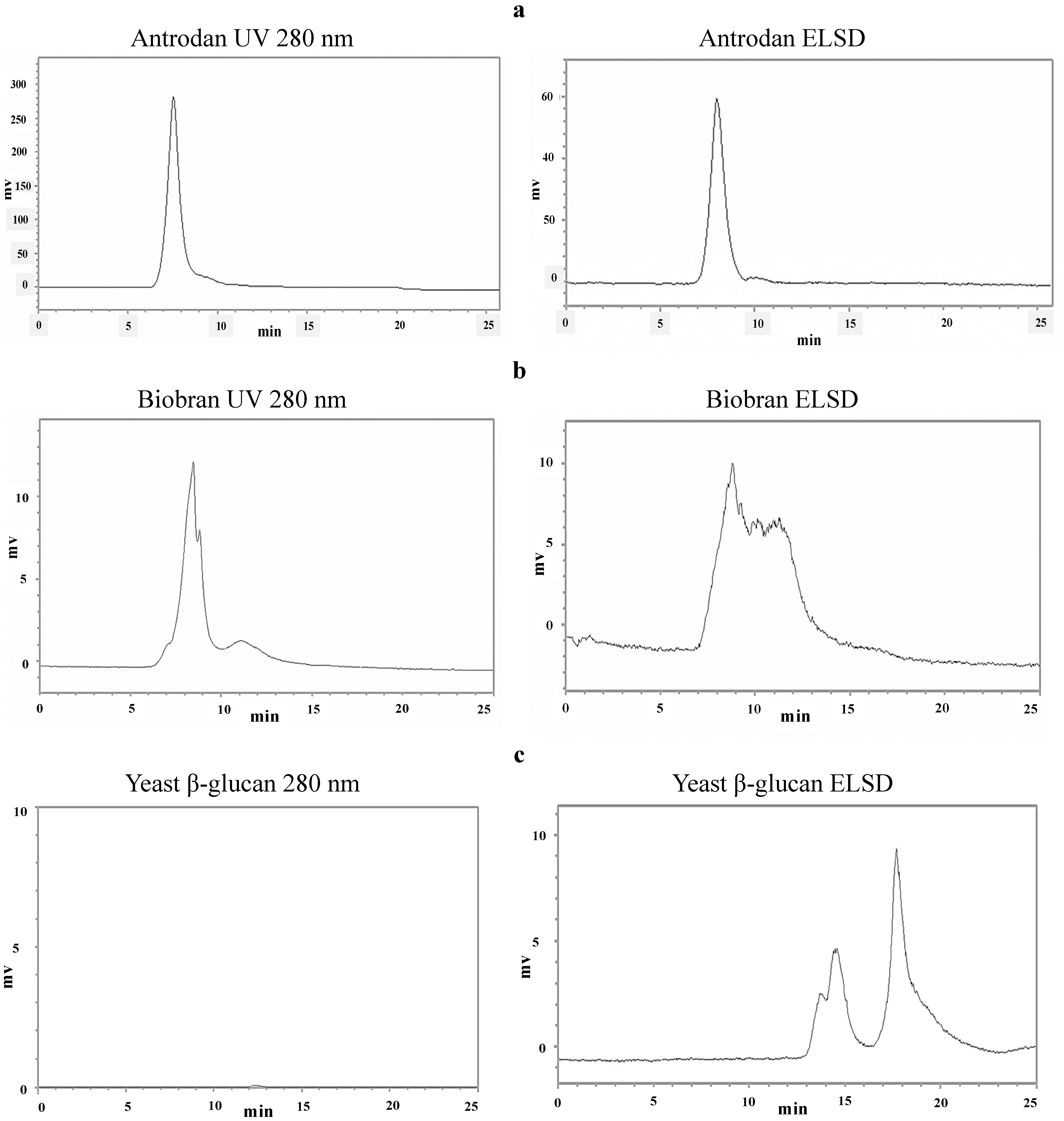
| Sample | Average MW (kDa) 2 | Uronic acid (mg/g) 3 | Glucan content (%) | ||
|---|---|---|---|---|---|
| α-glucan | β-glucan | Total | |||
| antrodan | 442 | 152.6 ± 0.8 | 1.45 | 14.20 | 15.65 |
| biobran | 29 | 146.0 ± 1.0 | ND 4 | ND | ND |
| yeast β-glucan | ~ 1 | 710.7 ± 2.3 | 1.70 | 45.47 | 47.17 |
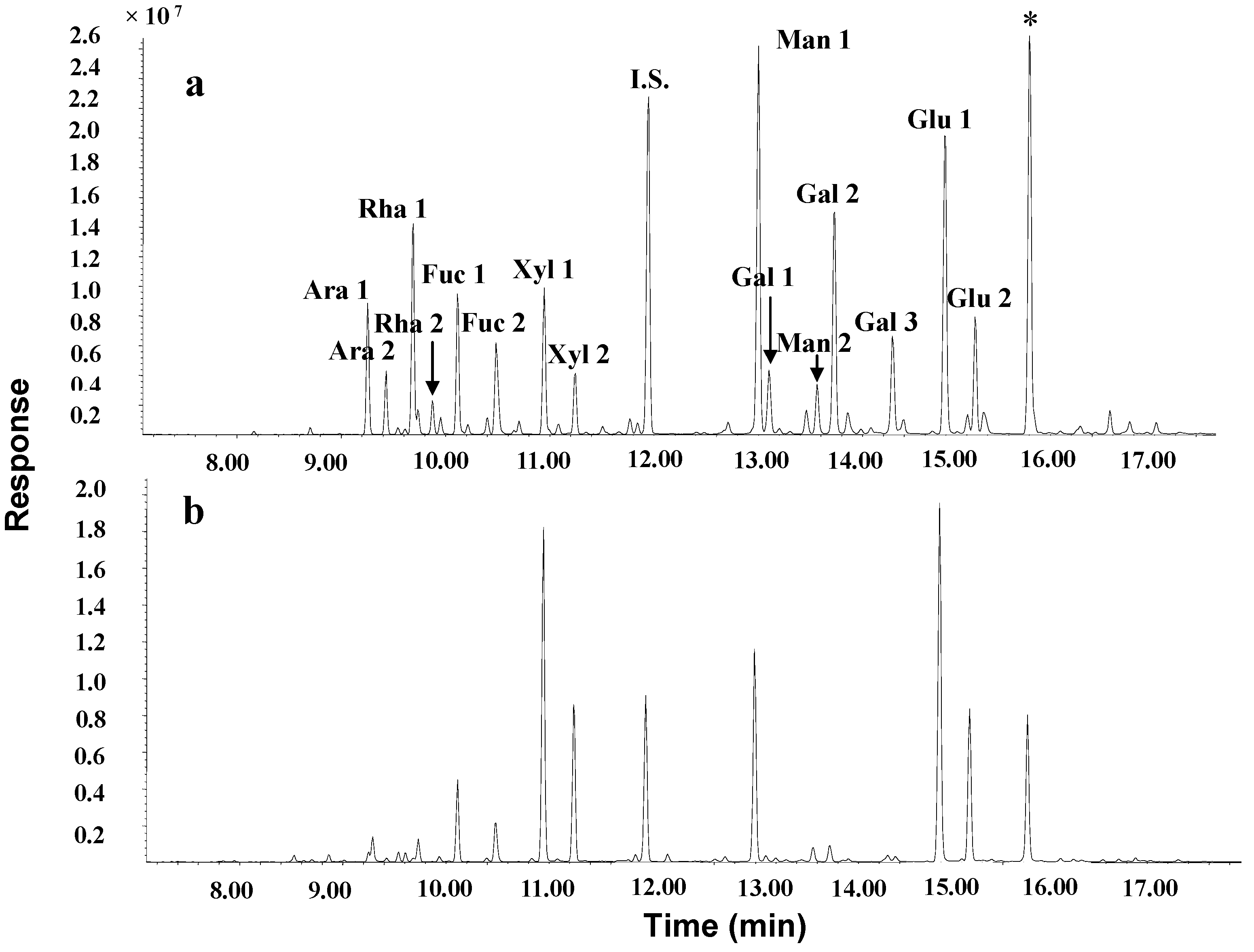
| Carbohydrate (%) | Protein (%) | Sugar component (%) | ||||||
|---|---|---|---|---|---|---|---|---|
| Arabinose | Rhamnose | Fucose | Xylose | Mannose | Galactose | Glucose | ||
| 14.10 | 71.00 | 0.96 | 0.32 | 8.40 | 33.71 | 16.61 | 1.80 | 38.20 |
| Amino acid | % a | Amino acid | % a |
|---|---|---|---|
| Histidine b | 0.23 | Asparagine | 2.23 |
| Isoleucine b | 10.53 | Glutamic acid | 6.60 |
| Leucine b | 17.62 | Glutamine | 1.28 |
| Lysine b | 1.24 | Glycine | 6.21 |
| Methionine b | 0.13 | Hydroxyproline | 0.05 |
| Phenylalanine b | 9.85 | Ornithine c | 1.35 |
| Threonine b | 0.85 | Proline | 4.40 |
| Valine b | 13.21 | Serine | 3.61 |
| Alanine | 9.12 | Tyrosine | 4.70 |
| Aspartic acid | 6.73 | - | - |
| Total | 99.94 |
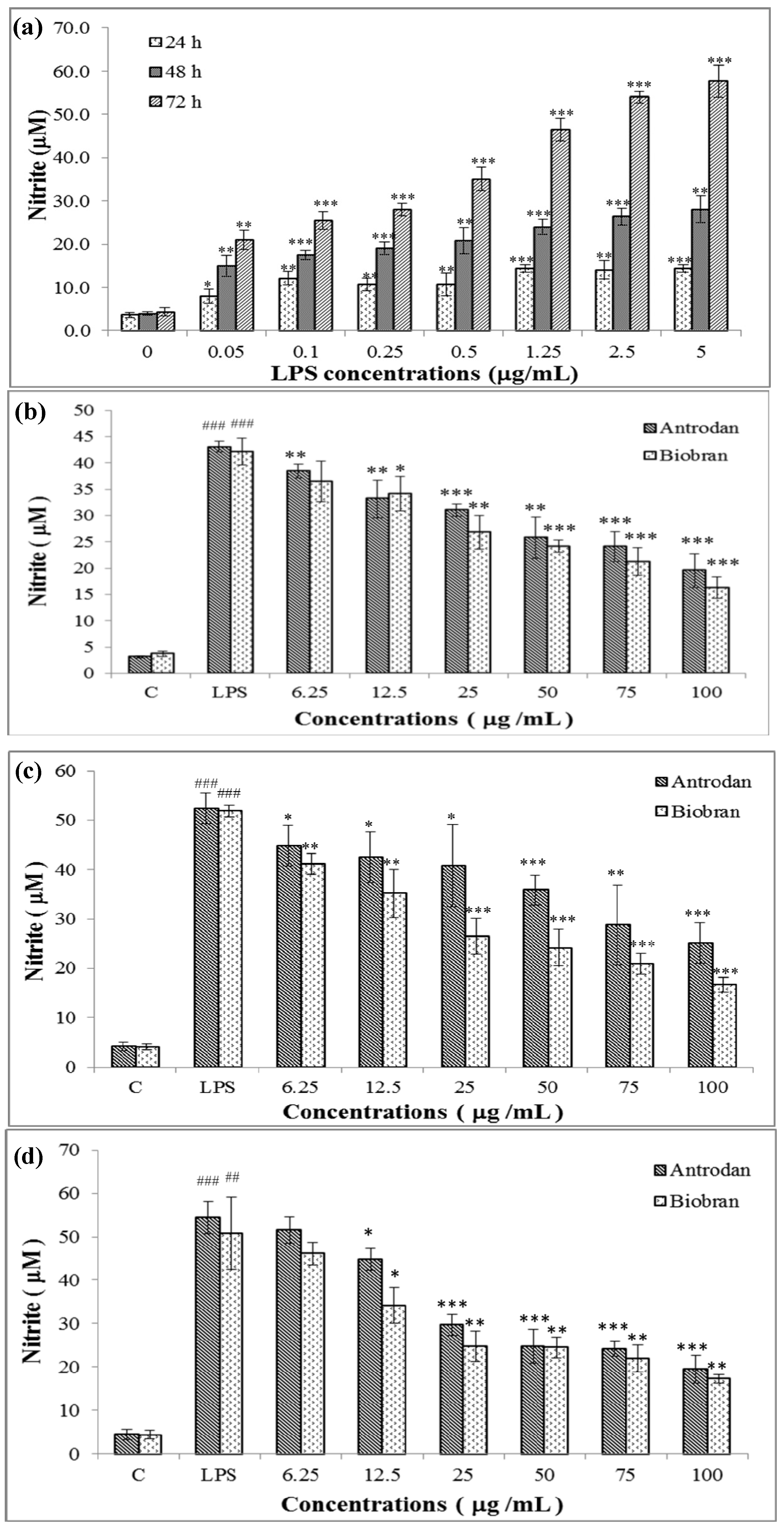
2.2. Cell Viability Affected by Antrodan and LPS

2.3. Antrodan Inhibited the in vitro LPS-Induced NO Production in RAW 264.7 Cell Line
3. Experimental
3.1. Chemicals and Reagents
3.2. Isolation and Preparation of A. cinnamomea Polysaccharides
3.3. Isolation of Antrodan by Gel Permeation Chromatography
3.4. Preparation of Biobran Polysaccharide
3.5. Physicochemcial Characterization of Antrodan
3.5.1. Structural Analyses
3.5.2. Determination of Total Sugar Content
3.5.3. Determination of Total Protein Content
3.5.4. Determination of Monosaccharide Composition
3.5.5. Analysis of Amino Acid Composition
3.5.6. Determination of Uronic Acid Content
3.5.7. Determination of Glucans
3.5.7.1. Total Glucans
3.5.7.2. α-Glucans
3.6. Macrophage RAW 264.7 Cell Cultivation
3.7. Effect of Antrodan and Biobran on the Cell Viability
3.8. Effect of LPS on the Cell Viability
3.9. Effect of LPS and Antrodan on NO Production in Cell Line
3.10. Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Chang, T.T.; Chou, W.N. Antrodia cinnamomea sp. nov. on Cinnamomum kanehirai in Taiwan. Mycol. Res. 1995, 99, 756–758. [Google Scholar] [CrossRef]
- Hsu, F.L.; Chou, C.J.; Chang, Y.C.; Chang, T.T.; Lu, M.K. Promotion of hyphal growth and underlying chemical changes in Antrodia camphorata by host factors from Cinnamomum camphora. Int. J. Food Microbiol. 2006, 106, 32–38. [Google Scholar] [CrossRef]
- Geethangili, M.; Tzeng, Y.M. Review of pharmacological effects of Antrodia camphorata and its bioactive compounds. Evid. Based Complement. Alternat. Med. 2011. [Google Scholar] [CrossRef]
- Yu, Y.L.; Chen, I.H.; Shen, K.Y.; Huang, R.Y.; Wang, W.R.; Chou, C.J.; Chang, T.T.; Chu, C.L. A triterpenoid methyl antcinate K isolated from Antrodia cinnamomea promotes dendritic cell activation and Th2 differentiation. Eur. J. Immunol. 2009, 39, 2482–2491. [Google Scholar] [CrossRef]
- Huang, C.C.; Hsu, M.C.; Huang, W.C.; Yang, H.R.; Hou, C.C. Triterpenoid-rich extract from Antrodia camphorata improves physical fatigue and exercise performance in mice. Evid. Based Complement. Alternat. Med. 2012. [Google Scholar] [CrossRef]
- Huang, C.H.; Chang, Y.Y.; Liu, C.W.; Kang, W.Y.; Lin, Y.L.; Chang, H.C.; Chen, Y.C. Fruiting body of Niuchangchih (Antrodia camphorata) protects livers against chronic alcohol consumption damage. J. Agric. Food Chem. 2010, 58, 3859–3866. [Google Scholar] [CrossRef]
- Cherng, I.H.; Wu, D.P.; Chiang, H.C. Triterpenoids from Antrodia cinnamomea. Phytochemistry 1996, 41, 263–267. [Google Scholar] [CrossRef]
- Chen, C.C.; Liu, Y.W.; Ker, Y.B.; Wu, Y.Y.; Lai, E.Y.; Chyau, C.C.; Hseu, T.H.; Peng, R.Y. Chemical characterization and anti-inflammatory effect of polysaccharides fractionated from submerge-cultured Antrodia camphorata mycelia. J. Agric. Food Chem. 2007, 58, 5007–5012. [Google Scholar]
- Meng, L.M.; Pai, M.H.; Liu, J.J.; Yeh, S.L. Polysaccharides from extracts of Antrodia camphorata mycelia and fruiting bodies modulate inflammatory mediator expression in mice with polymicrobial sepsis. Nutrition 2012, 28, 942–949. [Google Scholar] [CrossRef]
- Lu, M.C.; El-Shazly, M.; Wu, T.Y.; Du, Y.C.; Chang, T.T.; Chen, C.F.; Hsu, Y.M.; Lai, K.H.; Chiu, C.P.; Chang, F.R.; et al. Recent research and development of Antrodia cinnamomea. Pharmacol. Ther. 2013, 139, 124–156. [Google Scholar] [CrossRef]
- Liu, K.J.; Leu, S.J.; Su, C.H.; Chiang, B.L.; Chen, Y.L.; Lee, Y.L. Administration of polysaccharides from Antrodia camphorata modulates dendritic cell function and alleviates allergen-induced T helper type 2 responses in a mouse model of asthma. Immunology 2010, 129, 351–362. [Google Scholar] [CrossRef]
- Yang, C.M.; Zhou, Y.J.; Wang, R.J.; Hu, M.L. Anti-angiogenic effects and mechanisms of polysaccharides from Antrodia cinnamomea with different molecular weights. J. Ethnopharmacol. 2009, 123, 407–412. [Google Scholar] [CrossRef]
- Chen, Y.J.; Cheng, P.C.; Lin, C.N.; Liao, H.F.; Chen, Y.Y.; Chen, C.C.; Lee, K.M. Polysaccharides from Antrodia camphorata mycelia extracts possess immunomodulatory activity and inhibits infection of Schistosoma mansoni. Int. Immunopharmacol. 2008, 8, 458–467. [Google Scholar] [CrossRef]
- Wu, Y.Y.; Chen, C.C.; Chyau, C.C.; Chung, S.Y.; Liu, Y.W. Modulation of inflammation-related genes of polysaccharides fractionated from mycelia of medicinal basidiomycete Antrodia camphorata. Acta. Pharmacol. Sin. 2007, 28, 258–267. [Google Scholar] [CrossRef]
- Liu, J.J.; Huang, T.S.; Hsu, M.L.; Chen, C.C.; Lin, W.S.; Lu, F.J.; Chang, W.H. Antitumor effects of the partially purified polysaccharides from Antrodia camphorata and the mechanism of its action. Toxicol. Appl. Pharmacol. 2004, 201, 186–193. [Google Scholar] [CrossRef]
- Han, H.F.; Nakamura, N.; Zuo, F.; Hirakawa, A.; Yokozawa, T.; Hattori, M. Protective effects of a neutral polysaccharide isolated from the mycelium of Antrodia cinnamomea on Propionibacterium acnes and lipopolysaccharide induced hepatic injury in mice. Chem. Pharm. Bull. 2006, 54, 496–500. [Google Scholar] [CrossRef]
- Maehara, Y.; Tsujitani, S.; Saeki, H.; Oki, E.; Yoshinaga, K.; Emi, Y.; Morita, M.; Kohnoe, S.; Kakeji, Y.; Yano, Y.; et al. Biological mechanism and clinical effect of protein-bound polysaccharide K (KRESTIN): Review of development and future perspectives. Surg. Today 2012, 42, 8–28. [Google Scholar] [CrossRef]
- Akagi, J.; Baba, H. PSK may suppress CD57+ T cells to improve survival of advanced gastric cancer patients. Int. J. Clin. Oncol. 2010, 15, 145–152. [Google Scholar] [CrossRef]
- El-Din, N.K.B.; Noaman, E.; Ghoneum, M. In vivo tumor inhibitory effects of nutritional rice bran supplement MGN-3/biobran on Ehrlich carcinoma-bearing mice. Nutr. Cancer 2008, 60, 235–244. [Google Scholar] [CrossRef]
- Aguilar-Uscanga, B.; Francois, J.M. A study of the yeast cell wall composition and structure in response to growth conditions and mode of cultivation. Lett. Appl. Microbiol. 2003, 37, 268–274. [Google Scholar] [CrossRef]
- Anumula, K.R. Advances in fluorescence derivatization methods for high-performance liquid chromatographic analysis of glycoprotein carbohydrates. Anal. Biochem. 2006, 350, 1–23. [Google Scholar] [CrossRef]
- Hajto, T.; Kirsch, A. Case reports of cancer patients with hepatic metastases treated by standardized plant immunomodulatory preparations. J. Can. Res. Updat. 2013, 2, 1–9. [Google Scholar]
- Kim, Y.T.; Kim, E.; Cheong, C.; Williams, D.L.; Kim, C.W.; Lim, S.T. Structural characterization of β-d-(1→3,1→6)-linked glucans using NMR spectroscopy. Carbohydr. Res. 2000, 328, 331–341. [Google Scholar] [CrossRef]
- Sugawara, T.; Takahashi, S.; Osumic, M.; Ohnoe, N. Refinement of the structures of cell-wall glucans of Schizosaccharomyces pombe by chemical modification and NMR spectroscopy. Carbohydr. Res. 2004, 339, 2255–2265. [Google Scholar] [CrossRef]
- Bubb, W.A. NMR spectroscopy in the study of carbohydrates: Characterizing the structural complexity. Concepts Magn. Reson. 2003, 19, 1–19. [Google Scholar] [CrossRef]
- Rehm, T.; Huber, R.; Holak, T.A. Application of NMR in structural proteomics: Screening for proteins amenable to structural analysis. Structure 2002, 10, 1613–1618. [Google Scholar] [CrossRef]
- Moradali, M.F.; Mostafavi, H.; Ghods, S.; Hedjaroude, G.A. Immunomodulating and anticancer agents in the realm of macromycetes fungi (macrofungi). Int. Immunopharmacol. 2007, 7, 701–724. [Google Scholar] [CrossRef]
- Chan, G.C.; Chan, W.K.; Sze, D.M. The effects of beta-glucan on human immune and cancer cells. J. Hematol. Oncol. 2009, 2, 25–35. [Google Scholar] [CrossRef] [Green Version]
- Rice, P.J.; Adams, E.L.; Ozment-Skelton, T.; Gonzalez, A.J.; Goldman, M.P.; Lockhart, B.E.; Barker, L.A.; Breuel, K.F.; Deponti, W.K.; Kalbfleisch, J.H.; et al. Oral delivery and gastrointestinal absorption of soluble glucans stimulate increased resistance to infectious challenge. J. Pharmacol. Exp. Ther. 2005, 314, 1079–1086. [Google Scholar] [CrossRef]
- Masuko, T.; Minami, A.; Iwasaki, N.; Majima, T.; Nishimura, S.; Lee, Y.C. Carbohydrate analysis by a phenol-sulfuric acid method in microplate format. Anal. Biochem. 2005, 339, 69–72. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of micro quantities of protein utilizing the principle of protein-dye binding. Anal. Chem. 1976, 72, 248–254. [Google Scholar]
- Sobolevsky, T.G.; Revelsky, A.I.; Miller, B.; Oriedo, V.; Chernetsova, E.S.; Revelsky, I.A. Comparison of silylation and esterification/acylation procedures in GC-MS analysis of amino acids. J. Sep. Sci. 2003, 26, 1474–1478. [Google Scholar] [CrossRef]
- Galambos, J.T. The reaction of carbazole with carbohydrates. I. Effect of borate and sulfamate on the carbazole color of sugars. Anal. Biochem. 1967, 19, 119–132. [Google Scholar] [CrossRef]
- Green, L.C.; Wagner, D.A.; Glogowski, J.; Skipper, P.L.; Wishnok, J.S.; Tannenbaum, S.R. Analysis of nitrate, nitrite, and [15N]nitrate in biological samples. Anal. Biochem. 1982, 126, 131–138. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the Antrodia cinnamomea mycelia are available from the authors.
© 2013 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chiu, C.-H.; Peng, C.-C.; Ker, Y.-B.; Chen, C.-C.; Lee, A.; Chang, W.-L.; Chyau, C.-C.; Peng, R.Y. Physicochemical Characteristics and Anti-Inflammatory Activities of Antrodan, a Novel Glycoprotein Isolated from Antrodia cinnamomea Mycelia. Molecules 2014, 19, 22-40. https://doi.org/10.3390/molecules19010022
Chiu C-H, Peng C-C, Ker Y-B, Chen C-C, Lee A, Chang W-L, Chyau C-C, Peng RY. Physicochemical Characteristics and Anti-Inflammatory Activities of Antrodan, a Novel Glycoprotein Isolated from Antrodia cinnamomea Mycelia. Molecules. 2014; 19(1):22-40. https://doi.org/10.3390/molecules19010022
Chicago/Turabian StyleChiu, Chun-Hung, Chiung-Chi Peng, Yaw-Bee Ker, Chin-Chu Chen, Arwen Lee, Wan-Lin Chang, Charng-Cherng Chyau, and Robert Y. Peng. 2014. "Physicochemical Characteristics and Anti-Inflammatory Activities of Antrodan, a Novel Glycoprotein Isolated from Antrodia cinnamomea Mycelia" Molecules 19, no. 1: 22-40. https://doi.org/10.3390/molecules19010022






