Two New Phenolic Compounds from the Heartwood of Caesalpinia sappan L.
Abstract
:1. Introduction
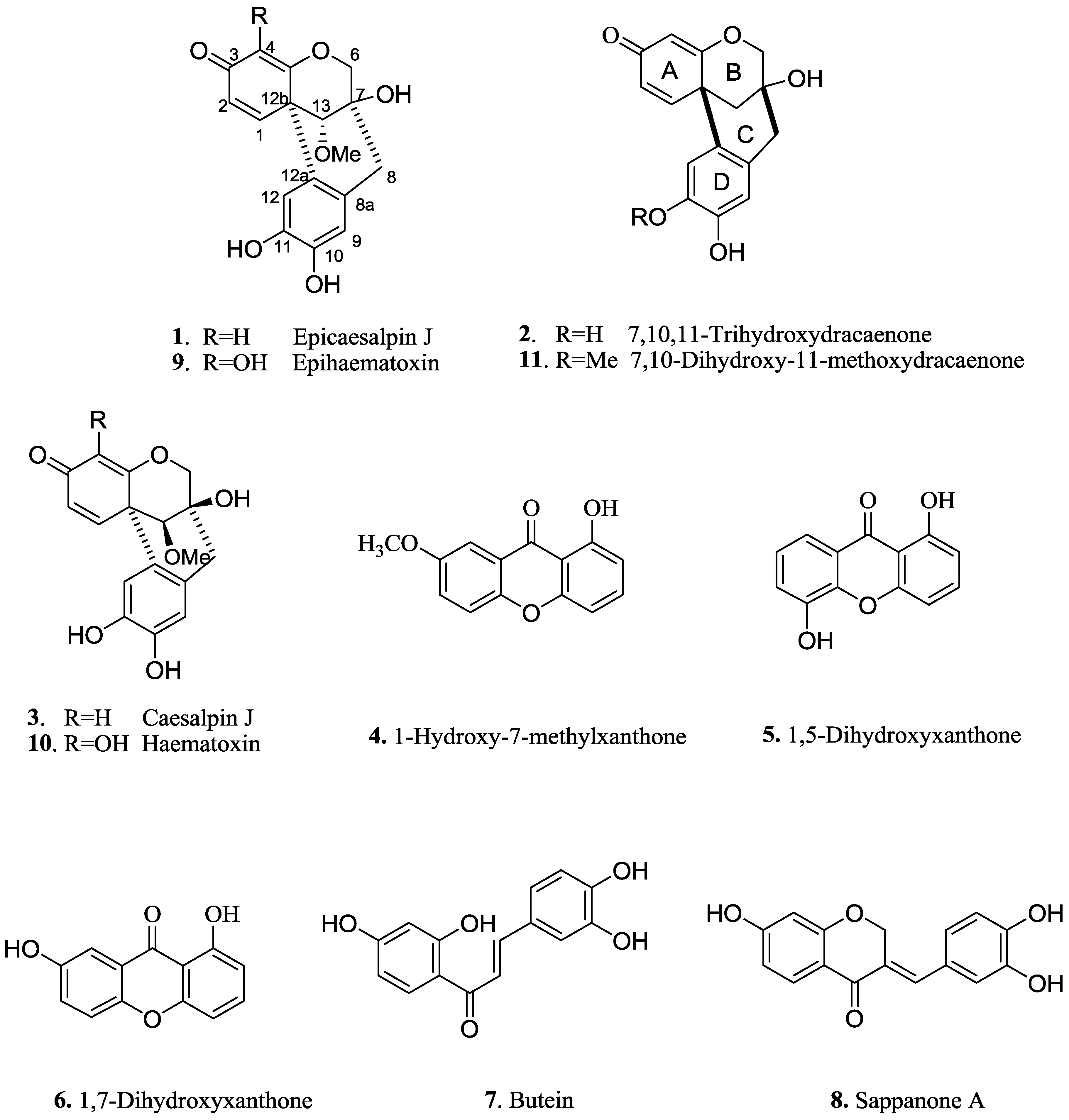
2. Results and Discussion
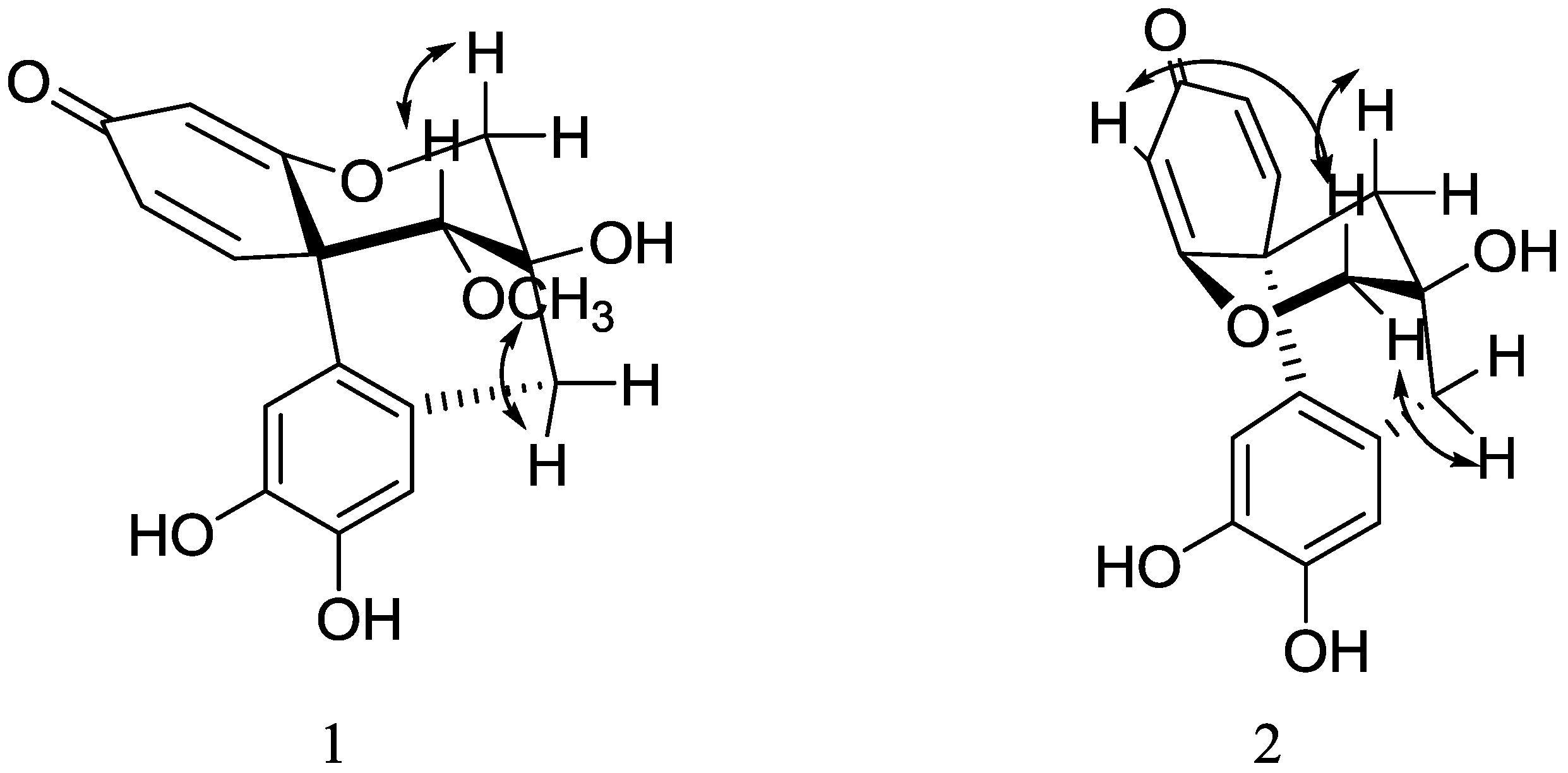
 +317.3 (c 0.30, MeOH), UV λmax (MeOH) nm: 212, 242. Its molecular formula was determined as C17H16O6 by HR-ESI-MS (found 317.1021 [M+H]+, calcd. 317.1025). The IR spectrum of 1 showed α,β-unsaturated ketone (1649 cm−1), aromatic ring (1595 cm−1) and hydroxyl group (3443 cm−1) absorptions. The 1H- and 13C-NMR spectra of 1 displayed the characteristic signals of a methoxyl group (δ 3.62), an oxymethylene (δ 4.18 and 3.79), an oxymethine (δ 3.48), an α,β-unsaturated ketone group, and a tetrasubstituted benzene ring, respectively. All protons and carbons were unambiguously assigned by 1D and 2D NMR experiments, including 1H-1H COSY, HSQC, and HMBC (Table 1, Figure 2). Comparison of the NMR data with those of the known compound, caesalpin J (3) [11,21], revealed that 1 possessed a similar skeleton. The major difference is the upfield shift of H-13 from δ 3.84 in 3 to δ 3.48 in 1, and the same phenomenon was also observed in the compounds haematoxin (10) and epihematoxin (9) [22], which suggested that 1 was a stereoisomer of 3. The absolute configuration of 3 had been established in [12] by an X-ray crystallographic study of its triacetate. According to this paper, the ring B and C in 3 were both in a chair conformation. The optical rotation of 1 (+371.3) was consistent with that of 3 (+445.0), which suggested that 1 had the same configuration. In the NOESY spectrum of 1, the cross peaks between H-13 and H-6β, and OMe-13 and H-8 unambiguously confirmed that 1 was a C-13 epimer of 3, and the methoxyl group at C-13 was α-oriented (Figure 3). Thus, the structure of 1 was fully elucidated, and it was named epicaesalpin J.
+317.3 (c 0.30, MeOH), UV λmax (MeOH) nm: 212, 242. Its molecular formula was determined as C17H16O6 by HR-ESI-MS (found 317.1021 [M+H]+, calcd. 317.1025). The IR spectrum of 1 showed α,β-unsaturated ketone (1649 cm−1), aromatic ring (1595 cm−1) and hydroxyl group (3443 cm−1) absorptions. The 1H- and 13C-NMR spectra of 1 displayed the characteristic signals of a methoxyl group (δ 3.62), an oxymethylene (δ 4.18 and 3.79), an oxymethine (δ 3.48), an α,β-unsaturated ketone group, and a tetrasubstituted benzene ring, respectively. All protons and carbons were unambiguously assigned by 1D and 2D NMR experiments, including 1H-1H COSY, HSQC, and HMBC (Table 1, Figure 2). Comparison of the NMR data with those of the known compound, caesalpin J (3) [11,21], revealed that 1 possessed a similar skeleton. The major difference is the upfield shift of H-13 from δ 3.84 in 3 to δ 3.48 in 1, and the same phenomenon was also observed in the compounds haematoxin (10) and epihematoxin (9) [22], which suggested that 1 was a stereoisomer of 3. The absolute configuration of 3 had been established in [12] by an X-ray crystallographic study of its triacetate. According to this paper, the ring B and C in 3 were both in a chair conformation. The optical rotation of 1 (+371.3) was consistent with that of 3 (+445.0), which suggested that 1 had the same configuration. In the NOESY spectrum of 1, the cross peaks between H-13 and H-6β, and OMe-13 and H-8 unambiguously confirmed that 1 was a C-13 epimer of 3, and the methoxyl group at C-13 was α-oriented (Figure 3). Thus, the structure of 1 was fully elucidated, and it was named epicaesalpin J.| No. | 1 (in DMSO-d6) | 1 (in CD3OD) | 2 (in CD3OD) | |||
|---|---|---|---|---|---|---|
| δC | δH | δC | δH | δC | δH | |
| 1 | 146.3 | 7.03 d (10.0) | 149.0 | 7.16 d (10.0) | 151.5 | 6.88 d (10.0) |
| 2 | 129.2 | 6.46 dd (10.0, 1.5) | 130.4 | 6.51 dd (10.0, 1.5) | 128.5 | 6.45 dd (10.0, 1.5) |
| 3 | 187.1 | 190.9 | 191.4 | |||
| 4 | 108.5 | 5.48 d (1.5) | 109.8 | 5.57 d (1.5) | 108.1 | 5.58 d (1.5) |
| 4a | 175.0 | 178.6 | 179.7 | |||
| 6 | 77.8 | 4.12 d (11.0) | 79.8 | 4.18 d (11.0) | 81.0 | 3.88 dd (11.0) |
| 3.72 d (11.0) | 3.79 d (11.0) | 4.27 dd (11.0) | ||||
| 7 | 69.4 | 71.3 | 66.8 | |||
| 8 | 37.6 | 3.16 d (16.0) | 39.0 | 3.30 d (16.0) | 43.5 | 3.16 d (16.0) |
| 2.83 d (16.0) | 2.88 d (16.0) | 3.10 d (16.0) | ||||
| 8a | 122.6 | 124.2 | 129.5 | |||
| 9 | 115.4 | 6.52 s | 116.6 | 6.59 s | 116.9 | 6.64 s |
| 10 | 145.0 | 146.6 | 146.8 | |||
| 11 | 143.6 | 145.2 | 145.1 | |||
| 12 | 112.9 | 6.26 s | 114.1 | 6.35 s | 113.4 | 6.44 s |
| 12a | 126.6 | 128.5 | 125.5 | |||
| 12b | 51.1 | 53.5 | 47.8 | |||
| 13 | 82.7 | 3.44 s | 84.8 | 3.48 s | 41.1 | 1.99 dd (11.5) |
| 2.34 dd (11.5) | ||||||
| OCH3 | 61.4 | 3.53 s | 62.6 | 3.62 s | - | - |
 −152.6 (c 0.54, MeOH). Its molecular formula was determined as C16H14O5 by HR-ESI-MS (found 285.0765 [M−H]+, calcd. 285.0769). The 1H- and 13C-NMR data of 2 were similar to those of the known compound 7,10-dihydroxy-11-methoxydracaenone (11), except that 2 showed no methoxyl signals. Considering that the molecular weight of 2 was 30 Da less than that of 11, compound 2 was identified as 7,10,11-trihydroxydracaenone. According to [23], the absolute configuration of 11 was different from those of 3 and 1, and 11 had a C ring boat conformation. The optical rotation of 2 was −152.6, which was similar to that of 11 (-465.9). Moreover, in the NOESY spectrum, the cross peaks between H-4 and H-6β, and H-8 and H-6α unambiguously confirmed that 2 had the same configuration as 11, which was opposite of that of 1 and 3 (Figure 3). All protons and carbons were unambiguously assigned by 1D and 2D NMR experiments, including 1H-1H COSY, NOESY, HSQC, and HMBC (Table 1, Figure 2 and Figure 3). Thus, the structure of 2 was confirmed as 7,10,11-trihydroxydracaenone.
−152.6 (c 0.54, MeOH). Its molecular formula was determined as C16H14O5 by HR-ESI-MS (found 285.0765 [M−H]+, calcd. 285.0769). The 1H- and 13C-NMR data of 2 were similar to those of the known compound 7,10-dihydroxy-11-methoxydracaenone (11), except that 2 showed no methoxyl signals. Considering that the molecular weight of 2 was 30 Da less than that of 11, compound 2 was identified as 7,10,11-trihydroxydracaenone. According to [23], the absolute configuration of 11 was different from those of 3 and 1, and 11 had a C ring boat conformation. The optical rotation of 2 was −152.6, which was similar to that of 11 (-465.9). Moreover, in the NOESY spectrum, the cross peaks between H-4 and H-6β, and H-8 and H-6α unambiguously confirmed that 2 had the same configuration as 11, which was opposite of that of 1 and 3 (Figure 3). All protons and carbons were unambiguously assigned by 1D and 2D NMR experiments, including 1H-1H COSY, NOESY, HSQC, and HMBC (Table 1, Figure 2 and Figure 3). Thus, the structure of 2 was confirmed as 7,10,11-trihydroxydracaenone.

| Compound | Viability (at 50 µM, %) | IC50 (µM) | Compound | Viability (at 50 µM, %) | IC50(µM) |
|---|---|---|---|---|---|
| 1 | 96.45 | 52.62 | 6 | 101.64 | 21.46 |
| 2 | 99.29 | 56.71 | 7 | 94.00 | 15.46 |
| 3 | 96.13 | 45.87 | 8 | 99.29 | 8.26 |
| 4 | 98.94 | 14.23 | quercetin | 23.42 | |
| 5 | 101.19 | 28.65 |
3. Experimental
3.1. General
3.2. Plant Material
3.3. Extraction and Isolation
3.4. Spectral Data
 +371.3° (c 0.30, MeOH), UV λmax (MeOH) nm: 212, 242. IR νmax (KBr) cm−1: 3443, 2957, 1649, 1595, 1454, 1395, 1016. HR-ESI-MS m/z: 317.1021 [(M+H)+, calcd. for C17H17O6 317.1025]. 1H-NMR and 13C-NMR (CD3OD and DMSO-d6) see Table 1.
+371.3° (c 0.30, MeOH), UV λmax (MeOH) nm: 212, 242. IR νmax (KBr) cm−1: 3443, 2957, 1649, 1595, 1454, 1395, 1016. HR-ESI-MS m/z: 317.1021 [(M+H)+, calcd. for C17H17O6 317.1025]. 1H-NMR and 13C-NMR (CD3OD and DMSO-d6) see Table 1. −152.6° (c 0.54, MeOH). UV λmax (MeOH) nm: 214, 240. IR νmax (KBr) cm−1: 3381, 1653, 1591, 1522, 1451, 1395, 1065. HR-ESI-MS m/z: 285.0765 [(M−H)−, calcd. for C16H14O5 285.0769]. 1H-NMR and 13C-NMR (CD3OD) see Table 1.
−152.6° (c 0.54, MeOH). UV λmax (MeOH) nm: 214, 240. IR νmax (KBr) cm−1: 3381, 1653, 1591, 1522, 1451, 1395, 1065. HR-ESI-MS m/z: 285.0765 [(M−H)−, calcd. for C16H14O5 285.0769]. 1H-NMR and 13C-NMR (CD3OD) see Table 1.3.5. Inhibition of NO Production in LPS-Stimulated BV-2 Microglia
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Flora of China Editorial Committee. Flora of China; Science Press: Beijing, China, 1988; Volume 35, p. 105. [Google Scholar]
- Chinese Pharmacopoeia Commission. Chinese Pharmacopoeia, 2010 ed.; Chemical Industry Press: Beijing, China, 2010; Part I; p. 153. [Google Scholar]
- Saitoh, T.; Sakashita, S.; Nakata, H.; Shimokawa, T.; Kinjo, J.; Yamahara, J.; Yamasaki, M.; Nohara, T. 3-Benzylchroman derivatives related to Brazilin from Sappan Lignum. Chem. Pharm. Bull. 1986, 34, 2506–2511. [Google Scholar] [CrossRef]
- Namikoshi, M.; Nakata, H.; Saitoh, T. Homoisoflavonoids from Caesalpinia sappan. Phytochemistry 1987, 26, 1831–1833. [Google Scholar] [CrossRef]
- Namikoshi, M.; Saitoh, T. Homoisoflavonoids and related compounds. IV. Absolute configurations of homoisoflavonoids from Caesalpinia sappan. Chem. Pharm. Bull. 1987, 35, 3597–3602. [Google Scholar] [CrossRef]
- Namikoshi, M.; Nakata, H.; Nuno, M.; Ozawa, T.; Saitoh, T. Homoisoflavonoids and related compounds. III. Phenolic constituents of Caesalpinia japonica SIEB. et ZUCC. Chem. Pharm. Bull. 1987, 35, 3568–3575. [Google Scholar] [CrossRef]
- Namikoshi, M.; Nakata, H.; Yamada, H.; Nagai, M.; Saitoh, T. Homoisoflavonoids and related compounds. II. Isolation and absolute configurations of 3,4-dihydroxylated homoisoflavans and brazilin from Caesalpinia sappan. Chem. Pharm. Bull. 1987, 35, 2761–2773. [Google Scholar] [CrossRef]
- Fu, L.C.; Huang, X.A.; Lai, Z.Y.; Hu, Y.J.; Liu, H.J.; Cai, X.L. A new 3-benzylchroman derivative from Sappan Ligum (Caesalpinia sappan). Molecules 2008, 13, 1923–1930. [Google Scholar] [CrossRef]
- Zhao, H.X.; Bai, H.; Wang, Y.S. A new homoisoflavan from Caesalpinia sappan. J. Nat. Med. 2008, 62, 325–327. [Google Scholar] [CrossRef]
- Yodsaoue, O.; Cheenpracha, S.; Karalai, C.; Ponglimanont, C.; Chantrapromma, S.; Fun, H.K.; Kanjana-Opas, A. Phanginin A–K, diterpenoids from the seeds of Caesalpinia sappan Linn. Phytochemistry 2008, 69, 1242–1249. [Google Scholar] [CrossRef]
- Shimokawa, T.; Kinjo, J.; Yamahara, J.; Yamasaki, M.; Nohara, T. Two novel aromatic compounds from Caesalpinia sappan. Chem. Pharm. Bull. 1985, 33, 3545–3547. [Google Scholar] [CrossRef]
- Miyahara, K.; Kawasaki, T.; Kinjo, J.; Shimokawa, T.; Yamahara, J.; Yamasaki, M.; Harano, K.; Noara, T. The X-ray analysis of caesalpin J from Sappan Lignum. Chem. Phram. Bull. 1986, 34, 4166–4169. [Google Scholar] [CrossRef]
- Nagai, M.; Nagumo, S.; Lee, S.M.; Eguchi, I.; Kawai, K.I. Protosappanin A, a novel biphenyl compound from Sappan Lignum. Chem. Pharm. Bull. 1986, 34, 1–6. [Google Scholar] [CrossRef]
- Nagai, M.; Nagumo, S. Protosappanin B, a new dibenzoxocin derivative from Sappan Lignum. Heterocycles 1986, 24, 601–605. [Google Scholar] [CrossRef]
- Nagai, M.; Nagumo, S. Protosappanin C from Sappan Lignum and absolute configuration of protosappanins. Chem. Pharm. Bull. 1987, 35, 3002–3005. [Google Scholar] [CrossRef]
- Nagai, M.; Nagumo, S. Protosappanin E-1 and E-2, stereoisomeric dibenzoxocins combined with brazilin from Sappan Lignum. Chem. Pharm. Bull. 1990, 38, 1490–1494. [Google Scholar] [CrossRef]
- Namikoshi, M.; Nakata, H.; Saitoh, T. Homoisoflavonoids and related compounds.V. a novel dibensoxocin derivative from Caesalpinia sappan L. Chem. Pharm. Bull. 1987, 35, 3615–3619. [Google Scholar] [CrossRef]
- Nguyen, M.T.T.; Awale, S.; Tezuka, Y.; Tran, Q.L.; Kadota, S. Neosappanone A, a xanthine oxidase (XO) inhibitory dimeric methanodibenzoxocinone with a new carbon skeleton from Caesalpinia sappan. Tetrahedron Lett. 2004, 45, 8519–8522. [Google Scholar] [CrossRef]
- Yang, B.O.; Ke, C.Q.; He, A.S.; Yang, Y.P.; Ye, Y. Brazilide A, a novel lactone with an unprecedented skeleton from Caesalpinia sappan. Tetrahedron Lett. 2002, 43, 1731–1733. [Google Scholar] [CrossRef]
- Chen, Y.P.; Liu, L.; Zhou, Y.H.; Wen, J.; Jiang, Y.; Tu, P.F. Chemical constituents from Sappan Lignum. J. Chin. Pharm. Sci. 2008, 17, 82–86. [Google Scholar]
- Cai, C.Q.; Zhao, M.B.; Tang, L.; Tu, P.F. Study on chemical constituents from heartwood of Caesalpinia sappan. Chin. Trad. Herb. Drug 2012, 43, 230–233. [Google Scholar]
- Lin, L.G.; Xie, H.; Li, H.L.; Tong, L.G.; Tang, C.P.; Ke, C.Q.; Liu, Q.F.; Lin, L.P.; Geng, M.Y.; Jiang, H.L.; et al. Naturally occurring homoisoflavonoids function as potent protein tyrosine kinase inhibitors by c-Src-Based High-Throughput Screening. J. Med. Chem. 2008, 51, 4419–4429. [Google Scholar] [CrossRef]
- Meksuriyen, D.; Cordell, G.A. Traditional medicinal plants of Thailand, IX. 10-hydroxy-11-methoxydracaenone and 7,10-dihydroxy-11-methoxydracaenone from Dracaena Loureiri. J. Nat. Prod. 1987, 50, 1118–1125. [Google Scholar] [CrossRef]
- Blasko, G.; Cordell, G.A. Total synthesis of (±)-11-O-methyl-caesalpin J and its C-13 epimer. Tetrahedron 1989, 45, 6361–6365. [Google Scholar] [CrossRef]
- Wang, S.; Li, J.; Sun, J.; Zeng, K.W.; Cui, J.R.; Jiang, Y.; Tu, P.F. NO inhibitory guaianolide-derived terpenoids from Artemisia argyi. Fitoterapia 2013, 85, 169–175. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
© 2013 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhao, M.-B.; Li, J.; Shi, S.-P.; Cai, C.-Q.; Tu, P.-F.; Tang, L.; Zeng, K.-W.; Jiang, Y. Two New Phenolic Compounds from the Heartwood of Caesalpinia sappan L. Molecules 2014, 19, 1-8. https://doi.org/10.3390/molecules19010001
Zhao M-B, Li J, Shi S-P, Cai C-Q, Tu P-F, Tang L, Zeng K-W, Jiang Y. Two New Phenolic Compounds from the Heartwood of Caesalpinia sappan L. Molecules. 2014; 19(1):1-8. https://doi.org/10.3390/molecules19010001
Chicago/Turabian StyleZhao, Ming-Bo, Jun Li, She-Po Shi, Chen-Qiu Cai, Peng-Fei Tu, Li Tang, Ke-Wu Zeng, and Yong Jiang. 2014. "Two New Phenolic Compounds from the Heartwood of Caesalpinia sappan L." Molecules 19, no. 1: 1-8. https://doi.org/10.3390/molecules19010001




