Synthesis and Properties of Annulated 2-(Azaar-2-yl)- and 2,2'-Di(azaar-2-yl)-9,9'-spirobifluorenes
Abstract
:1. Introduction
2. Results and Discussion
2.1. Synthesis


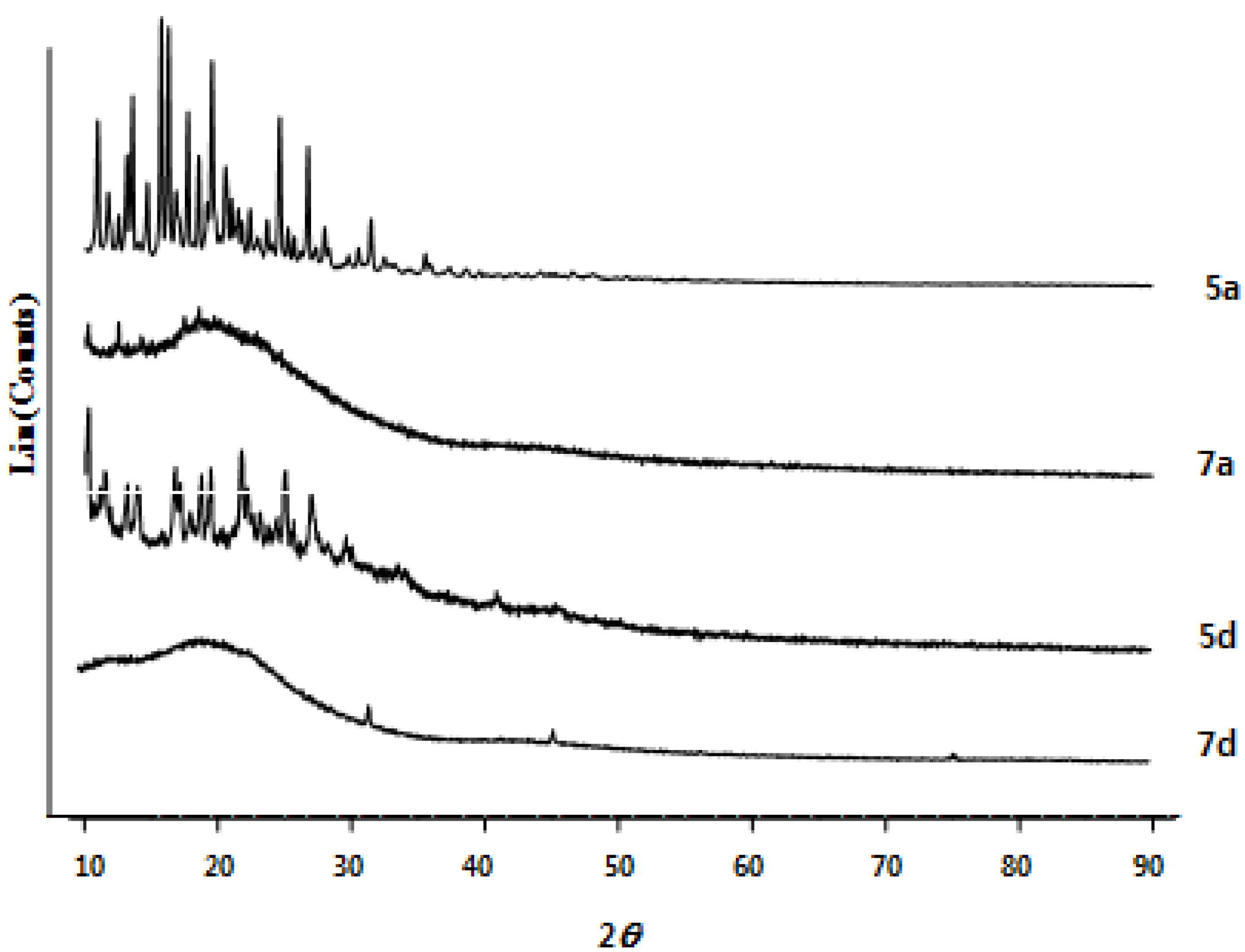
2.2. Structural and Thermal Properties
2.3. Spectroscopic Properties
| H1 a | H4 | H8 | H1' | H7'' | H8'' | H9'' | H10'' | |
|---|---|---|---|---|---|---|---|---|
| 1 | 7.63 | 7.84 [29] | - | - | - | - | - | - |
| 5a | 6.62 | 9.01 | 6.72 | 6.78 | - | 8.21 | - | - |
| 5b | 6.60 | 9.25 | 6.72 | 6.78 | 9.08 | - | - | - |
| 5c | 6.52 | 9.22 | 6.72 | 6.78 | - | 7.72 | 7.84 | 9.62 |
| 5d | 6.62 | 9.34 | 6.71 | 6.78 | 8.25 | 7.64 | 9.26 | - |
| 7a | 6.67 | 9.05 | 6.79 | - | - | 8.22 | - | - |
| 7b | 6.69 | 9.28 | 6.80 | 6.69 | 9.09 | - | - | - |
| 7c | 6.74 | 9.25 | 6.87 | 6.74 | 8.18 | 7.73 | 7.84 | 9.63 |
| 7d | 6.68 | 9.38 | 6.80 | - | 8.26 | 7.65 | 9.28 | - |
| Comp | λmax/nm (1 × 10−5 M in 95% EtOH, log ε) | λem/nm |
|---|---|---|
| 1 | 225 (4.65) 239 (4.40) 272 (4.29) 296 (3.92) 308 (4.11) | 388 (308) |
| 5a | 213 (4.97) 265 (5.00) 296 (4.53) 308 (4.46) 343 (sh, 4.28) 357 (4.44) | 390 (357) |
| 5b | 219 (4.99) 258 (4.95) 296 (4.45) 308 (4.38) 326 (4.45) 367 (4.48) | 430 (367) |
| 5c | 214 (4.91) 246 (sh, 4.96) 280 (4.70) 293 (4.66) 309 (4.96) 324 (4.31) 355 (4.35) 373 (4.50) | 392 (373) |
| 5d | 212 (4.96) 234 (4.90) 243 (4.93) 252 (4.91) 297 (4.77) 308 (4.63) 351 (4.34) 367 (4.43) | 395 (367) |
| 7a | 214 (4.90) 265 (5.00) 295 (4.48) 343 (sh, 4.80) 359 (4.63) | 395 (359) |
| 7b | 216 (4.92) 258 (4.98) 293 (sh, 4.48) 328 (4.40) 370 (4.67) | 395 (370) |
| 7c | 214 (4.81) 244 (4.95) 283 (4.67) 315 (4.41) 323 (4.40) 357 (4.40) 374 (4.22) | 390 (357) |
| 7d | 214 (4.84) 242 (4.97) 252 (4.94) 297 (4.82) 352 (4.45) 369 (4.61) | 397 (369) |

3. Experimental
General
4. Conclusions
Supplementary Materials
Acknowledgments
Conflicts of Interest
References
- Clarkson, R.G.; Gomberg, M. Spirans with four aromaatic radicals on the spiro carbon atom. J. Am. Chem. Soc. 1930, 52, 2881–2891. [Google Scholar] [CrossRef]
- Haas, G.; Prelog, V. Optisch aktive 9,9'-Spirobifluoren-Derivate. Helv. Chim. Acta 1969, 52, 1202–1218. [Google Scholar] [CrossRef]
- Prelog, V.; Kovačević, M.; Egli, M. Liphophilic tartaric acid esters as enantioselective ionophores. Angew. Chem. Int. Ed. Engl. 1989, 28, 1147–1152. [Google Scholar] [CrossRef]
- Alcázar Montero, V.; Tomlinson, L.; Houk, K.N.; Diederich, F. Selective α,ω-dicarboxylic acid recognition in a chiral cleft shaped by the 9,9'-spirobifluorene unit. Tetrahedron Lett. 1991, 32, 5309–5312. [Google Scholar] [CrossRef]
- Alcázar, V.; Diederich, F. Enantioselective complexation of chiral dicarboxylic acids in clefts of functionalized 9,9'-spirobifluorenes. Angew. Chem. Int. Ed. Engl. 1992, 31, 1521–1523. [Google Scholar] [CrossRef]
- Diederich, F.; Felber, B. Supramolecular chemistry of dendrimers with functional cores. Proc. Natl. Acad. Sci. USA 2002, 99, 4778–4781. [Google Scholar] [CrossRef]
- Weber, E.; Ahrendt, J.; Czugler, M.; Csöregh, I. Selective inclusion and separation of isomeric homologous hydrocarbons by hydrocarbon host lattices. Angew. Chem. Int. Ed. Engl. 1986, 25, 746–748. [Google Scholar] [CrossRef]
- Das, G.; Hamilton, A.D. Carbohydrate recognition: Enantioselective spirobifluorene diphosphonate receptors. Tetrahedron Lett. 1997, 38, 3675–3678. [Google Scholar] [CrossRef]
- Hernández, J.V.; Almaraz, M.; Raposo, C.; Martín, M.; Lithgow, A.; Crego, M.; Caballero, C.; Morán, J.R. Chiral recognition of tartaric acid derivatives with chromenone-benzoisoxazole receptors with a spirobifluorene spacer. Tetrahedron Lett. 1998, 39, 7401–7404. [Google Scholar] [CrossRef]
- Tejeda, A.; Oliva, A.I.; Simón, L.; Grande, M.; Caballero, C.; Morán, J.R. A macrocyclic receptor for the chiral recognition of hydroxycarboxylates. Tetrahedron Lett. 2000, 41, 4563–4566. [Google Scholar] [CrossRef]
- Poriel, C.; Rault-Berthelot, J.; Thirion, D. Modulation of the electronic properties of 3π-2spiro compounds derived from bridged oligophenylenes: A structure-property relationship. J. Org. Chem. 2013, 78, 886–898. [Google Scholar] [CrossRef]
- Huang, J.; Yang, X.; Wang, J.; Zhong, C.; Wang, L.; Qin, J.; Li, Z. New tetraphenylethene-based efficient blue luminophors: Aggregation induced emission and partially controllable emitting color. J. Mater. Chem. 2012, 22, 2478–2484. [Google Scholar] [CrossRef]
- Müller, C.D.; Falcou, A.; Reckefuss, N.; Rojahn, M.; Wiederhirn, V.; Rudati, P.; Frohne, H.; Nuyken, O.; Becker, H.; Meerholz, K. Multicolor organic light-emitting displays by solution processing. Nature 2003, 421, 829–833. [Google Scholar] [CrossRef]
- Grimsdale, A.C.; Chan, K.L.; Martin, R.E.; Jokisz, P.G.; Holmes, A.B. Synthesis of light-emitting conjugated polymers for applications in electroluminescent devices. Chem. Rev. 2009, 109, 897–1091. [Google Scholar] [CrossRef]
- Omer, K.M.; Ku, S.-Y.; Wong, K.-T.; Bard, A.J. Green electrogenerated chemluminescence of highly fluorescent benzothiadiazole and fluorene derivatives. J. Am. Chem. Soc. 2009, 131, 10733–10741. [Google Scholar] [CrossRef]
- Saragi, T.P.I.; Spehr, T.; Siebert, A.; Fuhrmann-Lieker, T.; Salbeck, J. Spiro compounds for organic optoelectronics. Chem. Rev. 2007, 107, 1011–1065. [Google Scholar] [CrossRef]
- Moreau, F.; Audebrand, N.; Poriel, C.; Moizan-Baslé, V.; Ouvry, J. A 9,9′-spirobifluorene based metal–organic framework: Synthesis, structure analysis and gas sorption properties. J. Mater. Chem. 2011, 21, 18715–18722. [Google Scholar] [CrossRef] [Green Version]
- Ferrand, Y.; Poriel, C.; Le Maux, P.; Rault-Berthelot, J.; Simonneaux, G. Asymmetric heterogeneous carbene transfer catalyzed by optically active ruthenium spirobifluorenylporphyrin polymers. Tetrahedron Asymmetry 2005, 16, 1463–1472. [Google Scholar] [CrossRef]
- Murase, S.; Tominaga, T.; Kitazawa, D. Organic Electroluminescent Device. Japan Patent 093425, 2005. [Google Scholar]
- Wu, F.; Riesgo, E.C.; Thummel, R.P.; Juris, A.; Hissler, M.; Elghayoury, A.; Ziessel, R. Closely-spaced chelating centers: Synthesis of novel spiro-bridged bis-phenathrolines and bis-indole derivatives. Tetrahedron Lett. 1999, 40, 7311–7314. [Google Scholar] [CrossRef]
- Liao, L.-S.; Begley, W.J.; Pellow, C.A. Phosphorscent OLED having double-blocking layers having different triplet energies. U.S. Patens Appl. Publ. 191427, 2009. [Google Scholar]
- Tominaga, T.; Kitazawa, D.; Makiyama, A.; Kohama, A. Light-emitting device material and light-emitting devices. PCT Int. Appl. WO 043449, 2005. [Google Scholar]
- Jahng, Y.; Rahman, A.F.M.M. Synthesis and properties of 2,2'-Di(heteroaryl)-9,9'-spirobifluorenes. Bull. Chem. Soc. Jpn. 2010, 83, 672–677. [Google Scholar] [CrossRef]
- Qu, Y.-W.; Shi, K.-H.; Liu, Q.-C. Synthesis of quinolinyl spirobifluorene derivatives. Chin. J. Synth. Chem. 2008, 16, 15–18. [Google Scholar]
- Chea, J.M.; Jahng, Y. Synthesis and properties of 9,9'-spirobifluorene-based heterocycles. Heterocycles 2009, 78, 1573–1580. [Google Scholar] [CrossRef]
- Opie, J.W.; Smith, L.I. o-Aminobenzaldehyde. Org. Synth. 1955, 56–58. [Google Scholar]
- Majewicz, G.; Caluwe, O.A. Facile synthesis of 2-aminonicotinaldehyde. J. Org. Chem. 1974, 39, 720–721. [Google Scholar] [CrossRef]
- Riesgo, E.C.; Jin, X.; Thummel, R.P. Introduction of benzo[h]quinoline and 1,10-phenanthroline subunits by friedländer methodology. J. Org. Chem. 1996, 61, 3017–3022. [Google Scholar] [CrossRef]
- Thirion, D.; Poriel, C.; Métivier, R.; Rault-Berthelot, J.; Barrière, F.; Jeannin, O. Violet-to-blue tunable emission of aryl-substituted dispirofluorene–indenofluorene isomers by conformationally-controllable intramolecular excimer formation. Chem. Eur. J. 2011, 17, 10272–10287. [Google Scholar] [CrossRef]
- Pretsch, E.; Seibl, J.; Simon, W. Tables of Spectral Data for Structural Determination of Organic Compounds, 2nd ed.; Springer-Verlag: Heidelberg, Germany, 1989. [Google Scholar]
- Sample Availability: Samples of the compounds 5a–d and 7a–d are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Liang, J.L.; Cha, H.; Jahng, Y. Synthesis and Properties of Annulated 2-(Azaar-2-yl)- and 2,2'-Di(azaar-2-yl)-9,9'-spirobifluorenes. Molecules 2013, 18, 13680-13690. https://doi.org/10.3390/molecules181113680
Liang JL, Cha H, Jahng Y. Synthesis and Properties of Annulated 2-(Azaar-2-yl)- and 2,2'-Di(azaar-2-yl)-9,9'-spirobifluorenes. Molecules. 2013; 18(11):13680-13690. https://doi.org/10.3390/molecules181113680
Chicago/Turabian StyleLiang, Jing Lu, Hyochang Cha, and Yurngdong Jahng. 2013. "Synthesis and Properties of Annulated 2-(Azaar-2-yl)- and 2,2'-Di(azaar-2-yl)-9,9'-spirobifluorenes" Molecules 18, no. 11: 13680-13690. https://doi.org/10.3390/molecules181113680




