Synthesis of New Macrocyclic Polyamides as Antimicrobial Agent Candidates
Abstract
:1. Introduction
2. Results and Discussion
2.1. Chemistry
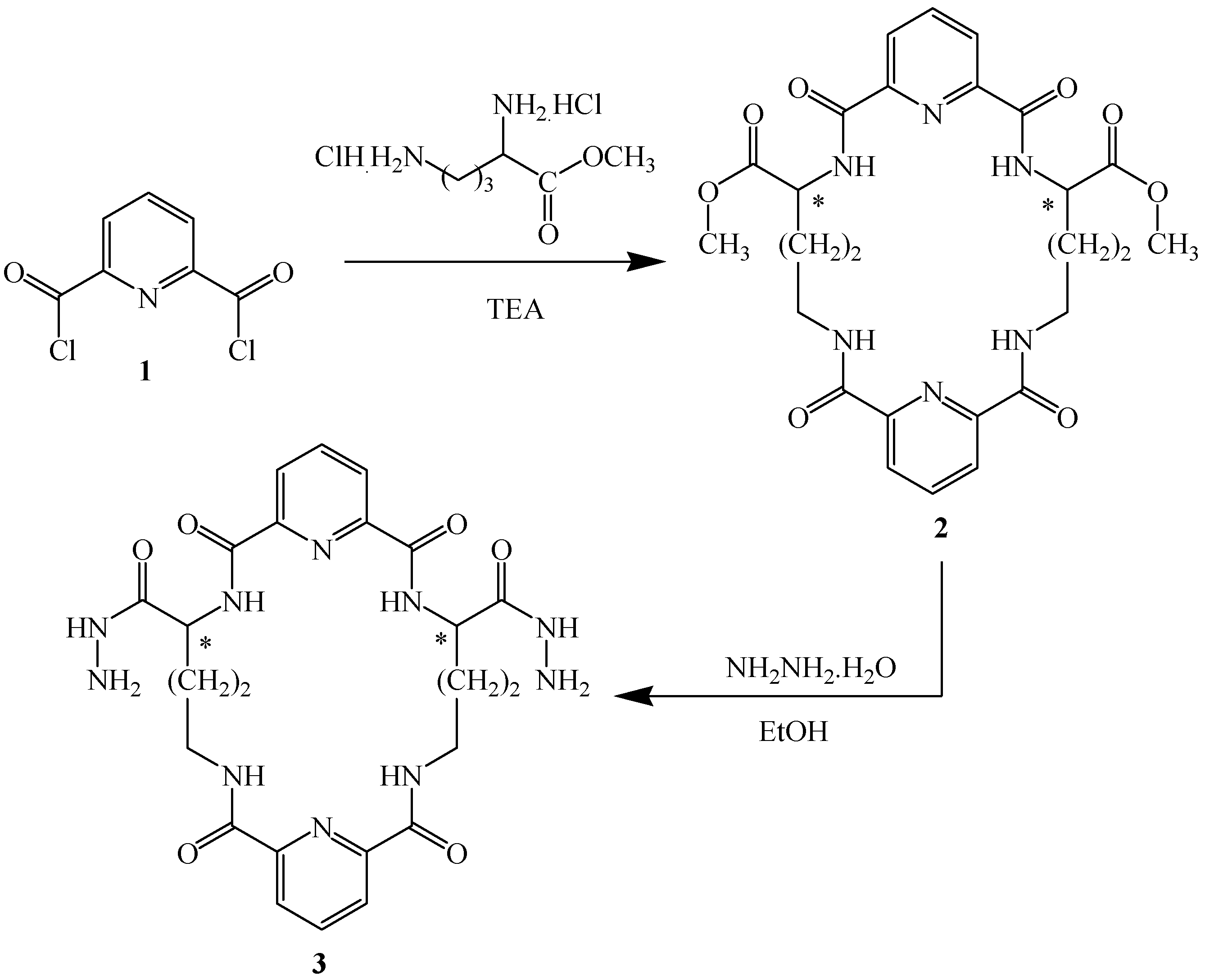
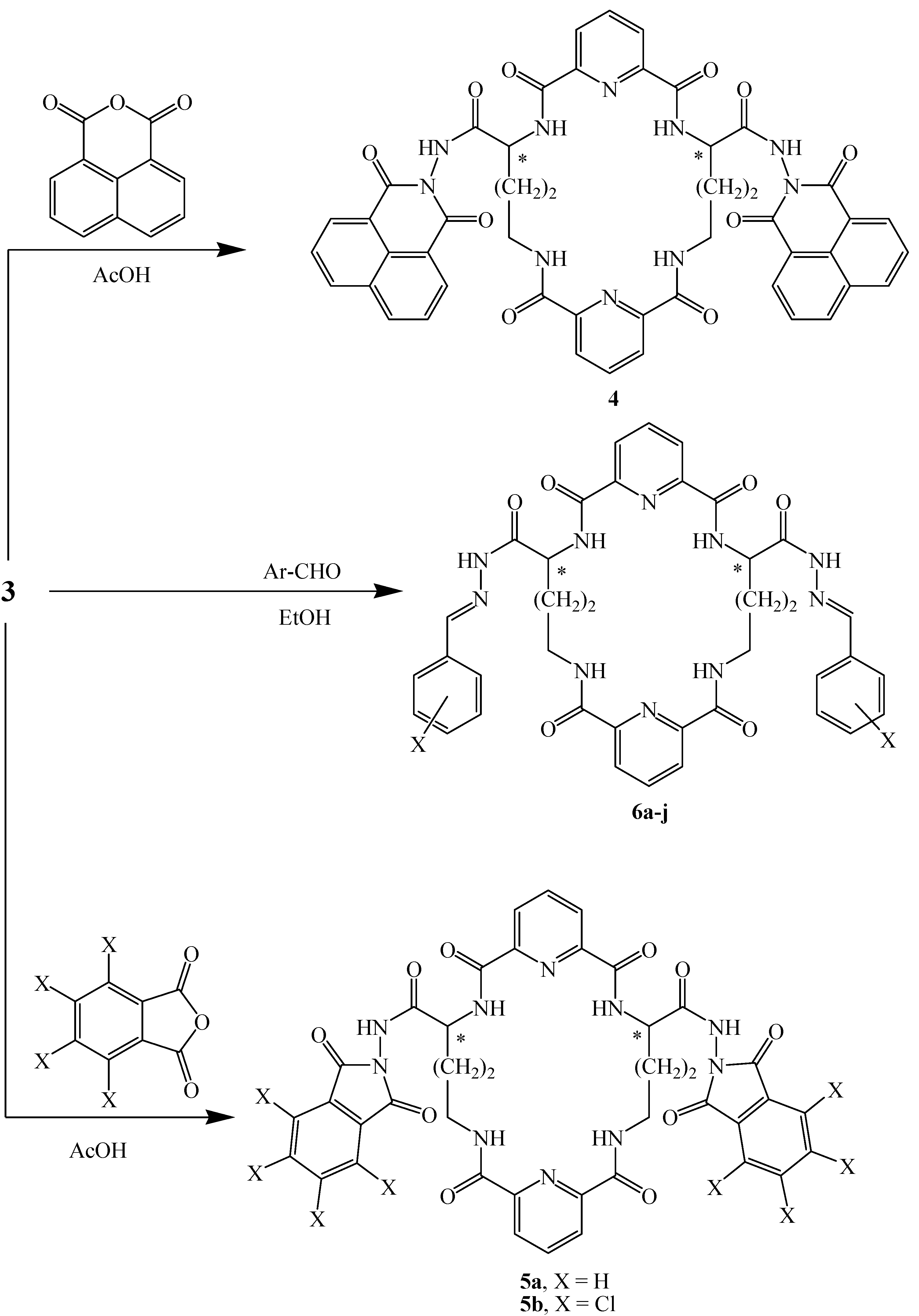
| Comp. No. | X | M.p. (°C) | Cryst. Solv. | Yield (%) | Molecular Formula (Mol. Wt.) |
|---|---|---|---|---|---|
| 4 | - | 276–278 | DMF/H2O | 65 | C48H38N10O10 (914.87) |
| 5a | H | 243–245 | DMF/H2O | 72 | C40H34N10O10 (814.75) |
| 5b | Cl | 296–298 | DMF/H2O | 88 | C40H26Cl8N10O10 (1090.31) |
| 6a | H | 178–180 | EtOH/Ether | 85 | C38H38N10O6 (730.77) |
| 6b | 3-Br | 232–234 | MeOH | 79 | C38H36Br2N10O6 (888.56) |
| 6c | 4-Br | 254–256 | Dioxane | 87 | C38H36Br2N10O6 (888.56) |
| 6d | 2,6-Cl2 | 198–200 | EtO | 68 | C38H34Cl4N10O6 (868.55) |
| 6e | 3,4-Cl2 | 188–190 | EtOH/Ether | 78 | C38H34Cl4N10O6 (868.55) |
| 6f | 2-Cl-6-F | 168–170 | AcOH/H2O | 84 | C38H34Cl2F2N10O6 (835.64) |
| 6g | 4-CH3 | 155–157 | EtOH/H2O | 82 | C40H42N10O6 (758.82) |
| 6h | 2-OCH3 | 210–212 | AcOH/H2O | 90 | C40H42N10O8 (790.82) |
| 6i | 4-OCH3 | 216–218 | EtOH/H2O | 80 | C40H42N10O8 (790.82) |
| 6j | 3,4,5-(OCH3)3 | 235–257 | AcOH/H2O | 75 | C44H50N10O12 (910.92) |
2.2. Antimicrobial Testing
| Comp. No. | Inhibition zone (mm) | ||||
|---|---|---|---|---|---|
| Gram +ve bacteria | Gram −ve bacteria | Fungi | |||
| B. subtilis | Staph. aureus | E. coli | C. albicans | A. niger | |
| 3 | 1.75 | 1.55 | 0.80 | - | 1.65 |
| 4 | 1.55 | 1.60 | 0.80 | 0.65 | 1.60 |
| 5a | 1.35 | 1.75 | 0.60 | 0.75 | 1.90 |
| 5b | 1.80 | 1.48 | - | - | 1.65 |
| 6a | 1.20 | 1.70 | - | - | 1.85 |
| 6b | 1.45 | 1.45 | 0.80 | - | 1.60 |
| 6c | 0.90 | 1.30 | - | 0.65 | 1.70 |
| 6d | 1.80 | 1.25 | 0.60 | - | 1.80 |
| 6e | 1.75 | 0.85 | - | - | 1.75 |
| 6f | 1.30 | 1.50 | - | 0.70 | 2.05 |
| 6g | 0.85 | 1.30 | 0.75 | 0.65 | 1.95 |
| 6h | 1.80 | 1.25 | 0.70 | 0.60 | 1.65 |
| 6i | 1.70 | 1.20 | 0.75 | - | 1.55 |
| 6j | 1.60 | 1.45 | 0.70 | 0.55 | 1.75 |
| Ampicillin | 1.15 | 1.30 | 0.75 | - | - |
| Ketaconazole | - | - | - | 0.80 | 2.30 |
3. Experimental
3.1. General
3.2. Chemistry
3.3. Antimicrobial Testing
4. Conclusions
Acknowledgements
References
- Krakowiak, K.E.; Bradshaw, J.S.; Zamecka-Krakowiak, D.J. Synthesis of aza-crown ethers. Chem. Rev. 1989, 89, 929–972. [Google Scholar] [CrossRef]
- Bradshaw, J.S.; Krakowiak, K.E.; Izatt, R.M. Preparation of diamino ethers and polyamines. Tetrahedron 1992, 48, 4475–4515. [Google Scholar] [CrossRef]
- Bradshaw, J.S.; Krakowiak, K.E.; Izatt, R.M. Azacrown Macrocycles. In The Chemistry of Heterocyclic Compounds; Taylor, E.C., Ed.; John Wiley & Sons: New York, NY, USA, 1993; p. 51. [Google Scholar]
- Molina, P.; Tarraga, A.; Gaspar, C.; Espinosa, A. Synthesis of a novel class of macrocyclic compounds containing 1,3,4-thiadiazole rings as subunits. J. Org. Chem. 1994, 59, 3665–3669. [Google Scholar] [CrossRef]
- Izatt, R.M.; Pawlak, K.; Bradshaw, J.S.; Bruening, R.L. Thermodynamic and kinetic data for macrocycle interaction with cations, anions, and neutral molecules. Chem. Rev. 1995, 95, 2529–2586. [Google Scholar] [CrossRef]
- Izatt, R.M.; Pawlak, K.; Bradshaw, J.S.; Bruening, R.L.; Tarbet, B. Thermodynamic and kinetic data for macrocycle interaction with neutral molecules. J. Chem. Rev. 1992, 92, 1261–1354. [Google Scholar] [CrossRef]
- Elwahy, A.H.M. New trends in the chemistry of condensed heteromacrocycles Part A: Condensed azacrown ethers and azathiacrown ethers. J. Heterocycl. Chem. 2003, 40, 1–23. [Google Scholar] [CrossRef]
- Chu, T.D.W.; Plattner, J.J.; Kotz, L. New directions in antibacterial research. J. Med. Chem. 1996, 39, 3853–3874. [Google Scholar] [CrossRef]
- Hirschmann, R.; Smith, A.B.; Sprengeler, P.A. New Perspectives in Drug Design; Academic: New York, NY, USA, 1995. [Google Scholar]
- Amr, A.E.; Mohamed, A.M.; Ibrahim, A.A. Synthesis of some new chiral tricyclic and macrocyclic pyridine derivatives as antimicrobial agents. Z. Naturforsch. 2003, 58b, 861–868. [Google Scholar]
- Amr, A.E. Synthesis of some new linear and chiral macrocyclic pyridine carbazides as analgesic and anticonvulsant agents. Z. Naturforsch. 2005, 60b, 990–998. [Google Scholar]
- Abou-Ghalia, M.H.; Amr, A.E.; Abdalah, M.M. Synthesis of some new (Nα-dipicolinoyl)-bis-L-leucyl-DL-norvalyl linear tetra and cyclic octa bridged peptides as new anti-inflammatory agents. Z. Naturforsch. 2003, 58b, 903–910. [Google Scholar]
- Amr, A.E.; Abo-Ghalia, M.H.; Abdalah, M.M. Synthesis of novel macrocyclic peptido-calix[4]arenes and peptidopyridines as precursors for potential molecular metallacages, chemosensors and biologically active candidates. Z. Naturforsch. 2006, 61b, 1335–1345. [Google Scholar]
- Amr, A.E.; Abo-Ghalia, M.H.; Abdalah, M.M. Synthesis of new (Nα-dipicolinoyl)-bis-L-valyl-L-phenylalanyl linear and macrocyclic bridged peptides as anti-inflammatory agents. Arch. Pharm. Chem. Life Sci. 2007, 340, 304–309. [Google Scholar] [CrossRef]
- Hassan, S.S.M.; Abo-Ghalia, M.H.; Amr, A.E.; Mohamed, A.H.K. New lead (II) selective membrane potentiometric sensors based on chiral 2,6-bis-pyridinecarboximide derivatives. Talanta 2003, 60, 81–91. [Google Scholar] [CrossRef]
- Hassan, S.S.M.; Abo-Ghalia, M.H.; Amr, A.E.; Mohamed, A.H.K. Novel thiocyanate-selective membrane sensors based on di-, tetra-, and hexaimidopyridine ionophores. Anal. Chem. Acta 2003, 482, 9–18. [Google Scholar] [CrossRef]
- Al-Mohizea, A.M.; Al-Omar, M.A.; Abdalla, M.M.; Amr, A.E. 5α-Reductase inhibitors, antiviral and anti-tumor activities of some steroidal cyanopyridinone derivatives. Int. J. Biol. Macromol. 2012, 50, 171–179. [Google Scholar] [CrossRef]
- Abdalla, M.M.; Al-Omar, M.A.; Bhat, M.A.; Amr, A.E.; Al-Mohizea, A.M. Steroidal pyrazolines evaluated as aromatase and quinone reductase-2 inhibitors for chemoprevention of cancer. Int. J. Biol. Macromol. 2012, 50, 1127–1132. [Google Scholar] [CrossRef]
- Fakhr, I.M.; Amr, A.E.; Sabry, N.M.; Abdalah, M.M. Anti-inflammatory and analgesic activities of newly synthesized chiral peptide derivatives using (3-benzoyl-4,5-dioxo-2-phenyl-pyrrolidin-1-yl)acetic acid ethyl ester ass material. Arch. Pharm. Chem. Life Sci. 2008, 341, 174–180. [Google Scholar] [CrossRef]
- Amr, A.E. Anticonvulsant and antiparkinsonian evaluation of some synthesized chiral peptide derivatives using 3-benzoyl-4,5-dioxo-2-phenylpyrrolidin-1-yl acetic acid. World J. Chem. 2010, 5, 1–6. [Google Scholar]
- Al-Salahi, R.A.; Al-Omar, M.A.; Amr, A.E. Synthesis of chiral macrocyclic or linear pyridine carboxamides from pyridine-2,6-dicarbonyl dichloride as antimicrobial agents. Molecules 2010, 15, 6588–6597. [Google Scholar] [CrossRef]
- Al-Omar, M.A.; Amr, A.E. Synthesis of some new pyridine-2,6-carboxamide-derived Schiff bases as potential antimicrobial agents. Molecules 2010, 15, 4711–4721. [Google Scholar] [CrossRef]
- Attia, A.; Abdel-Salam, O.I.; Amr, A.E.; Stibor, I.; Budesinsky, M. Synthesis and antimicrobial activity of some new chiral bridged macrocyclic pyridines. Egypt. J. Chem. 2000, 43, 187–201. [Google Scholar]
- Amr, A.E.; Abdel-Salam, O.I.; Attia, A.; Stibor, I. Synthesis of new potential bis-intercallators based on chiral pyridine-2,6-dicarbox-amides. Collect. Czech. Chem. Commun. 1999, 64, 288–298. [Google Scholar] [CrossRef]
- Boxworth, D.M. The treatment of tuberculous lesions of bones and joints with iproniazid (marsilid). N.Y. State J. Med. 1956, 56, 1281–1284. [Google Scholar]
- Schroder, H.; Lubke, K. Synthesis, Occurrence and Action of Biologically Active Peptides, Volume II, Translated by Erhard Gross; Academic Press: New York, NY, USA and London, UK, 1966; p. 405. [Google Scholar]
- Rollas, S.; Küçükgüzel, S.G. Biological activities of hydrazone derivatives. Molecules 2007, 12, 1910–1939. [Google Scholar] [CrossRef]
- National Committee for Clinical Laboratory for Standards (NCCLS), Performance Standards for Antimicrobial Testing, Eighth information supplement; Publication No. NCCLS M 100–58; NCCLS: Villanova, PA, USA, 1998.
- National Committee for Clinical Laboratory for Standards (NCCLS), Methods for Determining Bactericidal Activity of Antimicrobial Agents, Tentative guidelines; Publication No. NCCLS M26-T; NCCLS: Villanova, PA, USA, 1992.
- Furtado, G.L.; Medeiros, A.A. Single-disk diffusion testing (Kirby-Bauer) of susceptibility of Proteus mirabilis to chloramphenicol: Significance of the intermediate category. J. Clin. Microbiol. 1980, 12, 550–553. [Google Scholar]
- Sample Availability: Samples of the compounds are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
El-Salam, O.I.A.; Al-Omar, M.A.; Fayed, A.A.; Flefel, E.M.; Amr, A.E.-G.E. Synthesis of New Macrocyclic Polyamides as Antimicrobial Agent Candidates. Molecules 2012, 17, 14510-14521. https://doi.org/10.3390/molecules171214510
El-Salam OIA, Al-Omar MA, Fayed AA, Flefel EM, Amr AE-GE. Synthesis of New Macrocyclic Polyamides as Antimicrobial Agent Candidates. Molecules. 2012; 17(12):14510-14521. https://doi.org/10.3390/molecules171214510
Chicago/Turabian StyleEl-Salam, Osama I. Abd, Mohamed A. Al-Omar, Ahmed A. Fayed, Eman M. Flefel, and Abd El-Galil E. Amr. 2012. "Synthesis of New Macrocyclic Polyamides as Antimicrobial Agent Candidates" Molecules 17, no. 12: 14510-14521. https://doi.org/10.3390/molecules171214510
APA StyleEl-Salam, O. I. A., Al-Omar, M. A., Fayed, A. A., Flefel, E. M., & Amr, A. E.-G. E. (2012). Synthesis of New Macrocyclic Polyamides as Antimicrobial Agent Candidates. Molecules, 17(12), 14510-14521. https://doi.org/10.3390/molecules171214510




