A Regioselective Synthesis of E-Guggulsterone
Abstract
:1. Introduction
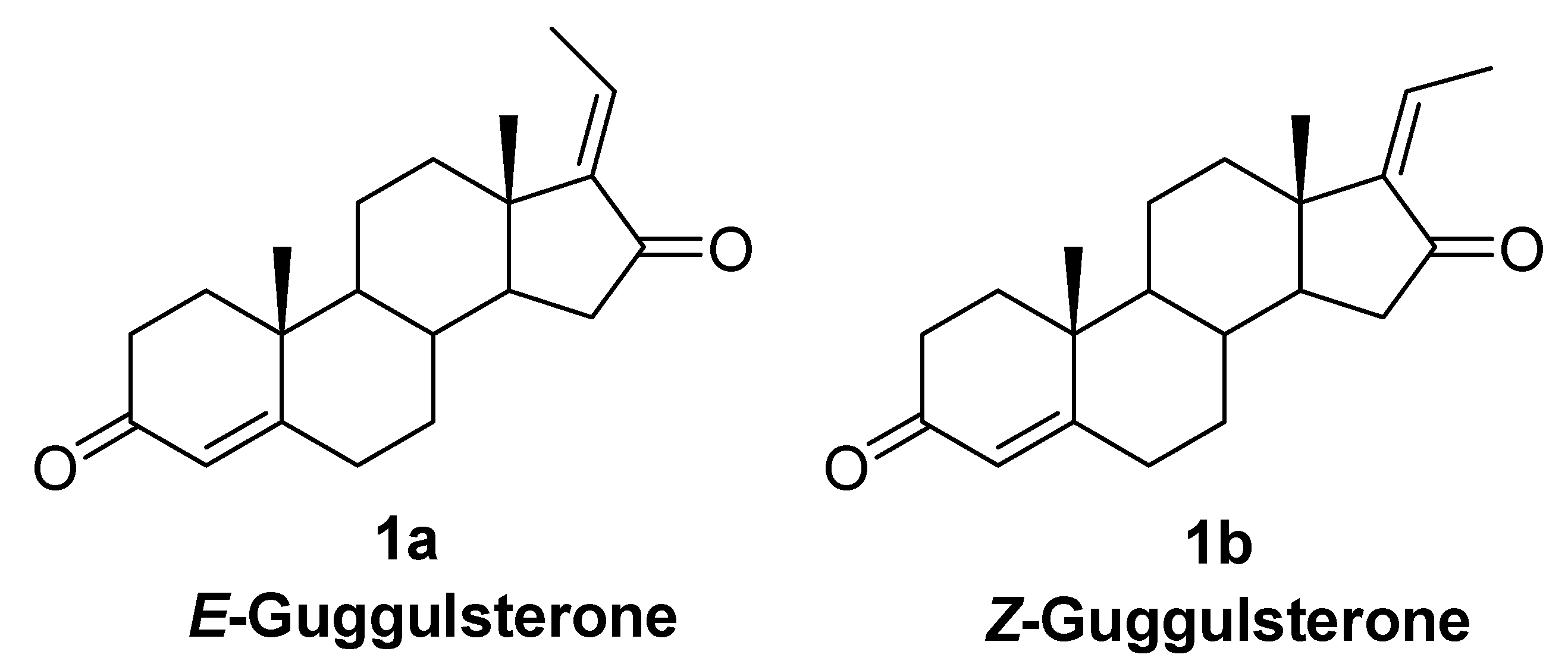
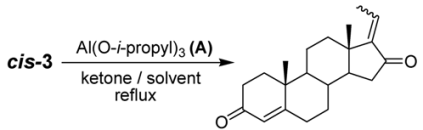
2. Results and Discussion
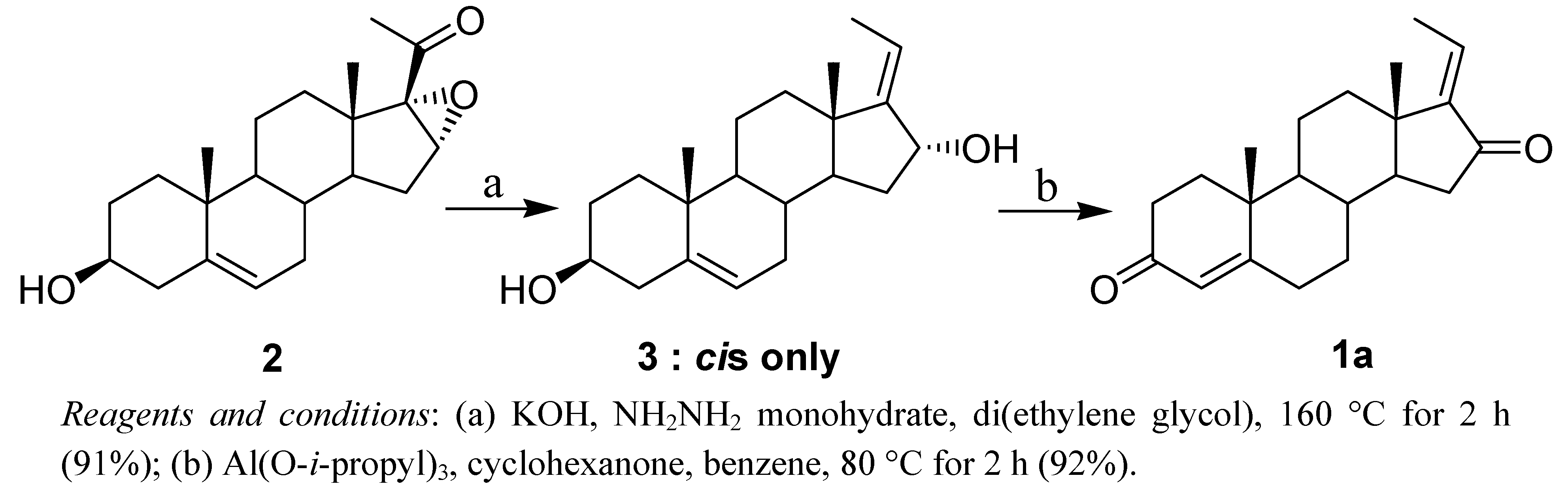
| Entry | Equiv of A | Ketone | Solvent | Time (h) | % Yield b | E:Z Ratio c |
|---|---|---|---|---|---|---|
| 1 | 0.5 | cyclohexanone | toluene | 2 | 94 | 67:33 |
| 2 | 1.0 | cyclohexanone | toluene | 2 | 96 | 86:14 |
| 3 | 0.5 | cyclohexanone | benzene | 2 | 92 | only E |
| 4 d | 1.0 | acetone | benzene | 7 | no reaction | - |
| 5 | 0.5 | 2-butanone | benzene | 4 | 32 | only E |
| Entry | Driving force | Solvent/Temp. | Time (h) | E(1a):Z(1b) Ratio b |
|---|---|---|---|---|
| 1 | heat | toluene/110 °C | 2 | 95:5 |
| 2 | heat | mesitylene/170 °C | 2 | 65:35 |
| 3 | heat | toluene/sealed tube,140 °C | 2 | 45:55 |
| 4 | light c | MeOH/25 °C | 12 | 50:50 |
| 5 | p-TsOH | benzene/80 °C | 1 | 40:60 |
| 6 | 2 N-HCl | acetonitrile/36 °C | 18 | 60:40 |
3. Experimental
3.1. General
4. Conclusions
Acknowledgments
References and Notes
- Patil, V.D.; Nayak, U.R.; Dev, S. Chemistry of Ayurvedic crude drugs. Tetrahedron 1972, 28, 2341–2352. [Google Scholar] [CrossRef]
- Satyavati, G.V. Gum guggul (Commiphora mukul) - the success story of an ancient insight leading to a modern discovery. Indian J. Med. Res. 1988, 87, 327–335. [Google Scholar]
- Dev, S. Ethno therapeutics and modern drug development: The potential of auerveda. Curr. Sci. 1997, 73, 909–928. [Google Scholar]
- Mesrob, B.; Nesbitt, C.; Misra, R.; Pandey, R.C. High-performance liquid chromatographic method for fingerprinting and quantitative determination of E- and Z-guggulsterones in Commiphora mukul resin and its products. J. Chromatogr. B 1998, 720, 189–196. [Google Scholar]
- Nityanand, S.; Kapoor, N.K. Cholesterol lowering activity of the various fractions of guggul. Indian J. Exp. Biol. 1973, 11, 395–398. [Google Scholar]
- Nityanand, S.; Srivastava, J.S.; Asthana, O.P. Clinical trials with gugulipid: A new hypolipidaemic agent. J. Assoc. Physicians India 1989, 37, 323–328. [Google Scholar]
- Singh, R.B.; Niaz, M.A.; Ghosh, S. Hypolipidemic and antioxidant effects of Commiphora mukul as an adjunct to dietary therapy in patients with hypercholesterolemia. Cardiovasc. Drugs Ther. 1994, 8, 659–664. [Google Scholar] [CrossRef]
- Wang, X.; Greilberger, J.; Ledinski, G.; Kager, G.; Paigen, B.; Jurgens, G. The hypolipidemic natural product Commiphora mukul and its component guggulsterone inhibit oxidative modification of LDL. Atherosclerosis 2004, 172, 239–246. [Google Scholar]
- Cheon, J.H.; Kim, J.S.; Kim, J.M.; Kim, N.; Jung, H.C.; Song, S.S. Plant sterol guggulsterone inhibits nuclear factor-kappaB signaling in intestinal epithelial cells by blocking IkappaB kinase and ameliorates acute murine colitis. Inflamm. Bowel Dis. 2006, 12, 1152–1161. [Google Scholar]
- Singh, S.V.; Choi, S.; Zeng, Y.; Hahm, E.R.; Xiao, D. Guggulsterone-induced apoptosis in human prostate cancer cells is caused by reactive oxygen intermediate-dependent activation of c-Jun NH2-terminal kinase. Cancer Res. 2007, 67, 7439–7449. [Google Scholar]
- Benn, W.R.; Dodson, R.M. The Synthesis and Stereochemistry of Isomeric 16-Hydroxy-17(20)-pregnenes. J. Org. Chem. 1964, 29, 1142–1148. [Google Scholar] [CrossRef]
- Owsley, E.; Chiang, J.Y.L. Guggulsterone antagonizes farnesoid X receptor induction of bile salt export pump but activates pregnane X receptor to inhibit cholesterol 7alpha-hydroxylase gene. Biochem. Bioph. Res. Commun. 2003, 304, 191–195. [Google Scholar]
- Hamied, Y.K. A Process for the preparation of pharmacologically active synthetic Z and E steroisomeric mixture of guggulsterones. Eur. Patent 0447706 A1.
- Kesser, S.V.; Rampal, A.L. Synthetic studies in steroidal sapogenins and alkaloids. III. Synthesis and stereochemistry of isomeric 16-hydroxy- and 16-oxo-5,17(20)-pregnadien-3-ols. Tetrahedron 1968, 24, 887–892. [Google Scholar]
- Djerassi, C. Oppenauer oxidation. Org. React. 1951, VI, 207–272. [Google Scholar]
- de Graauw, C.F.; Peters, J.A.; van Bekkum, H.; Huskens, J. Meerwein-Ponndorf-Verley Reductions and Oppenauer Oxidations: An Integrated Approach. Synthesis 1994, 10, 1007–1017. [Google Scholar]
- Sample Availability: E-Guggulsterone is available from the authors.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ham, J.; Chin, J.; Kang, H. A Regioselective Synthesis of E-Guggulsterone. Molecules 2011, 16, 4165-4171. https://doi.org/10.3390/molecules16054165
Ham J, Chin J, Kang H. A Regioselective Synthesis of E-Guggulsterone. Molecules. 2011; 16(5):4165-4171. https://doi.org/10.3390/molecules16054165
Chicago/Turabian StyleHam, Jungyeob, Jungwook Chin, and Heonjoong Kang. 2011. "A Regioselective Synthesis of E-Guggulsterone" Molecules 16, no. 5: 4165-4171. https://doi.org/10.3390/molecules16054165