Introduction
Melanin is the general term assigned to a group of phenolic pigments produced from tyrosine and related compounds by an oxidative polymerization. Tyrosinase (EC 1. 14. 18. 1), widely distributed in Nature, is a copper-containing enzyme that catalyzes two different reactions: a hydroxylation of monophenols to
o-diphenols (monophenolase activity) and an oxidation of
o-diphenols to
o-quinones (diphenolase activity), both using molecular oxygen [
1]. It is well known that tyrosinase is a key enzyme for melanin biosynthesis in plants and animals. Therefore, tyrosinase inhibitors have been reported and tested as therapeutic agents for the treatment of melanin hyperpigmentation and as cosmetic materials for whitening after sunburn [
2]. Melanin formation is also considered to be deleterious to the color quality of fruits and vegetables, so an inhibition of this enzyme should be useful for browning control of plant-derived foods in the food industry [
3]. Furthermore, tyrosinase is one of the most important key enzymes in insect molting process, and investigating its inhibitors may be important in finding alternative insect control agents [
4,
5].
Many efforts have been addressed to the search for effective and safe tyrosinase inhibitors from natural materials, and flavonoids are one of the most numerous and best-studied group of plant polyphenols. It is reported that some flavonoids inhibited tyrosinase activity by active chelation and others acted as cofactors and/or substrates towards tyrosinase [
6,
7,
8,
9]. Baicalein, 5,6,7-trihydroxyflavone (
1), originally isolated from
Scutellaria baicalensis, has reportedly been used as a depigmentation agent in cosmetics [
10,
11]. In the previous study, the inhibitory effects by
1 on tyrosinase and melanin formation in skin were evaluated, but the structure-activity relationship of
1 towards tyrosinase remains unknown.
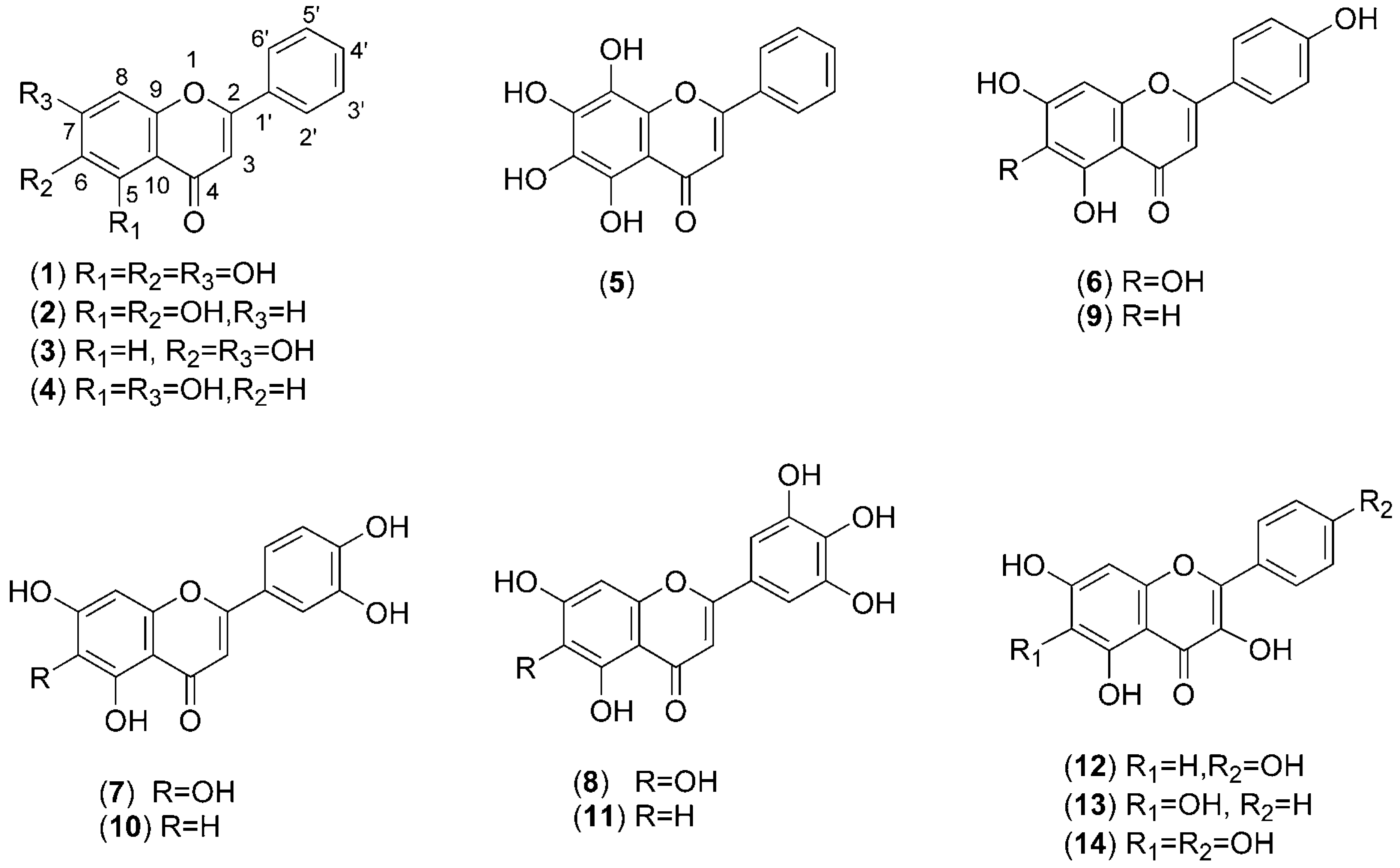
Baicalein (
1,
Figure 1) is a unique flavone bearing a 6-hydroxyl group together with the standard 5,7-dihydroxyl substituents, and it shows a variety of biological and pharmacological effects including antioxidant [
12], antimutagenic [
13], anti-inflammatory [
14], antimicrobial [
15], and anticarcinogenic properties [
16]. We previously reported the inhibitory effect and mechanism of the interaction of
1 and related 5,6,7-trihydroxyflavones towards rat intestinal α-glucosidase [
17,
18,
19,
20,
21]. As a part of our research on the new biological and physiological properties of 6-hydroxyflavones, we are interested in the inhibitory effect on melanin formation by
1 and its related compounds. In order to evaluate the inhibitory effect on melanin biosynthesis by 5,6,7-trihydroxyflavones, we synthesized a series of natural hydroxyflavones (
Figure 1) and conducted the structure-activity relationship study for their tyrosinase inhibition, together with demonstrating the inhibitory mechanism in the present study. In addition, we evaluated their antioxidant potency, which can contribute to prevent pigmentation due to non-enzymatic auto-oxidation.
Results and Discussion
Inhibitory effects on
l-DOPA oxidizing diphenolase activity of tyrosinase of baicalein (
1) and related hydroxyflavones
2-
14 are summarized in
Table 1. At a concentration of 400 μM,
1 inhibited 69% of tyrosinase activity with respect to
l-DOPA as a substrate, the where 50% inhibitory concentration (IC
50) was calculated to be 273 μM. On the other hand,
4, which lacks a 6-hydroxyl group, did not show any activity. Hence, this result indicates that the 6-hydroxyl substituent of
1 is an important factor for the tyrosinase inhibition activity. Further evidence supporting this rationale is that, compounds
9-
11, all of which do not possess 6-hydroxyl group, showed no activity; however,
6,
7 and
8, all of which possess 6-hydroxyl group, showed a higher activity than their unsubstituted counterparts,
9,
10 and
11. Furthermore, removal of any hydroxyl group at positions 5,6,7 (eg.
2 and
3) led to a dramatic loss of potency, and compounds
9-
11 having no 5,6,7-trihydroxyflavone structure also did not inhibit tyrosinase activity. Hence, these results indicate that all of the three hydroxyl substituents of
1 are important for an inhibition of the tyrosinase activity.
Figure 1.
Chemical structures of baicalein (1) and related hydroxyflavones 2-14.
Figure 1.
Chemical structures of baicalein (1) and related hydroxyflavones 2-14.
Table 1.
Mushroom tyrosinase inhibitory activities of hydroxyflavones (1-14).
Table 1.
Mushroom tyrosinase inhibitory activities of hydroxyflavones (1-14).
| compound | | | IC50 (μM) | |
| | diphenolasea | | | monophenolaseb |
| 1 | | 273 | | cofactor |
| 2 | | NIc | | NT |
| 3 | | NI | | NT |
| 4 | | NI | | NT |
| 5 | | NI | | NT |
| 6 | | 193 | | cofactor |
| 7 | | 400 (38%) | | NT |
| 8 | | 400 (43%) | | NT |
| 9 | | NI | | NT |
| 10 | | NI | | NT |
| 11 | | 400 (33%) | | NT |
| 12 | | 262 | | cofactor |
| 13 | | 182 | | NT |
| 14 | | 124 | | cofactor |
Having identified the importance of the 5,6,7-trihydroxyl group of 1, we decided to explore the incorporation of an additional hydroxyl group onto position 8 of compound 1 to afford 5. Unexpectedly, this led to a reduction in potency. Among the B-ring-hydroxylated 5,6,7-trihydroxyflavones, it is interesting to note that the highest activity was observed for monohydroxylated 6 (IC50=193 μM), compared to dihydroxylated 7 (38% inhibition at the concentration of 400 μM) and trihydroxylated 8 (43% inhibition at the concentration of 400 μM). Hence, it was suggested that the enhanced inhibitory activity of 1 is solely due to the 4’-hydroxylation of the B-ring, and extra hydroxyl group substituents of the A- or B-rings of 1 are not always advantageous. It is unclear, however, why the additive effect of the 4’-OH is different from that of the other hydroxyl substitution.
Kubo and coworkers have reported the diphenolase-inhibitory activity of flavonols, and indicated the importance of a hydroxyl group at position 3 [
6]. In order to estimate the effect on the inhibitory activity against diphenolase of the introduction of a 3-hydroxyl group in
1, flavonols
12-
14 were tested (
Table 1). Compounds
13 (IC
50=182 μM) and
14 (IC
50=124 μM) showed higher activity than that of
1, while even
12 (IC
50=262 μM) showed the comparable activity to
1, which validates the importance of the 3-hydroxyl group of
1 for the activity as well as the basic 5,6,7-trihydroxyflavone structure.
Having demonstrated the diphenolase-inhibitory activity of 5,6,7-trihydroxyflavones
1,
6,
13 and
14, we then evaluated their
l-tyrosine oxidizing monophenolase activity of mushroom tyrosinase. Monophenolase activity of tyrosinase is characterized by a lag time, derived from oxidative hydroxylation of monophenolic substrates to
o-diphenols. As shown in
Figure 2, the lag time was not observed for
1,
6,
13 or
14. It was reported that quercetin (
o-diphenols) acted as an alternative cofactor to initiate monophenolase activity [
7]. Judging from the structure of
1,
6,
13 or
14 (which possesses 5,6,7-trihydroxyflavone structure), we can conclude that
1,
6,
13 or
14 are co-factors to tyrosinase monophenolase.
Figure 2.
Lag period of l-dopaquinone formation. l-tyrosine (0.8 mM) was incubated with tyrosinase in the absence (control) or with the addition of baicalein (1, 320 μM), 6-hydroxyapigenin (6, 200 μM), 6-hydroxygalangin (13, 320 μM) and 6-hydroxykaempferol (14, 100 μM).
Figure 2.
Lag period of l-dopaquinone formation. l-tyrosine (0.8 mM) was incubated with tyrosinase in the absence (control) or with the addition of baicalein (1, 320 μM), 6-hydroxyapigenin (6, 200 μM), 6-hydroxygalangin (13, 320 μM) and 6-hydroxykaempferol (14, 100 μM).
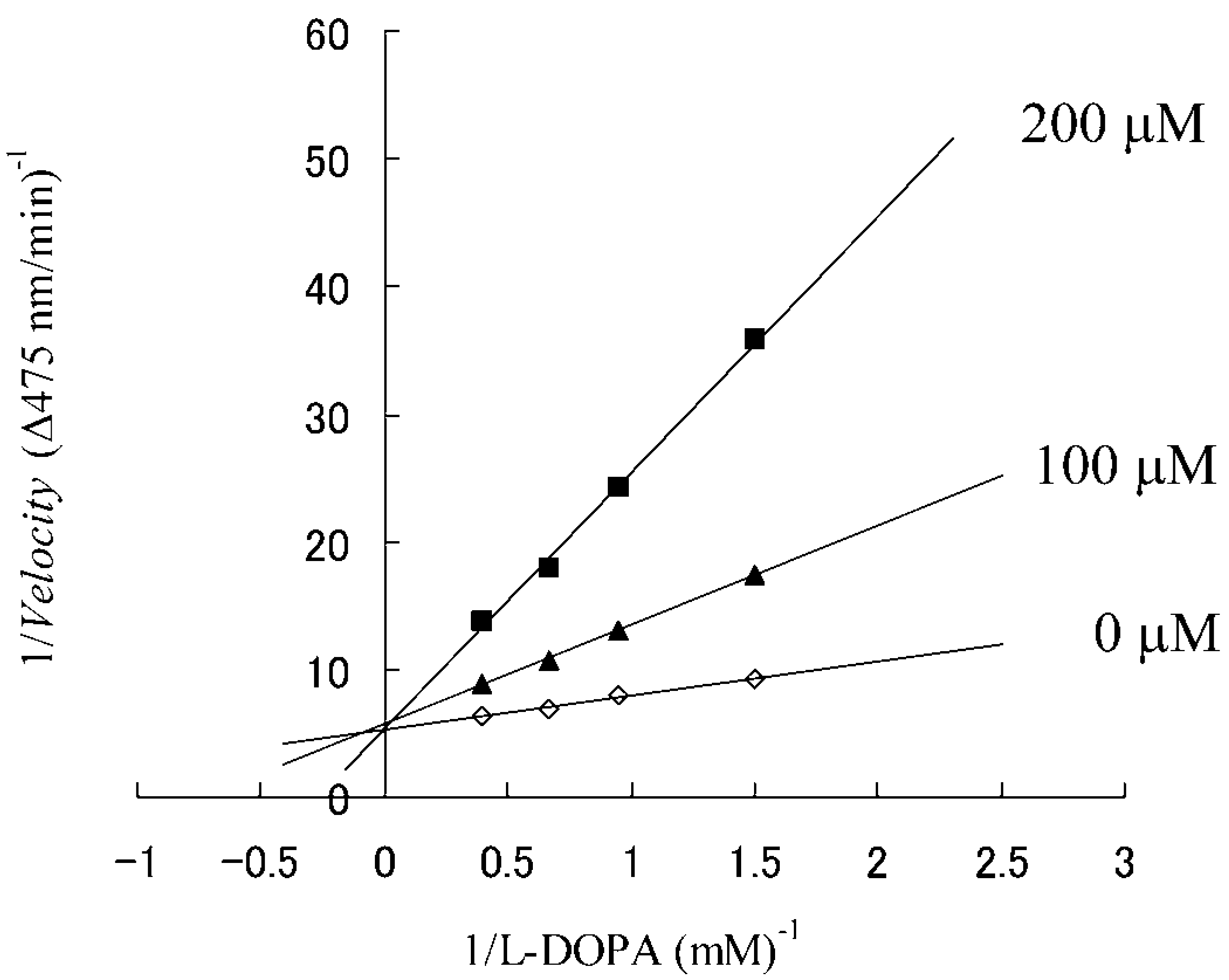
Among the tested hydroxyflavones
1-
14, compound
14 showed the highest diphenolase-inhibitory activity, and hence, we carried out the steady-state kinetic analysis of
14 for tyrosinase inhibition with respect to
l-DOPA as a substrate. Lineweaver-Burk plots for the inhibition of tyrosinase (diphenolase) by
14 were obtained with variable concentrations of
14 and the substrate (
Figure 3). The intersection of these lines on the vertical axis indicated that
14 was a competitive inhibitor of tyrosinase with respect to
l-DOPA as a substrate, with a
Ki value of 148 μM. These data strongly suggested that
14 effectively inhibited the activity by binding to the active site of this enzyme.
Figure 3.
Lineweaver-Burk plots for the inhibition of tyrosinase by 6-hydroxykaempferol (14) with respect to l-DOPA as substrate.
Figure 3.
Lineweaver-Burk plots for the inhibition of tyrosinase by 6-hydroxykaempferol (14) with respect to l-DOPA as substrate.
In previous studies, flavonoids were reported to form a chelate with the copper in tyrosinase and thus irreversibly inactivate this enzyme [
6,
7]. In particular, it has been reported recently that the ability of flavonoids to form the chelate with copper ions plays an important role in the tyrosinase inhibition [
22]. To further confirm the inhibitory mechanism of
14, the copper-ion chelating ability of
14 was examined. A solution of
14 (50 μM) in phosphate buffer was scanned with or without CuSO
4 (100 μM), and the spectra obtained was compared (data not shown). It was observed that the characteristic absorption maxima at 272 nm and 359 nm shifted to 287 nm and 386 nm, respectively, with the addition of copper ion; and returned to their original values 272 nm and 359 nm of the addition of 250 μM EDTA. This copper chelating ability can be further supported by the observation of a noticeable shift (358 →303 nm) when
14 was incubated with enzyme. The characteristic hypsochromic shift was observed in the absorption spectrum of
14 and tyrosinase complex, similar to that observed in the other flavonols [
7], such as quercetin. These results suggested that the mechanism of inhibition by
14 could be due to its chelation with copper ion in the active site of the enzyme so as to irreversibly inactivate the enzyme, although the detailed three-dimensional structure of the mushroom tyrosinase and the binding-model of
14 with this enzyme blocked.
Table 2.
ABTS radical scavenging activity and superoxide dismutase (SOD)-like activity of baicalein (1), 6-hydroxyapigenin (6), 6-hydroxygalangin (13) and 6-hydroxy-kaempferol (14).
Table 2.
ABTS radical scavenging activity and superoxide dismutase (SOD)-like activity of baicalein (1), 6-hydroxyapigenin (6), 6-hydroxygalangin (13) and 6-hydroxy-kaempferol (14).
| Compounds | EC50 (μM) |
| ABTS radical scavenging activity | SOD-like activity |
| 1 | 4.89 | 48.6 |
| 6 | 4.43 | 36.7 |
| 13 | 3.33 | 46.9 |
| 14 | 2.59 | 34.3 |
| Trolox®,a | 10.86 | NT b |
| l-Ascorbic acid c | NT | 42.5 |
Melanin biosynthesis is caused by the production of dopachrome from dopa catalyzed by tyrosine and concurrent auto-oxidative polymerization of dopachrome [
1]. Moreover, compounds with reducing properties can have depigmentation effects by interaction with
o-quinones, thus avoiding the oxidative polymerization of melanin intermediates [
1]. In addition, these compounds contain phenolic hydroxyls, which can prevent pigmentation resulting from auto-oxidative processes [
23,
24]. Therefore, we tested the antioxidant ability of 5,6,7-trihydroxyflavones
1,
6,
13 and
14 by measuring the ABTS radical scavenging as well as superoxide dismutase (SOD)-like activity, closely related to the auto-oxidation. ABTS radical scavenging activity (EC
50) of
1,
6,
13 and
14 ranged from 2.6 μM to 4.9 μM, which is higher than that of Trolox
® (EC
50=10.9 μM) as a positive control (
Table 2). Superoxide dismutase (SOD)-like activity was estimated by suppression of the superoxide anion radical (O
2-) generated from the hypoxanthine-xanthine oxidase system in the presence of test samples. As shown in
Table 2, 5,6,7-trihydroxyflavones
1,
6,
13 and
14 exhibited strong SOD-like activity with EC
50s at the order of 10
-5 M, which is comparable to that of
l-ascorbic acid as a positive control. These results indicate that the 5,6,7-trihydroxyflavones may contribute to the inhibition of melanin biosynthesis in cells by their potent antioxidant activities.
Conclusions
We have disclosed considerable information about the structure-activity relationships with respect of the number and relative position of the hydroxyl groups of baicalein (1) for enhancing the inhibitory activity against tyrosinase, and found that 6-hydroxyapigenin (6), 6-hydroxygalangin (13) and 6-hydroxykaemferol (14) all exhibited high inhibitory effects towards tyrosinase. Moreover, 14 showed the highest activity among the tested flavonoids, and 14 was a competitive inhibitor of tyrosinase with respect to l-DOPA. 5,6,7-Trihydroxyflavones 1, 6, 13 and 14 also showed high antioxidant activities. Hence, we may conclude that the 5,6,7-trihydroxyflavones are useful as good depigmentation agents with inhibitory effects on tyrosinase in addition to their antioxidant properties.
Experimental Section
General
NMR spectra were recorded with a Bruker AMX500 (1H-, 500 MHz; 13C-, 125 MHz) instrument. Chemical shifts are expressed in ppm referenced to the residual solvent signals of methanol-d4 (δH 3.30 ppm), chloroform-d (δH 7.24 ppm) and dimethyl sulfoxide-d6 (δH 2.49 and δC 39.5 ppm). Coupling constants (J) are reported in Hz. Fast atom bombardment (FAB), Field desorption (FD), FD-high resolution (HR), electron ionization (EI), and EI-HR mass spectra (MS) were obtained on a Jeol JMS-SX102A instrument. Melting points were measured on a hot stage and are uncorrected. The following experimental conditions were used for chromatography: normal phase column chromatography: Silica gel Wakogel C-300 (Wako Pure Chem. Co., Osaka, Japan, 40-64 mesh), reverse phase ODS column chromatography; Cosmosil 75C18-OPN (Nacalai Tesque, Inc., Kyoto, Japan). TLC, precoated TLC plates with Silica gel 60 F254 (Merck, 0.25 mm or 0.5 mm thickness, normal phase) and Silica gel RP-18 F254s (Merck, 0.2 mm thickness, reverse phase). Detection was done with a UV lamp (254 nm). Preparative HPLC was done with an Inertsil PREP-ODS column (20.0×250 mm, GL-Science). The appropriate analytical conditions are mentioned in each section. All reagents were of reagent grade and were purchased from Wako Pure Chem. Co., Osaka, Japan, unless otherwise stated. Acetone and tetrahydrofuran were dried by storage over 3Å molecular sieves. All solvents were distilled before use. All nonaqueous reactions were done in dry glassware.
Test Compounds
Among the tested hydroxyflavones
1-
14 (
Figure 1), baicalein (
1), 5,6-dihydroxyflavone (
2), 6,7-dihydroxyflavone (
3), 5,6,7,8-tetrahydroxyflavone (
5), 6-hydroxyapigenin (
6), 6-hydroxylutelin (
7), 3’,4’,5’,5,6,7-hexahydroxyflavone (
8) and 6-hydroxygalangin (
13) were synthesized as described previously [
19,
20,
21]. 5,7-Dihydroxyflavone (
4) was purchased from Tokyo Kasei Kogyo Co., Tokyo, Japan. Apigenin (
9), luteolin (
10) and kaempferol (
12) were purchased from Sigma Aldrich Japan Co.,Tokyo, Japan. 3’,4’,5,5’,7-Pentahydroxyflavone (
11) and 6-hydroxykaempferol (
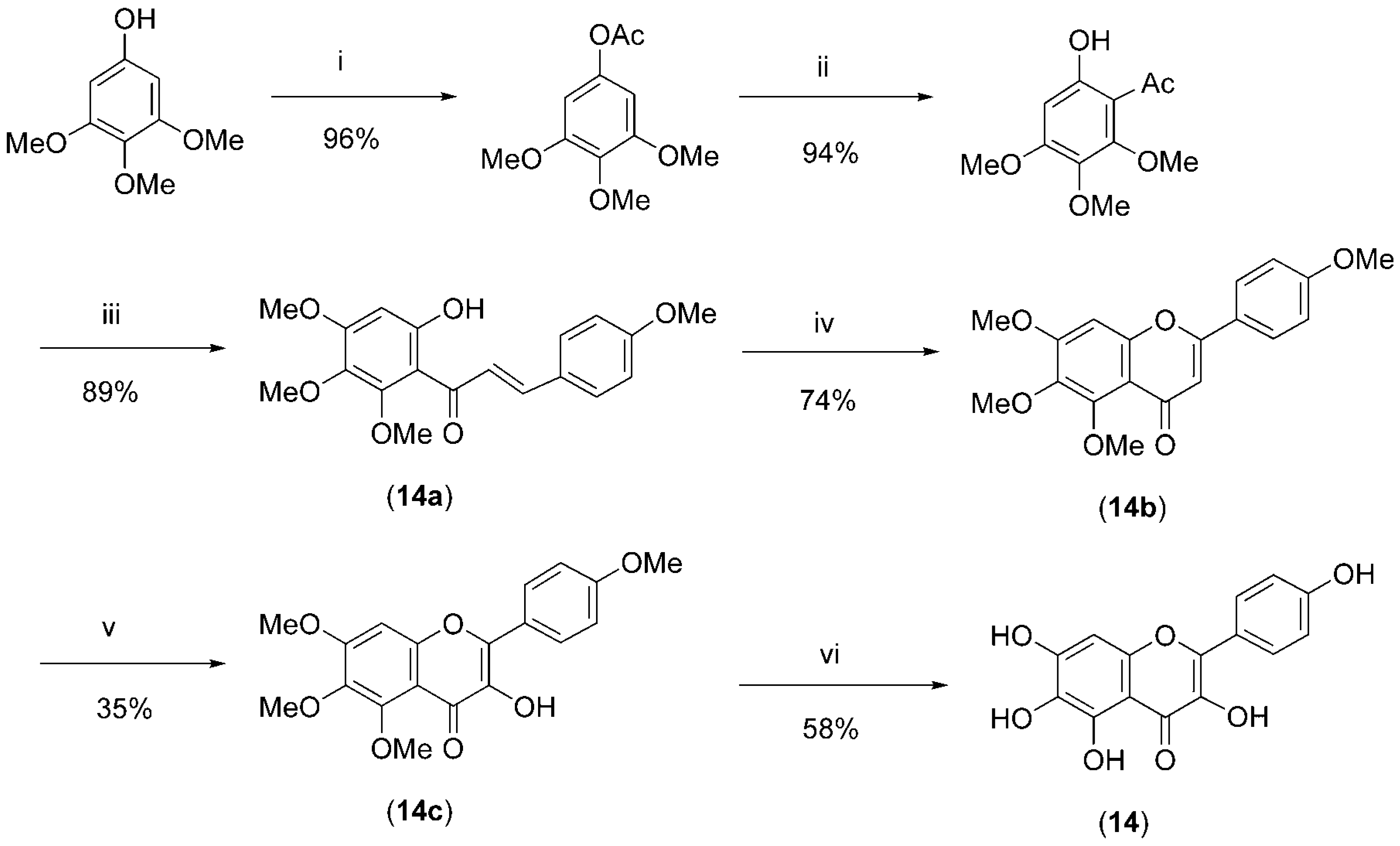
14, Scheme 1) were synthesized as described below.
3’,4’,5,5’,7-Pentahydroxyflavone (11).
A mixture of 5,7-dihydroxy-3’,4’,5’-trimethoxyflavone (112 mg, 0.33 mmol) [
20], 47% aqueous hydrobromic acid (1.1 mL, 9.9 mmol) and acetic acid (4 mL) was heated under reflux for 23 h . After cooling to room temperature, the reaction mixture was extracted with 1-butanol and concentrated to dryness. The residue was dissolved in a small amount of methanol and subjected to reverse-phase column chromatography, using water-methanol-formic acid (40:60:0.1) as the eluent to yield crude
11. This was further purified by preparative HPLC (mobile phase, water-methanol-formic acid (40:60:0.1); flow rate, 5.0 mL/min; detection, UV 254 nm) to give
11 (
tR 21 min, 35 mg, 38%) as pale yellow powders: mp >300 °C; FAB-MS (negative)
m/
z (%) 301 ([M-H]
-, 100);
1H-NMR δ (DMSO-
d6): 6.17 (1H, d,
J=1.6, 6-H), 6.39 (1H, d,
J=1.6, 8-H), 6.52 (1H, s, 3-H), 6.95 (2H, s, 2’ and 6’-H), 12.96 (1H, s, 5-OH).
Synthesis of 6-hydroxykaempferol (14): 6’-Hydroxy-2’,3’,4,4’-tetramethoxychalcone (14a).
To 6’-hydroxy-2’,3’,4’-trimethoxyacetophenone [
25] (6.1 g, 26.6 mmol) and 4-methoxy-benzaldehyde (3.3 mL, 27 mmol) in ethanol (32 mL) was added 50% KOH (15 g) at 0 °C and the mixture was then stirred under argon gas at room temperature for 32 h. The reaction was quenched with ice water (300 g) and acidified with 6
m HCl. The resulting mixture was extracted with ethyl acetate (300 mL x 3) and the combined organic extracts were washed with brine, dried over anhydrous Na
2SO
4 and concentrated. The residue was purified by silica gel column chromatography using hexane-ethyl acetate (8:2) as the eluent to give
14a (8.1 g, 89%) as a pale yellow powder: mp 137-139 °C (lit. [
25] mp 139-141°C); FDHRMS
m/
z 344.1235 (calcd. for C
19H
20O
6, 344.1260);
1H-NMR δ (chloroform-
d): 3.82, 3.85, 3.89, 3.91 (each 3H, 2’,3’,4,4’-OMe), 6.28 (1H, s, 5’-H), 6.93 (2H, d,
J=8.9, 3 and 5-H), 7.59 (2H, d,
J=8.9, 2 and 6-H), 7.82-7.84 (2H, m, α and β-H), 13.75 (1H, s, 2’-OH).
Scheme 1.
Synthesis of 6-hydroxykaempferol (14).
Scheme 1.
Synthesis of 6-hydroxykaempferol (14).
Reagents and conditions: (i) Ac2O, AcONa, 110 °C, 3 h; (ii) BF3-Et2O, 70 °C, 2 h; (iii) 4-methoxybenzaldehyde, EtOH, KOH, R.T, 36 h; (iv) I2, DMSO, 190-200 °C, 2h; (v) LiN(i-Pr)2, (MeO)3B, AcOH, 20% H2O2, THF; (vi) BBr3, CH2Cl2, -80 °C for 1 h and R.T for 12 h.
4’,5,6,7-Tetramethoxyflavone (14b)
A mixture of
14a (7.3 g, 21.2 mmol) and iodine (250 mg) in DMSO (25 mL) was heated at 190 – 200 °C for 2 h, and then poured onto ice water (300 g). This mixture was partitioned with ethyl acetate and water. The organic phase was washed with brine, dried over anhydrous Na
2SO
4 and concentrated. The residue was purified by silica gel column chromatography using hexane-ethyl acetate (7:3) as the eluent to give
14b (5.4 g, 74%) as a pale yellow powder: mp 144-146 °C (lit. [
26] mp 142 °C); FDHRMS
m/
z 342.1094 (calcd. for C
19H
18O
6, 342.1103);
1H-NMR δ (DMSO-
d6): 3.75, 3.79, 3.84, 3.94 (each 3H, 4’,5,6,7-OMe), 6.70 (1H, s, 3-H), 7.08 (1H, dd,
J=6.8, 2.3, 3’ and 5’-H), 7.20 (1H, s, 8-H), 8.01 (2H, dd,
J=6.8, 2.3, 2’ and 6’-H).
3-Hydroxy-4’,5,6,7-tetramethoxyflavone (14c)
n-Butyllithium (1.6
m solution in hexane, 5 mL, 8 mmol) was added to diisopropylamine (1.2 mL, 8 mmol) in dry THF (40 mL) at –80 °C under argon gas. The resulting lithium diisopropylamide (LDA) solution was warmed to 0 °C for 30 min and re-cooled to –80 °C. Compound
14b (1.4 g, 4 mmol) in THF (40 mL) was added dropwise to the LDA solution and stirred for 1 h. After an addition of trimethyl borate (0.9 mL, 8 mmol) in THF (20 mL), the resulting solution was then stirred at –80 °C for 30 min. The reaction mixture was acidified with acetic acid (0.7 mL), stirred for 15 min, and then oxidized with 30% hydrogen peroxide (1.2 mL, 8 mmol). The solution was allowed to warm slowly to room temperature and was then shaken with aqueous saturated sodium bicarbonate solution (50 mL). The aqueous mixture was extracted with ethyl acetate (100 mL x 3). The combined organic extracts were dried over anhydrous Na
2SO
4 and concentrated. The residue was purified by silica gel column chromatography using chloroform-methanol (10:0.2) as the eluent to give
14c (0.5 g, 35%) as a yellow powder: mp 132-134 °C (lit. [
27] mp 131-132 °C); FDHRMS
m/z 358.1068 (calcd. for C
19H
18O
7, 358.1052);
1H-NMR δ (DMSO-
d6): 3.31, 3.76, 3.84, 3.93 (each 3H, s, 4’,5, 6 and 7-OMe), 7.10 (2H, d,
J=9.1, 3’ and 5’-H), 7.16 (1H, s, 8-H), 8.16 (2H, d,
J=9.1, 2’ and 6’-H), 9.08 (1H, s, 3-OH).
6-Hydroxykaempferol (14).
To a stirred solution of
14c (358 mg, 1 mmol) in dry CH
2Cl
2 (10 mL) at -80 °C was added BBr
3 (1M CH
2Cl
2 solution, 8 mL, 8 mmol). The reaction mixture was then stirred for 1 h at –80 °C and 12 h at room temperature. The reaction was stopped by adding ice-cooled water (50 mL). To this mixture was added 1-butanol (200 mL). The organic phase was separated, washed with brine, and evaporated to dryness. The residue was dissolved in a small amount of methanol and subjected to reverse-phase column chromatography, using water-methanol-formic acid (40:60:0.1) as the eluent to yield crude
14. This was further purified by preparative HPLC (mobile phase, water-methanol-formic acid (30:70:0.1); flow rate, 4.0 mL/min; detection, UV 254 nm) to give
14 (
tR 14.9 min, 176 mg, 58%) as a pale yellow powder: mp 284-286 °C (lit. [
28] mp 280-282 °C); FDHRMS
m/
z 302.0452 (calcd. for C
15H
10O
7, 302.0426);
1H-NMR δ (DMSO-
d6): 6.51 (1H, s, 8-H), 6.91 (2H, d,
J=8.9, 3’ and 5’-H), 8.02 (2H, d,
J=8.9, 2’ and 6’-H).
Tyrosinase inhibition assays
Tyrosinase inhibition assays were performed according to a modified method described by Kubo [
6]. Mushroom tyrosinase (EC 1. 14. 18. 1, Sigma Product T3824 with an activity of 3320 units/mg) was used for the bioassay in this study. The tested compounds were first dissolved in DMSO and diluted 25 times with water in each experiment before use. The activity was expressed as the sample concentration that gave a 50% inhibition in the enzyme activity (IC
50). For the measurement of diphenolase inhibition, a sample solution (0.1 mL) and 1.5 M
l-DOPA solution (1.5 mL, 0.1M phosphate buffer, pH 6.8) were mixed with H
2O (0.4 mL) and preincubated at 25 °C for 5 min. Then, tyrosinase solution (0.5 mL, 125 units) was added and the formation of dopachrome was monitored at 475 nm from 0.5 to 4 min using a Hitachi UV-3210 spectrophotometer. For the measurement of monophenolase inhibition, a sample solution (0.1 mL) and 2
m l-tyrosine solution (1 mL) were mixed with of 0.1 M phosphate buffer (0.9 mL, pH 6.8), and pre-incubated at 25 °C for 5 min. Then, tyrosinase solution (0.5 mL, 250 units) was added and the dopachrome formed was monitored at 475 nm for 20 min.
Chelation ability of 5,6,7-trihydroxyflavones towards copper ions
A solution of test compounds (10 mM) was prepared in DMSO. Then 50 μM solution was prepared in a cuvette containing phosphate buffer (0.1 M, pH 6.8) and the sample absorbance was recorded in the 200-540 nm range using Shimadzu UV-1600 spectrophotometer. A repeated scan was recorded after addition of 100 μM CuSO4 and a third spectrum after addition of 250 μM EDTA. To check if 5,6,7-trihydroxyflavones can chelate copper in the enzyme, the UV-visible spectra (200-540 nm) was measured. A mixture containing 0.1 M phosphate buffer (1.5 mL, pH 6.8), H2O (0.4 mL), a test solution (0.1 mL) and tyrosinase solution (0.5 mL, 125 units/mL), was incubated at 25 °C for 30 min, and then the spectra was recorded.
ABTS radical scavenging activity
ABTS radical scavenging activity was determined according to a modification of the method of reference [
29]. In brief, 2,2’-azinobis-(3-ethylbenzothiazoline-6-sulfonic acid diammonium salt, ABTS, 19 mg) was reacted with potassium persulfate (3.3 mg) overnight in the dark at room temperature. The working solution was prepared by diluting it with water to get an absorbance around 0.70 at 734 nm. Test sample (30μL in DMSO) was reacted with diluted ABTS (2.97 mL) and absorbance at 734 nm was recorded within 30 min. Trolox
® was used as a positive control, and the control was prepared without the samples. The activity was expressed as the concentration of sample necessary to give a 50% reduction in the original absorbance (EC
50).
SOD (superoxide dismutase)-like activity
SOD (superoxide dismutase)-like activity was measured by the nitrite method [
24,
30]. In brief, a mixture of the sample solution (0.01 mL), EDTA-phosphate buffer solution (pH 8.2, 0.6 mL), reagent A (0.5 mM xanthine and 10 mM hydroxylammonium chloride in buffer, 0.2 mL) and xanthine-oxidase solution (10 mU/mL in buffer) was incubated at 37 °C for 30 min, and then coloring reagent was added (25 μM
N-(1-naphthyl)ethylenediamine, 2 mM sulfanilic acid and 16.7% acetic acid, 2 mL). The resulting mixture was allowed to stand for 30 min at room temperature and the optical absorbance was measured at 550 nm. SOD-like activity was expressed as a value of EC
50 against the production of superoxide anion from hypoxanthine-xanthine-oxidase.