A Handy and Solventless Direct Route to Primary 3-[3-Aryl)-1,2,4-oxadiazol-5-yl]propionamides Using Microwave Irradiation
Abstract
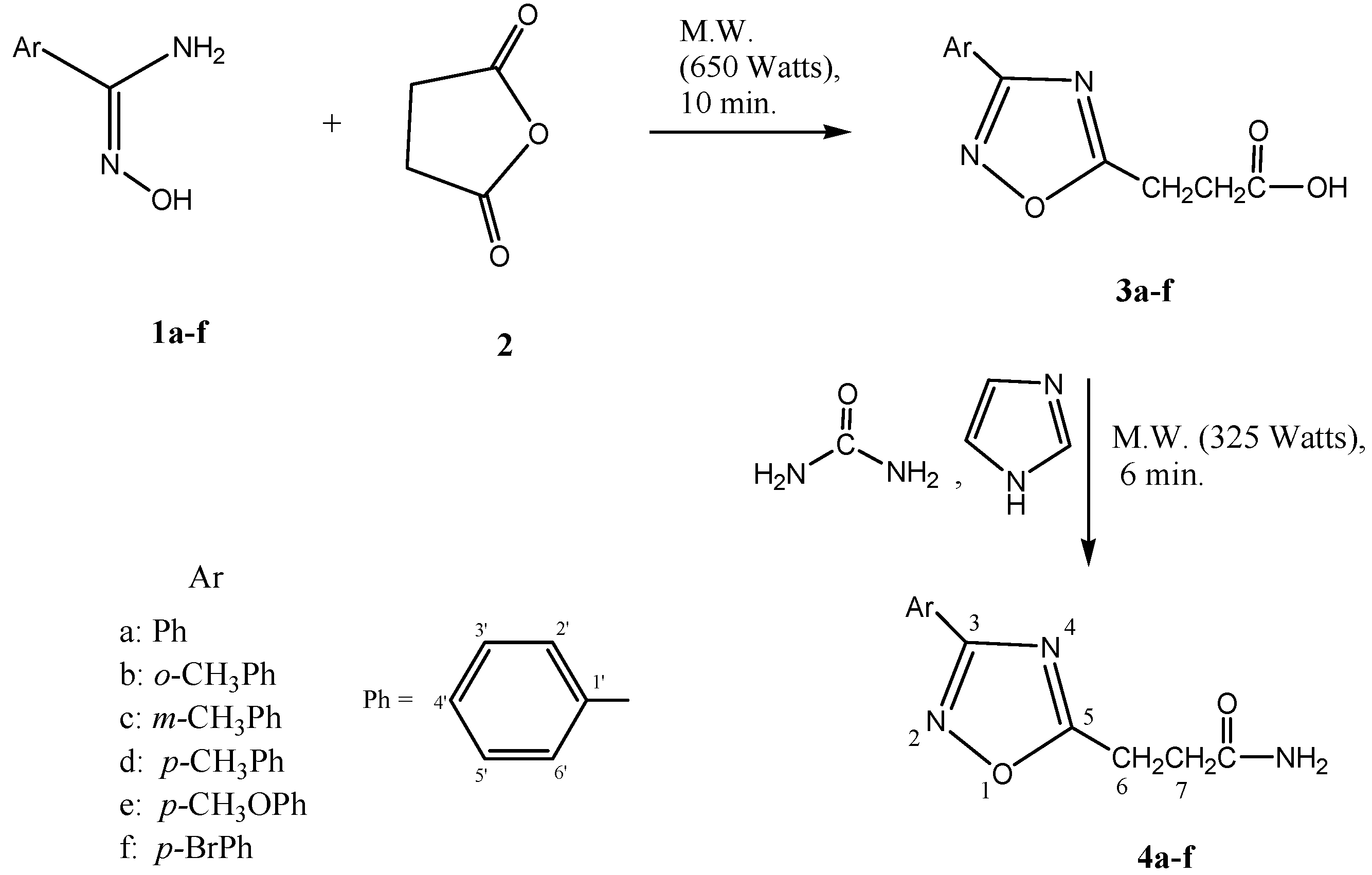
:Introduction
| Compound | ReactionTemp.(°C) a | Yield (%) b | Rf values c | M.p (° C) d |
|---|---|---|---|---|
| 4a | 130 | 73 | 0.22 | 175-176 |
| 4b | 110 | 83 | 0.28 | 124-125 |
| 4c | 117 | 85 | 0.26 | 122-123 |
| 4d | 115 | 87 | 0.26 | 168-169 |
| 4e | 110 | 84 | 0.19 | 174-175 |
| 4f | 120 | 87 | 0.24 | 161-162 |
Results and Discussion
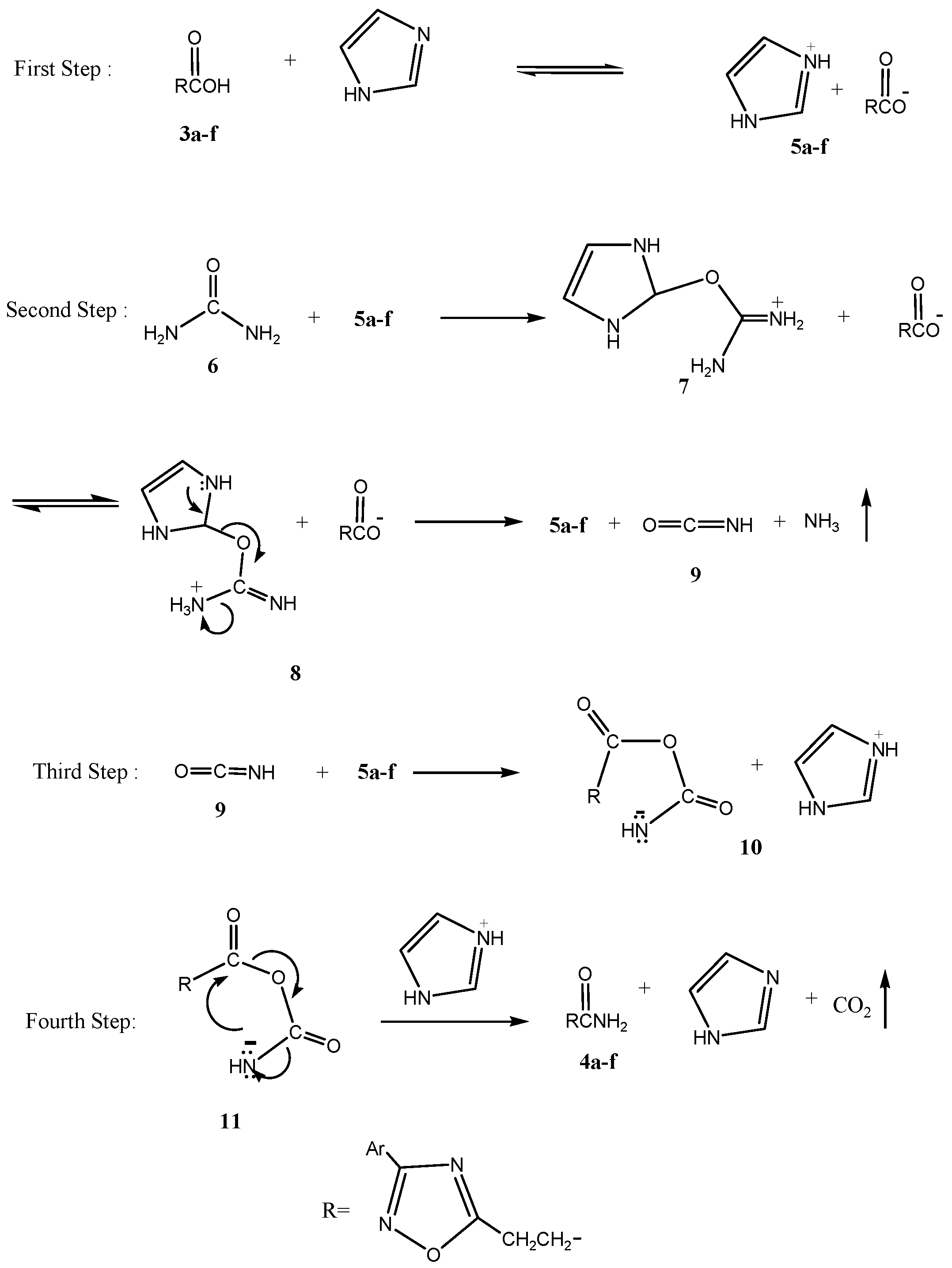
Conclusions
Experimental Section
General
General Synthetic Procedure
Spectral and Analytical Data of Compounds 4a-f
Acknowledgments
References and Notes
- Clapp, L.B. “Advances in Heterocyclic Chemistry”; Katritzky, A.R., Ed.; Academic Press: New York, 1976; Volume 20, pp. 65–116. [Google Scholar]
- Clapp, L.B. “Comprehensive Heterocyclic Chemistry”; Katritzky, A.R., Rees, C.W., Eds.; Pergamon Press: Oxford, 1984; Volume 6, pp. 365–391. [Google Scholar]
- Jochims, J.C. “Comprehensive Heterocyclic Chemistry II”; Katritzky, A.R., Rees, C.W., Scriven, E.F.D., Eds.; Elsevier Science: Oxford, 1996; Volume 4, pp. 179–228. [Google Scholar]
- Hemming, K. Recent developments in the synthesis, chemistry and applications of the fully unsaturated 1,2,4- oxadiazoles. J. Chem. Res. 2001, 216, 209–216. [Google Scholar] [CrossRef]
- Antunes, R.; Batista, H.; Srivastava, R.M.; Thomas, G.; Araújo, C.C.; Longo, R.L.; Magalhães, H.; Leão, M.B.C.; Pavão, A.C. Synthesis, characterization and interaction mechanism of new oxadiazolo-phthalimides as peripheral analgesics. IV. J. Mol. Struct. 2003, 600, 1–13. [Google Scholar] [CrossRef]
- Srivastava, R.M.; Morais, L.P.F.; Catanho, M.T.J.A.; Souza, G.M.L.; Seabra, G.M.; Simas, A.M.; Rodriguez, M.A.L. Synthesis and antiinflammatory activity of 3-aryl-5-isopropyl-1,2,4-oxadiazoles. Heterocycl. Commun. 2000, 6, 41–48. [Google Scholar]
- Dahlgren, S.E.; Dalhamn, T. Antiinflammatory action of phenyl-methyl-oxadiazole (pmo) - experimental study on guinea-pig trachea. Acta Pharmacol. Toxicol. 1972, 31, 193–202. [Google Scholar] [CrossRef]
- Afiatpour, P.; Srivastava, R.M.; de Oliveira, M.L.; Barreiro, E.J. Analgesic and antiinflammatory effects of 3-[3-(phenyl)-1,2,4-oxadiazol-5-yl] propionic acid. Braz. J. Med. Biol. Res. 1994, 27, 1403–1406. [Google Scholar] [PubMed]
- Glushkov, V.A.; Anikina, L.V.; Vikharev, Y.B.; Stryapunina, O.G.; Shklyaev, Y.V.; Tolstikov, A.G. Synthesis and antiinflammatory activity of N-[2-(p-hydroxyphenyl)-1,1-dialkylethyl]-α-dialkylamino acetamides. Pharm. Chem. J. 2004, 38, 86–89. [Google Scholar] [CrossRef]
- Sladowska, H.; Sieklucka-Dziuba, M.; Rajtar, G.; Sodowski, M.; Kleinrok, Z. Investigations on the synthesis and pharmacological properties of amides of 7-methyl-3-phenyl-1-[2-hydroxy-3-(4-phenyl-1-piperazinyl)propyl]-2,4-dioxo-1,2,3,4-tetrahydropyrido[2,3-d]-pyrimidine-5-carboxylic acid. Il Farmaco 1999, 54, 773–779. [Google Scholar]
- Sanders-Bush, E.; Mayer, S.E. “Goodman & Gilman’s” The Pharmacological Basis of Therapeutics, 10th International Edition; Hardman, J.G., Linbird, L.E., Eds.; McGraw-Hill: New York, 2001; pp. 284–285. [Google Scholar]
- Benz, G. Comprehensive Organic Synthesis; Trost, B.M., Fleming, I., Eds.; Pergamon Press: Oxford, 1991; Volume 6, pp. 381–418. [Google Scholar]
- Smith, M.B.; March, J. March’s Advanced Organic Chemistry. Reactions, Mechanisms and Structure, Fifth Edition; John Wiley & Sons: New York, 2001; pp. 506–513. [Google Scholar]
- Baldwin, B.W.; Hirose, T.; Wang, Z.-H. Improved microwave oven synthesis of amides and imides promoted by imidazole; convenient transport agent preparation. Chem. Commun. 1996, 2669–2670. [Google Scholar] Vasquez-Tato, M.P. Microwave-mediated synthesis of amides. Synlett 1993, 506. [Google Scholar] Gelens, E.; Smeets, L.; Sliedregt, L.A.J.M.; van Steen, B.J.; Kruse, C.G.; Leurs, R.; Orru, R.V.A. An atom efficient and solvent-free synthesis of structurally diverse amides using microwaves. Tetrahedron Lett. 2005, 45, 3751–3754. [Google Scholar] Gorelski, C.; Krlej, A.; Steffens, C.; Ritter, H. Microwave-assisted single-step synthesis of various (meth)acrylamides and poly(meth)acrylamides directly from (meth)acrylic acid and amines. Macromol. Rapid Commun. 2004, 25, 513–516. [Google Scholar]
- Varma, R.S.; Naicker, K.P. Solvent-free synthesis of amides from non-enolizable esters and amines using microwave irradiation. Tetrahedron Lett. 1999, 40, 6177–6180. [Google Scholar] [CrossRef]
- Chen, J.J.; Deshpande, S.V. Rapid synthesis of alpha-ketoamides using microwave irradiation-simultaneous cooling method. Tetrahedron Lett. 2003, 44, 8873–8876. [Google Scholar] [CrossRef]
- Perreux, L.; Loupy, A.; Volatron, F. Solvent-free preparation of amides from acids and primary amines under microwave irradiation. Tetrahedron 2002, 58, 2155–2162, and references cited therein. [Google Scholar]
- Sauer, D.R.; Kalvin, D.; Phelan, K.M. Microwave-assisted synthesis utilizing supported reagents: a rapid and efficient acylation procedure. Org. Lett. 2003, 5, 4721–4724. [Google Scholar] [CrossRef] [PubMed]
- Khalafi-Nezhad, A.; Mokhtari, B.; Rad, M.N.S. Direct preparation of primary amides from carboxylic acids and urea using imidazole under microwave irradiation. Tetrahedron Lett. 2003, 44, 7325–7328. [Google Scholar]
- Pasha, M.A.; Jayashankara, V.P. Synthesis of amides from carboxylic acids and urea in the presence of pyridine under microwave irradiation. J. Ind. Chem. Soc. 2005, 82, 675–676. [Google Scholar]
- a) InterBioScreen Ltd., P.O. Box 218, Moscow, Russia, publication date: Mar. 15, 2005; b) ChemBridge Corporation, 16981 Via Tazon, Suite G, San Diego, CA, 92127, U.S.A. ChemBridge Screening Library, publication date: Jan. 12, 2005.
- We have taken precautions to ensure that the results are reproducible. We first determined the region inside the microwave oven where the heating was the maximum using water samples and recording the temperatures with an infrared thermometer. After doing this, we repeated the reaction and got the same results. In our opinion, the reactions can be repeated easily without any problem by any chemist
- Rault, P.; Pilard, J.; Touaux, B.; Texier-Boullet, F.; Hamelin, J. Rapid generation of amines by microwave irradiation of ureas dispersed on clay. Synlett 1994, 935–936. [Google Scholar]
- Srivastava, R.M.; Seabra, G.M. Preparation and reactions of 3-[3-(aryl)-1,2,4-oxadiazol-5-yl] propionic acids. J. Braz. Chem. Soc. 1997, 8, 397–405. [Google Scholar]
- Sample availability: Please contact the authors.
© 2006 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Neves, R.A.W., Filho; Srivastava, R.M. A Handy and Solventless Direct Route to Primary 3-[3-Aryl)-1,2,4-oxadiazol-5-yl]propionamides Using Microwave Irradiation. Molecules 2006, 11, 318-324. https://doi.org/10.3390/11050318
Neves RAW Filho, Srivastava RM. A Handy and Solventless Direct Route to Primary 3-[3-Aryl)-1,2,4-oxadiazol-5-yl]propionamides Using Microwave Irradiation. Molecules. 2006; 11(5):318-324. https://doi.org/10.3390/11050318
Chicago/Turabian StyleNeves, Ricardo A.W., Filho, and Rajendra M. Srivastava. 2006. "A Handy and Solventless Direct Route to Primary 3-[3-Aryl)-1,2,4-oxadiazol-5-yl]propionamides Using Microwave Irradiation" Molecules 11, no. 5: 318-324. https://doi.org/10.3390/11050318




