Introduction
Convective cooling of surface finds wide application in industry. In this case, cooling of the surface (i) improves mechanical and metallurgical properties of the substrate material, such as quenching, and (ii) enhances the life cycle of the equipment or parts such as cooling of electronic components. Moreover, during convective heating or cooling process, thermal stress is generated in the surface region of the substrate material. Depending on the temperature gradient in the region heated convectively, stress levels above or around yielding limit of the substrate material can occur. Consequently, investigation into thermal stress levels during the convective heating/cooling becomes necessary. Moreover, the temperature and stress fields are involved with thermodynamic irreversibility in the system. Therefore, study into thermodynamic irreversibility associated with the temperature and stress fields gives insight into the thermodynamic heating of the substrate material.
Considerable research studies were carried out to investigate the convective cooling/heating of the surfaces. Lumped capacitance analysis for the quenching of the metallic substrate was carried out by Campo and Hernandez-Morales [
1]. They showed that for convective cooling, two primary dimensionless groups existed, which included solid fluid thermal conductivity ratio and modified Rayleigh number. The contact problems with frictional heat generation were studied by Kul'chyts'kyi – Zhyhailo and Evtushenko [
2] – for semi-bonded bodies of revolution by taking into account the effect of convective cooling of the free surface. They determined the conditions under which the influence of convective heat exchange on the level and distribution of contact stresses could be neglected. Numerical analysis of partially heated vertical plates in natural convective cooling was carried out by Campo et al [
3]. They obtained the correlations between the induced mass flow rate as well as the maximum wall temperatures, the Rayleigh number, and the extension ratio. Transient two-dimensional thermal analysis of electronic packages by the boundary element method was examined by Guven et al [
4 ]. They indicated that the results captured the singular flux field arising form the mismatched in the thermal conduction coefficients and geometric discontinuity. Three-dimensional convective cooling in a vertical channel with flush-mounted heat sources was carried out by Gunes and Laikopoulos [
5]. They presented temperature and velocity field distributions as well as maximum temperature, maximum velocity, and local Nusselt number distributions in terms of Grashof number. Thermal characteristics of back cooled impingement cooler for packaging of high power semiconductor laser diode array were studied by Mirji and Ahmad [
6]. They showed that experimental thermal resistance values closely match with theoretical calculations.
Considerable research studies were carried out to explore thermal stresses developed in solid bodies. The numerical simulation of thermal stresses due to grinding was carried out by Moulik et al [
7]. They indicated that the predicted thermal stress distributions were consistent with experimentally measured residual stresses on ground surface. Green's function for thermal stress mixed boundary value problem of an infinite plate was introduced by Hann and Hasebe [
8]. They obtained an explicit solution including a hybergeometrical function by explaining the mapping technique and complex variable method. The thermal stress around a penny-shaped crack in a thermopiezoelectiric solid was studied by Shang and Kuna [
9]. They presented exact expression for the stress-intensity factor. Thermal stress analysis and residual stresses in a thermoplastic composite beam were studied by Sayman et al [
10]. They showed that for the same plastic distribution, the intensity of the transverse displacement was the largest for that orientation angle. Finite element modeling of stress development during deposition of ion assisted coating was carried out by Ward and Arnell [
11]. They indicated that the predicted trends of mean stress and distribution were similar to those observed in the literature. Thermal stresses in an actively cooled two-piece rail structure were investigated by Satapathy and Persad [
12]. They showed that use of thin resistive overlays minimized the thermal stresses on the structure.
Irreversibility of the thermal system can be quantified by the entropy analysis. A second law analysis of the optimum design and operation of thermal energy storage systems were considered by Robert et al [
13]. He showed that an entire cycle of storage and removal should be considered when optimizing the design and performance of a thermal system. Thermodynamics analysis of thermo-mechanical coupling in Couette flow was studied by Demirel [
14]. He indicated that since the irreversibility was related to loss of energy, the thermodynamic analysis could facilitate the optimal conditions which produced less entropy for an existing design. The second law analysis in fundamental convective heat transfer problems was carried out by Mahmud and Fraser [
15]. They derived analytical expression for entropy generation and Bejan numbers from velocity and temperature profiles. Minimization of entropy generation by asymmetric convective cooling was investigated by Ibanez et al [
16]. They showed that when the Biot numbers were different, the entropy function displayed a minimum for specific conditions. Yilbas [
17] formulated entropy generation in nonequilibrium energy transport systems. He indicated that entropy generation enhanced as nonequilibrium energy transport increased across the two-nonequilibrium sub-systems.
Convective cooling of the surface and stress field due to temperature field were formulated previously [
18]. However, the irreversibility associated with the system was left obscured. Consequently, the present study is conducted to present closed form solution for temperature and stress fields due to convective heating of the surface and previously obtained solution for the temperature and stress distributions are enhanced. Entropy generation due to temperature and stress fields is formulated and the magnitude of entropy generation is predicted for various heat transfer coefficients.
Mathematical Model
Consider a semi-infinite solid body heated by convection heat transfer at the surface as shown in
Figure 1. A steep temperature gradient is generated in the surface region of substrate material during this process. The problem of thermal shock on solid surface can be examined in half space. The closed form solution of the unsteady temperature field
T(x,t) in the half space takes the form, as given by Shuja and Yilbas [
18 ],
where
To,
θ, α, and
γ are the initial surface temperature (
x = 0,
t = 0 ), heat source temperature, thermal diffusivity, and the ratio of the heat transfer coefficient (
h) to the thermal conductivity (
k), respectively. erfc stands for the complementary error function, which is 1- erf.
From the stress analysis of one dimensional thermoelasticity problem, the thermal strain, ε
x, in a solid substrate along the x axis as a function of the stress,
σx, and temperature, ∆
T= (T- To) , can be written as
where
ν,
E, and
αT are the Poisson's ratio, elastic modulus, and thermal expansion of a solid material, respectively.
Upon solving the equation of motion referenced to [
18] the closed form solution of the stress field for which the surface (
x = 0 ) is stress free as well as when
x is very large (
x = ∞ ) is given as
where
where
c =
E ( 1-
ν ) / [ ( 1+
ν) ( 1- 2
ν )
ρ ] is the velocity of propagation of elastic longitudinal wave and
ρ is the density of the solid material.
In the case of entropy calculations, entropy generation in the solid is a function of temperature and volume change due to thermal expansion, in this case
s = s ( T, υ ) , therefore one can write
where
and
By definition: and where αT, and β are thermal expansion coefficient and compressibility coefficient, respectively.
Considering the Helmholtz function, F = u –T s
and dF = du –T ds– s dT,
and knowing that du = T ds – Pdυ ,
therefore dF= -s dT – Pds.
Using the following Maxwell relation
one can reach to the relation
Also, the relation:
du = T ds – Pdυ reduces to
and knowing that
Therefore
Now equation (6) becomes
which is a unique solution for entropy generation in solid obtained from Maxwell relations.
Looking at the second term in equation (10)
where (
, specific volume and
ε displacement ). Therefore
Equation (11) can be written in non-dimensional form by introducing the following non-dimensional parameters
Integrating equation (12) yields:
The displacement,
ε, can be obtained after integration of equation (2), i. e.:
A mathematica software is used to compute temperature, displacement, and entropy generation during convective heating of the surface.
Results and Discussions
Heating of solid substrate due to convective boundary at the surface is formulated. The closed form solution for the thermal stress field is presented. The entropy analysis due to temperature and stress fields are carried out to demonstrate the temperature, displacement, and entropy fields, steel is employed in the simulation.
Figure 2 shows temperature profiles inside the substrate material for three different heat transfer coefficients. The sharp increase of the temperature occurs in the surface region of the substrate material, which is particularly true for high heat transfer coefficients ( h > 10
5 W/m
2 K). Temperature decay along the depth appears to be almost linear despite the fact that the heating situation is transient. This is because of: (i) long period of heating duration, steady heating situation is resulted, and (ii) low heat transfer coefficient ( h ≤ 10
7 W/m
2 K); i. e., impulsive like heating (shock) at the surface is omitted. The influence of heat transfer coefficient on the temperature rise is considerable, provided that high heat transfers coefficient (10
7 W/m
2 K) results in rapid change of temperature gradient in the substrate material, i.e. temperature gradient reduces with progressing heating duration.
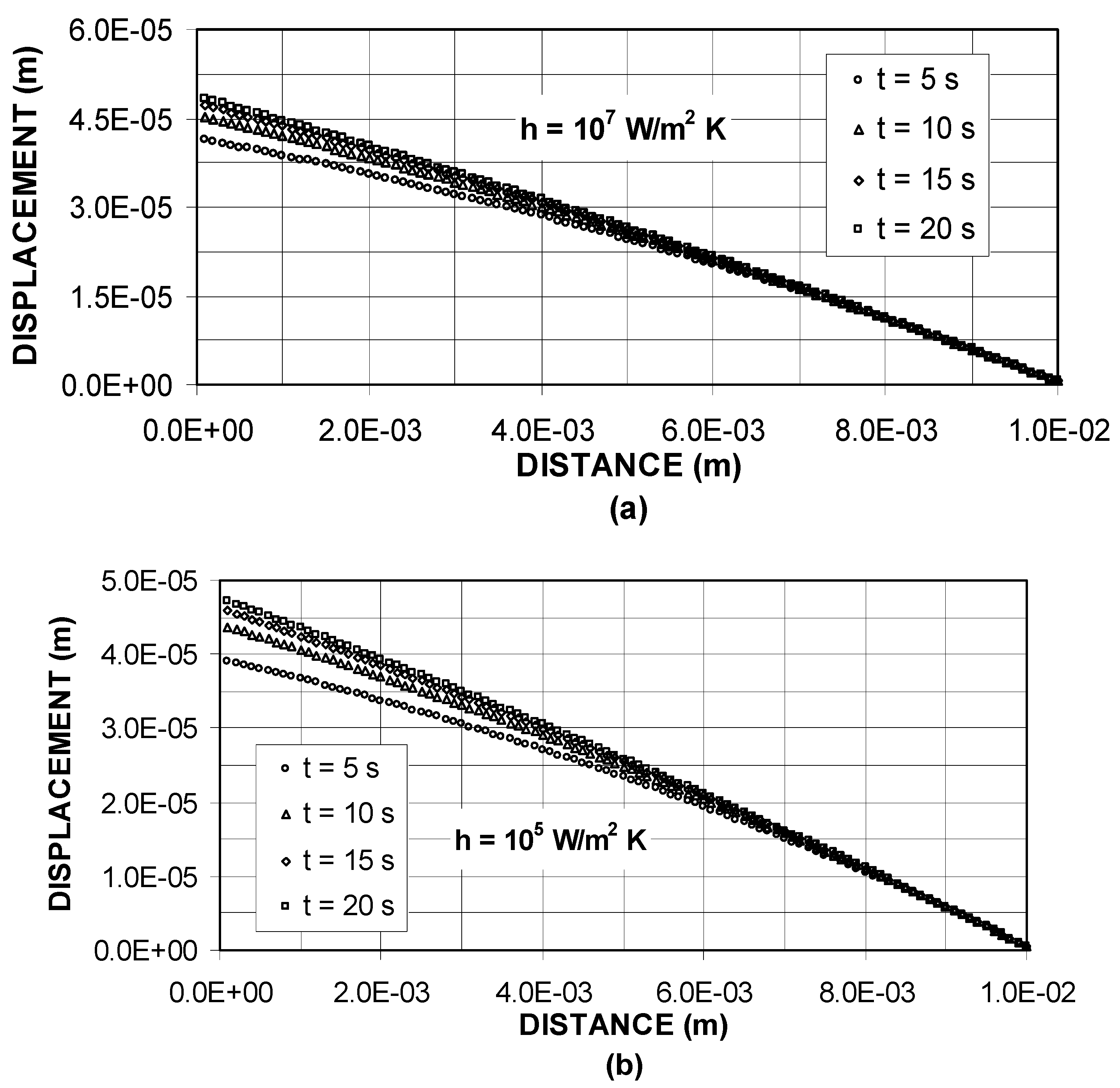
Figure 3 shows the thermal displacement inside the substrate material for different heating durations. The displacement is maximum at the surface and almost zero at some depth below the surface. The thermal displacement does not follow exactly the temperature profiles despite the fact that the substrate material is considered to be elastic. This is because of the strain developed in the surface region which does not vary linearly along the depth like temperature profiles, i.e. expansion of the substrate material below the surface is suppressed by the thermal strain developed in this region. As similar to temperature behavior, increasing heat transfer coefficient results in large magnitude of displacement particularly at the surface of substrate material. The displacement gradient attains slightly higher values at low heat transfer coefficients as compared to that corresponding to high heat transfer coefficients. This is due to the temperature profiles, provided that the variation in temperature gradient is larger than its counterpart corresponding to the displacement.
Entropy analysis covers the entropy generation due to temperature field and thermal strain. However, thermal strain can be formulated through thermal displacement. Consequently, equation (13) is written in terms of temperature field and thermal displacement. Therefore, entropy generation due to temperature field and thermal displacement are segregated.
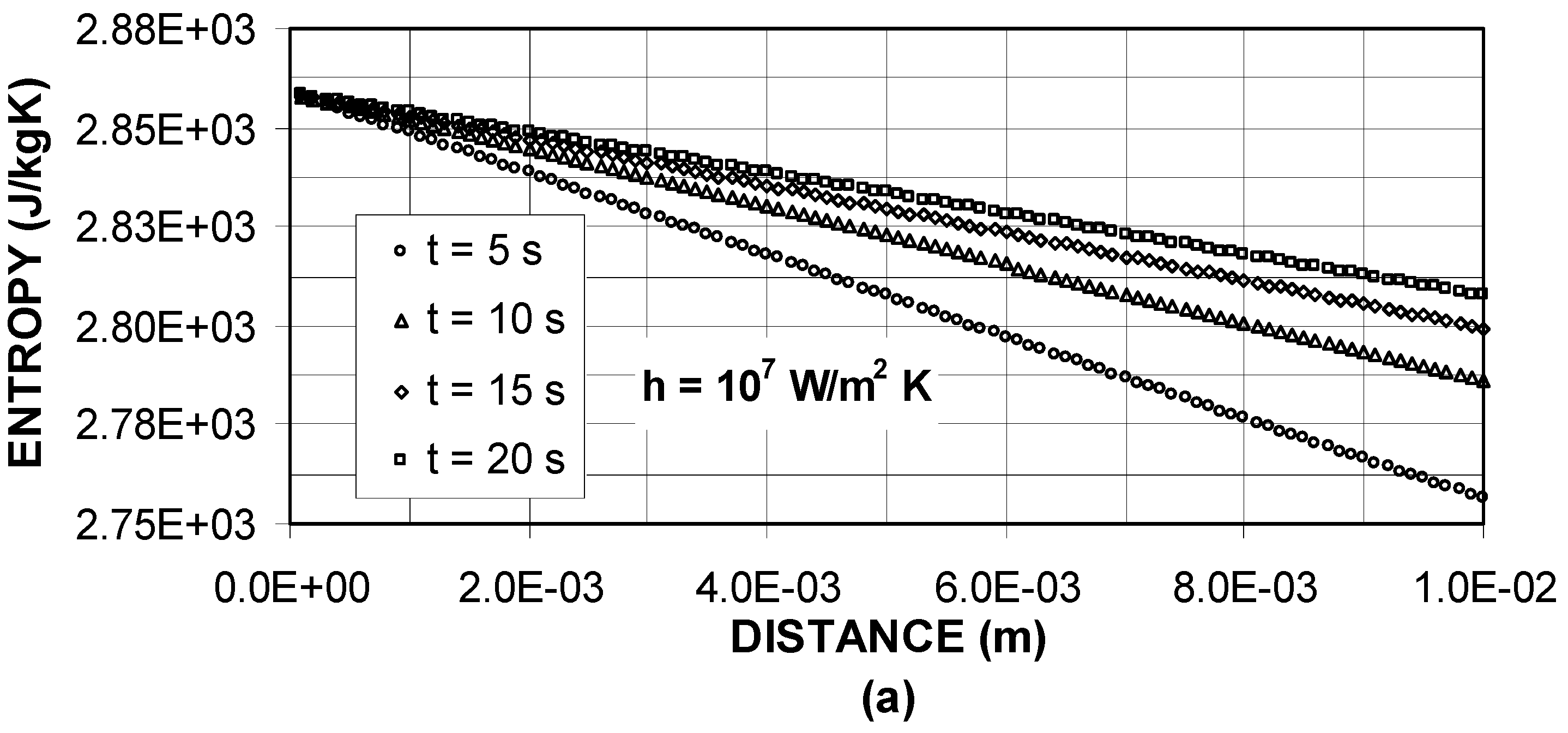
Figure 4 shows entropy generation due to temperature field inside the substrate material for different heating periods. Entropy attains maximum value in the surface vicinity and as the distance increases away from the surface it reduces. The entropy profiles follow almost the temperature profiles provided that the slopes of both curves differ. In the early heating periods entropy production is low and as the heating duration progresses, it increases. Moreover, for high heat transfer coefficients (h ≥ 10
5 W/m
2K), entropy production increases. This is because of the heat transfer rate to the solid material, which increases with improving heat transfer coefficients.
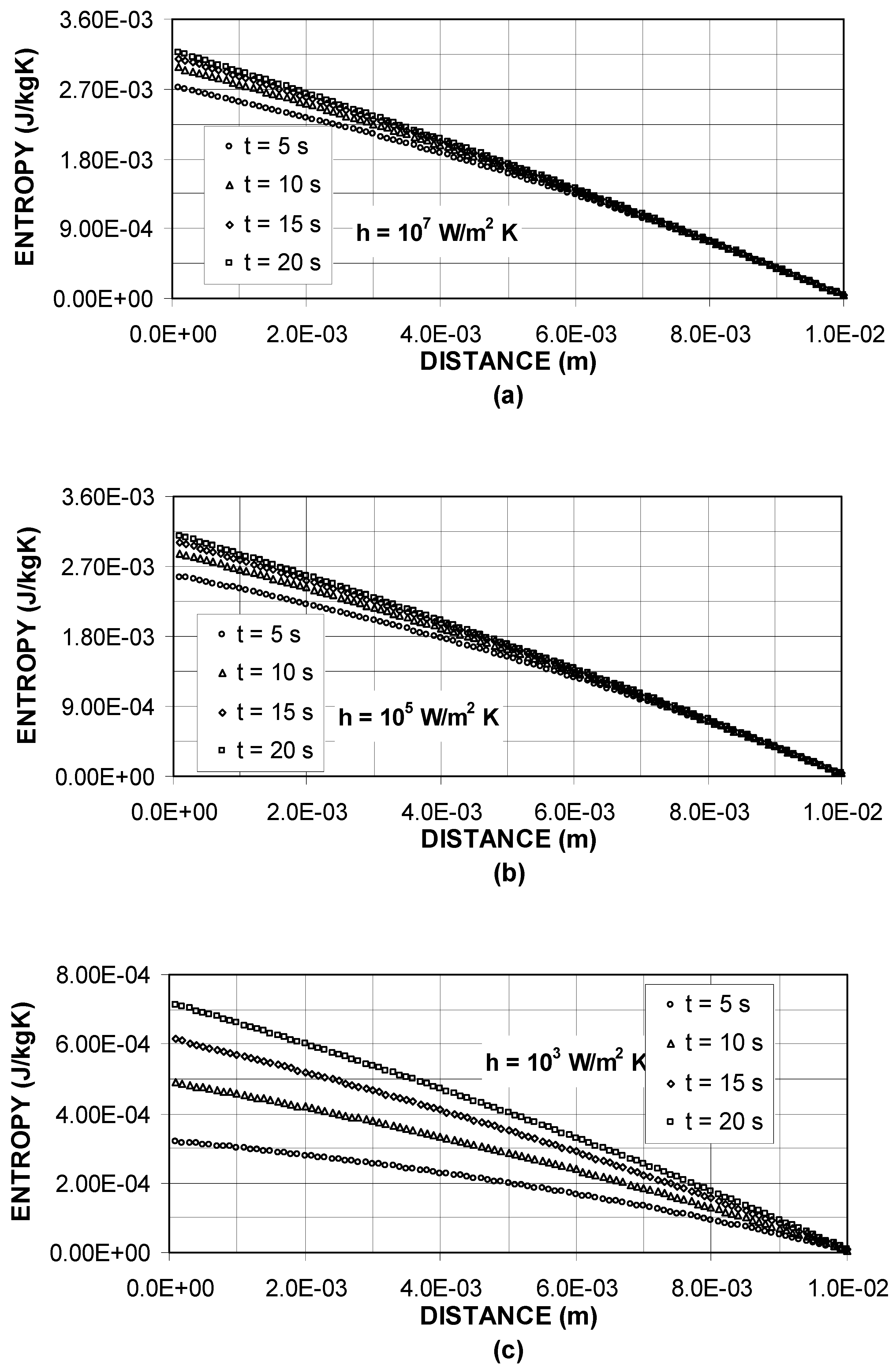
Figure 5 shows entropy generation due to the stress field inside the solid substrate for different heating periods. Entropy generation attains high values in the surface region and it reduces with increasing depth below the surface. Entropy profiles follow almost the displacement profiles, provided that their magnitudes are different. Moreover, entropy generation increases with progressive heating periods. This occurs because of the thermal strain, which enhances with progressive heating period. Entropy generation also increases with increasing heat transfer coefficient. In this case increasing heat transfer rates at the surface enhances the displacement in the surface region; consequently, entropy generation increases in this region. When comparing
figure 4 and
figure 5, it can be observed that entropy generation due to temperature field attains considerably higher values in the surface region as compared to its counterpart corresponding to stress field. This indicates that entropy generated due to internal energy gain of the substrate material dominates over the mechanical work done during the displacement of the surface.