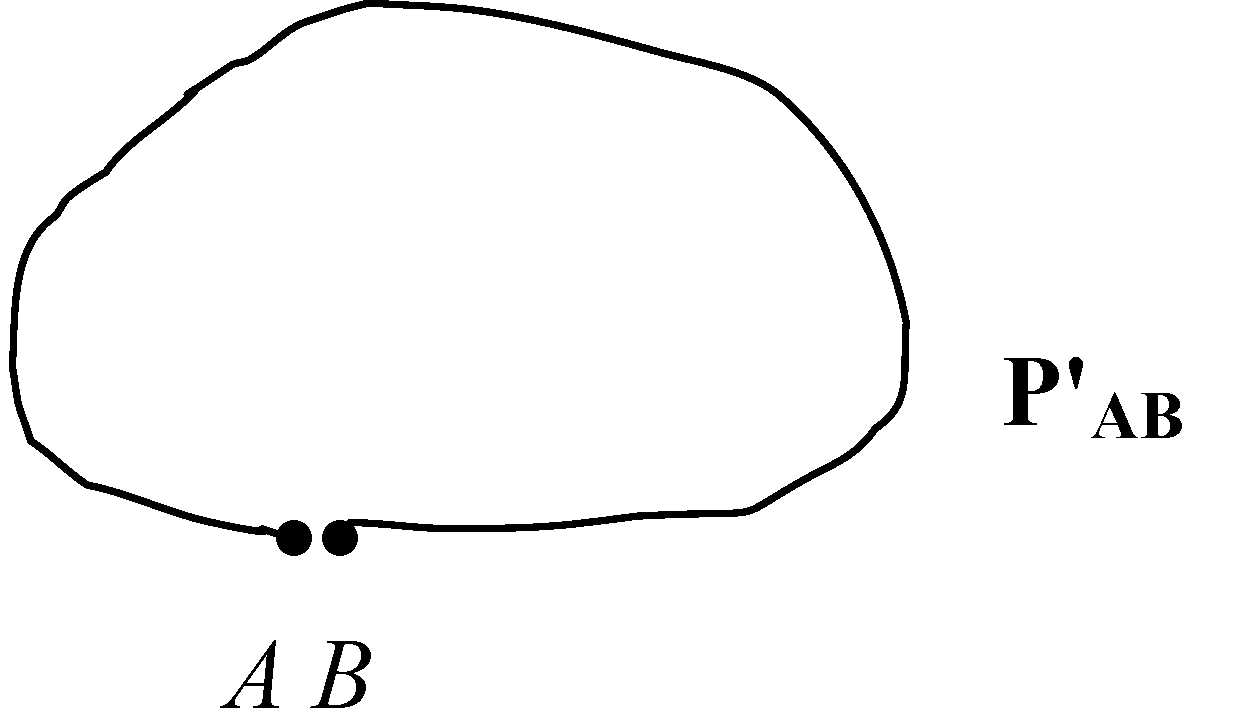1. Introduction and Semantics
In order to determine potential “flaws” in the pioneering 19
th century development of thermodynamics, one must highlight the well known presuppositions (stated below in axiomatic form) concerning the system under study. The axioms apply to closed systems.
For what follows, the systems are closed, such as obtains for the original Carnot engine; generalization to open systems were quantitatively and intuitively carried out most notably by Gibbs [
8] in his pioneering work, which did not resort to the rigorous consideration of cyclical changes which lead to the entropy function of state definition in the first place, which was presumed to obtain for heterogeneous equilibria involving open systems as well.
We now illustrate by some examples how the traditional interpretation of the Kelvin-Clausius theorems, based on the above axiomatics differ from those recently postulated [
9]; an instance is afforded by the Clausius definition of equivalent transformations. It turns out that the problem seems to be a divergence in how systems are categorized and understood.
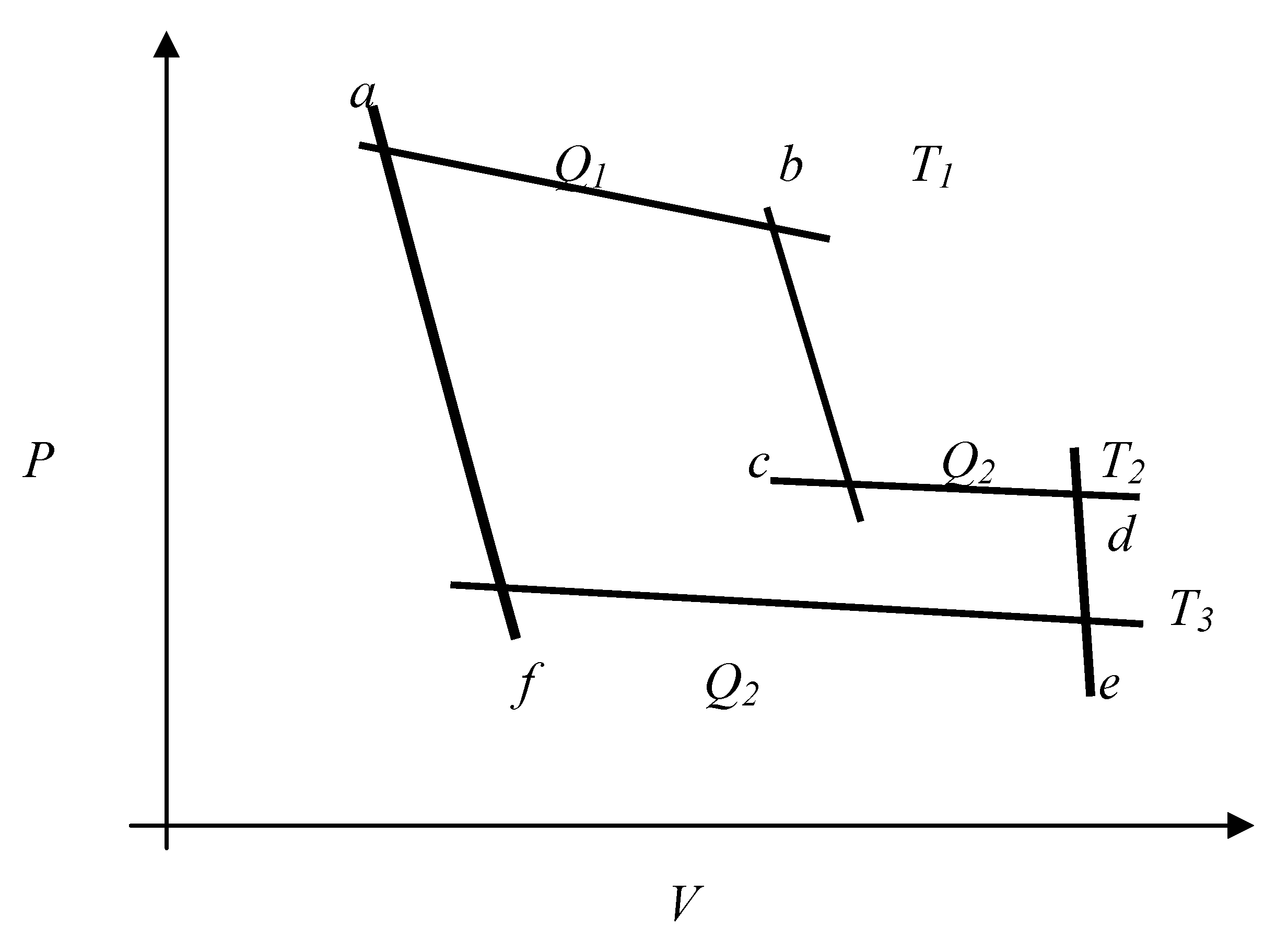
Figure 1.
A closed system utilizing three heat reservoirs undergoing a cyclical change by changes in pressure (P), temperature (T) and volume (V). The adiabatic steps are represented by the vertical curves such as a-f, b-c and d-e.
Figure 1.
A closed system utilizing three heat reservoirs undergoing a cyclical change by changes in pressure (P), temperature (T) and volume (V). The adiabatic steps are represented by the vertical curves such as a-f, b-c and d-e.
Fig. 1 depicts a closed system undergoing a cyclical transformation absorbing heat
and
at temperatures
and releasing amount
at temperature
in isothermal transitions involving heat reservoirs [
10].
Fig. 1 above reduces to
Fig. 1 of reference [
11] if the adiabatic step
b-c is reduced to zero measure (zero external work done). Our
Fig. 1 is adapted from a diagram concerned with a standard analysis of Clausius’ equivalence transformations [
10]. Let
denote the Clausius equivalence value of the generation of heat
at temperature
from mechanical energy, and
as the equivalence value of the flow of heat energy
from
to
. Clausius, probably relying on Kelvin’s 1854 paper on thermoelectric currents, makes the identities [
10]
and
, so that this equation for the total or net equivalence
between a flow of heat energy and the conversion of heat into mechanical energy becomes
for the above reversible cycle of
Fig. 1;
in retrospect may be identified with the entropy. If the Universe
is viewed as a set composed of the union of the closed system
and
thermal reservoirs
with the set of all reservoirs given by
, then our Universe may be loosely denoted by
. Clausius views the heat increments as changes in the reservoirs, rather than the system, where for reversible transitions involving heat exchanges with the reservoirs,
in set theory notation. Early workers, including Clausius, defined the heat absorbed according to Eq.(1) (or
Fig.1 in [
11]) as positive relative to the reservoirs, i.e. the entropy is measured in terms of heat absorptions experienced by the reservoirs, i.e.
. The generalization of Eq.(1) as the number of reservoirs tend to infinity leads to the Second law deduction for closed systems over a cyclical loop-like transition between the same end-points written as
The implication sign leading to the path integral in Eq.(3) requires a separate analysis which will not be attempted here, suffice to say that it has been the standard assumption throughout the history of thermodynamic analysis concerning the imperfect heat increment , where the thermodynamical path is Riemann integrable with respect to this heat variable , which is assumed to be a continuous function.
Never in the traditional understanding and development has ‘entropy’ or equivalent transformations been interpreted as pertaining to , i.e. the entire Universe taken as a whole[
12,
13] and all logical and algebraic developments have assumed that
is not the whole Universe but the variables connected to the system (or the respective reservoirs, but not both). There is therefore a danger of extrapolating the basic thermodynamical development beyond its intentions if this and other basic presuppositions are not borne in mind. For instance,
[
11] writes for his
Fig.1 transformations the following “So, the values of the transformations occurring in a reversible cyclical process instead of being
as the Clausius principle of the equivalence of transformations demand, they both turn out to be, as eqs. 1 and 2 show, equal to zero.” However, his
eqs.1 and 2 refer to the algebraic sum of contributions from
both system and reservoir (i.e. the entropy is essentially defined as
in our notation) and therefore differs in interpretation from the traditional development, including the definitions and axioms used traditionally. Indeed, the real significance of the non-composite sum
from the Clausius and traditional point of view is that it is equal to zero; the generalization of which leads to the Second law statement of the existence of the entropy state function. Since the
development does not use the same basic premises and axiomatics of the traditional Kelvin-Clausius thermodynamics, they are not comparable, and it is therefore questionable to speak of “flaws” in the traditional development when a common basis is absent. In isothermal exchanges of heat, clearly the algebraic sum of entropy increments must always cancel for system and reservoirs taken together, but no significant consequences may be deduced from this observation.
There have been attempts to create an irreversible thermodynamics with an entropy perfect differential based on considerations of “compensated heat” [
14], which has been disproved [
15] for isothermal processes; the analysis for the general case is given here in a subsequent section. Generally, concepts used in the attempts to extend thermostatics to nonequilibrium systems still rely on the structure and definitions used in equilibrium theory. In such developments, it is generally assumed that the Clausius inequality
obtains where the heat exchange is isothermal in the limit, even for irreversible transitions; clearly if this were not the case, then the ‘compensated heat’ entropy of the composite system would certainly break down, since the reservoir temperature variables are free to vary in this case [
14]. It will be shown below that the statements concerning the law of increasing entropy for systems obeying the stated conditions is not jeopardized by non-isothermal heat transfers between system and reservoir. It is interesting that at least one text [
7] has discussed the possibility of non-isothermal system-reservoir heat transfer as the basis or cause of irreversibility and the Clausius inequality, but in general the arguments seem to assume that the temperature parameter
is that registered at the heat reservoirs only which is the same as the system temperature when there is energy exchange with the reservoirs.
Another example of departure from tradition is due to the definition of quantities. Definitions (almost always) are not provable, but the effectiveness of definitions in scientific analysis is dependent on the rationality of the arguments used to create the definitions. For instance, whilst it is acknowledged that [
16] the efficiency
of a cyclical process associated with heat transfers between temperatures
and
is given by
, it is also stated that if the “availability of energy for external purposes” is defined
u as its quality, then the quality of loss
will be proportional to the temperature of its cold reservoir, i.e.
since its “capacity for further work production will be lesser the lesser the temperature of the cold reservoir...”. Elementary considerations show that as
, so that the capacity for further work production increases as
. We note that there is a danger in utilizing ratios, proportions and reciprocals without establishing the value of the exponents γ associated with a particular variable in expressions such as
; there is no proof why unity (±1) should be the preferred exponent from the arguments presented for proportions, etc. (although it may eventually be proved as the correct choice). Notational confusion can also add to the obscurity [
17] as when it is stated: “The true criterion of reversibility is embedded in eq.8 in the equation:
.... For every process that can in no way be completely reversed, it follows that:
”. From the fundamental theorem of integration, if
S possesses a differential
dS (in multidimensional space) it follows that
, irrespective of pathway (whether irreversible or not, provided
dS represents the entropy increment). Hence it is apparent [
17] that the writer concerned has at the very least used an inappropriate notation in his explanations. Unreviewed grammar may also complicate the situation, when subject and predicate is not clearly distinguished, as when one writes [
18]: “...it is learned that while the entropy change for the Universe of the reversible transfer of an amount of heat
from
to
is equal to zero, that of the irreversible transfer is equal to
. Looking back now at the principle of the equivalence of transformations one can only qualify as peculiar the fact that the entropy change
in it associated to the reversible transfer of heat taking place in the
reversible cyclical process to which such a principle refers, instead of being
zero, as it should correspond to a
reversible process, be
which is the one associated to an
irreversible heat transfer” (italics mine).
It is difficult to relate the above from the traditional perspective. Restricting the argument to closed systems delivering work W in one complete cycle, the entropy change of the reservoirs is given by whereas for a completely dissipating system, W=0, and so . Thus the intentions of the writer is not so evident here.
(Needless to mention perhaps, vague and imprecise writing which does not trouble to relate historically to previously accepted practices and to persons, and which does not take pains to point out areas of departure from pre-existing or prevailing practices, could potentially masquerade as a TOE -“theory of everything”- by claims that what was mooted could subsume whatever another creates or postulates, thereby jeopardizing the anamnesis sequence within the living communion of investigators, so vital for its non-nihilistic functioning).
Apart from ambiguity of terminology, we next examine whether it is possible to extend the equilibrium Clausius entropy principle to irreversible systems.
2. Discussion of the Clausius Inequality and Some Definitions
The derivation of the Clausius inequality
for heat increments
absorbed by a closed system in a closed loop transition is based on superimposing in the limit an infinite number of Carnot engines with common adiabatic steps between any two infinitesimal small loop transitions, such as discussed in [
7]. Two general cases may be considered, when the thermal reservoirs are in the limit at the same temperature of the system (subparagraph a) below and when they are not (subparagraph b).
a) When system and reservoirs are in contact at almost the same temperature: Associated system
For conductive thermal energy transfer between system at temperature and thermal reservoir at the Fourier inequality implies for arbitrarily small positive (where q is the heat current vector). On the other hand, a common assumption in the Carnot analysis is that there can be “isothermal” transfer of heat, so that For what follows, we shall assume that these limits obtain.
For any one Carnot engine cycle in this summation process, let the optimum heat absorbed be (at temperature ) and that expelled be (at temperature ) where and δW is the positive work done by the system (engine). The following definition will be required in due course.
Definition 1. Internally driven engine (system): An internally driven engine (system) i is one where for any engine Ei where the work refers to a cycle, and if the work increment for an element of path , the system or engine is internally driven along that path increment.
Definition 2. Externally driven engine (system): An externally driven engine (system) i is one where for any engine Ei where the work refers to a cycle, and if the work increment for an element of path , the system or engine is externally driven along that path increment.
Hence one necessary condition is that the system is not functioning as a refrigerator (where
) for
for an internally driven engine and where the system can never act as a source of work for an externally driven engine. From Axiom 1, the only way in which dissipation could be achieved is when work
, representing the Carnot maximum work (see
Fig. 2 below) is partly dissipated back by amounts
and
- where the subscripts (1,2) refer specifically to the interfacial ports of heat absorption and expulsion respectively - and held at temperatures
and
respectively.
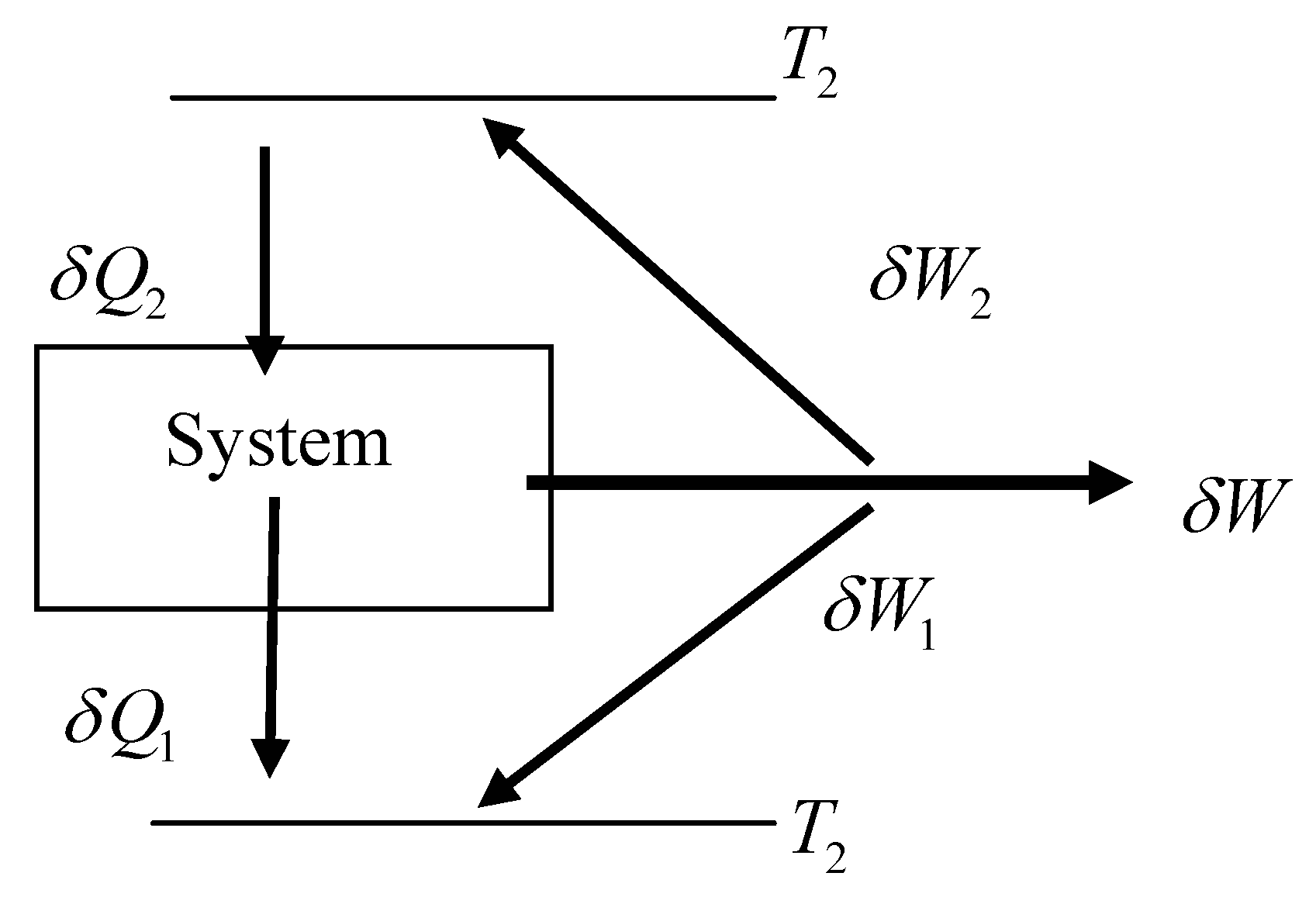
Figure 2.
Typical energy exchange of a Carnot engine during a cycle with dissipation.
Figure 2.
Typical energy exchange of a Carnot engine during a cycle with dissipation.
For internally driven engines,
, and for externally driven engines (e.g. refrigerating cycles),
where
is the actual work done on the system. For the above, it will be noticed that the work term, as it so far stands, is a globally relevant term, in that it represents the sum of all work increments over the cycle. It has been rigorously proven [
19] that any transition along the adiabatic steps in the Carnot cycle (a device used to ensure that the working substance can absorb heat at at least two different temperatures) itself conforms to the Carnot theorem, and furthermore, it has been suggested [
19] and proven [
20] that the “isothermal”” steps involving conductive heat with vanishingly small heat gradients also conforms to a Carnot theorem involving a “disintegrating” system [
19,
20]. Hence it might be possible to derive the above inequalities by
not concatenating “global” engines, but by a process involving local transitions, but this analysis is for the future; at present, on an
ad hoc basis, one can suppose –without jeopardizing the traditional format– that the system is coupled to a “work reservoir” that can store pure work energy at each stage in the cycle, and at different times, and this work is available at all times to the system. Since the energy
is unchanged over a cyclic transition, then over the cycle
where
is the total work done on the system and
the total heat absorbed; i.e.
in the notation of
Fig. 2.
Therefore, for net work to be done to the environment,
; hence as
for an infinitesimally small (arbitrary) cycle, an internally driven engine will always have the
capacity to do net work on the environment at any stage where the energy reservoir has zero work energy if
provided the cycle begins at the upper temperature isotherm. Denoting the entropy differential
, with superscripts labeling the system or heat reservoir, and the subscripts denote whether the increment is due to an irreversible or reversible transition. Then according to the notation of
Fig.1
for the system. If there is dissipation (irreversible change) we get
where
represents imperfect differentials and
is the potential work that is dissipated as heat to the thermal reservoir concerned through the system surface concerned. From Axioms {1-7}, a fundamental presupposition made for the Clausius inequality to obtain
for the system is that the potential work energy must be dissipated to the thermal reservoirs interacting with the system,
and that the dissipation of energy must be considered to flow through the system boundary to the thermal reservoir, or else inequality (5) would
never obtain and so likewise the Clausius inequality
would also not obtain; by definition, we have excluded thermal energy transfer for the adiabatic steps, although in a more flexible theory, one might distinguish between system heat absorption through its own boundary and heat transfer from a work reservoir to the thermal reservoirs, through another boundary as distinct (and therefore allow for a specialized form of heat transfer during an adiabatic transition). Here we adhere to the conventional definitions. From inequality (5), since the
’s are positive, the extra entropy gain for the reservoirs
would be
over the cycle rather than zero; in particular even if the work were dissipated to another heat reservoir external to the system, the total entropy change of the reservoirs and system would be positive, as is conventionally expected, since both the
δW′s and
T′s are positive. For permitted or accessible transitions from state A to B (denoted AB, and vice-versa BA for B to A transitions) the Clausius inequality reduces to
over a specified pathway. Integration yields
and
where the sign of
β cannot change for a BA irreversible transition (unlike α since the transition is irreversible). Thus, inequality (6) yields
Now, if we should allow Axiom 3 to obtain for the moment then the reservoirs involved in the heat interchange would be
. For the AB transition, we have that the total entropy change of system and reservoir is given by
from (6). This result is to be contrasted with Iñiguez who writes [
21]
“…That the entropy change for the universe of a process can be negative, a fact defined by Clausius work, is the reason the name chosen –negentropic—for the formulation herein presented.” The inequality is zero for reversible cyclic transitions, but it can never be negative.
b) When system and reservoirs are in contact at not the same temperature: Non−Associated system
Here,
where
is any finite non-vanishing number for thermal energy exchange. The above result is not jeopardized if the condition given by Axiom 3 is relaxed, such as what Moore alleges [
7], when he writes for the expression
(irreversible) , the following: “We should note that the
T ... is the temperature of the reservoir that supplies the heat, and not the temperature of the body to which the heat is supplied.” Clearly, this is in apparent contradiction to Axiom 3 as understood by other authorities; the above shows that for associated systems, (i.e. those for which the system and reservoir temperatures when in mutual contact possess a vanishingly small temperature gradient), Clausius’ inequality can still obtain if we postulate a back transfer of heat through the system surface due to work degradation; Moore’s postulate seems to not preclude the case where if the
T variable where almost exactly the same for the system and reservoir, then the Clausius inequality would not obtain in general if the above back transfer suggestion is not included. But Moore’s definition may not be too helpful since if
T were not the system temperature, then the inequality need not apply without further assumptions concerning the flow of (thermal) heat; hence it is imperative to consider Moore’s suggestion to define the limits of its applicability. If conductive heat transfer is envisaged [
22,
23] where the inequality
obtains locally, (
is the thermal heat conduction vector), then this fundamental inequality may be applied to any portion of a Carnot engine, at any stage in a cycle. Thus relative to
Fig.3 below,
Figure 3.
System absorbing thermal energy from heat reservoirs with dissimilar temperatures.
Figure 3.
System absorbing thermal energy from heat reservoirs with dissimilar temperatures.
is a positive increment of heat absorbed by the system whenever
and vice-versa, and
would be a negative quantum of heat absorbed (i.e. heat lost) of
. Now, in the traditional understanding, minute temperature differences
must exist with the condition
for heat absorption, or for work degradation into heat – by convention through the system boundary – whenever
at the same interface. Clearly the back transfer of work energy as dissipated heat through the same boundary as when heat was absorbed would involve the change of sign of
and these processes
cannot be simultaneously accomplished. If there exists a non-vanishing temperature difference
, where
is fixed, then the
isothermal absorption of heat by the system at temperature
and its conversion to work (e.g. in the isothermal expansion of a gas ) along a segment AB of a thermodynamical path where
>0 (for the system) implies
; if the transition along AB is along a pathway where the system boundary temperature is always less than the heat reservoir so that no work back-transfer can occur, then the curios result
would obtain; the heat absorbed would correspond to the reversible heat absorbed. However, for such a transition, the entropy of both system and reservoir would increase since for each increment of heat
absorbed by the system from the thermal reservoir,
for each
increment. For the segment of the system transition CD for which
<0, represented in general by
Fig.3 by the heat transition
between temperatures
and
,the gradient is such that if
represents the reversible transition increment, then by the external dissipation of work from the environment with increment
, there is the actual transfer of heat
, so that the entropy increment follows the overall entropy increment for the system
(in accordance with the Clausius inequality derived from cyclical considerations) and the total entropy increase of the Universe
for each increment
because
. This illustration may be generalized.
For a reversible transition between states A and B, the heat increments along a pathway can be represented by a sequence
, where
is a generalized path parameter completely specifying the path taken. From the First law statement
(with
Q and
W representing the heat and work functions respectively), it is clear that an irreversible transition need not correspond to the same
sequence; a particular transitional pathway may be specified by the set
with heat absorption increments
;
are all the other thermodynamical variables other than the temperature
T and total heat content Q for transitions between states A and B; the temperature of the system is given by the sequence
and time of transition by the sequence
; the temperature of the reservoirs is denoted by the sequence
where for the associated case
For this case
as demonstrated previously. We partition the sequence
where
represents positive increments for heat absorption
by the system with a similar notation for
, which represents the ordered (infinite) sequence with net heat loss by the system, the mechanism in both cases being purely Fourier heat conduction across a diathermal boundary[
22,
23]. The corresponding partitioned temperature sequences will be denoted
and similarly
are the corresponding time increments associated with the direction of heat transfer. For ‘associated’ heat transfers
for all members in
, and for such a case the reservoir entropy
clearly obeys
For the same specified path
to be traversed for non-associated systems subjected to the Fourier inequality, it is sufficient to maintain the temperature gradients in the same direction as in the associated case where
is a (small) finite and non-vanish quantity for a reservoir held at
and this reservoir temperature is maintained until the amount of heat
has been transferred; hence only the
sequence will differ for the associated and non-associated case. Since (Axiom 1) the system is instantaneously defined, and
for any
(it is as small as is specified),we can write
. Thus for the same system pathway
, the reservoir entropies would obey the inequality
Since
, for any definite pathway
, and in particular, Eq.(6) which yields
clearly implies
for the same AB transition and so even here the entropy of the Universe is positive. For such constructs, the Iñiguez result [
21] cannot obtain. The reservoir variables are free to vary in Moore’s characterization, so it is not necessarily true –subjected to further elaboration- to suppose that a “compensated entropy” may be described to create a new function of state because of the ambiguity of the temperature value, where we suppose for the time being that such theories are true [
14] for associated systems. However, even for associated systems, it has been proved that such a scheme does not exist for isothermal systems with no work transitions [
15]. Hence it is imperative to study the case of non-equilibrium transitions for associated systems to determine whether any new function(s) of state are implicated as a generalization of Clausius’ result.
3. Investigation on whether a Nonequilibrium Entropy may be Deduced from the Clausius Inequality of Equilibrium Thermodynamics
(a) Formal theoretical construct
The lucid arguments of Benofy and Quay [
22] make a case for the global nature of the descriptions provided by the traditional Kelvin-Clausius development, which must be contrasted to its reduction to a supposed local form from total system interactions involving circular integrals [
24]; the resolution of these conflicting viewpoints is attempted here. Writing the Clausius integral as –
N and integrating between thermodynamical variable space points A and B located on the thermodynamical path of the loop yields the following “global” [
22] result
where
is the reversible entropy change between states A and B; where
denotes a reversible path and
an irreversible pathway, and the state variables for the system is
Q for each equilibrium state.
Lemma 1: The variable must be a functional of the variable A,B and path , i.e. .
Proof: Since is the integral of a perfect differential, it is a function of the endpoints of the integral, and the irreversible integration along is path dependent, hence the result.
In order to develop the global properties of the Kelvin-Clausius theory, we need to state some elementary theorems, where the first theorem is more obvious than the second.
Theorem 1: There exists an infinite number of irreversible heat exchange pathways even if during the irreversible transition from A to B, the system traverses arbitrarily closely along a reversible pathway where the pathway is described by a sequence of equilibrium state variables Q.
Proof: From Axiom 1 the system state is instantaneously defined, so that the isothermal heat exchanges are due to the dissipation of heat about the system boundary due to the system work or the external work reservoir, and likewise at each point in the time sequence, the virtual reversible transition may be characterized by an equilibrium state variable, the entire set constituting
. In accordance with the well-known Axiom 8, the dissipation of work to heat through the boundaries of the system is bounded by
Wop, the optimum work which the cycle can perform. However, if the external environment were to dissipate energy in addition to
Wop, the result below would still obtain by considering the dissipation inequalities below. Now, consider the loop formed by the reversible path
followed by the irreversible transition
, such as depicted in
Fig. 4 below. By Axiom 1 and 8, we may consider one elementary Carnot cycle with corners at (
a1, b1, b2, a2) where heat
Q1 is absorbed along reversible segment a
1-b
1 and
ejected about irreversible segment
a2-b2.
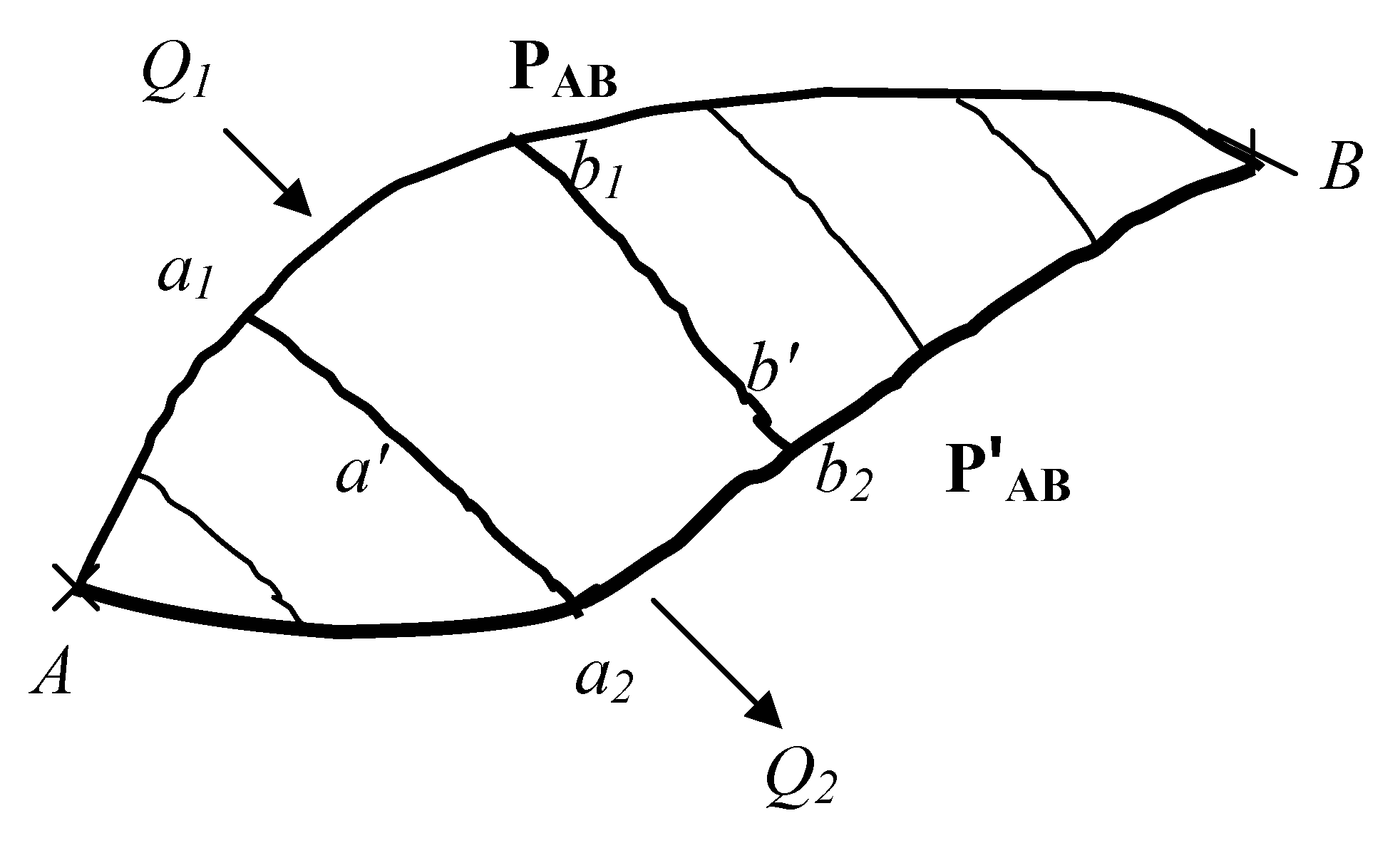
Figure 4.
A thermodynamical circular pathway with the reversible portion maintained at a higher temperature than the irreversible segment, and where heat is absorbed, with elementary Carnot engine superimposed with isotherms and adiabats meeting at a1, b1, b2 and a2.
Figure 4.
A thermodynamical circular pathway with the reversible portion maintained at a higher temperature than the irreversible segment, and where heat is absorbed, with elementary Carnot engine superimposed with isotherms and adiabats meeting at a1, b1, b2 and a2.
The system here corresponds to an internally driven engine; the arguments for an externally driven engine are similar (and will be omitted). At the optimum (op) level, , where is the work done on the system for the reversible cycle about (a1,b1,b2,a2) and as stated previously. Since segment (a1,b1) is reversible, the amount of heat ejected about the (a2,b2) segment would be where is the dissipation function about segment ((a2,b2) where and if there is extra work provided externally, then the preceding condition reduces to .Clearly is not dependent on the or sequence of path variables and an infinite number of separate values for exists for each infinitesimal segment (a2,b2). Hence an entire set of dissipation functions obtain for this infinitesimal segment, and thus for the entire pathway which corresponds to the system having a particular set of equilibrium variable sequences, we can write , where are the variables along connected to the dissipation function about the boundary of the system.
Theorem 2: Subject to the (system) conditions of the preceding theorem, it is impossible for the irreversible pathway (for any finite length) to contain the same sequence of points as (in the opposite direction) for any fixed, predetermined path in a closed loop Clausius integral.
Remark: The above theorem precludes the localization of the irreversible and reversible connected segments of the Clausius closed loop integral.
Proof: Any segment
(see
Fig. 4) may be completed by the reversible pathway
to form a closed loop, where
possesses at each point the state variables belonging to the reversible pathway
(
in terms of the sequence of points ) as in the preceding theorem. We now distort the contour
such that
, where
as described previously. Then for each virtual Carnot cycle, the adiabatic paths
b' and
a' → 0 in length, so that
because
and
(along the entire irreversible segment)
Hence the opposite direction pathway
is a reversible pathway with
for each infinitesimal isotherm, and since
,
cannot therefore be irreversible.
Corollary 1: The Clausius inequality of the closed loop integral under the conditions of Axiom 8 precludes the non-trivial existence of a local infinitesimal excess quantity dN for any of the following definitions of dN: (a) , (b) and (c) where N is defined as in Eqs.(10a,b).
Proof: Case (a) If
, then since
,we have
The general possibilities for (11) imply a cusp-like topology as in
Fig. 5 (or its generalizations to intersecting looped chains) or line for the path.
Figure 5.
A simple cusp or loop path as .
Figure 5.
A simple cusp or loop path as .
If a cusp or cusps are implied, then in the limit as , we get dN = N(P'AB), where dN is a finite, non-vanishing number if in (11) and the length of the path is bounded, and so dN is dependent on P'AB and is clearly not a differential element since it is a finite number. If the condition applies, then from Theorem 2, since is irreversible, it cannot contain the same sequence of points as , and so is a non-local path dependent parameter. Writing the heat absorption increment as where dR is the element of length in thermodynamic space and H a bounded function specific to the pathway implies that as , dR→ 0, so dN→ 0 and in the limit, dN = 0.
Case (b): From (a), if , then from Theorem 2, trivially since an irreversible path would not exist.
Case (c): From (a) and (b) , .
Corollary 2: N is at most a constant function locally since when there is a coincidence of paths from Corollary 1 and Theorems 1 and 2.
Corollary 3: It is not possible to have a (continuous) sequence of states which are known to be nonequilibrium states be described by a continuous sequence of thermodynamical equilibrium states for if this was the case, Theorem 2 would be contradicted.
We now investigate whether (10a) rewritten as
and the expression
(circular integral) leading to
implies the existence of a new entropy exact differential
such that
The formal development above indicates clearly that
if a cusp-like non-local loop is involved from Corollary 1 where dN is a finite number and is not a differential, and therefore
is not a differential. If locality (implying coincidence of the thermodynamical variables) is imposed as in Corollary 2, then
is a perfect differential, where
must be defined as the reversible heat increment, which is
not obvious from (13b). Hence the formal development based on traditional axiomatics does not in general support (13b) as a candidate for a new irreversible entropy differential [
24]. In order to confirm the above for the general case, we resort to a direct evaluation.
(b) Direct analysis of irreversible entropy increment
The integral
is not arbitrary but has a form which must always conform to the defining relations (10a), where the pathways for the reversible and irreversible portion of the integral must be carefully distinguished, i.e.
Hence generalizations of (14) include expressions such as
for a cyclical
reversible path AA and a similar expression obtains for irreversible paths, i.e.
Eqs.(15-16) makes it clear that Eqs.(10a,b) are not arbitrary circular integrals over all thermodynamical spaces such that
, but that
must be defined always in terms of the reversible and irreversible paths traversed, i.e.
leading to Eq.(10a)
Reference [
14] for instance has
( Eq.(10) there, denoted (10') here) and
(Eq.(9) there, denoted (9') here) so that substitution of (10') into (9') apparently yields
From (14), retracing the substitutions leading to (19) leads exactly to
So, exactly the same equations are subtracted i.e.
, from which a perfect differential was deduced in reference [
14]; this tautology cannot imply that
are conjugate variables, in the sense that
are in thermostatics, (which incidentally are experimentally distinguishable and defined differently theoretically). It is deducted that since
, then
is a perfect differential. This is true if
were defined over all completely arbitrary pathways, but since
dQ ,
N and
dN are all specifically characterized by the pathways and points (
A, B, PAB, P'AB), there being different functionals
dQ,
N and
dN for different (
A, B, PAB, P'AB), it does not follow in general that the same infinitesimal quantities (e.g.
dQ and
dN) obtain for different pathways; it they did, i.e. if these quantities were defined independently of the variation of thermodynamical space, then
would be a perfect differential, but this has not been explicitly demonstrated. Further, it is clear that the infinitesimals such as
are not simultaneously defined for exactly the same thermodynamical space coordinate, which must obtain if a test is to be performed for their sum being a perfect differential; Corollary 2 give examples of the resulting value of
dN when the contour is localized. In particular, if points A and B are considered fixed then (10b) yields
from which it would be not true to state that
for every irreversible segment, despite
since the irreversible heat exchange has indeterminate sign. Hence the statement "The differential ….
for every irreversible segment, or it would be possible to contrive…" [
14] need not obtain for non-local situations, whereas Corollary 2 shows that for the limits mentioned, the local result reduces to
, where clearly
must represent the
reversible heat increment for
to be a perfect differential.