1. Introduction
In the first half of the 19th Century, von Mayer and Joule discovered the energy conservation principle, demonstrating that heat (internal energy) and work are inter-convertible, and that the total energy of the universe is constant. The Second Law forbids the realization of perpetual motion machines and limits the effectiveness of heat engines and cold machines (heat pumps and refrigerators). Based on Carnot’s theorems (in the more analytical form developed by Clapeyron), Thomson and Clausius greatly improved the theory of heat engines. Clausius eventually introduced the concept of entropy to represent the transformation or
Verwandlung of heat into work and vice versa [
1,
2]. The year 2015 marked the 150th anniversary of this definition, but it continues to be a confusing concept in thermodynamics. It is one of the challenges encountered in the attempt to reconcile the microscopic view of matter offered by statistical thermodynamics with the macroscopic view of classical thermodynamics. In Clausius’ work [
2,
3], the term emerges from his attempt to quantify the transformation of heat into mechanical work and mechanical work into heat. It should be noted that, although the equality of work and heat was established in the First Law, the physical units were often not the same, so that a conversion factor was often needed in quantifying the mechanical equivalence of heat. Work was generally given in units of kilogram-meters (kgm), from which in modern terms, the corresponding potential energy in newton-meter or joule would be obtained as the product of that quantity in kilogram-meter and the acceleration due to gravity. On the other hand, heat was often quantified in terms of the work-equivalence of heat, whereby 423.55 kgm (or about 4155 J) of work are needed to raise the temperature of 1 kg of water by 1
C [
2].
Confusion about the meaning of entropy is not a new problem [
4,
5,
6]. A few years after its definition, Tait proposed a redefinition of entropy as the useful part of energy [
7]. This idea was initially taken up by Maxwell [
8]. Tait seemed to have pursued this line of thought in order to demonstrate that the entropy definition proposed by Clausius had initially been obtained by Thomson in the latter’s earlier publications [
7,
9,
10]. The widespread misconception about entropy continued into the 20th century with von Neumann allegedly advising Shannon to adopt the name entropy in his information theory, with the justification that “... nobody really knows what entropy is, so in a debate, you will always have the advantage” (adapted from [
6]).
Clausius’ analysis focused on the interaction of a heat engine cycle with heat reservoirs and he arrived at the cyclic integral,
, that equals zero for a reversible cycle. One of the critics of the foundations of Thermodynamics, Truesdell, attacks the rather circular definition of irreversible processes offered by Clausius: irreversible processes are those processes that are not reversible [
11]. One needs to understand irreversible processes in order to understand reversible ones. Truesdell is looking for a mathematical expression for irreversible processes since he criticizes Thermodynamics as a subject with an unusually high ratio of words to equations. In modern thermodynamics textbooks, this difficulty is circumvented by defining reversible, internally reversible, and externally or fully reversible processes [
12,
13]. Uffink, in his criticism of the liberal extrapolation of the Second Law to the concept of the Arrow of Time, points out the problem with the opaque distinction between reversible and irreversible processes [
6]. It should also be noted that little attention is paid to the heat reservoirs in Clausius’ derivation in modern discussions of entropy. Difficulties in reconciling classical thermodynamics with statistical thermodynamics [
14,
15,
16] also seem to be related to differences in the conceptual understanding of entropy in these two fields. The need to clarify classical entropy continues to attract attention with a number of new articles devoted to this topic.
Another area where problems arise owing to the current view of entropy is in metrology, where it is used in defining the thermodynamic temperature scale [
17]. Recommendations have been made to the international system of units to consider changing the temperature unit from kelvin to a unit based on the Boltzmann constant, an energy parameter. The argument for this change is that precise temperature measurement based on the kelvin is still dependent on the equilibrium properties of a given material, whereas relating temperature to the more universal energy constant,
, will make temperature measurement independent of the material, method of realization, and temperature range. Could some of these conceptual problems be resolved by only including energy variables in entropy definition and dispensing with the central role of temperature? Furthermore, in quantum metrology, some authors argue that the conditions for thermodynamic equilibrium and thermal contact are not met; this renders energy quantification through the kelvin meaningless [
18,
19]. These issues are all related to entropy definition in the sense that its definition was seen as a basis for the realization of a thermodynamic temperature scale. In the process, the implicit energy unit was lost and the notion of absolute temperature was given more significance.
In its historical context, entropy is wedded to the concepts of heat engines and cold machines. It is important today to clarify the physical meaning of this important property, since thermodynamics is increasingly used in the analysis of a wide range of problems that are very remote from the concept of heat engines. Examples include biological processes and quantum mechanical systems.
In this work, we offer a modified definition of entropy aimed at clarifying its physical meaning. We suggest that heat transfer between two systems initially at different temperatures is a better physical model for entropy explanation than the usual heat engines. From this perspective, entropy is closely related to non-equilibrium thermodynamics: we start with two systems at equilibrium but such that their temperatures are different; their interaction leads to the flow of heat in the direction of the temperature gradient. This, together with the heat exchange sign convention, leads to the principle of entropy increase. Alternatively, the entropy increase principle could be associated with the evolution of an isolated system from a prepared non-equilibrium state toward a new equilibrium state. We also show that further modification of the suggested entropy definition makes it possible to arrive at the proposition of the Third Law or Nernst Theorem, without the need for a separate law. In essence, the Third Law establishes the entropy difference for systems or processes at two temperatures close to zero.
We start by restating the definition of entropy and the related heat engine analysis. We then discuss some of the conceptual difficulties arising from this definition. This is followed by a presentation of our modification. We further assess the use of this modified definition in the analysis of model processes. We then discuss how the modified version of entropy aligns with statistical thermodynamics entropy and the Third Law.
2. Clausius Approach to Entropy Definition
The kinetic theory, thermodynamic process relations, and the First Law were well established before Clausius’ work on entropy.
The ideal gas provided a link between macroscopic and microscopic thermodynamics. The pressure exerted by an ideal gas is related to the average kinetic energy of a constituent particle, the system volume, and the number of gas particles:
In terms of temperature:
where the purely thermodynamic property, temperature, is taken to be related to the average kinetic energy of the particles as:
One could define as an energy variable to obtain the gas law as . More will be said about later, but we note here that this is equivalent to in statistical mechanics. For the purpose of this paper, β is not preferable; it grows unbound at low temperatures, complicating the new physical interpretation we offer. It should also be noted that, in statistical mechanics, β is motivated by a constraint on the total energy as entropy is maximized. This role could as well be played by ϵ, taken as . In Gibbsian statistical mechanics, we shall comment that ϵ is similar to an energy parameter that Gibbs called the modulus θ. The equation of state above is obtained using the kinetic theory, which is part of statistical mechanics. This attests to the central role of statistical physics in the foundation of classical thermodynamics.
For an adiabatic process from state 1 to state 2, if constant specific heats and ideal gas behavior are assumed, then two of the three state variables, pressure, temperature, and specific volume may be related as:
The efficiency of a heat engine is generally given as the ratio of net mechanical work to the heat supplied:
For a four-stroke heat engine cycle with two adiabatic processes, heat is added during one of the four processes and rejected during another. The heat exchange can be determined from the First Law for closed systems:
where
U is the internal energy,
is the heat added,
is work added, so that the internal energy of the system can increase on account of heat and/or work addition. For an ideal gas, internal energy is a function of temperature only, and
, so that for an adiabatic process:
Isothermal heat exchange, therefore, implies that the internal energy stays constant and
For an isothermal process in which intermediate states can be assumed to be in quasi-equilibrium, pressure and volume are related as
One can make use of the ideal gas law to express
p in terms of
V and
T or
V and
ϵThe efficiency of this heat engine cycle can be shown to be
What is lost in establishing Equation (
12) is that the heat added to a system translates to internal energy that is characteristic of microscopic motion and can be measured in terms of
. This is the motivating idea that led to the discovery of entropy, namely, the search for a property of the system that scales with the heat added to a system. If we sought to maintain the energy connection as we replace
and
, we should have the efficiency as
However, with our intensive energy variable,
, this takes the form
Clausius’ entropy definition draws from Equations (
6) and (
12), while ours draws from Equations (
6) and (
14), motivated by the emphasis on the energy variable as the best physical quantity to connect heat to a mechanical property of the system receiving or rejecting heat.
Tait criticized Clausius’ entropy definition, alleging that it was a restatement of Thomson’s earlier work. This view seems to stem from a 1854-paper in which Joule and Thomson sum up their quest for an absolute temperature as:
If any substance whatever, subjected to a perfectly reversible cycle of operations, takes in heat only in a locality kept at a uniform temperature, and emits heat only in another locality kept at a uniform temperature, the temperatures of these localities are proportional to the quantities of heat taken in or emitted at them in a complete cycle of operations.
This is equivalent to saying that .
As mentioned before, Clausius’ path to entropy definition starts from the Carnot cycle and seems to be prompted by the fact that heat and work are interchangeable but did not have a common physical unit at the time. Transformations of work to heat and vice versa are such that the following relation holds,
The equivalence value of the transformation of heat to work (
original German: Äquivalenzwert der Umwandlung von Wärme in die mechanische Arbeit) can be determined as
For a series of reversible heat engines in contact with many reservoirs, the equivalence value of all transformations can be determined from
For a very large number of reversible heat engines, the summation can be replaced by integration
The term
entropy is then introduced such that its differential corresponds to the transformation
Clausius then seeks a suitable form of the function
τ and
, where he considers that
. Drawing from the previous derivation of the thermal efficiency of a Carnot heat engine using an ideal gas with constant specific heat, the equality
, is used to suggest that
. However, Clausius recognizes that this choice is not unique, given that the ideal gas uses many assumptions and a number of temperature functions could equally be acceptable [
3].
One of the weaknesses of Clausius’ approach to entropy definition is the sole focus on the reversible heat engine (Carnot) cycle, for which the net entropy in a cycle is zero. The fact that the efficiency is independent of material properties, is often presented as a special feature of the Carnot cycle. However, it is known from the analysis of four-process heat engine cycles with two adiabats and heat exchange at constant volume, temperature, or pressure, that the thermal efficiency is
. Here,
is the temperature at which the working medium loses contact with the heat sink and
is the temperature at which the working medium establishes contact with the heat source [
12,
13]. Such cycles include the Otto cycle with heat exchange processes at constant volume, the Brayton or Joule cycle of a gas turbine with heat exchange processes at constant pressure, and the Carnot cycle with heat exchange at constant temperature. It happens that in the case of the Carnot heat engine,
and
are also the minimum and maximum temperatures of the cycle, respectively, while the maximum temperatures in Otto and Brayton cycles are higher than
. Clearly, their efficiencies can be represented without invoking the materials of their working media. Analyzed without explicit focus on entropy changes of the heat reservoirs, all of these heat engine cycles have no net entropy generation. Therefore, focusing on the cycles, and not including the heat reservoirs or the walls enclosing the working medium, obscures the origin of entropy generation. Including the analysis of the heat source and heat sinks in entropy analysis is more complicated than would be achieved with a simple explanatory model process.
Furthermore, in theoretical analysis of heat engines, no attention is paid to the walls of the device, but they tend to be sources of entropy generation through friction, heat storage, and heat transfer. It is therefore difficult to transfer analysis of heat engines to thermodynamic systems that are not concerned with work and heat interconversion. Furthermore, heat engines and refrigerators rely on thermal non-equilibrium and the associated tendency to effect heat transfer in quest for thermal equilibrium. Heat transfer is a more appropriate thermodynamic model process to explain entropy generation in heat engine systems as well as in processes that are not related to heat engines but potentially subjected to temperature gradients.
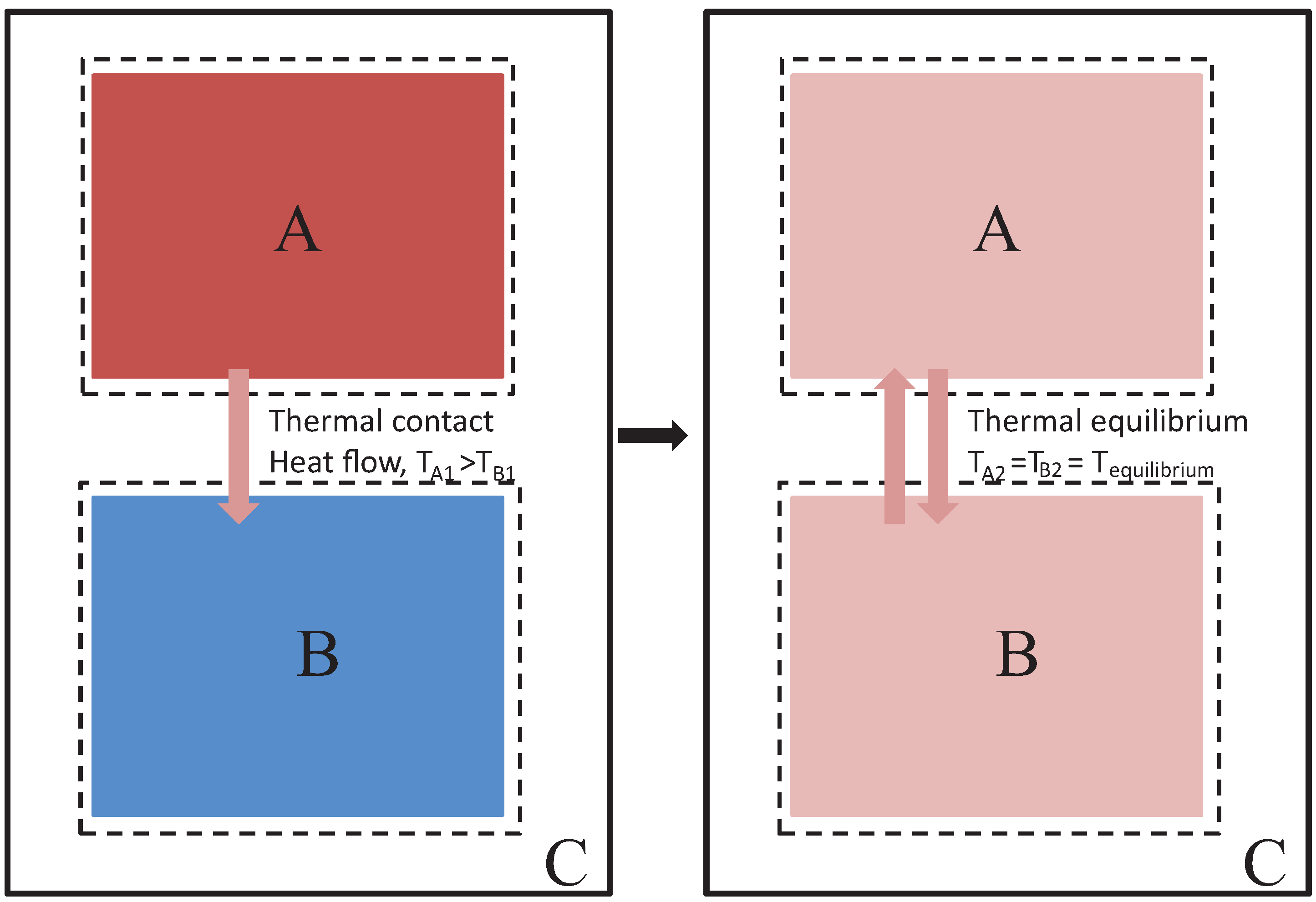
- 1
We start with a simple heat transfer problem. Two systems, 1 and 2, are separately in thermal equilibrium but at different temperatures, that is
(see
Figure 1).
- 2
We allow the systems to interact, and we seek a parameter whose sign will unambiguously indicate the natural tendency of heat to flow from hot to cold.
Since Clausius’ initial motivation is to seek an equivalence between heat and its transformation to mechanical work, he presents an interesting conceptual scheme according to which heat transfer proceeds through initial conversion of heat from the source to mechanical work, followed by conversion of this mechanical work back to heat at the lower temperature region. That is, heat going to work corresponds to reducing the intensity of random microscopic motion (corresponding to very high degrees of freedom) by transferring the related energy to macroscopically ordered motion that is observable to us as work (bulk motion with up to three degrees of freedom); conversely, work going to heat is equivalent to transforming the energy of macroscopically ordered motion to more intensified random microscopic motion that becomes unavailable to directional bulk motion. If we leave aside Clausius’ mechanistic picture of heat transfer, we simply view the hotter region as one with average particles that are faster and the lower region as one with average particles that are slower. Allowed to interact by collisions, the slower particles gain energy while the faster ones become less energetic.
- 3
A fundamental indicator of the natural tendency of this transformation is the specific entropy change. It is preferable that it takes the form of a non-dimensional energy parameter to correspond to our notion of invigorating or reducing microscopic motion (a relative assessment):
where
s is the specific entropy,
q is the amount of heat added per unit particle of the system,
k is the Boltzmann constant and
T is the temperature. The convention is such that for heat added
is positive; hence,
s increases.
We use the microscopic energy variable,
, playing the role of an intensive energy of the system, with units of joule per particle. A derived energy unit for
ϵ could be named after Clausius or to avoid confusion between specific internal energy and our new intensive energy,
ϵ, the latter could be called Clausius energy. This microscopic energy is basically the average kinetic energy of a representative particle without the 1/3 factor that accounts for the three translational degrees of freedom:
This modification does not change existing thermodynamic relations, but the units are now aligned to give better insight to the physical meaning of specific entropy. The fundamental thermodynamic potential becomes in specific terms and in units of energy, where . Here, is a driving force with as the associated coordinate.
- 4
In order to determine the sign of entropy change associated with natural heat transfer from a hot to a colder region, we consider heat transfer from the hot body, 1, to the colder body, 2. For simplicity, let both have the same mass,
m, and the same temperature-independent specific heat capacity at constant volume,
. Let us further assume that no volume-change work occurs, so that all heat transfer only results in changes in the internal energy of each system and
. The common final temperature, hence final intensive energy, can be determined from:
We note here that or , correspondingly.
The specific entropy change,
, associated with the process can be determined from:
The last expression in Equation (30) can be rewritten as
That is, two systems initially at different temperatures, such that 1 is hotter than 2, if allowed to interact by heat exchange will reach a new equilibrium state with a temperature intermediate between the initial body temperatures. The associated entropy change is always positive. Thus, the process is that of the non-equilibrium interaction of systems seeking a new thermal equilibrium state.
The problem considered above imposed a direction by requiring that both systems achieve an equilibrium temperature; heat must flow from the hotter to the colder until thermal equilibrium is attained. We found that this natural process is associated with an increase in entropy. Now, suppose we have two bodies, 1 and 2, of equal mass and heat capacity, but we are not told which of them is hotter, and we seek to establish the condition that has to be fulfilled for the associated entropy change to be positive as they go from states 1 to 1’ and from 2 to 2’. Assuming that
decreases by
, then by virtue of energy conservation,
increases by
. Similarly,
and
:
From Equation (34), two observations can be made. Firstly, for natural heat transfer, that is, , it is necessary that , bearing in mind that it was assumed that body 1 transfers heat to body 2. Secondly, if , we are dealing with fluctuations around thermal equilibrium, so that . The result, , can be directly connected to the statistical mechanics result, , where, for a naturally occurring process, the number of ways of realizing the microscopic energy distribution or multiplicity of the final state, , must be greater than that of the initial state, .
- 5
Having freed the entropy increase principle from the unnecessary analysis of heat engines and refrigerators by favoring the heat transfer model, we now focus on how the heat transfer model can be used to explain the performance of heat engines and refrigerators as well as two common formulations of the Second Law.
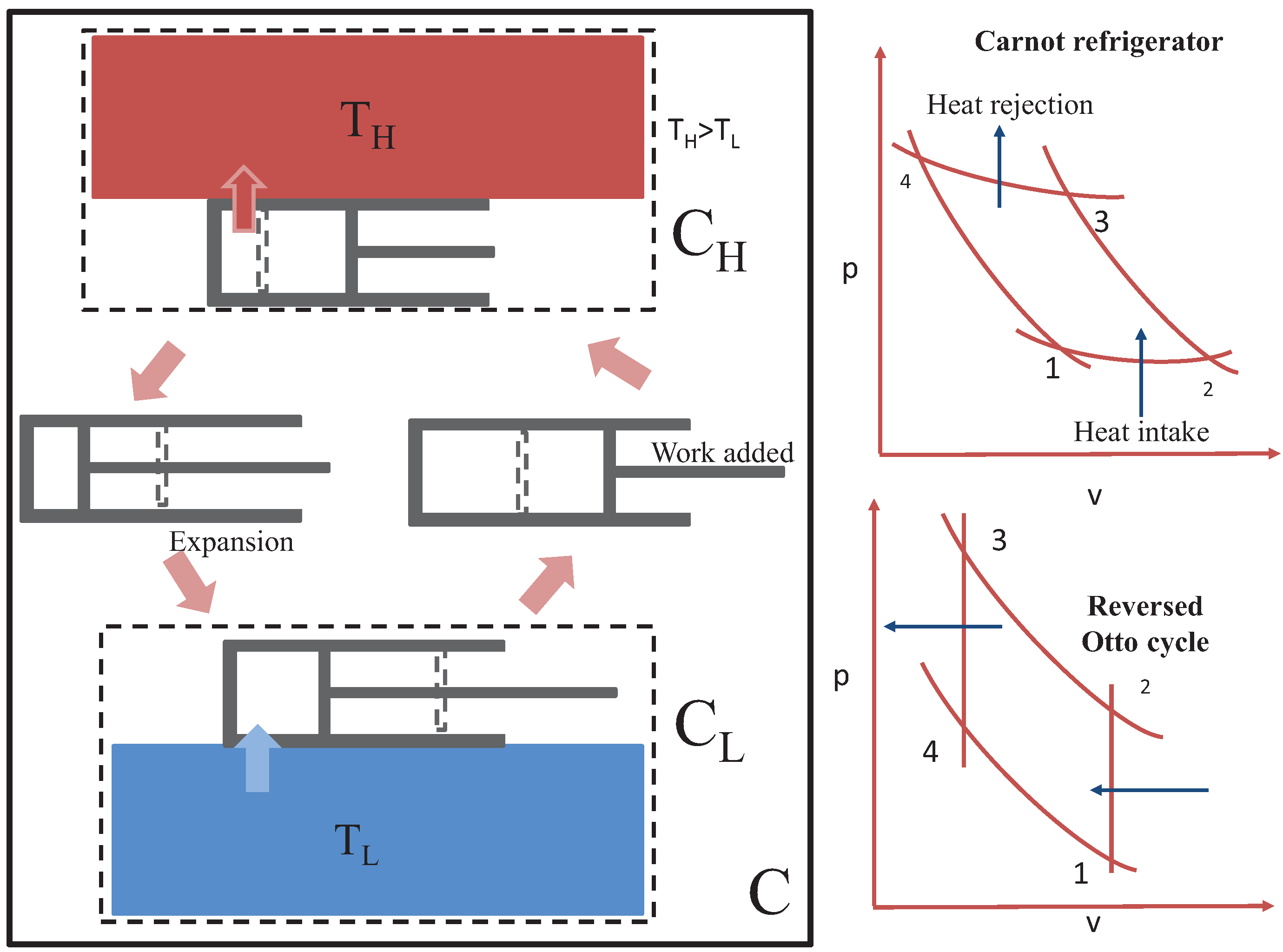
Figure 2 is a schematic of a heat engine embedded within a natural heat transfer process, with a hot reservoir maintained at
and a heat sink maintained at
. By means of a piston-cylinder device working in a cycle, mechanical work can be obtained from the configuration, with the effect that less heat is transferred to the heat sink. The working medium leaves the heat sink at state 1 and is raised adiabatically to a higher pressure and temperature until it reaches state 2, where it establishes contact with the heat source. It loses contact with the heat source at state 3 and adiabatically expands to yield useful work before re-establishing contact with the heat sink at state 4. If the heat transfer process is infinitely fast, the heat exchange processes occur at constant volume, and the device establishes contact with the source at
and with the heat sink at
. Viewed from the “universes”
and
, all heat transfer processes are natural, and thus, are accompanied by entropy increase so that from the perspective of the “overall universe”
C, the entropy increase principle is fulfilled. In the most efficient case, heat transfer occurs such that
and
, which requires infinite time for the isothermal heat exchange process (will have to rely on equilibrium fluctuations). In this limiting case, no entropy is generated in the universes
.
The operation of cold machines (heat pumps and refrigerators) is illustrated in
Figure 3. The processes take place in the reverse order of those of heat engines. However, the externally perceived transfer of heat from a region of low temperature to one of high temperature is assured by embedded heat transfer processes that obey the natural law. Net entropy is generated in the universe,
C, in all realizable devices, with the exception of the ideal case where heat exchange between the working medium and the reservoirs occur as fluctuations around thermal equilibrium at all times.
The apparent counter-intuitive flow of heat from a region of lower temperature to one of higher temperature relies on the conversion of work to internal energy so that the working medium attains a temperature that is higher than that of the hotter region to which heat is rejected. In other words, a thermal non-equilibrium condition is brought about by converting mechanical energy into energy of random microscopic motion.
From the foregoing discussion, we can combine the two commonly stated formulations of the Second Law into a single heat transfer principle and discuss the various formulations of the Second Law as implications of this main principle. We recall that the heat engine or Kelvin–Planck formulation [
22] is stated as: “
It is impossible to devise an engine which, working in a cycle, shall produce no effect other than the extraction of heat from a reservoir and the performance of an equal amount of mechanical work”.Similarly, the refrigerator or Clausius statement is often stated as: “It is impossible to devise a machine which, working in a cycle, shall produce no effect other than the transfer of heat from a colder to a hotter body”.
These two formulations can be taken to arise from a general heat transfer principle, that is, the Main Principle expressed by the Second Law is: “In the absence of other driving forces, heat flows from a region of higher temperature to one of lower temperature, as a consequence of which the net entropy of the two regions increases”.
Implication 1: Heat engines (cyclic devices that convert heat to mechanical work) operate by intercepting some of the heat as it flows from a hot to a colder body. The inner workings of such devices are such that the heat transfer principle holds at all stages of heat exchange. The most effective heat engine is one in which heat intake and heat rejection occur while the working medium is in thermal equilibrium with constant temperature heat source and sink, respectively. This limiting case corresponds to net zero entropy generation in the universe by the action of the device and its heat reservoirs.
Implication 2: Cold machines or refrigerators/heat pumps (cyclic devices that use mechanical work to transfer heat against a temperature gradient) operate by incorporating heat exchange processes between the heat reservoirs so that the principle of heat transfer holds true. The heat exchange is enabled by converting added mechanical work to internal energy. The most effective refrigerator is one in which heat intake and heat rejection occur while the working medium is in thermal equilibrium with the constant temperature heat source and sink, respectively. This limiting case corresponds to net zero entropy generation in the universe by the action of the device.
Thus, it is clear that the Second Law of Thermodynamics should properly be called the law of natural heat transfer processes; it is clearly about non-equilibrium thermodynamics, and not thermostatics. Specific entropy changes capture the existing non-equilibrium potential of heat flow.
We notice that the law focuses on heat transfer but is silent concerning the direction of adiabatic processes induced by purely mechanical non-equilibrium interactions.
- 6
Ideal adiabatic or isentropic processes are induced by non-thermal forces or gradients. Mechanical non-equilibrium results from an imbalance of forces or pressures acting on a system. The direction of a natural response of such non-equilibrium system or interacting systems can be determined using Newton’s First and Second Axioms. In the case of concentration gradients between two systems in thermal equilibrium, diffusion proceeds in accordance with Fick’s theory [
23], seeking a new equilibrium concentration state.
The mixing problem and Gibbs’ mixing paradox are related to diffusion due to concentration non-equilibrium. They are usually analyzed using statistical mechanics, but, in my view, entropy has been tacitly redefined to measure the diffusion interaction. Let us consider the mixing of two ideal gases. Since for the process, the differential of the fundamental potential becomes , that is, the classical entropy term does not even feature in the equation. If the only driving force arises from concentration differences, then this force is the chemical potential, , and the associated coordinate is the number density of one of the gases, . The statistical mechanical problem thus seems to evaluate an implicitly modified entropy definition, . Viewed from this perspective, classical thermodynamics would suggest that the Gibbs mixing paradox is a non-problem because if the gases separated by the membrane are dissimilar, there is a non-zero driving force, namely, the chemical potential, which leads to non-equilibrium interaction. For similar gases, however, the chemical potential is zero and no measurable interaction/mixing occurs. This shows that modified definitions of entropy as measures of various non-equilibrium interactions of systems can be defined, but they may not be measuring thermal non-equilibrium interactions as initially intended by Clausius. Such problems would not be correctly handled by the heat transfer model of the Second Law as proposed here, but the delineation it offers is of epistemic value.
There is a tendency among some scientists and philosophers to try to bring the concept of entropy to bear on cosmological debates such as the nature of time and the dynamics of the universe. Entropy as a measure of thermal non-equilibrium interactions will undoubtedly face challenges related to system definition and the other non-thermal non-equilibrium processes mentioned. We need to know how non-thermal driving forces give rise to thermal non-equilibrium of a given system, from which deductions about entropy changes can be made. A rigorous reconstruction of processes from the present to the remote past would also inevitably bring us to the problem of infinite regress.
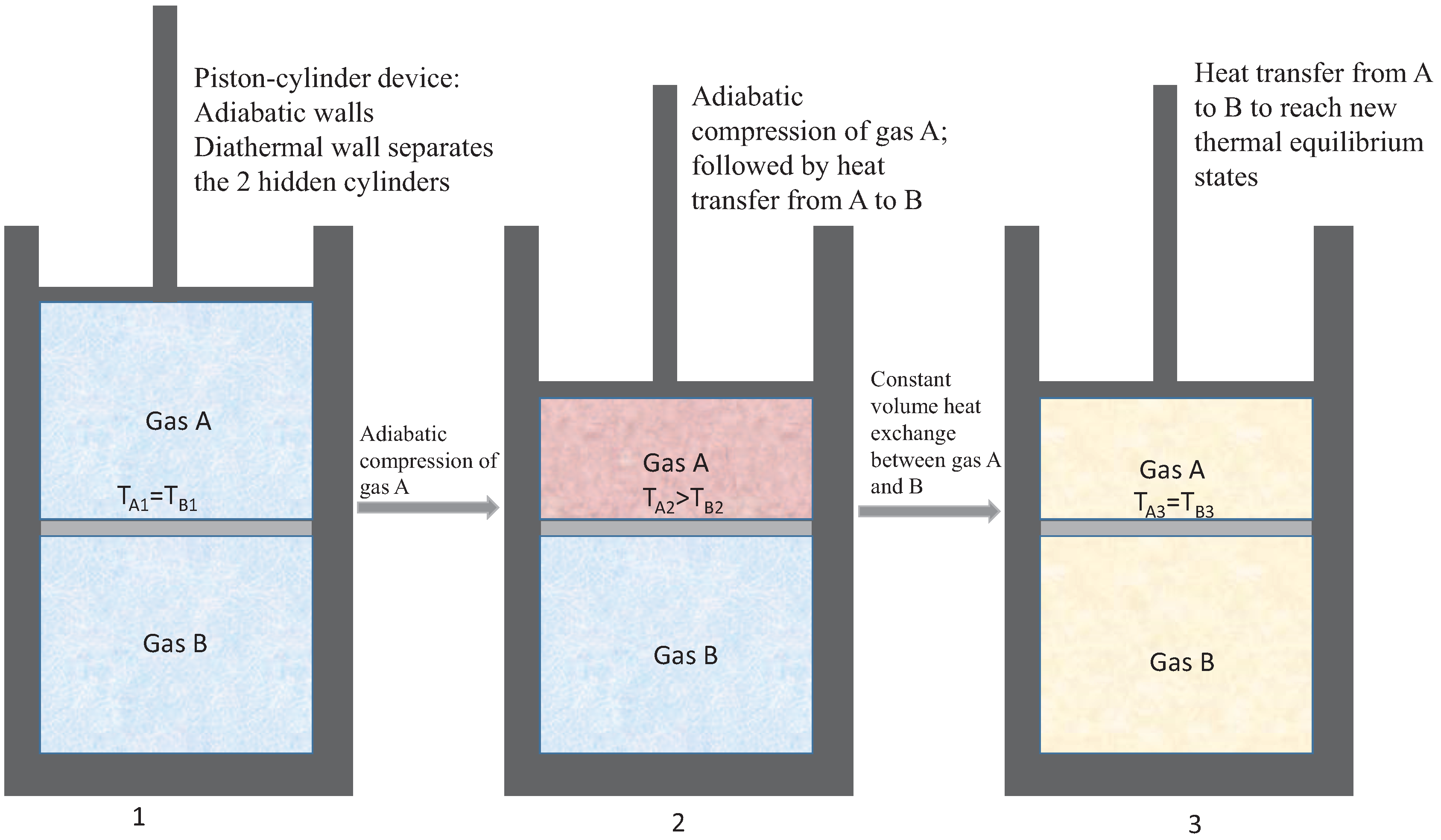
How can non-thermal non-equilibrium lead to conditions of thermal non-equilibrium, and hence induce entropy generation through heat transfer? Consider the sequence of processes shown in
Figure 4. The piston-cylinder device consists of two cylinders separated by a diathermal wall, while the walls of the enclosure are perfectly insulated. If we focus on the friction-free adiabatic compression from state 1 to 2, we see that the system can be returned to state 1 by friction-free expansion, giving off exactly the same amount of mechanical work as was applied during compression. This speaks to the fact that process 1 to 2 is not accompanied by entropy generation.
If we now focus on the compression from state 1 to 2, unbeknownst to us that the system comprises two cylinders separated by a diathermal wall, we may continue to hold the view that the compression process has not increased the entropy of the universe. We do see that after the compression, gas A is at a higher temperature and therefore not in thermal equilibrium with gas B. If, after compression, both gases are held at constant volume, and have the same mass and heat capacity, then the resulting heat transfer is analogous to the heat transfer model process previously discussed.
Figure 4 thus summarizes a scenario in which, if it were not known to us that the device comprises two cylinders separated by a diathermal wall, we would conclude that no entropy is generated, whereas the partition leads to the creation of thermal non-equilibrium and subsequent relaxation to a new thermal equilibrium, with an overall increase in the entropy of the universe.
The example discussed here is connected to Implication 2 above, namely, the operation of cold machines by means of mechanical work addition. The equivalence of heat and work means that mechanical energy can be transformed to microscopic thermal motion, which is manifested as internal energy and higher temperature or intensive energy,
ϵ. Since the Second Law is mostly concerned with thermal non-equilibrium and the direction of heat transfer, one can add as an Auxiliary Statement to the Main Principle expressed by the Second Law:
Although entropy is operationally defined through heat transfer, and would therefore be absent in interactions of two subsystems initially in thermal equilibrium, thermal non-equilibrium can be brought about in such a system of subsystems by mechanical work addition, whereby the internal energy of one subsystem is raised. This accords with the principle of equivalence of work and heat as they relate to changes in the energy state of a given system.
One more thing to consider is whether the emphasized role of heat transfer in the formulation of the Second Law and the entropy increase principle can be used to establish the impossibility of perpetual motion machines. Two classical thought experiments bring out the essence of most machines alleged to be capable of violating the Second Law. One is the Maxwell’s Demon [
24] and the other is the Feynman–Smoluchowski (Brownian) ratchet [
25]. These thought experiments shift their focus from macroscopic thermal non-equilibrium to the mechanical non-equilibrium of the microscopic particle motion. This microscopic mechanical non-equilibrium differs from that of macroscopic systems in the randomness of particle velocity magnitudes and directions (equal probability distribution).
The Maxwell’s Demon thought experiment concerns the transfer of heat from a colder to a hotter region by taking advantage of the Maxwellian particle velocity distribution. This system is supposed to succeed thanks to a special mechanical agent capable of detecting approaching high-velocity particles from the colder region and preferentially letting these pass through a gate to the hotter system, without suffering an equal or greater flux of particles from the hotter to the colder system. Were such an agent practically realizable, work would have to be expended to detect and regulate the opening and closing of the gate. The non-equilibrium process would have to be decided by classical or quantum mechanics; the randomness, however, precludes the possibility of selective directional motion without enormous energy expenditure on particle sorting. Macroscopically considered, according to the heat transfer model, it is impossible to effect transfer of mass and heat from a cold to a hotter region without expenditure of mechanical work, be it by a demon or a mechanical contraption.
The Feynman–Smoluchowski engine is supposed to be able to extract work from a single heat reservoir, contradicting the Kelvin–Planck statement and the view that heat engines merely intercept and convert to work part of the heat as it undergoes natural transfer down a temperature gradient. The device is supposed to succeed by tapping into mechanical non-equilibrium of constituent particles (velocity distribution); it restricts motion in one direction and turns a shaft in the other, when hit by more energetic particles on one side than on the other. Such a device will necessarily establish thermal equilibrium with the system of particles and the restricting ratchet and pawl system will fail to secure directional preference as pointed out by Feynman. These two thought experiments fail to tap into mechanical non-equilibrium of the microscopic particles on account of their randomness and uniform mix with slower particles. Thus, the focus on interaction of non-equilibrium systems, such as heat transfer and mechanical non-equilibrium of microscopic particles, serves as a simple and clear explanatory framework for all issues related to the Second Law.
2.1. The Third Law of Thermodynamics
The Third Law of thermodynamics states that the change in entropy between two states approaches zero as both states approach the absolute zero temperature. In statistical thermodynamics; however, entropy is evaluated at a given state. The question arises of whether, in this instance, statistical thermodynamics recovers the result of classical thermodynamics.
The Third Law, otherwise known as the Nernst theorem, grew out of investigations of systems at low temperatures, and the recognition that specific heats tend to zero as the absolute zero temperature is approached. The fact that the entropy change between two states close to absolute zero is zero does not arise directly from the current entropy definition. However, we can explore whether it is possible to arrive at this result if we adopt the temperature function in Clausius’ equation,
, to be
, where
is the microscopic or intensive energy at absolute zero for a given substance. If
is small but non-zero, this definition can render the Third Law of Thermodynamics (Nernst Theorem) unnecessary. That is, it follows that specific entropy differences tend to zero as
. Assuming that we are cooling from
to
, so that
<
and
where we have used
in the last equation. This does not eliminate the need to define an entropy value at absolute zero. The question is whether a law is needed for this, seeing that similar definitions are used for enthalpy, internal energy, Gibbs function, and other energies for pragmatic reasons. The validity of this analysis relies on the assumption that
, which may initially be considered inapplicable to the ideal gas. However, near absolute zero temperature, it is more realistic to assume that
for all substances. The assumptions on which the ideal gas model is based fail at very low temperatures, so that it is reasonable to assume that
is more general. At temperatures where ideal gas assumptions hold,
, so that
can be neglected.
It has been pointed out by one of the reviewers that the modified definition explains the entropy differential or entropy change, but it does not say what entropy is. One way to work around this is to explain entropy at a given state with reference to a hypothetical heat addition process that brings the system from absolute zero to the state in question. The entropy at the state is, therefore, a measure of the invigoration of microscopic motion associated with heat addition, bringing the system from absolute zero kelvin to the given state. This follows from , where can be set to zero by definition (neither true nor false and not empirically demonstrable).
2.2. Connection to Entropy in Statistical Mechanics
In his approach as presented in [
26] (pp. 33, 44), Gibbs seeks a canonical distribution in phase space, with the requirement that its probability be single-valued, and, for each phase space, it should neither be negative nor imaginary:
The options,
and
, are considered impossible while the Boltzmann factor is taken as the simplest conceivable case that meets all requirements:
where
η is the average probability index,
ψ is a constant corresponding to the energy for which the probability is unity,
ϵ is the energy of the phase space differential volume, and
θ is called the modulus. The modulus is considered to play the same role as temperature in classical thermodynamics. The subject is further developed to arrive at the differential of the average energy:
which is then compared to the differential form of the Second Law, after ignoring the negative signs arising from the definition of
η:
That is, it is compared to the fundamental potential,
:
where
u is the specific internal energy,
s—the specific entropy,
—the generalized force, and
, the generalized coordinate. In Gibbs’ presentation, the average probability index,
η, taken with its negative sign, is thought to correspond to entropy in classical thermodynamics. There are clearly conceptual issues with this approach that need further clarifications, but a striking point is the difference in the dimensions of T and
θ—the former being temperature and the latter, energy per unit of amount of matter. One of the challenges in statistical mechanics is the axiomatic acceptance of previous results established in classical thermodynamics. The proposed modification here identifies the Gibbs modulus
θ as analogous to
, instead of
T. The connection is natural because in classical thermodynamics,
is the statistical average energy of the microscopic constituents and in Gibbs’ approach to statistical mechanics, the modulus
θ is a normalizing energy quantity for an elemental volume of phase space.
The case is slightly different for the Boltzmann entropy. Entropy enters in his statistical mechanics through the H-theorem. According to Boltzmann, H is the sum of all values of the logarithm of the distribution function
f corresponding to designated molecules in a volume element
:
Mathematically, H is the first moment of . However, specific entropy is , corresponding to the first moment of . Increase in H denotes an increase in the weighted mean of . It is well known that the transition from Equation (45) to Equation (46) involves very subtle arguments, some of which are still debatable. The constant in Equation (46) was introduced by Planck and named after Boltzmann (k or ). Its units are chosen such that they are consistent with classical thermodynamics.
Since our modified entropy definition views specific entropy as a non-dimensional energy variable, the constant in Equation (46) becomes unity and one simply interprets the increase in the number of ways upon addition of heat to the canonical ensemble to be similar to the present notion of invigorating and increasing the average microscopic energy variable, ϵ. Implicit in Equation (45) is the assumption that ; otherwise, it should be in order to highlight the fact that the probability is assessed with reference to absolute rest. If we carry out the integration in Equation (36) in the limits from to an arbitrary ϵ, we obtain . We see that , if it is assumed or set by convention that . In essence, Boltzmann’s entropy captures the invigoration or reduction of microscopic motion through counting the number of ways energy distribution can be realized in a given macro state with implicit reference to a single possible distribution at absolute zero temperature; classical entropy on the contrary, focuses on the average intensive energy of the particles, which can increase or reduce based on heat transfer. However, since the average energy of a system at a given state can be obtained from the number of ways the microscopic energy is distributed, there is a connection between statistical and classical entropy centered on the idea that . In an isothermal heat exchange process (constant ϵ or T), the notion of invigoration or reduction of microscopic motion following heat exchange is retained by properly interpreting the associated volume and pressure change. In the case of an ideal gas, the entropy change is simply . Here, entropy as a non-dimensional energy measure indicates that isothermal heat addition amounts to invigoration of microscopic motion in order to maintain the same intensive energy (temperature) but in a new state characterized by a larger volume or a lower pressure. In statistical mechanics, this isothermal entropy change would be reflected in the multiplicity, W, as well. Much of statistical mechanics is developed with the assumption of a correct classical thermodynamics framework. It is not surprising, then, that concepts of entropy go back and forth between these two branches. We have shown that, this notwithstanding, a clear conceptual connection can be found once we focus on differential entropy as a non-dimensional measure of thermal non-equilibrium interactions. In classical thermodynamics, the differential is non-dimensional energy and in statistical thermodynamics, it can be energy or number of ways of realizing energy distribution among microstates.