Hierarchical Decomposition Thermodynamic Approach for the Study of Solar Absorption Refrigerator Performance
Abstract
:1. Introduction
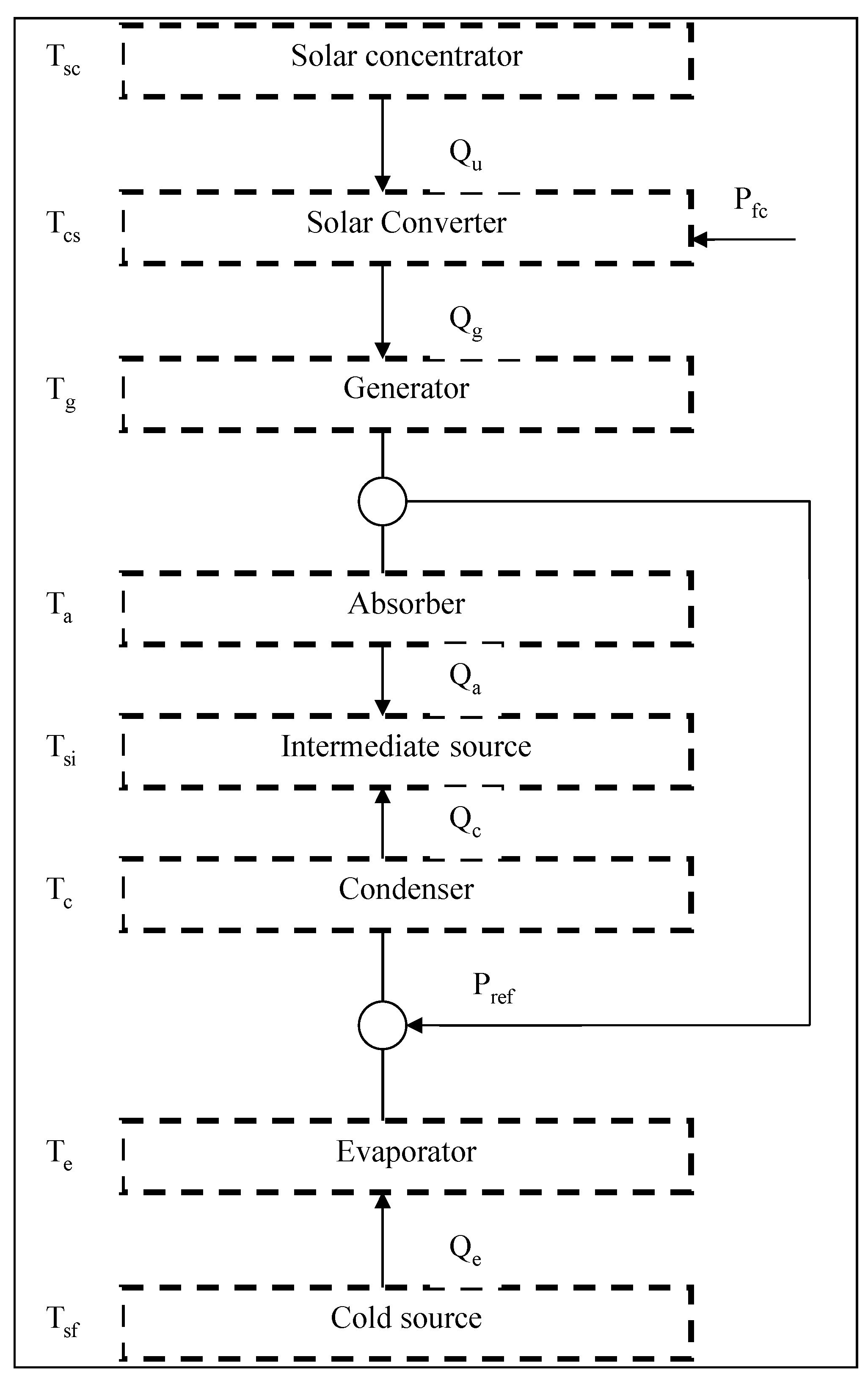
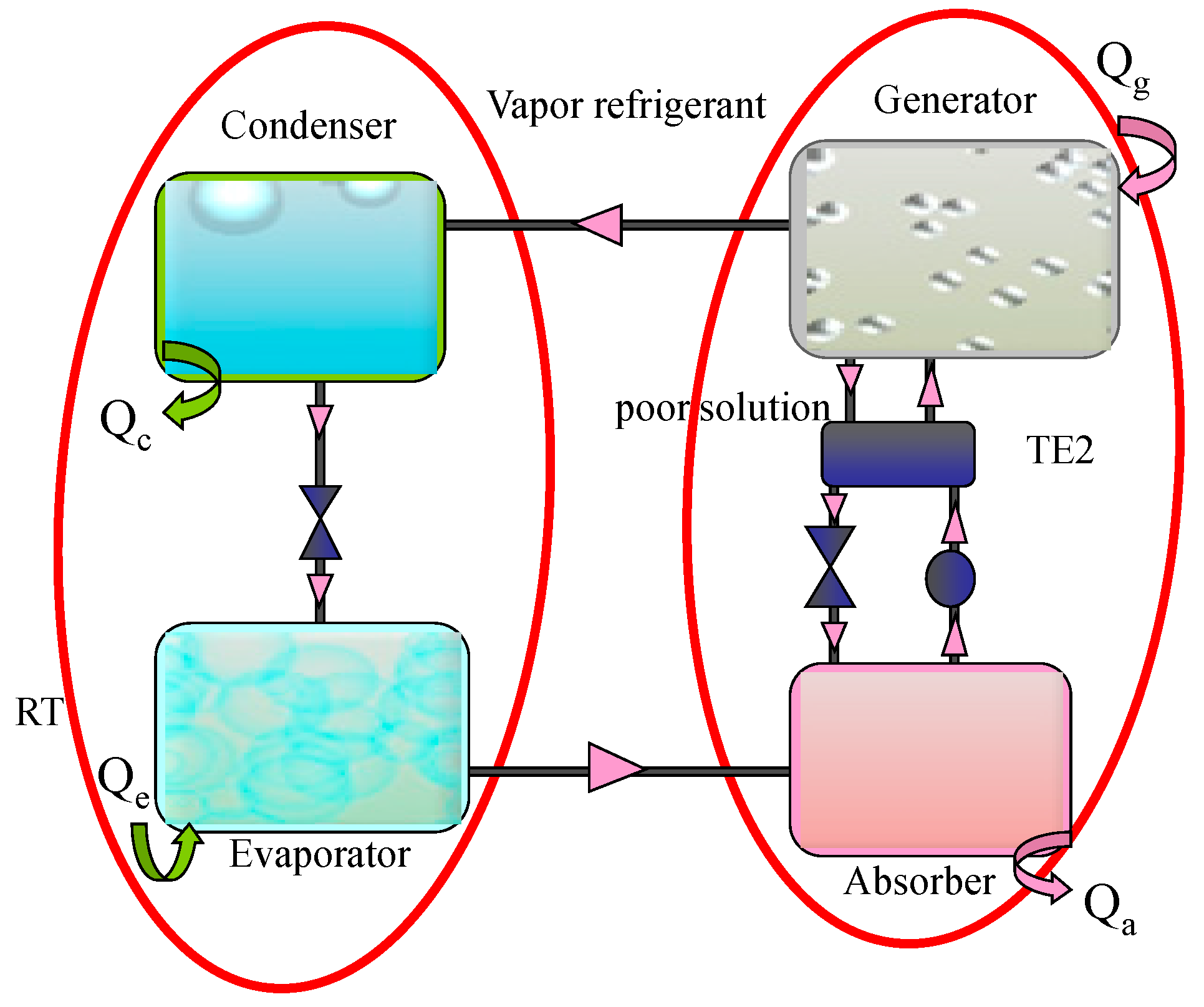
2. Thermodynamic Approach and Hierarchical Decomposition
- (1)
- The heat sources are thermostats (reservoirs with finite heat source capacitance).
- (2)
- The heat transfers with the sources are supposed to be linear. This linear heat transfer law can be written as follows:where U is the heat transfer coefficient, A is the area and ΔT is the temperature difference according to a level or a sub-level.
- (3)
- Any inlet quantity to the system is supposed positive and any outlet quantity is negative (Convention).
- (4)
- All the subsystems of the structure are endoreversible. Consequently, only the heat transfers between sub-systems, through the borders are sources of irreversibility.
3. Results and Discussion
3.1. Solar Concentrator Effect on Entropy and Coefficient of Performance
3.2. Solar Converter Temperature Effect on the Coefficient of Performance
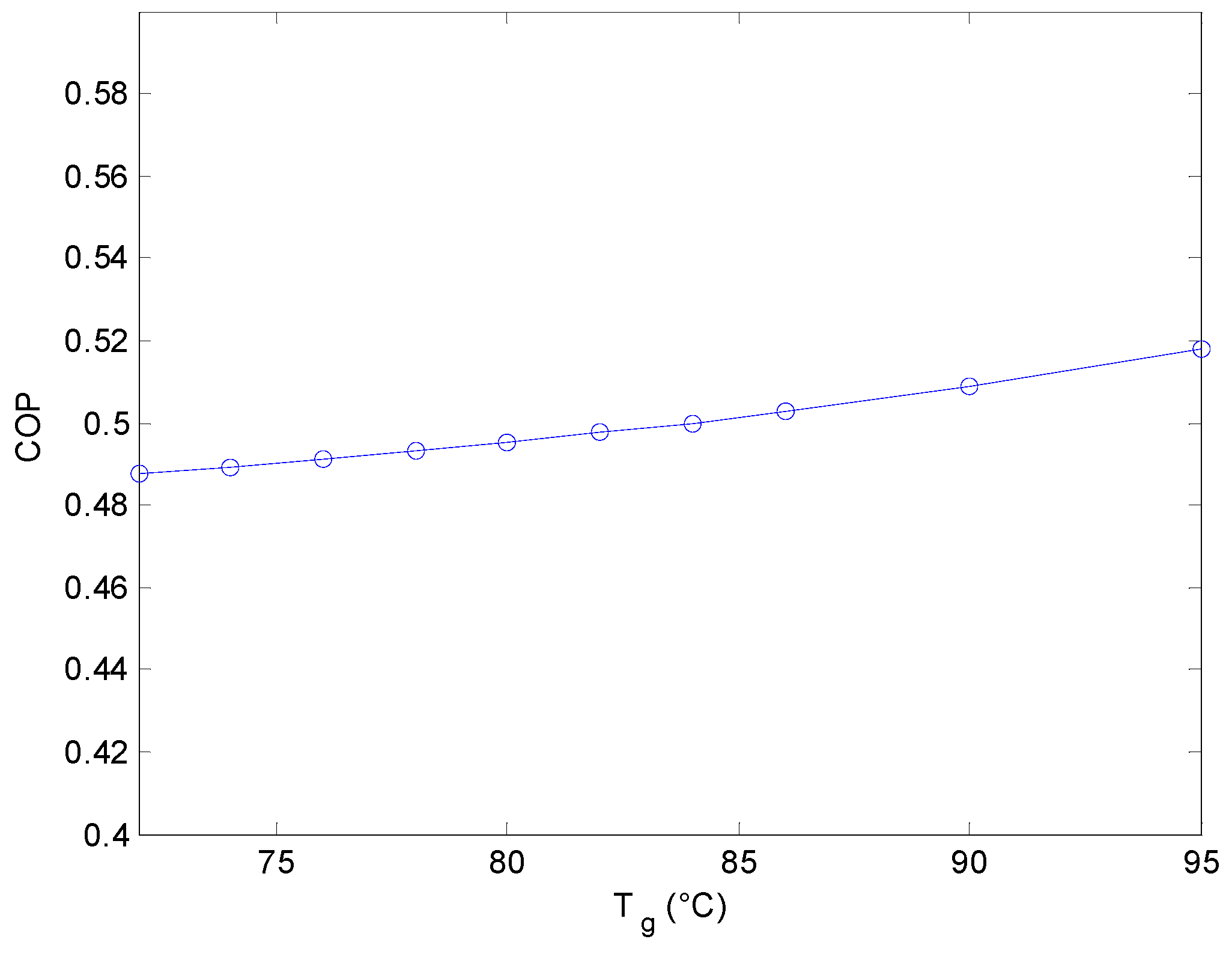
3.3. Generator Temperature Effect on the Coefficient of Performance
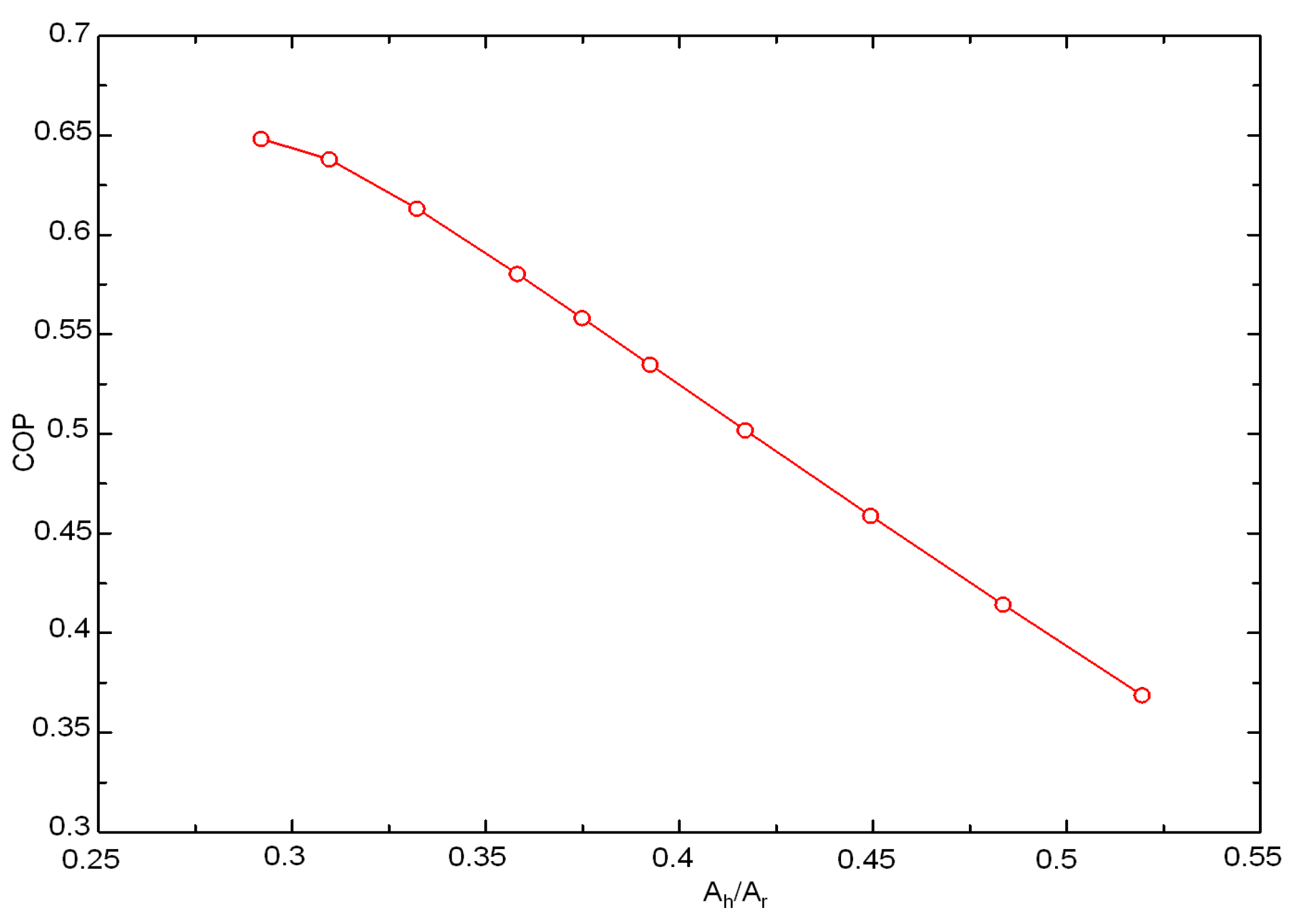
3.4. Area Distribution Effect on the Coefficient of Performance
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Goth, Y.; Feidt, M. Conditions optimales de fonctionnement des pompes à chaleur ou machines à froid associeés à un cycle de Carnot endoreversible. CRAS 1986, 303, 19–24. (In French) [Google Scholar]
- Wijeysundera, N.E. An irreversible thermodynamic model for solar powered absorption cooling systems. Sol. Energy 2000, 68, 69–75. [Google Scholar] [CrossRef]
- Kaushik, S.C.; Kumar, P.; Jain, S. Performance evaluation of irreversible cascaded refrigeration and heat pump cycles. Energy Convers. Manag. 2002, 43, 2405–2424. [Google Scholar] [CrossRef]
- Chen, L.; Zheng, T.; Sun, F.; Wu, C. Optimal cooling load and COP relationship of a four-heat-reservoir endoreversible absorption refrigeration Cycle. Entropy 2004, 6, 316–326. [Google Scholar] [CrossRef]
- Meeuse, F.M. On the Design of Chemical Processes with Improved Controllability Characteristics; DUP Science: Delft, The Netherlands, 2002. [Google Scholar]
- Berrich, E.; Fellah, A.; Ben Brahim, A.; Feidt, M. Etude Conceptuelle et Fonctionnelle d’un Cycle de Réfrigération Par Absorption Solaire Selon l’Approche d’Endoréversibilité. In Proceedings of the 17th Annual SFT Conference on Efficacité Energétique, Vannes, France, 2009; pp. 695–700. (In French)
- Fellah, A.; Ben Brahim, A.; Bourouis, M.; Coronas, A. Cooling loads analysis of an equivalent endoreversible model for a solar absorption refrigerator. Int. J. Exergy 2006, 3, 452–465. [Google Scholar] [CrossRef]
- Zheng, T.; Chen, L.; Sun, F.; Wu, C. Performance optimization of an irreversible four-heat-reservoir absorption refrigerator. Appl. Energy 2003, 76, 391–414. [Google Scholar] [CrossRef]
- Feidt, M. Thermodynamique et Optimisation Énergétique des Systèmes et Procédés; Lavoisier: Paris, France, 1987. (In French) [Google Scholar]
- Berrich, E.; Fellah, A.; Ben Brahim, A.; Feidt, M. Conceptual and functional study of a solar absorption refrigeration cycle. Exergy 2011, 8, 265–280. [Google Scholar] [CrossRef]
- Bejan, A. Method of entropy generation minimization, or modeling and optimization based on combined heat transfer and thermodynamics. Revue Générale de Thermique 1996, 35, 637–646. [Google Scholar] [CrossRef]
- Kaynakli, O.; Kilic, M. Theoretical study on the effect of operating conditions on performance of absorption refrigeration system. Energy Convers. Manag. 2007, 48, 599–607. [Google Scholar] [CrossRef]




© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Berrich Betouche, E.; Fellah, A.; Ben Brahim, A.; Aloui, F.; Feidt, M. Hierarchical Decomposition Thermodynamic Approach for the Study of Solar Absorption Refrigerator Performance. Entropy 2016, 18, 82. https://doi.org/10.3390/e18030082
Berrich Betouche E, Fellah A, Ben Brahim A, Aloui F, Feidt M. Hierarchical Decomposition Thermodynamic Approach for the Study of Solar Absorption Refrigerator Performance. Entropy. 2016; 18(3):82. https://doi.org/10.3390/e18030082
Chicago/Turabian StyleBerrich Betouche, Emma, Ali Fellah, Ammar Ben Brahim, Fethi Aloui, and Michel Feidt. 2016. "Hierarchical Decomposition Thermodynamic Approach for the Study of Solar Absorption Refrigerator Performance" Entropy 18, no. 3: 82. https://doi.org/10.3390/e18030082





