Int. J. Mol. Sci. 2022, 23(3), 1653; https://doi.org/10.3390/ijms23031653 - 31 Jan 2022
Cited by 7 | Viewed by 3575
Abstract
In the last several years, accumulating evidence indicates that noncoding RNAs, especially long-noncoding RNAs (lncRNAs) and microRNAs, play essential roles in regulating angiogenesis. However, the contribution of lncRNA-mediated competing-endogenous RNA (ceRNA) activity in the control of capillary sprouting from the pre-existing ones has
[...] Read more.
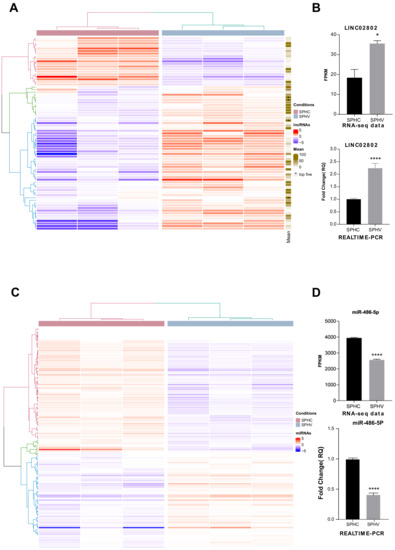
In the last several years, accumulating evidence indicates that noncoding RNAs, especially long-noncoding RNAs (lncRNAs) and microRNAs, play essential roles in regulating angiogenesis. However, the contribution of lncRNA-mediated competing-endogenous RNA (ceRNA) activity in the control of capillary sprouting from the pre-existing ones has not been described so far. Here, by exploiting the transcriptomic profile of VEGF-A-activated endothelial cells in a consolidate three-dimensional culture system, we identified a list of lncRNAs whose expression was modified during the sprouting process. By crossing the lncRNAs with a higher expression level and the highest fold change value between unstimulated and VEGF-A-stimulated endothelial cells, we identified the unknown LINC02802 as the best candidate to take part in sprouting regulation. LINC02802 was upregulated after VEGF-A stimulation and its knockdown resulted in a significant reduction in sprouting activity. Mechanistically, we demonstrated that LINC02802 acts as a ceRNA in the post-transcriptional regulation of Mastermind-like-3 (MAML3) gene expression through a competitive binding with miR-486-5p. Taken together, these results suggest that LINC02802 plays a critical role in preventing the miR-486-5p anti-angiogenic effect and that this inhibitory effect results from the reduction in MAML3 expression.
Full article
(This article belongs to the Special Issue Noncoding RNAs: Molecular Mechanisms and Physio-Pathological Implications)
►
Show Figures