Novel Gastric Cancer Stem Cell-Related Marker LINGO2 Is Associated with Cancer Cell Phenotype and Patient Outcome
Abstract
:1. Introduction
2. Results
2.1. LINGO2 is Differentially Expressed in Gastric Cancer Sphere Cells
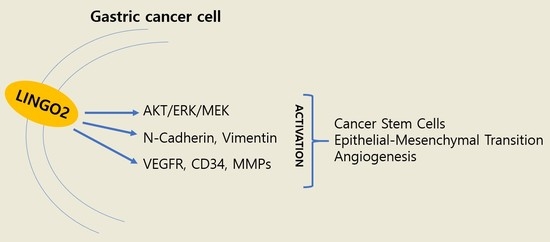
2.2. Increase in LINGO2 Expression Elevates Cancer Stem Cell Characteristics
2.3. Silencing LINGO2 Reduces Cell Proliferation and Motility
2.4. LINGO2 Alters Secretion of Matrix Metallopeptidases and Angiogenic Factors
2.5. Clinical Classification of Gastric Cancer Patients Based on LINGO2 Expression
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Sphere-Formation Assay
4.3. Microarray Analysis
4.4. Knockdown of LINGO2 by shRNA
4.5. Semiquantitative Reverse-Transcription Polymerase Chain Reaction (RT-PCR)
4.6. Western Blotting
4.7. Immunofluorescence Staining
4.8. Cell Proliferation Assays
4.9. Migration and Invasion Assays
4.10. Wound-Healing Assays
4.11. Clonogeinc Assays
4.12. Gel Zymography
4.13. Flow Cytometry
4.14. Tumorigenesis Assays
4.15. Tube Formation Assay
4.16. Patients
4.17. Immunohistochemistry
4.18. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| LINGO2 | leucine-rich repeat and immunoglobulin-like domain-containing nogo receptor-interacting protein 2 |
| CSC | cancer stem cells |
| EMT | epithelial to mesenchymal transition |
| TMA | tissue microarray analysis |
| SPEM | spasmolytic polypeptide-expressing metaplasia |
References
- Howlader, N.N.A.; Krapcho, M.; Garshell, J.; Neyman, N.; Altekruse, S.F.; Kosary, C.L.; Yu, M.; Ruhl, J.; Tatalovich, Z.; Cho, H.; et al. (Eds.) SEER Cancer Statistics Review, 1975–2010; National Cancer Institute: Bethesda, MD, USA, 2013. Available online: http://seer.cancer.gov/csr/1975_2010.
- Karimi, P.; Islami, F.; Anandasabapathy, S.; Freedman, N.D.; Kamangar, F. Gastric cancer: Descriptive epidemiology, risk factors, screening, and prevention. Cancer Epidemiol. Biomark. Prev. 2014, 23, 700–713. [Google Scholar] [CrossRef] [PubMed]
- Korea Central Cancer Registry, N.C.C. Annual Report of Cancer Statistics in Korea in 2014; Ministry of Health and Welfare: Taipei, Taiwan, 2016.
- Isobe, Y.; Nashimoto, A.; Akazawa, K.; Oda, I.; Hayashi, K.; Miyashiro, I.; Katai, H.; Tsujitani, S.; Kodera, Y.; Seto, Y.; et al. Gastric cancer treatment in Japan: 2008 annual report of the JGCA nationwide registry. Gastric Cancer 2011, 14, 301–316. [Google Scholar] [CrossRef] [PubMed]
- Clarke, M.F.; Dick, J.E.; Dirks, P.B.; Eaves, C.J.; Jamieson, C.H.; Jones, D.L.; Visvader, J.; Weissman, I.L.; Wahl, G.M. Cancer stem cells-perspectives on current status and future directions: AACR Workshop on cancer stem cells. Cancer Res. 2006, 66, 9339–9344. [Google Scholar] [CrossRef] [PubMed]
- Visvader, J.E.; Lindeman, G.J. Cancer stem cells in solid tumours: Accumulating evidence and unresolved questions. Nat. Rev. Cancer 2008, 8, 755–768. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Ping, Y.F.; Yu, X.; Qian, F.; Guo, Z.J.; Qian, C.; Cui, Y.H.; Bian, X.W. Gastric cancer stem-like cells possess higher capability of invasion and metastasis in association with a mesenchymal transition phenotype. Cancer Lett. 2011, 310, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Floor, S.; van Staveren, W.C.; Larsimont, D.; Dumont, J.E.; Maenhaut, C. Cancer cells in epithelial-to-mesenchymal transition and tumor-propagating-cancer stem cells: Distinct, overlapping or same populations. Oncogene 2011, 30, 4609–4621. [Google Scholar] [CrossRef]
- Mani, S.A.; Guo, W.; Liao, M.J.; Eaton, E.N.; Ayyanan, A.; Zhou, A.Y.; Brooks, M.; Reinhard, F.; Zhang, C.C.; Shipitsin, M.; et al. The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell 2008, 133, 704–715. [Google Scholar] [CrossRef]
- Santisteban, M.; Reiman, J.M.; Asiedu, M.K.; Behrens, M.D.; Nassar, A.; Kalli, K.R.; Haluska, P.; Ingle, J.N.; Hartmann, L.C.; Manjili, M.H.; et al. Immune-induced epithelial to mesenchymal transition in vivo generates breast cancer stem cells. Cancer Res. 2009, 69, 2887–2895. [Google Scholar] [CrossRef]
- Peng, Z.; Wang, C.X.; Fang, E.H.; Wang, G.B.; Tong, Q. Role of epithelial-mesenchymal transition in gastric cancer initiation and progression. World J. Gastroenterol. 2014, 20, 5403–5410. [Google Scholar] [CrossRef]
- Chambers, A.F.; Groom, A.C.; MacDonald, I.C. Dissemination and growth of cancer cells in metastatic sites. Nat. Rev. Cancer 2002, 2, 563–572. [Google Scholar] [CrossRef]
- Nguyen, D.X.; Bos, P.D.; Massague, J. Metastasis: From dissemination to organ-specific colonization. Nat. Rev. Cancer 2009, 9, 274–284. [Google Scholar] [CrossRef] [PubMed]
- Valastyan, S.; Weinberg, R.A. Tumor metastasis: Molecular insights and evolving paradigms. Cell 2011, 147, 275–292. [Google Scholar] [CrossRef] [PubMed]
- Takaishi, S.; Okumura, T.; Tu, S.; Wang, S.S.; Shibata, W.; Vigneshwaran, R.; Gordon, S.A.; Shimada, Y.; Wang, T.C. Identification of gastric cancer stem cells using the cell surface marker CD44. Stem Cells 2009, 27, 1006–1020. [Google Scholar] [CrossRef] [PubMed]
- Ryu, H.S.; Park, D.J.; Kim, H.H.; Kim, W.H.; Lee, H.S. Combination of epithelial-mesenchymal transition and cancer stem cell-like phenotypes has independent prognostic value in gastric cancer. Hum. Pathol. 2012, 43, 520–528. [Google Scholar] [CrossRef] [PubMed]
- Mi, S.; Lee, X.; Shao, Z.; Thill, G.; Ji, B.; Relton, J.; Levesque, M.; Allaire, N.; Perrin, S.; Sands, B.; et al. LINGO-1 is a component of the Nogo-66 receptor/p75 signaling complex. Nat. Neurosci. 2004, 7, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Vilarino-Guell, C.; Wider, C.; Ross, O.A.; Jasinska-Myga, B.; Kachergus, J.; Cobb, S.A.; Soto-Ortolaza, A.I.; Behrouz, B.; Heckman, M.G.; Diehl, N.N.; et al. LINGO1 and LINGO2 variants are associated with essential tremor and Parkinson disease. Neurogenetics 2010, 11, 401–408. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Delay, C.; Tremblay, C.; Brochu, E.; Paris-Robidas, S.; Emond, V.; Rajput, A.H.; Rajput, A.; Calon, F. Increased LINGO1 in the cerebellum of essential tremor patients. Mov. Disord. 2014, 29, 1637–1647. [Google Scholar] [CrossRef] [PubMed]
- Tran, C.P.; Cook, G.A.; Yeomans, N.D.; Thim, L.; Giraud, A.S. Trefoil peptide TFF2 (spasmolytic polypeptide) potently accelerates healing and reduces inflammation in a rat model of colitis. Gut 1999, 44, 636–642. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, C.S.; Wei, K.L.; Chou, J.L.; Lu, C.K.; Hsieh, C.C.; Lin, J.M.; Deng, Y.F.; Hsu, W.T.; Wang, H.M.; Leung, C.H.; et al. Aberrant JAK/STAT Signaling Suppresses TFF1 and TFF2 through Epigenetic Silencing of GATA6 in Gastric Cancer. Int. J. Mol. Sci. 2016, 17, 1467. [Google Scholar] [CrossRef]
- Kirikoshi, H.; Katoh, M. Expression of TFF1, TFF2 and TFF3 in gastric cancer. Int. J. Oncol. 2002, 21, 655–659. [Google Scholar] [CrossRef]
- Leung, W.K.; Yu, J.; Chan, F.K.; To, K.F.; Chan, M.W.; Ebert, M.P.; Ng, E.K.; Chung, S.C.; Malfertheiner, P.; Sung, J.J. Expression of trefoil peptides (TFF1, TFF2, and TFF3) in gastric carcinomas, intestinal metaplasia, and non-neoplastic gastric tissues. J. Pathol. 2002, 197, 582–588. [Google Scholar] [CrossRef] [PubMed]
- Dhar, D.K.; Wang, T.C.; Maruyama, R.; Udagawa, J.; Kubota, H.; Fuji, T.; Tachibana, M.; Ono, T.; Otani, H.; Nagasue, N. Expression of cytoplasmic TFF2 is a marker of tumor metastasis and negative prognostic factor in gastric cancer. Lab. Investig. 2003, 83, 1343–1352. [Google Scholar] [CrossRef]
- Carim-Todd, L.; Escarceller, M.; Estivill, X.; Sumoy, L. LRRN6A/LERN1 (leucine-rich repeat neuronal protein 1), a novel gene with enriched expression in limbic system and neocortex. Eur. J. Neurosci. 2003, 18, 3167–3182. [Google Scholar] [CrossRef] [PubMed]
- Haines, B.P.; Rigby, P.W. Expression of the Lingo/LERN gene family during mouse embryogenesis. Gene Expr. Patterns 2008, 8, 79–86. [Google Scholar] [CrossRef]
- Klorin, G.; Rozenblum, E.; Glebov, O.; Walker, R.L.; Park, Y.; Meltzer, P.S.; Kirsch, I.R.; Kaye, F.J.; Roschke, A.V. Integrated high-resolution array CGH and SKY analysis of homozygous deletions and other genomic alterations present in malignant mesothelioma cell lines. Cancer Genet. 2013, 206, 191–205. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, W.Q.; Pfeiffer, R.M.; Hyland, P.L.; Shi, J.; Gu, F.; Wang, Z.; Bhattacharjee, S.; Luo, J.; Xiong, X.; Yeager, M.; et al. Genetic polymorphisms in the 9p21 region associated with risk of multiple cancers. Carcinogenesis 2014, 35, 2698–2705. [Google Scholar] [CrossRef] [Green Version]
- Reya, T.; Morrison, S.J.; Clarke, M.F.; Weissman, I.L. Stem cells, cancer, and cancer stem cells. Nature 2001, 414, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.P.; Wen, J.; Bang, S.; Park, S.; Song, S.Y. CD44-positive cells are responsible for gemcitabine resistance in pancreatic cancer cells. Int. J. Cancer 2009, 125, 2323–2331. [Google Scholar] [CrossRef]
- Chen, T.; Yang, K.; Yu, J.; Meng, W.; Yuan, D.; Bi, F.; Liu, F.; Liu, J.; Dai, B.; Chen, X.; et al. Identification and expansion of cancer stem cells in tumor tissues and peripheral blood derived from gastric adenocarcinoma patients. Cell Res. 2012, 22, 248–258. [Google Scholar] [CrossRef]
- Han, M.E.; Jeon, T.Y.; Hwang, S.H.; Lee, Y.S.; Kim, H.J.; Shim, H.E.; Yoon, S.; Baek, S.Y.; Kim, B.S.; Kang, C.D.; et al. Cancer spheres from gastric cancer patients provide an ideal model system for cancer stem cell research. Cell. Mol. Life Sci. 2011, 68, 3589–3605. [Google Scholar] [CrossRef]
- Jiang, J.; Zhang, Y.; Chuai, S.; Wang, Z.; Zheng, D.; Xu, F.; Zhang, Y.; Li, C.; Liang, Y.; Chen, Z. Trastuzumab (herceptin) targets gastric cancer stem cells characterized by CD90 phenotype. Oncogene 2012, 31, 671–682. [Google Scholar] [CrossRef]
- Jiang, Y.; He, Y.; Li, H.; Li, H.N.; Zhang, L.; Hu, W.; Sun, Y.M.; Chen, F.L.; Jin, X.M. Expressions of putative cancer stem cell markers ABCB1, ABCG2, and CD133 are correlated with the degree of differentiation of gastric cancer. Gastric Cancer 2012, 15, 440–450. [Google Scholar] [CrossRef] [PubMed]
- Katsuno, Y.; Ehata, S.; Yashiro, M.; Yanagihara, K.; Hirakawa, K.; Miyazono, K. Coordinated expression of REG4 and aldehyde dehydrogenase 1 regulating tumourigenic capacity of diffuse-type gastric carcinoma-initiating cells is inhibited by TGF-beta. J. Pathol. 2012, 228, 391–404. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Li, C.; He, F.; Cai, Y.; Yang, H. Identification of CD44+CD24+ gastric cancer stem cells. J. Cancer Res. Clin. Oncol. 2011, 137, 1679–1686. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Du, P.; Zhao, J.; Hu, C.; Qin, Y.; Huang, G. Gastric Cancer Stem Cells: Mechanisms and Therapeutic Approaches. Yonsei Med. J. 2018, 59, 1150–1158. [Google Scholar] [CrossRef]
- Liu, J.; Ma, L.; Xu, J.; Liu, C.; Zhang, J.; Liu, J.; Chen, R.; Zhou, Y. Spheroid body-forming cells in the human gastric cancer cell line MKN-45 possess cancer stem cell properties. Int. J. Oncol. 2013, 42, 453–459. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.W.; Sung, J.S.; Park, Y.S.; Chung, S.; Kim, Y.H. Isolation of spheroid-forming single cells from gastric cancer cell lines: Enrichment of cancer stem-like cells. Biotechniques 2018, 65, 197–203. [Google Scholar] [CrossRef]
- Nam, K.T.; Lee, H.J.; Sousa, J.F.; Weis, V.G.; O’Neal, R.L.; Finke, P.E.; Romero-Gallo, J.; Shi, G.; Mills, J.C.; Peek, R.M., Jr.; et al. Mature chief cells are cryptic progenitors for metaplasia in the stomach. Gastroenterology 2010, 139, 2028–2037.e9. [Google Scholar] [CrossRef]
- Goldenring, J.R.; Nam, K.T.; Mills, J.C. The origin of pre-neoplastic metaplasia in the stomach: Chief cells emerge from the Mist. Exp Cell Res. 2011, 317, 2759–2764. [Google Scholar] [CrossRef] [Green Version]
- Nam, K.T.; Lee, H.J.; Mok, H.; Romero-Gallo, J.; Crowe, J.E., Jr.; Peek, R.M., Jr.; Goldenring, J.R. Amphiregulin-deficient mice develop spasmolytic polypeptide expressing metaplasia and intestinal metaplasia. Gastroenterology 2009, 136, 1288–1296. [Google Scholar] [CrossRef]






| Total n = 103 | LINGO2 Weak n = 75 | LINGO2 Strong n = 28 | p | |
|---|---|---|---|---|
| Mean age, years (SD) | 57.9 (10.7) | 58.3 (10.5) | 56.9 (11.3) | 0.549 |
| Sex (%) | ||||
| Male | 67 (65.0) | 50 (66.7) | 17 (60.7) | 0.573 |
| Female | 36 (35.0) | 25 (33.3) | 11 (39.3) | |
| CEA, ng/mL (SD) | 8.0 (26.3) | 8.0 (26.3) | 8.1 (27.1) | 0.992 |
| Median OS, months (range) | 96.9 (2.0–220.8) | 121.2 (2.0–220.8) | 25.5 (5.3–219.3) | 0.012 |
| pT stage (%) | 0.011 | |||
| T1 | 35 (34.0) | 29 (38.7) | 6 (21.4) | |
| T2 | 23 (22.3) | 20 (26.7) | 3 (10.7) | |
| T3 | 43 (41.7) | 24 (32.0) | 19 (67.9) | |
| T4 | 2 (1.9) | 2 (2.7) | 0 (0.0) | |
| T1/2 | 58 (56.3) | 49 (65.3) | 9 (32.1) | 0.003 |
| T3/4 | 45 (43.7) | 26 (34.7) | 19 (67.9) | |
| pN stage (%) | 0.005 | |||
| N0 | 45 (43.7) | 39 (52.0) | 6 (21.4) | |
| N1 | 27 (26.2) | 19 (25.3) | 8 (28.6) | |
| N2 | 24 (23.3) | 15 (20.0) | 9 (32.1) | |
| N3 | 7 (6.8) | 2 (2.7) | 5 (17.9) | |
| N0/1 | 72 (69.9) | 58 (77.3) | 14 (50.0) | 0.007 |
| N2/3 | 31 (30.1) | 17 (22.7) | 14 (50.0) | |
| Stage (%) | 0.015 | |||
| IA | 26 (25.2) | 23 (30.7) | 3 (10.7) | |
| IB | 28 (27.2) | 22 (29.3) | 6 (21.4) | |
| IIB | 22 (21.4) | 17 (22.7) | 5 (17.9) | |
| IIIA | 18 (17.5) | 9 (12.0) | 9 (32.1) | |
| IIIB | 9 (8.7) | 4 (5.3) | 5 (17.9) | |
| I/II | 76 (73.8) | 62 (82.7) | 14 (50.0) | 0.001 |
| III | 27 (26.2) | 13 (17.3) | 14 (50.0) |
| Univariate | Multivariate | |||
|---|---|---|---|---|
| Hazard Ratio (95% CI) | p | Hazard Ratio (95% CI) | p | |
| LINGO2strong (vs. LINGO2 weak) | 1.939 (1.146–3.279) | 0.014 | 1.245 (0.674–2.301) | 0.484 |
| Older Age | 1.053 (1.025–1.082) | <0.001 | 1.053 (1.023–1.083) | <0.001 |
| Male sex (vs. female) | 1.160 (0.689–1.956) | 0.576 | 1.433 (0.805–2.551) | 0.222 |
| Higher initial CEA | 1.007 (0.998–1.016) | 0.141 | 1.001 (0.991–1.011) | 0.828 |
| Stage (III vs. I or II) | 3.768 (2.235–6.354) | <0.001 | 3.693 (2.059–6.624) | <0.001 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jo, J.H.; Park, S.B.; Park, S.; Lee, H.S.; Kim, C.; Jung, D.E.; Song, S.Y. Novel Gastric Cancer Stem Cell-Related Marker LINGO2 Is Associated with Cancer Cell Phenotype and Patient Outcome. Int. J. Mol. Sci. 2019, 20, 555. https://doi.org/10.3390/ijms20030555
Jo JH, Park SB, Park S, Lee HS, Kim C, Jung DE, Song SY. Novel Gastric Cancer Stem Cell-Related Marker LINGO2 Is Associated with Cancer Cell Phenotype and Patient Outcome. International Journal of Molecular Sciences. 2019; 20(3):555. https://doi.org/10.3390/ijms20030555
Chicago/Turabian StyleJo, Jung Hyun, Soo Been Park, Semi Park, Hee Seung Lee, Chanyang Kim, Dawoon E. Jung, and Si Young Song. 2019. "Novel Gastric Cancer Stem Cell-Related Marker LINGO2 Is Associated with Cancer Cell Phenotype and Patient Outcome" International Journal of Molecular Sciences 20, no. 3: 555. https://doi.org/10.3390/ijms20030555