1. Introduction
With the acceleration of the size of the aging population, the world is now experiencing the “aging era” characterized by an increased incidence of neurodegenerative diseases like Alzheimer’s disease (AD) and Parkinson’s disease (PD) [
1]. Neurodegenerative diseases represent a heterogeneous group of disorders whose common characteristic is the progressive and selective loss of neurons. Although many studies have been carried out to understand the pathophysiology of neurodegenerative diseases over the years, more efforts are still needed to uncover the processes that trigger these disorders. Despite different aetiologies, neurodegenerative disorders share common features like abnormal protein aggregation, perturbed Ca
2+ homeostasis, excitotoxicity, impairment of mitochondrial functions, apoptosis, inflammation, and oxidative stress [
2,
3]. Oxidative stress is an imbalance between accumulation and removal of reactive oxygen species (ROS). The brain is especially susceptible to oxidative stress due to its high oxygen consumption, high content of oxidizable polyunsaturated fatty acids, and low level of endogenous antioxidants and antioxidant enzymes [
4,
5,
6]. It has been shown that oxidative stress is involved in the onset and progression of neurodegenerative diseases like PD [
2,
7,
8,
9,
10] and AD [
11,
12,
13].
Despite recent advances in the research of biomarkers, early diagnosis, and pharmacotherapy, there are no treatments that can halt the progression or reverse the brain changes of any age-associated neurodegenerative diseases. This is likely due to the multifactorial nature of these pathologies that arise from a confluence of multiple, toxic insults.
Diet is considered one of the most important factors in lifestyle. Diet strongly influences the occurrence of cardiovascular and neurodegenerative diseases and, hence, a healthy lifestyle can be related to healthy aging [
14]. Numerous epidemiological studies indicate that long-term intake of a Mediterranean diet, characterized by a high consumption of extra virgin olive oil, correlates with better cognition in aged populations [
15,
16,
17]. Olive oil phenolic compounds have been demonstrated to possess different biological activities like antioxidant [
18], antithrombotic [
19], and anti-inflammatory [
20] activities. Oleocanthal, a dialdehydic form of (−)-deacetoxyligstroside glycoside, is one of the phenolic components of extra virgin olive oil [
21]. Even though oleocanthal makes up only 10% of the olive’s phenols, it is getting more and more scientific attention due to its interesting biological activities [
22,
23]. Oleocanthal is responsible for the bitter and pungent taste of extra virgin olive oil, and has anti-inflammatory properties similar to the nonsteroidal anti-inflammatory drug ibuprofen [
24]. In vitro, it has been shown that oleocanthal is effective on the key mediators of AD pathogenesis, amyloid-β and hyper-phosphorylated tau proteins [
25,
26,
27,
28], which contribute significantly to neurodegeneration and memory loss [
29]. Moreover, oleocanthal reduces astrocyte activation and interleukin-1β levels in vivo [
21]. Studies on the antioxidant activities of oleocanthal are limited. Only one study carried out in isolated human monocytes showed that oleocanthal inhibits nicotinamide adenine dinucleotide phosphate oxidase (NOX) activity and reduces the intracellular level of superoxide anion [
30].
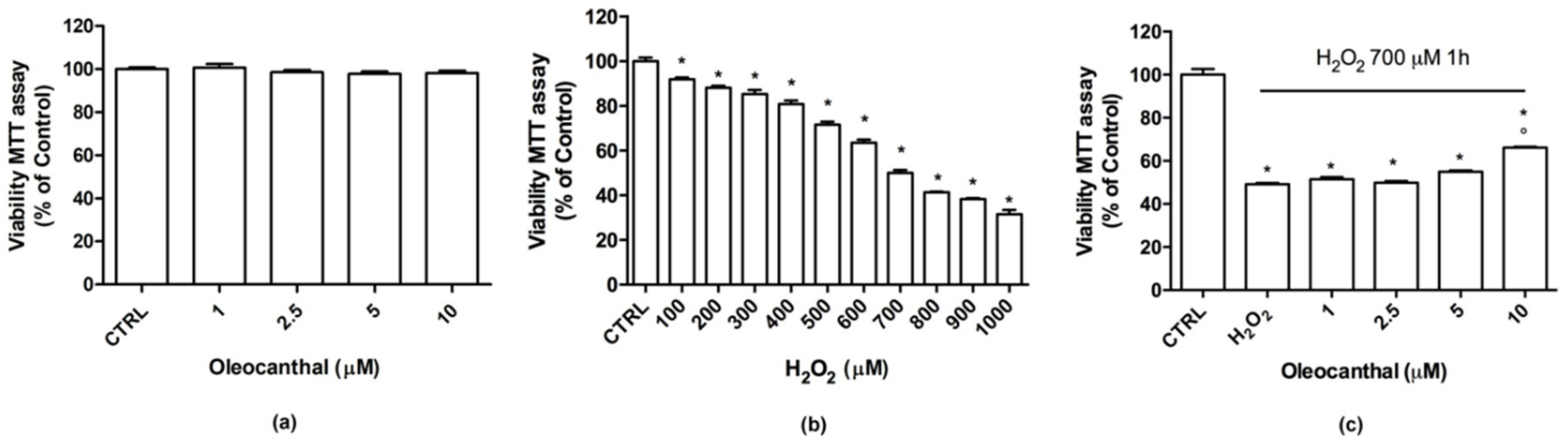
The aim of this research was to characterize the neuroprotective effects of oleocanthal against H2O2-induced oxidative stress in the neuron-like SH-SY5Y cell line. Moreover, protein expression profiling, combined with pathways analyses, was used to investigate the molecular events related to the protective effects, and to gain insight into the underlying mechanisms of neuroprotection and oxidative damage.
3. Discussion
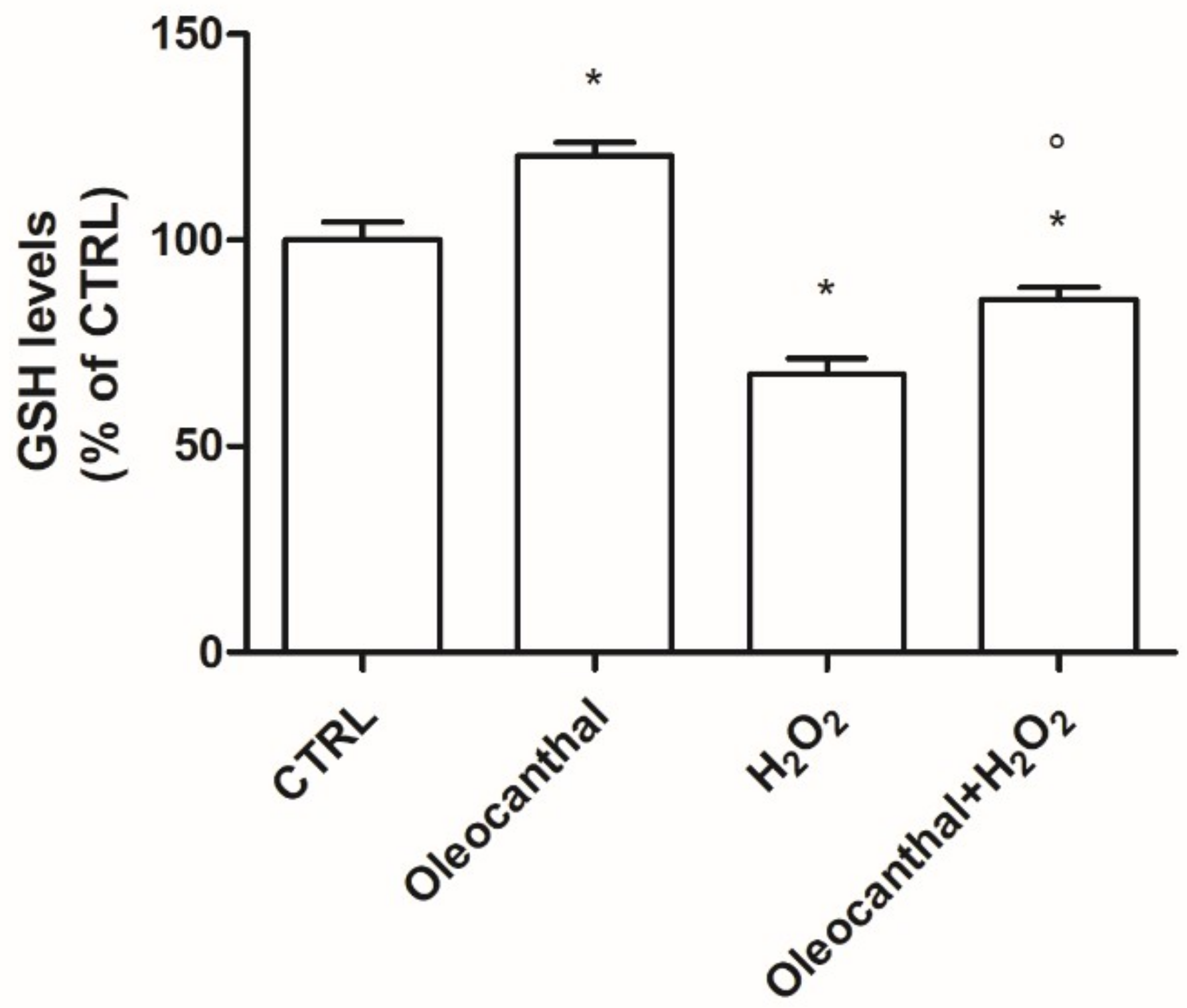
In this study, we demonstrated that oleocanthal significantly protects differentiated human neuroblastoma SH-SY5Y cell line from oxidative stress induced by H
2O
2. In particular, oleocanthal reduces intracellular ROS levels and increases endogenous GSH levels. To further elucidate the mechanisms behind oleocanthal’s neuroprotective activity we used proteomic analysis that revealed an involvement of the ubiquitin–proteasome system, heat shock proteins, pyruvate kinases, and peroxiredoxin 1. Proteomic data were validated by real-time PCR analysis in order to have a complementary and wider vision of oleocanthal effect on both gene and protein expression. Since the levels of transcription of the selected genes were similar to their translation, we considered the comparable change in mRNA levels as a validation of proteomic data. A fundamental issue in the study of nutraceutical compounds is their bioavailability. The pharmacokinetics and bioavailability of oleocanthal have been poorly investigated. Under simulated gastric acid conditions, it has been shown that oleocanthal is stable in acidic conditions at 37 °C and almost half of the oleocanthal diffused from the oil phase into the aqueous solution [
34]. Only one study has investigated the bioavailability of oleocanthal by identifying oleocanthal and its metabolites in urines, demonstrating oleocanthal metabolism in the human body [
35]. Oleocanthal is mainly metabolized through phase I metabolism, like hydroxylation, hydrogenation, and hydration. Some of the hydrogenated oleocanthal metabolites are further metabolized through phase II metabolism [
35]. Even if no studies directly examined the blood–brain barrier (BBB) permeability of oleocanthal, different studies indirectly demonstrated its ability to reach the brain. In particular, Qosa et al. reported a decreased amyloid load in the hippocampus of TgSwDI mice after oleocanthal treatment [
21]; meanwhile, in C57BL/6 mice, Abuznait et al. demonstrated that oleocanthal induces P-gp and LRP1, which are responsible for Aβ clearance across the BBB, and increases Aβ degradation by up-regulating Aβ-degrading enzymes [
25], strongly suggesting its ability to reach the brain.
The ubiquitin–proteasome system is the main proteolytic complex responsible for the removal of misfolded and damaged intracellular proteins, often produced upon oxidative stress [
36]. The proteasome is a non-lysosomal threonine protease [
37] that degrades both normal and damaged proteins into short peptides by the internal protease activities [
38,
39]. It has been observed that the impairment of the ubiquitin–proteasome system is implicated in the pathogenesis of many neurodegenerative diseases [
40] like Alzheimer’s disease, Parkinson’s disease, Lewy body dementia, Pick disease, frontotemporal dementia, and Huntington’s disease (HD) [
41]. On these bases, the induction of the proteasome system has been emerging as a therapeutic target to counteract these diseases. To our best knowledge, no studies have explored the effect of oleocanthal on the ubiquitin–proteasome system upon oxidative stress. Interestingly, our proteomic data, obtained in the presence of oxidative stress, indicate the up-regulation of two proteasome subunits, namely, the 26S proteasome non-ATPase regulatory subunit and the proteasome subunit β type-4, and the up-regulation of the ubiquitin carboxyl-terminal hydrolases 14 (Usp14). As Usp14 negatively regulates proteasome activity by ubiquitin chain disassembly as well as by a noncatalytic mechanism [
42], oleocanthal, in the presence of oxidative stress, has two opposite effects: on one side, it up-regulates the proteasome and, on the other side, it up-regulates Usp14, abrogating the suggested enhancement of the proteasome activity. For this reason, we examined the effect of oleocanthal in the absence of oxidative stress by real-time PCR. Intriguingly, in normal conditions, oleocanthal is able to induce proteasome activity as it up-regulates proteasome subunits Psmd1 and Psmb4 and does not affect Usp14 expression, suggesting that oleocanthal could have a role in the degradation of misfolded proteins rather than in the degradation of oxidized ones. Our observations are in agreement with different studies that demonstrated that oleocanthal counteracts neurodegenerative diseases by enhancing the clearance of misfolded proteins like amyloid-β, phosphorylated tau, and α-synuclein [
27,
43,
44].
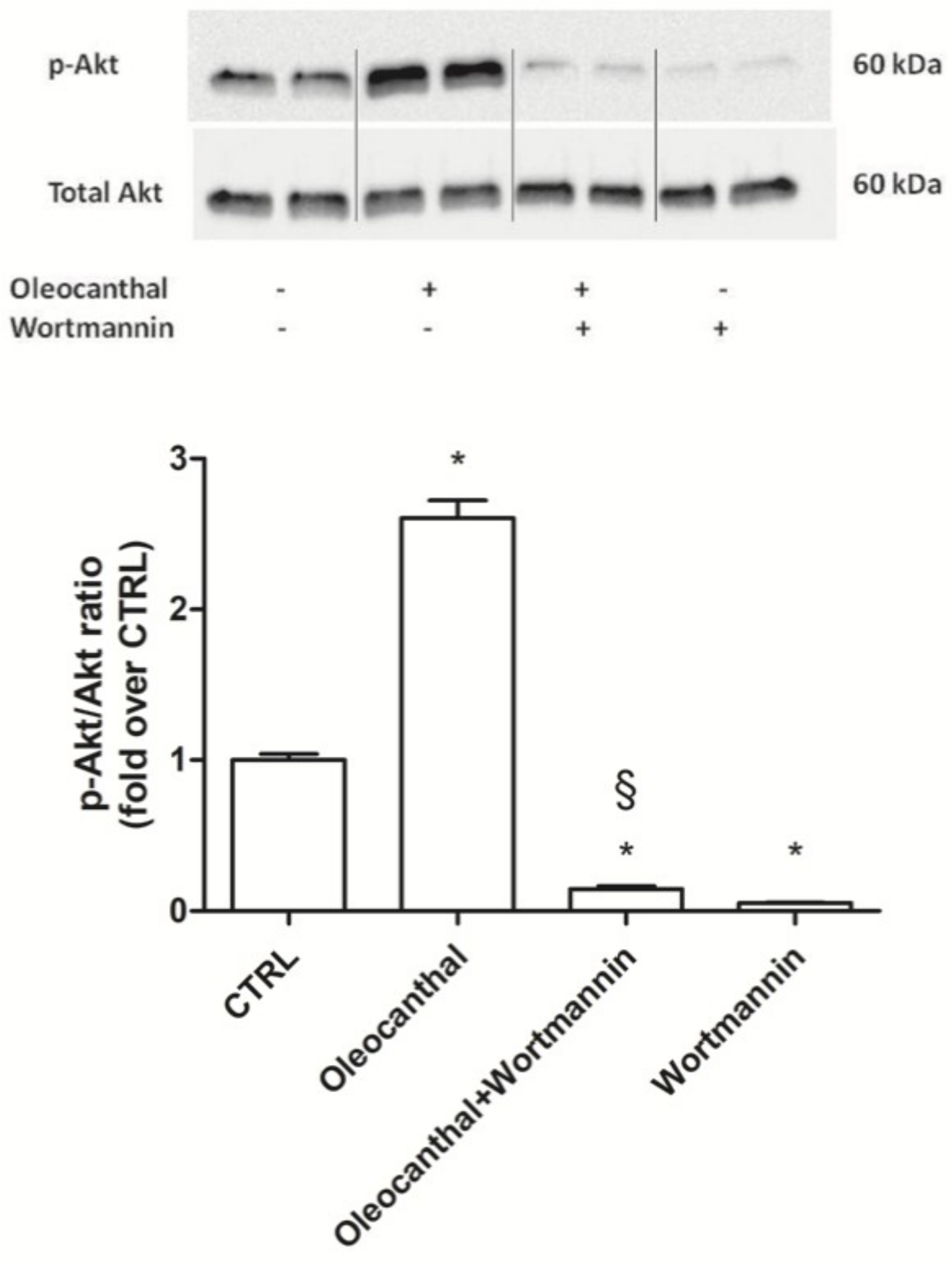
Hsp90 is a chaperone protein important for maintaining stability, maturation, and signaling of Hsp90 client proteins [
45]. It has been demonstrated that Hsp90 maintains cell proliferation and cell survival by different mechanisms. Hsp90 may inhibit apoptosis by binding to apoptotic peptidase-activating factor 1, resulting in the prevention of apoptosome formation, caspase activation, and apoptotic cell death [
46]. In addition, Taiyab et al. showed that inhibition of Hsp90 results in ER stress-induced apoptosis in rat histiocytoma [
47]. A strict correlation between Hsp90 and Akt has been demonstrated [
33], as the inhibition of the binding of Akt to Hsp90 inactivates Akt and makes cells more susceptible to apoptosis-inducing stimuli [
48]. Interestingly, Xie et al. observed that the inhibition of Akt activity led to a reduction of Hsp90 level, suggesting that Akt activation can elevate Hsp90 protein levels [
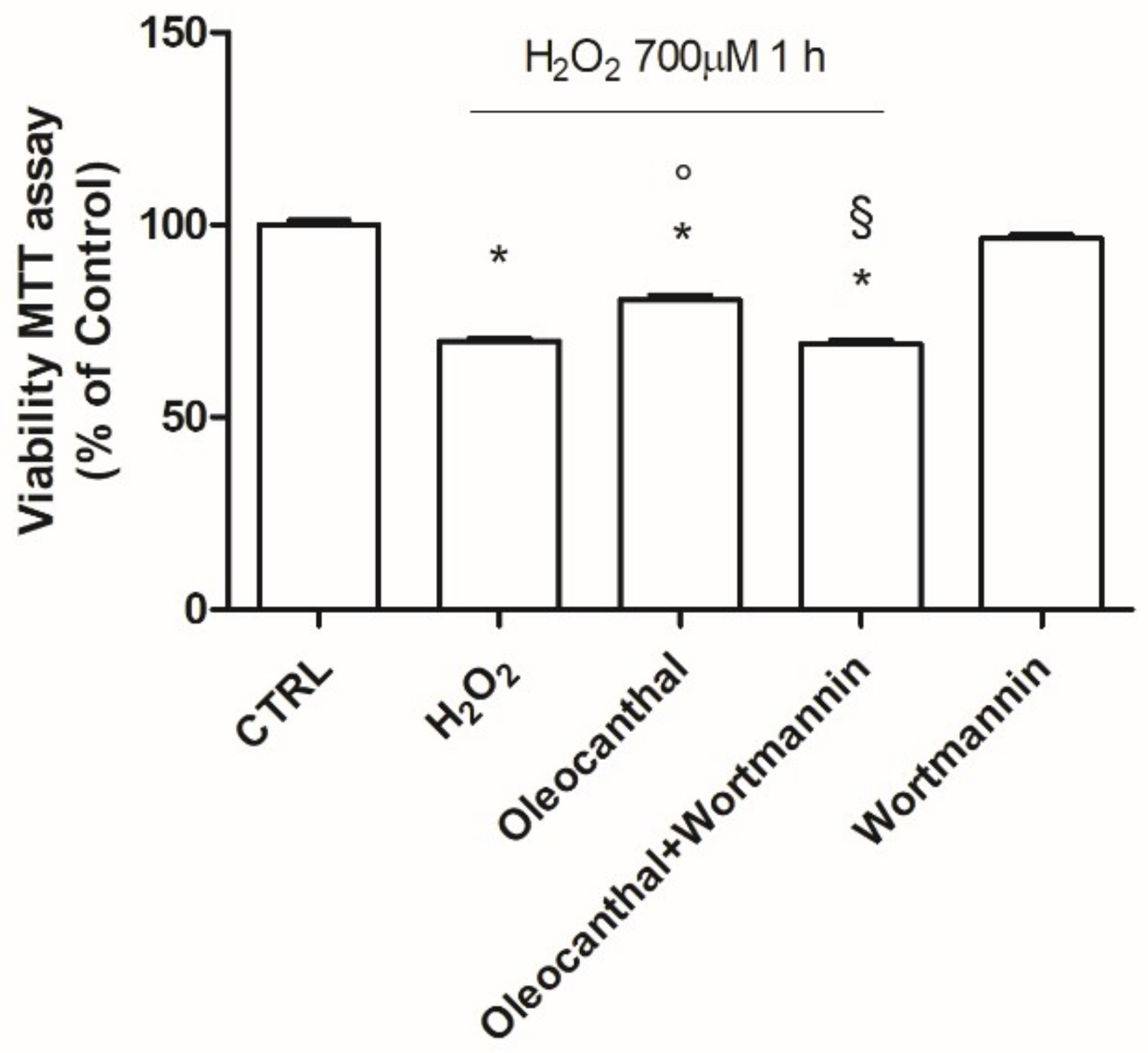
49]. Our data demonstrated that oleocanthal, upon oxidative stress, is able to up-regulate Hsp90. As Hsp90 acts with Akt, we also evaluated the effect of oleocanthal treatment on Akt activation (phosphorylation). As expected, we measured a strong and significative activation of Akt. Moreover, Akt inhibition by wortmannin led to the inhibition of the up-regulation of Hsp90 induced by oleocanthal, in agreement with the previous observation by Xie et al. Of note, the fact that wortmannin totally abrogated oleocanthal protection against oxidative stress suggests a fundamental role of Akt and/or Hsp90 in the protective mechanisms elicited by oleocanthal against oxidative stress in SH-SY5Y neuronal cells. As wortmannin can affect other signaling cascades apart from Akt, we could not exclude that oleocanthal regulates neuroprotective effects through other signaling pathways. Further studies are necessary to fully elucidate this aspect.
Our proteomic and expression data indicated a significative up-regulation of Pkm1 and Pkm2 by oleocanthal in the presence of oxidative stress. Pyruvate kinase, catalyzing the final rate-limiting step in glycolysis, is a crucial enzyme for glucose metabolism and energy production in the brain. Elevated levels of pyruvate kinase have been suggested to be protective in neurodegeneration as decreased aerobic glycolysis in the brain leads to a loss of cell survival mechanisms that counter pathogenic processes underlying neurodegeneration [
50]. Moreover, Luo et al. demonstrated that Pkm hinders the Aβ fibrillation and reduces the toxicity of Aβ aggregates in SH-SY5Y cells, suggesting that Pkm interferes with the stability of the Aβ oligomer by hydrophobic and hydrophilic interactions of its surface [
51].
Oleocanthal also induced a strong up-regulation of the antioxidant enzyme Prdx1 in the presence of H
2O
2, as indicated by our proteomic and expression results. Peroxiredoxins play an important role in cell proliferation, redox signaling, differentiation, and gene expression [
52,
53]. In particular, Prdx1 eliminates hydrogen peroxide produced during cellular metabolism [
54] and participates in cell survival by enhancing the expression of the pro-survival factor protein kinase B (PKB) [
55]. The role of Prdx1 in AD has been recently highlighted by Majd et al. who observed reduced levels of Prdx1 and 2 in postmortem brains of AD [
56]; meanwhile, Schreibelt et al. analyzed the functional role of Prdx1 using a brain endothelial cell line overexpressing Prdx1 and showed that enhanced Prdx1 expression in brain endothelial cells increased BBB integrity and reduced monocyte adhesion to and migration across a brain endothelial cell layer [
57].
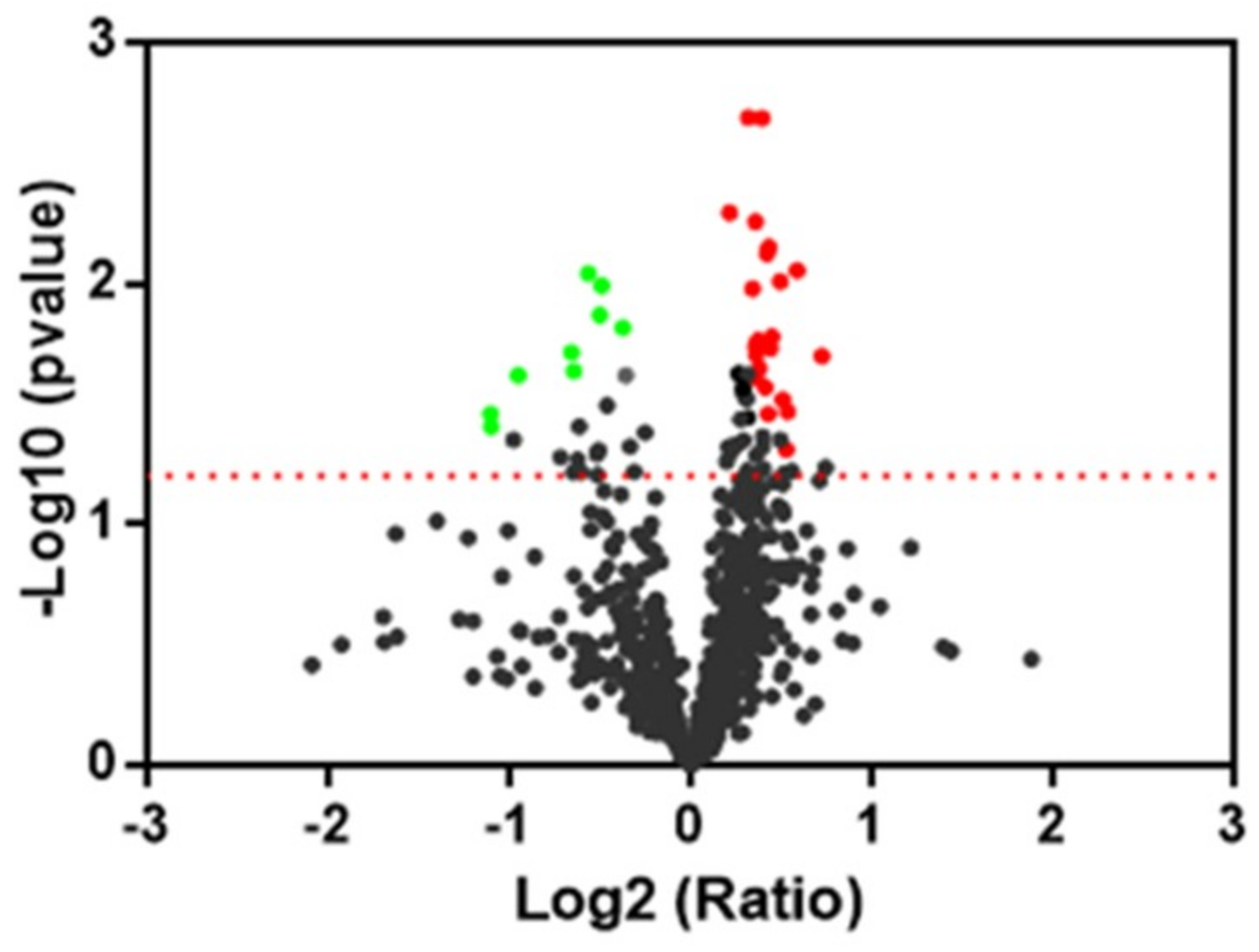
Focusing on proteomic results, the quantitative two-dimensional analysis showed that the insult induced by peroxide induced a significantly different expression of four Prdx in the human neuroblastoma SH-SY5Y cell line. Interestingly, two near spots, different for the pI value and for level of expression, were detected for PRDX1, 2, and 6 in our samples, suggesting the presence of post-translational modification. Post-translational modification and intracellular protein–protein interactions have the potential to influence Prdx catalytic activity and susceptibility to hyperoxidation [
58]. We found for all three Prdx an increase of expression of more acidic spots and a significant reduction of more basic spots in the H
2O
2 sample with respect to the control. The protective effect of oleocanthal takes place by way of an increase of more acidic spots of Prx1. The nature of this modification needs to be investigated and underlines a mechanism of control of protein activity.
Finally, all proteins found differentially expressed from a comparison of oleocanthal + H
2O
2 vs. H
2O
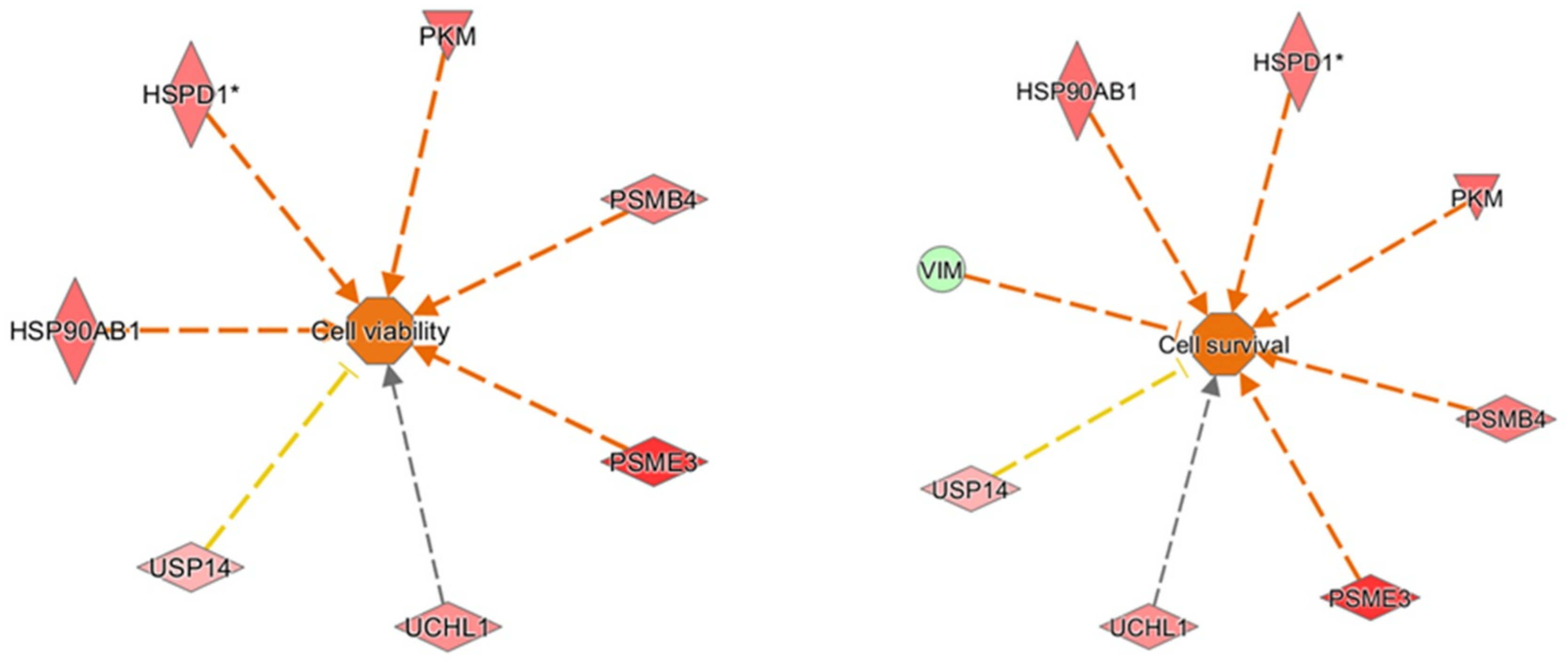
2 were analyzed by IPA to explore in depth the network, functions, and molecules (e.g., upstream regulators) that could play a role in the protection induced by oleocanthal. The molecular functions defined by our differentially expressed proteins were well arranged in protein synthesis, cell death and survival, protein degradation, and cellular function and maintenance. On the other hand, the up-regulation of heat shock protein and proteasome members in addition to ubiquitin enzymes and pyruvate kinase concurred to predict cellular viability and vitality (
Figure 12). As concerns upstream regulators, intriguing results arose from the IPA analysis. Along the list of noteworthy potential activated or inhibited upstreams were NFE2L2, HSF1, and ATF7.
NFE2L2, which encodes for the transcription factor Nrf2, is important for the coordinated up-regulation of genes in response to oxidative stress. Very recent studies showed neuroprotective actions [
59,
60] through the activation of Akt/Nrf2/antioxidant enzymes in neuronal cells. On the other hand, the significant up-regulation of the active form of Akt that we found in our results agrees with an activation state of Nrf2.
HSF1 works as a stress-inducible and DNA-binding transcription factor that plays a central role in the transcriptional activation of the heat shock response, leading to the expression of a large number of chaperone heat shock proteins. A central role of HSF1 in synaptic fidelity and memory consolidation has been recently suggested by Hooper and coworkers [
61] who showed that the activation of HSF1 alone augmented vesicle transport and synaptic scaffolding proteins. Meanwhile, HSF1 agonists can improve cognitive function in dementia models and activation of neuroprotective signaling pathways. Finally, HSF1 protected neurons from death caused by accumulation of misfolded proteins. This neuroprotection result was abrogated by inhibition of classical deacetylases such as SIRT1 [
62].
As concerns ATG7 (autophagy-related 7), a predicted inhibition was advanced by IPA analysis. ATG7 is an E1-like activating enzyme involved in the 2-ubiquitin-like systems required for cytoplasm-to-vacuole transport and autophagy. Recent findings reveal that selective neuronal deletion of ATG7 is strongly protective against neuronal death and overall brain hypoxic–ischemic injury [
63].
Overall, our results, combined with IPA analysis, suggest that the major part of protein targets of oleocanthal in response to peroxide insult belongs to the proteostasis network including proteasome, ubiquitin, and chaperone proteins which move to restore equilibrium between protein synthesis, folding, trafficking, secretion, and degradation in different cell compartments. Moreover, our findings add new evidence on the effects of oleocanthal on heat shock protein 90, Pkm1 and 2, and Prdx1.