On the Contrasting Effect Exerted by a Thin Layer of CdS against the Passivation of Silver Electrodes Coated with Thiols
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. ECALE Cycles for CdS Deposition
2.3. Self-Assembly of Alkanethiols on Ag(III) and on CdS-Covered Ag(III)
2.4. EQCM Measurement
2.5. Auger Electron Spectroscopy (AES) Analysis
3. Results
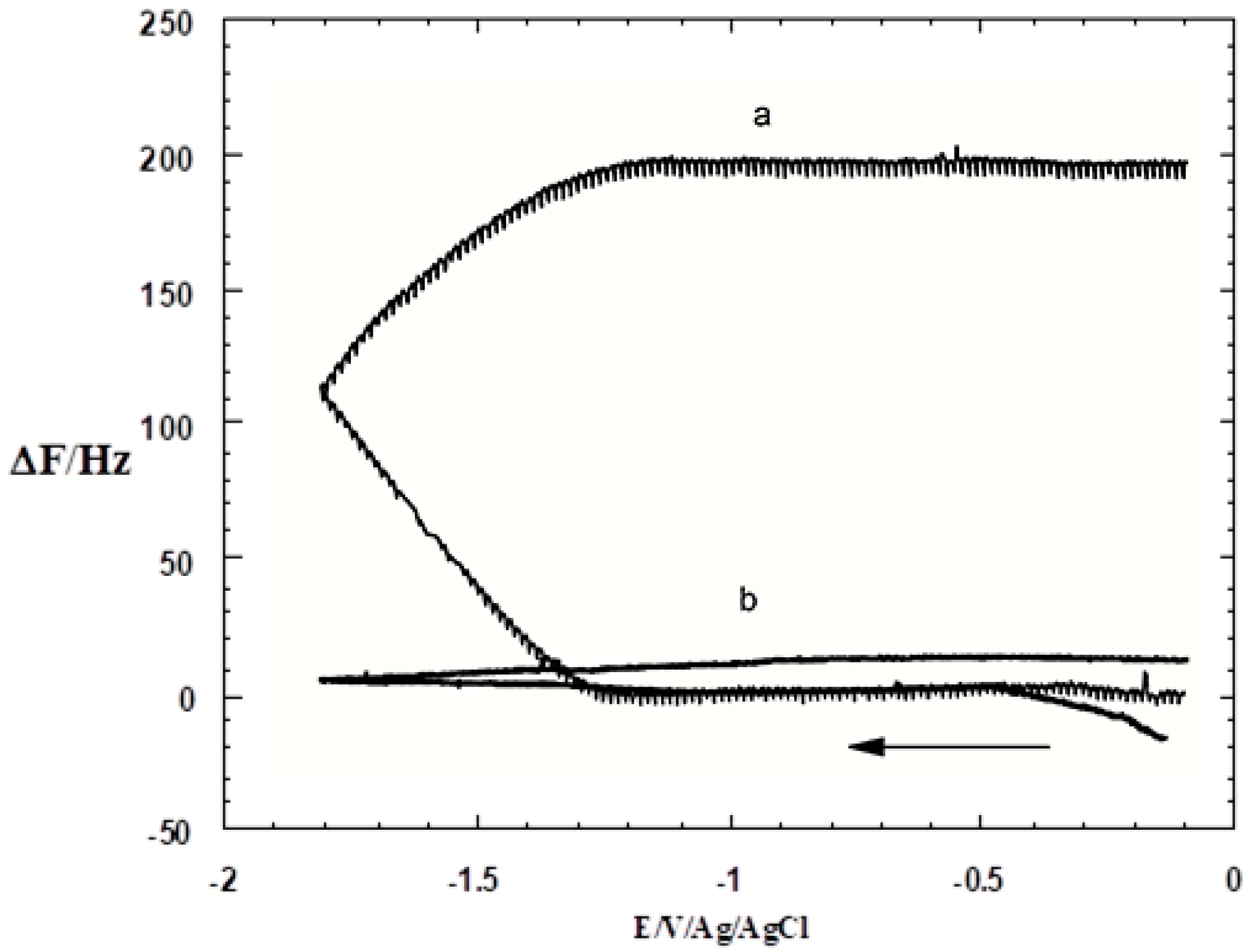
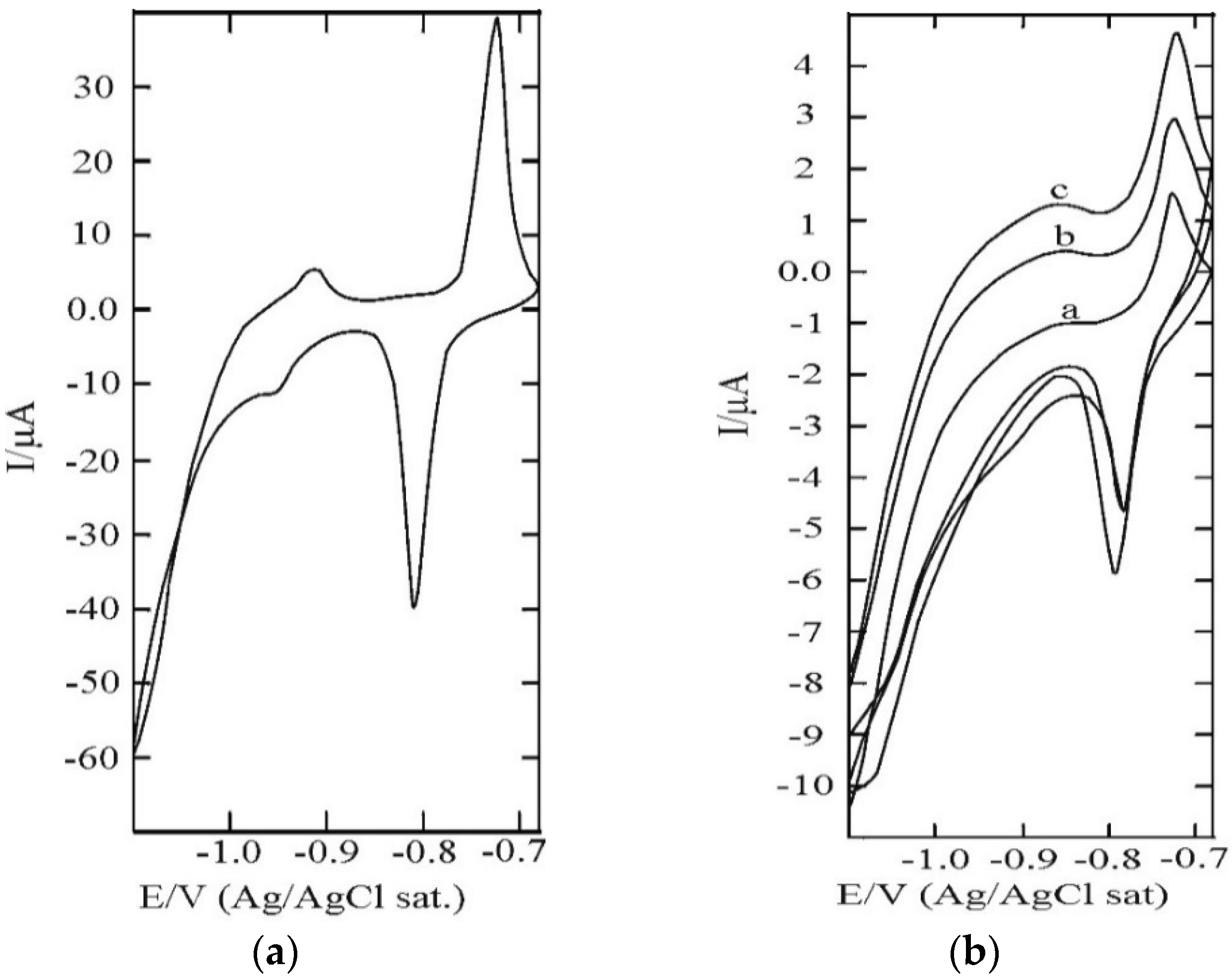
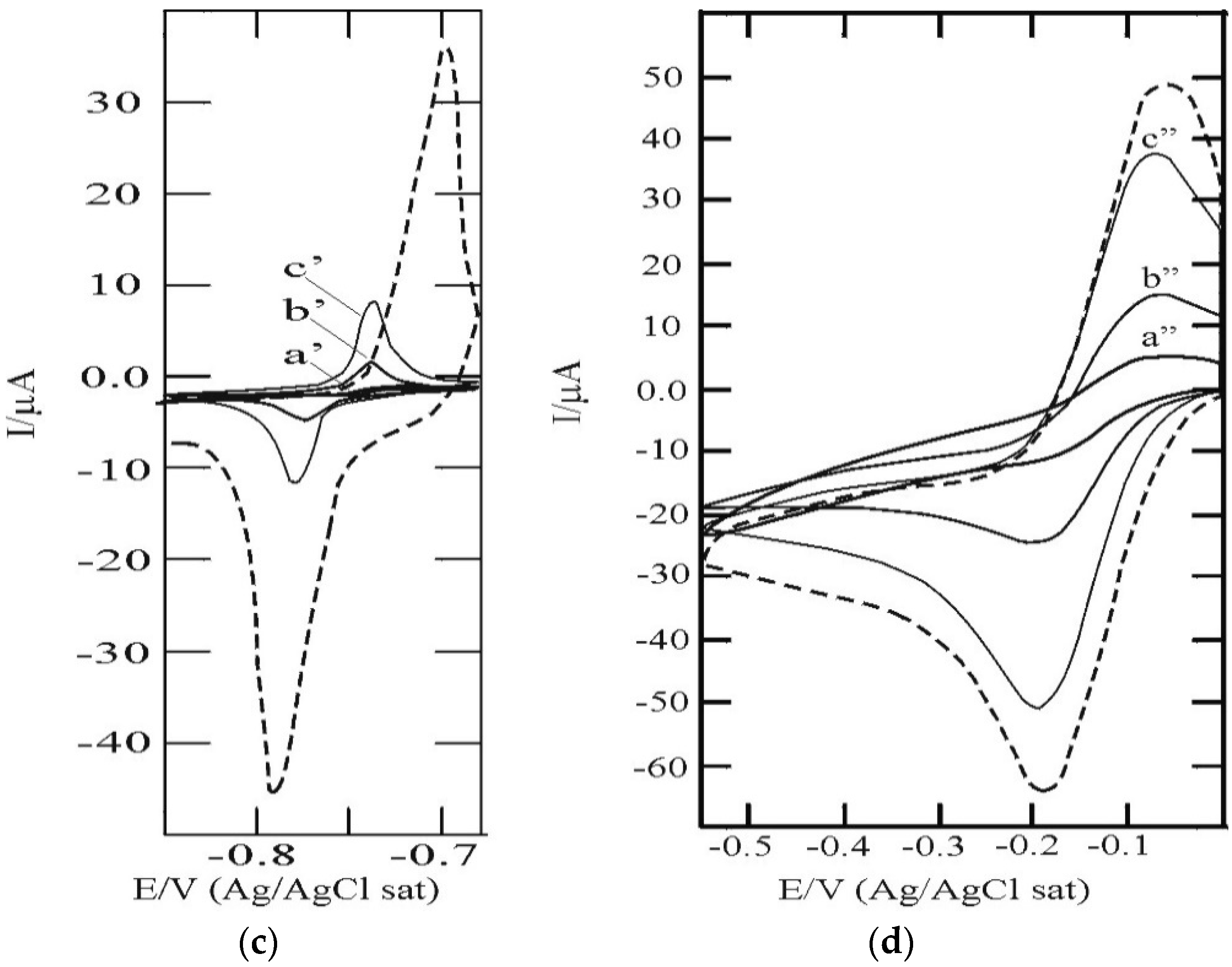
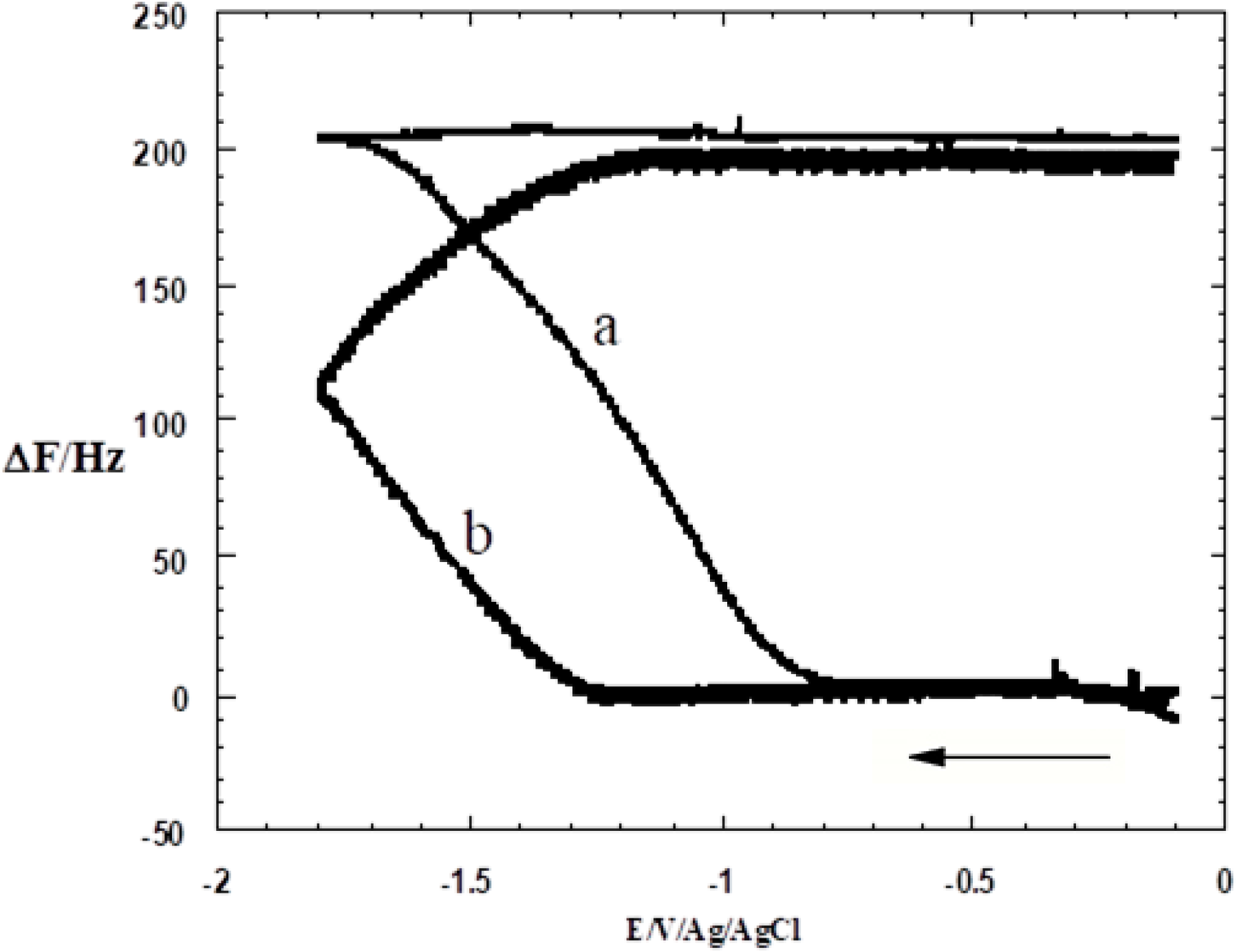
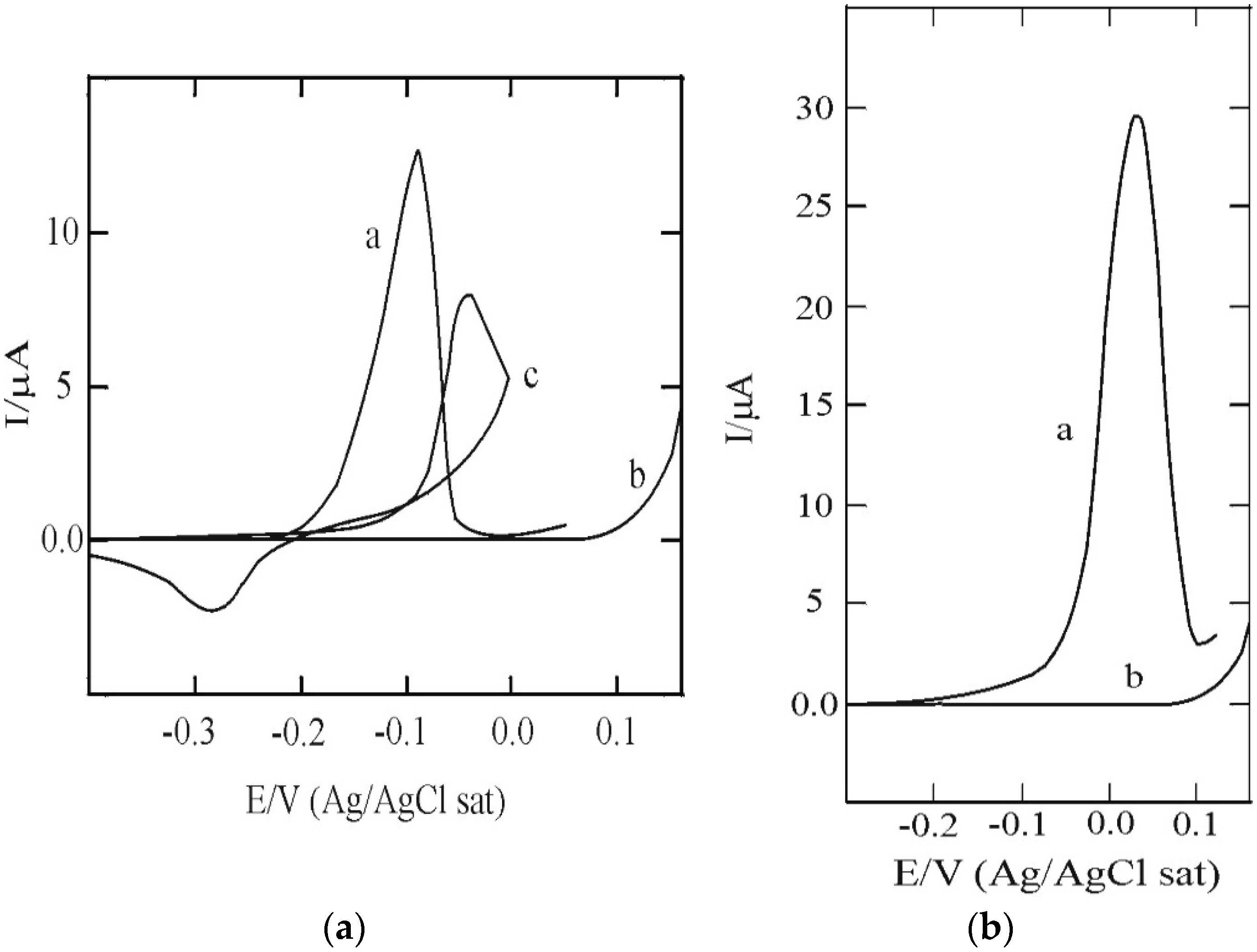
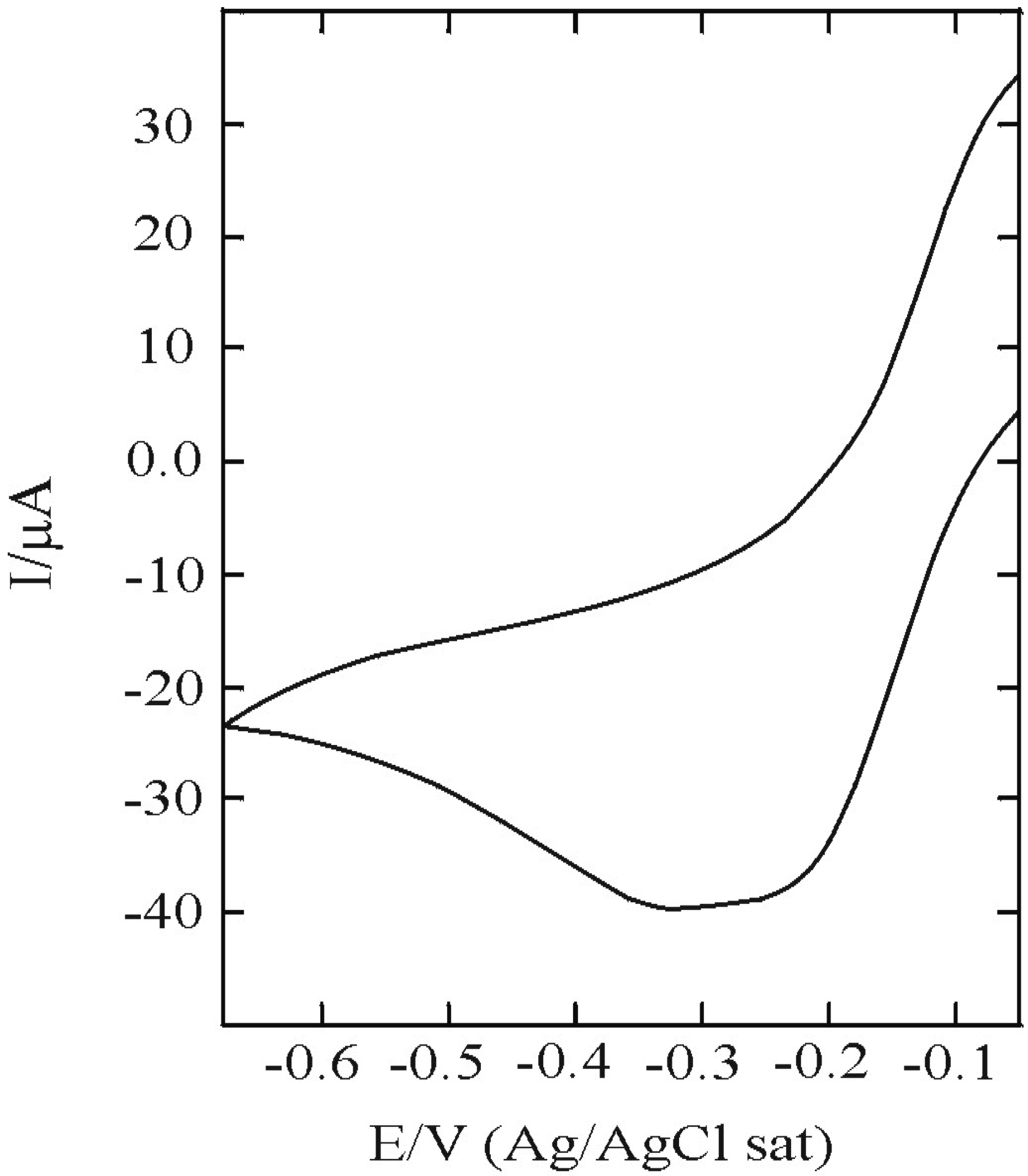
3.1. SAM of C16SH on Ag(III)
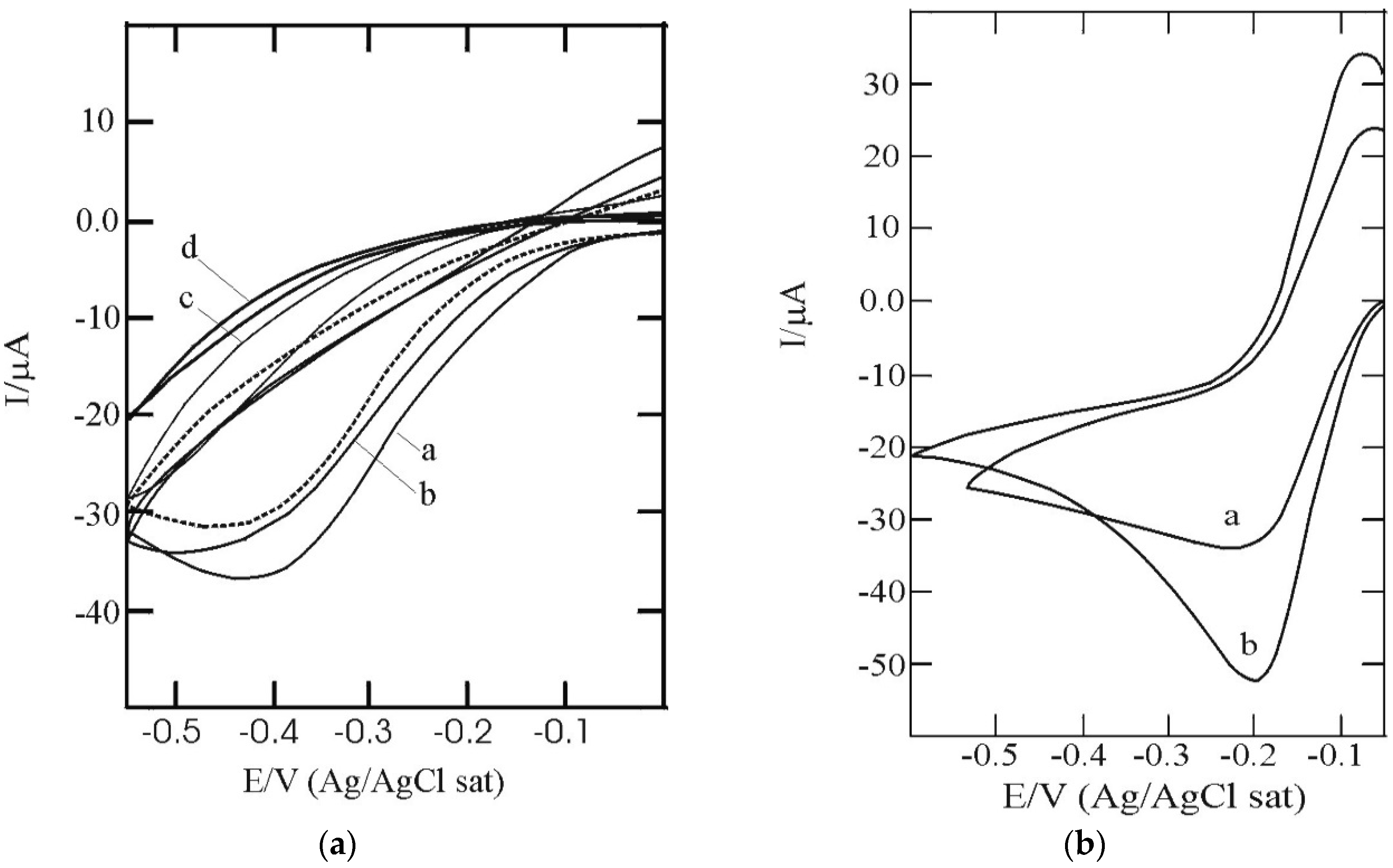
3.2. SAM of C16SH on Ag(III) Covered by an Ultrathin Film of CdS
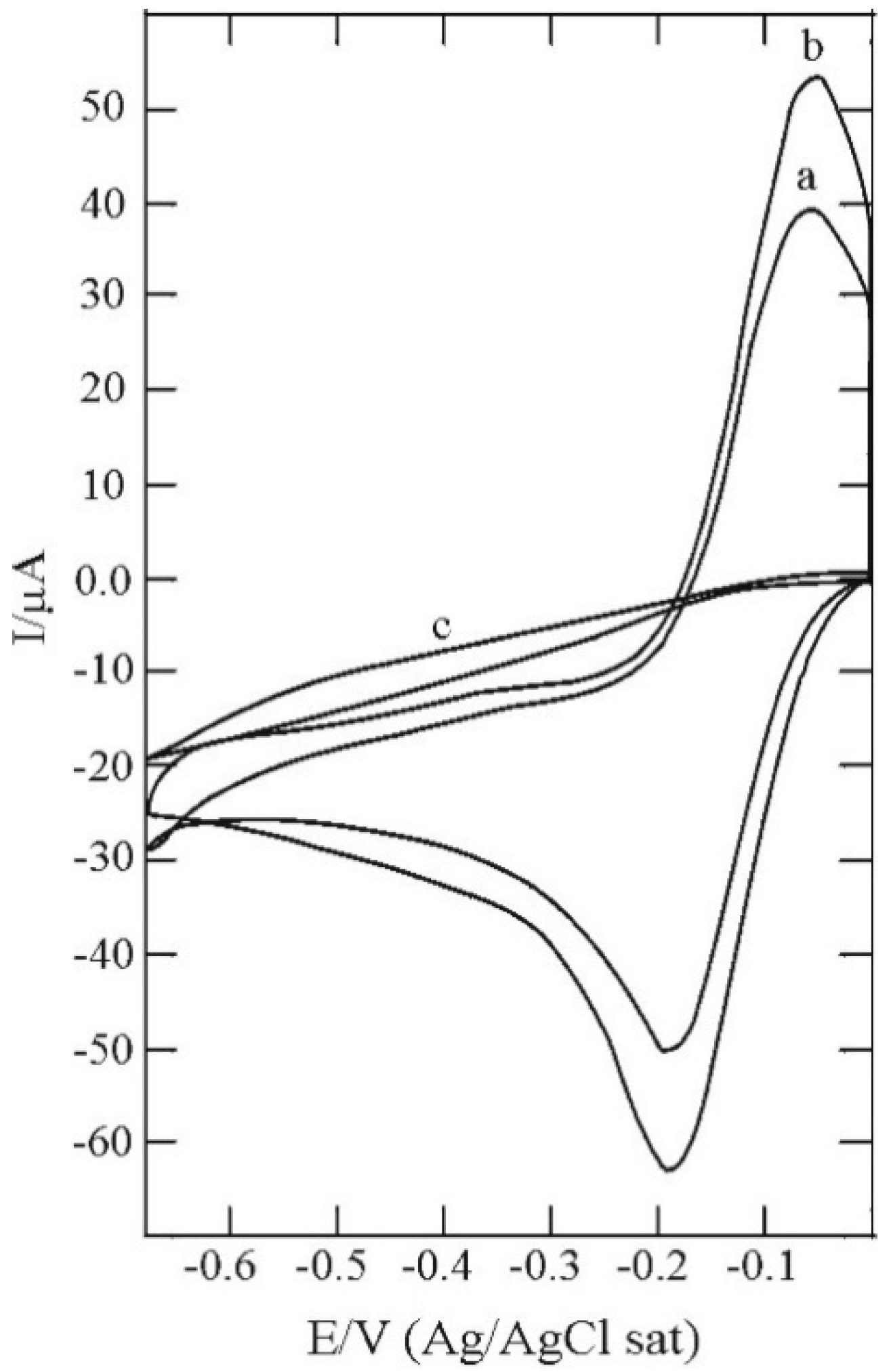
3.3. C16SH SAM Formation on CdS
3.4. Estimate of the SAM Layer Quality
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Schreiber, R. Structure and growth of self-assembled monolayers. Prog. Surf. Sci. 2000, 65, 151–256. [Google Scholar] [CrossRef]
- Ulman, A. Formation and structure of self-assembled monolayers. Chem. Rev. 1996, 96, 1533–1554. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Lin, Z.; Butera, R.A.; Smentkowski, V.S.; Waldeck, D.H. Preparation of Self-Assembled Monolayers on InP. Langmuir 1995, 11, 1849–1851. [Google Scholar] [CrossRef]
- Gu, Y.; Waldeck, D.H. Studies of electron tunneling at semiconductor electrodes. J. Phys. Chem. 1996, 100, 9573–9576. [Google Scholar] [CrossRef]
- Gu, Y.; Kumar, K.; Read, I.; Zimmt, M.B. Studies into the character of electronic coupling in ectron transfer. J. Photochem. Photobiol. A 1997, 105, 189–196. [Google Scholar] [CrossRef]
- Gu, Y.; Waldeck, D.H. Electron Tunneling at the Semiconductor−Insulator−Electrolyte Interface. Photocurrent Studies of the n-InP−Alkanethiol−Ferrocyanide System. J. Phys. Chem. B 1998, 102, 9015–9028. [Google Scholar] [CrossRef]
- Sheen, C.W.; Shi, J.X.; Martensson, J.; Parikh, A.N.; Allara, D.L. A New Class of Organized Self-Assembled Monolayers: Alkane Thiols on GaAs(100). J. Am. Chem. Soc. 1992, 114, 1514–1515. [Google Scholar] [CrossRef]
- Baum, T.; Ye, S.; Uosaki, K. Formation of Self-Assembled Monolayers of Alkanethiols on GaAs Surface with in Situ Surface Activation by Ammonium Hydroxide. Langmuir 1999, 15, 8577–8579. [Google Scholar] [CrossRef]
- Ye, S.; Li, G.; Noda, H.; Uosaki, K.; Osawa, M. Characterization of self-assembled monolayers of alkanethiol on GaAs surface by contact angle and angle-resolved XPS measurements. Surf. Sci. 2003, 529, 163–170. [Google Scholar] [CrossRef]
- Kosuri, M.R.; Cone, R.; Li, Q.; Han, S.M.; Bunker, B.C.; Mayer, T.M. Adsorption kinetics of 1-alkanethiols on hydrogenated Ge(111). Langmuir 2004, 20, 835–840. [Google Scholar] [CrossRef] [PubMed]
- Porter, M.D.; Bright, T.B.; Allara, D.L.; Chidsey, C.E.D. Spontaneously organized molecular assemblies. 4. Structural characterization of n-alkyl thiol monolayers on gold by optical ellipsometry, infrared spectroscopy, and electrochemistry. J. Am. Chem. Soc. 1987, 109, 3559–3568. [Google Scholar] [CrossRef]
- Miller, C.; Cuendet, P.; Graetzel, M. Adsorbed .omega.-hydroxy thiol monolayers on gold electrodes: Evidence for electron tunneling to redox species in solution. J. Phys. Chem. 1991, 95, 877–886. [Google Scholar] [CrossRef]
- Miller, C.; Graetzel, M. Electrochemistry at .omega.-hydroxythiol coated electrodes. 2. Measurement of the density of electronic states distributions for several outer-sphere redox couples. J. Phys. Chem. 1991, 95, 5225–5233. [Google Scholar] [CrossRef]
- Becka, A.M.; Miller, C.J. Electrochemistry at .omega.-hydroxy thiol coated electrodes. 3. Voltage independence of the electron tunneling barrier and measurements of redox kinetics at large overpotentials. J. Phys. Chem. 1992, 96, 2657–2668. [Google Scholar] [CrossRef]
- Terrettaz, S.; Becka, A.M.; Traub, M.J.; Fettinger, J.C.; Miller, C.J. .omega.-Hydroxythiol Monolayers at Au Electrodes. 5. Insulated Electrode Voltammetric Studies of Cyano/Bipyridyl Iron Complexes. J. Phys. Chem. 1995, 99, 11216–11224. [Google Scholar] [CrossRef]
- Hatchett, D.W.; Uibel, R.H.; Stevenson, K.J.; Harris, J.M.; White, H.S. Electrochemical measurement of the free energy of adsorption of n-alkanethiolates at Ag(111). J. Am. Chem. Soc. 1998, 120, 1062–1069. [Google Scholar] [CrossRef]
- Azzaroni, O.; Vela, M.E.; Andreasen, G.; Carro, P.; Salvarezza, R.C. Electrodesorption potentials of self-assembled alkanethiolate monolayers on Ag(111) and Au(111). An electrochemical, scanning tunneling microscopy and density functional theory study. J. Phys. Chem. B 2002, 106, 12267–12273. [Google Scholar] [CrossRef]
- Lee, T.; Wang, W.; Reed, M.A. Mechanism of Electron Conduction in Self-Assembled Alkanethiol Monolayer Devices. Ann. N. Y. Acad. Sci. 2003, 1006, 21–35. [Google Scholar] [CrossRef] [PubMed]
- Krysiński, P.; Brzostowska-Smolska, M. Three-probe voltammetric characterisation of octadecanethiol self-assembled monolayer integrity on gold electrodes. J. Electroanal. Chem. 1997, 424, 61–67. [Google Scholar] [CrossRef]
- Finklea, H.O.; Robinson, L.R.; Blackburn, A.; Richter, B.; Allara, D.; Bright, T. Formation of an Organized Monolayer by Solution Adsorption of Octadecyltrichlorosilane on Gold: Electrochemical Properties and Structural Characterization. Langmuir 1986, 2, 239–244. [Google Scholar] [CrossRef]
- Finklea, H.O.; Avery, S.; Lynch, M.; Furtsch, T. Blocking oriented monolayers of alkyl mercaptans on gold electrodes. Langmuir 1987, 3, 409–413. [Google Scholar] [CrossRef]
- Innocenti, M.; Pezzatini, G.; Forni, F.; Foresti, M.L. CdS and ZnS Deposition on Ag(111) by Electrochemical Atomic Layer Epitaxy. J. Electrochem. Soc. 2001, 148, C357–C362. [Google Scholar] [CrossRef]
- Cecconi, T.; Atrei, A.; Bardi, U.; Forni, F.; Innocenti, M.; Loglio, F.; Foresti, M.L.; Rovida, G. X-ray photoelectron diffraction (XPD) study of the atomic structure of the ultrathin CdS phase deposited on Ag(111) by electrochemical atomic layer epitaxy (ECALE). J. Electron Spectros. Relat. Phenom. 2001, 114–116, 563–568. [Google Scholar] [CrossRef]
- Innocenti, M.; Cattarin, S.; Cavallini, M.; Loglio, F.; Foresti, M.L. Characterisation of thin films of CdS deposited on Ag(111) by ECALE. A morphological and photoelectrochemical investigation. J. Electroanal. Chem. 2002, 532, 219–225. [Google Scholar] [CrossRef]
- Innocenti, M.; Forni, F.; Pezzatini, G.; Raiteri, R.; Loglio, F.; Foresti, M.L. Electrochemical behavior of As on silver single crystals and experimental conditions for InAs growth by ECALE. J. Electroanal. Chem. 2001, 514, 75–82. [Google Scholar] [CrossRef]
- Foresti, M.L.; Milani, S.; Loglio, F.; Innocenti, M.; Pezzatini, G.; Cattarin, S. Ternary CdSxSe1−x deposited on Ag(111) by ECALE: Synthesis and characterization. Langmuir 2005, 21, 6900–6907. [Google Scholar] [CrossRef] [PubMed]
- Loglio, F.; Innocenti, M.; D’Acapito, F.; Felici, R.; Pezzatini, G.; Salvietti, E.; Foresti, M.L. Cadmium selenide electrodeposited by ECALE: Electrochemical characterization and preliminary results by EXAFS. J. Electroanal. Chem. 2005, 575, 161–167. [Google Scholar] [CrossRef]
- Cavallini, M.; Facchini, M.; Albonetti, C.; Biscarini, F.; Innocenti, M.; Loglio, F.; Salvietti, E.; Pezzatini, G.; Foresti, M.L. Two-dimensional self-organization of CdS ultra thin films by confined electrochemical atomic layer epitaxy growth. J. Phys. Chem. C 2007, 111, 1061–1064. [Google Scholar] [CrossRef]
- Wang, L.; Bevilacqua, M.; Chen, Y.X.; Filippi, J.; Innocenti, M.; Lavacchi, A.; Marchionni, A.; Miller, H.; Vizza, F. Enhanced electro-oxidation of alcohols at electrochemically treated polycrystalline palladium surface. J. Power Sources 2013, 242, 872–876. [Google Scholar] [CrossRef]
- Gregory, B.W.; Stickney, J.L. Electrochemical atomic layer epitaxy (ECALE). J. Electroanal. Chem. Interfacial Electrochem. 1991, 300, 543–561. [Google Scholar] [CrossRef]
- Azzaroni, O.; Schilardi, P.L.; Salvarezza, R.C. Metal electrodeposition on self-assembled monolayers: A versatile tool for pattern transfer on metal thin films. Electrochim. Acta 2003, 48, 3107–3114. [Google Scholar] [CrossRef]
- Foresti, M.L.; Capolupo, F.; Innocenti, M.; Loglio, F. Visual Detection of Crystallographic Orientations of Face-Centered Cubic Single Crystals. Cryst. Growth Des. 2002, 2, 73–77. [Google Scholar] [CrossRef]
- White, R.E.; Bockris, J.O.; Conway, B.E. Modern Aspects of Electrochemistry; Springer: New York, NY, USA, 1995; ISBN 978-1-4899-1726-3. [Google Scholar]
- Diao, P.; Jiang, D.; Cui, X.; Gu, D.; Tong, R.; Zhong, B. Studies of structural disorder of self-assembled thiol monolayers on gold by cyclic voltammetry and ac impedance. J. Electroanal. Chem. 1999, 464, 61–67. [Google Scholar] [CrossRef]
- Bain, C.D.; Troughton, E.B.; Tao, Y.T.; Evall, J.; Whitesides, G.M.; Nuzzo, R.G. Formation of monolayer films by the spontaneous assembly of organic thiols from solution onto gold. J. Am. Chem. Soc. 1989, 111, 321–335. [Google Scholar] [CrossRef]
- Vericat, C.; Andreasen, G.; Vela, M.E.; Martin, H.; Salvarezza, R.C. Following transformation in self-assembled alkanethiol monolayers on Au(111) by in situ scanning tunneling microscopy. J. Chem. Phys. 2001, 115, 6672–6678. [Google Scholar] [CrossRef]
- Amatore, C.; Savéant, J.M.; Tessier, D. Charge transfer at partially blocked surfaces: A model for the case of microscopic active and inactive sites. J. Electroanal. Chem. Interfacial Electrochem. 1983, 147, 39–51. [Google Scholar] [CrossRef]
- Seah, P.M.; Dench, A.W. Quantitative electron spectroscopy of surfaces: A standard data base for electron inelastic mean free paths in solids. Surf. Interface Anal. 1979, 1, 2–11. [Google Scholar] [CrossRef]
- Woodruff, D.P.; Delchar, T.A. Modern Techniques of Surface Science; Cambridge Solid State Science Series; Cambridge Univercity Press: Cambridge, UK, 1986. [Google Scholar]
- Powell, C.J.; Jablonski, A. NIST Electron Inelastic-Mean-Free-Path Database; U.S. Department of Commerce: Washington, DC, USA, 2010.
- Finklea, H.O.; Snider, D.A.; Fedyk, J.; Sabatani, E.; Gafni, Y.; Rubinstein, I. Characterization of Octadecanethiol-Coated Gold Electrodes as Microarray Electrodes by Cyclic Voltammetry and ac Impedance Spectroscopy. Langmuir 1993, 9, 3660–3667. [Google Scholar] [CrossRef]

| Sample | Total Thickness (Å) | CdS Thickness (Å) | C16SH Thickness (Å) |
|---|---|---|---|
| 30 CdS/Ag(III) | 85 + 7 | 75 + 10 | - |
| C16SH/Ag(III) | 25 + 5 | - | 25 + 5 |
| C16SH + 10CdS/Ag(III) | 40 + 6 | 20 + 3 | 20 + 3 |
| C16SH + 30CdS/Ag(III) | 88 + 8 | 68 + 7 | 20 + 3 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salvietti, E.; Giurlani, W.; Foresti, M.L.; Passaponti, M.; Fabbri, L.; Marcantelli, P.; Caporali, S.; Martinuzzi, S.; Calisi, N.; Pedio, M.; et al. On the Contrasting Effect Exerted by a Thin Layer of CdS against the Passivation of Silver Electrodes Coated with Thiols. Surfaces 2018, 1, 29-42. https://doi.org/10.3390/surfaces1010004
Salvietti E, Giurlani W, Foresti ML, Passaponti M, Fabbri L, Marcantelli P, Caporali S, Martinuzzi S, Calisi N, Pedio M, et al. On the Contrasting Effect Exerted by a Thin Layer of CdS against the Passivation of Silver Electrodes Coated with Thiols. Surfaces. 2018; 1(1):29-42. https://doi.org/10.3390/surfaces1010004
Chicago/Turabian StyleSalvietti, Emanuele, Walter Giurlani, Maria Luisa Foresti, Maurizio Passaponti, Lorenzo Fabbri, Patrick Marcantelli, Stefano Caporali, Stefano Martinuzzi, Nicola Calisi, Maddalena Pedio, and et al. 2018. "On the Contrasting Effect Exerted by a Thin Layer of CdS against the Passivation of Silver Electrodes Coated with Thiols" Surfaces 1, no. 1: 29-42. https://doi.org/10.3390/surfaces1010004









