Clay Aerogel Supported Palladium Nanoparticles as Catalysts
Abstract
:1. Introduction
2. Results and Discussion
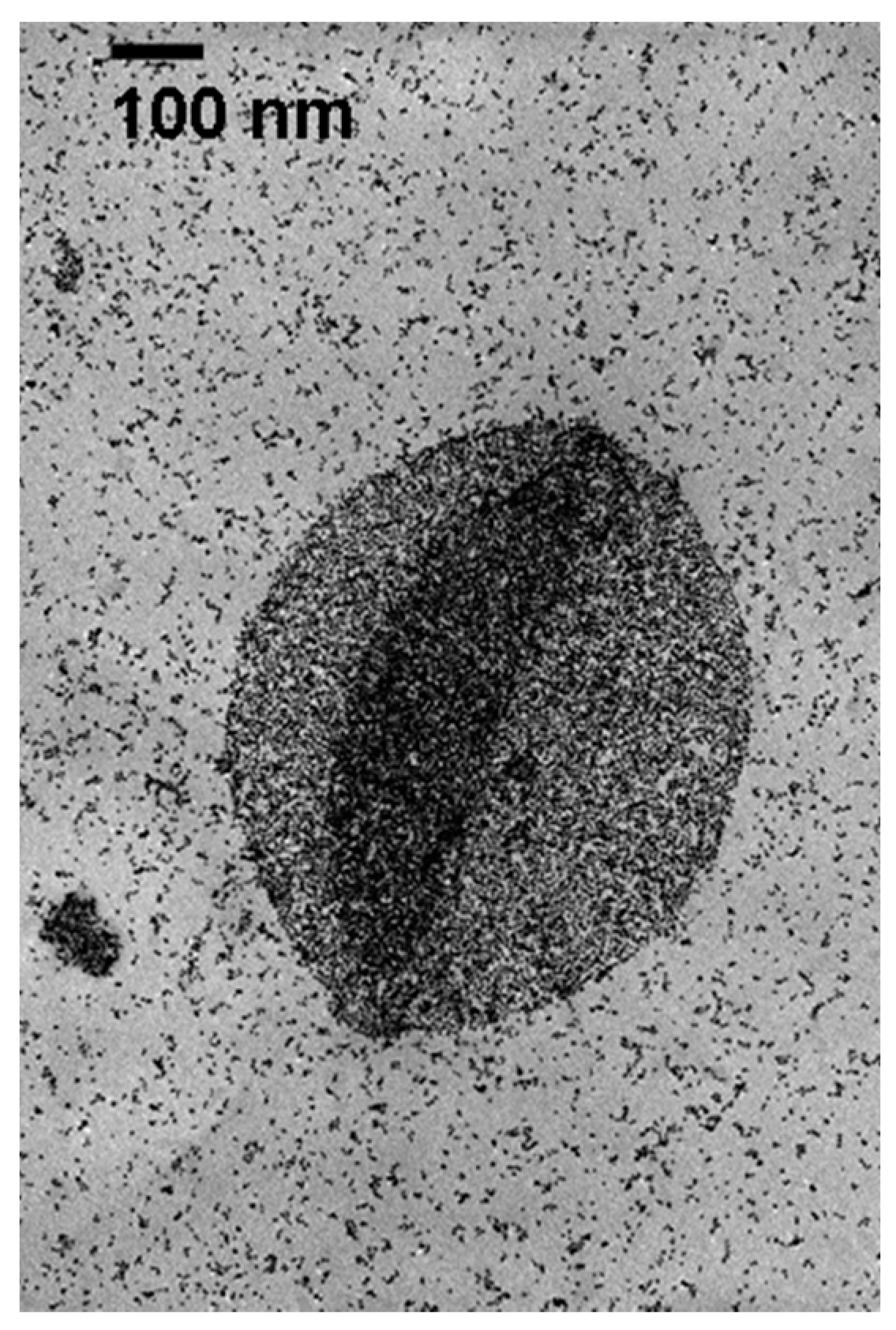
2.1. Catalyst Preparation and Characterization
2.2. Catalytic Reactions of the Pd/MMT Aerogel
3. Conclusions
4. Experimental Section
4.1. Materials
4.2. Nanoparticle Preparation
4.3. Aerogel Preparation
4.4. Characterization
4.5. Safety Precaution
4.6. Catalytic Reactions—Isomerization
4.7. Catalytic Reactions—Hydrogenation
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Timken, H.K.C. Homogeneous Modified-Alumina Fischer-Tropsch Catalyst Supports. U.S. 7,348,293, 25 March 2008. [Google Scholar]
- Cornils, B.; Herrmann, W.A. (Eds.) Applied Homogeneous Catalysis with Organometallic Compounds, a Comprehensive Handbook in Three Volumes; Wiley-VCH: Weinheim, Germany, 2002.
- Ertl, G.; Knőzinger, H.; Weitkamp, J. Handbook of Hetergenerous Catalysis; Wiley-VCH: Weinheim, Germany, 1997. [Google Scholar]
- Anderson, M.L.; Stroud, R.M.; Morris, C.A.; Merzbacker, C.I.; Rolison, D.R. Tailoring advanced nanoscale materials through synthesis of composite aerogel architectures. Adv. Eng. Mater. 2000, 2, 481–488. [Google Scholar] [CrossRef]
- Guha, A.; Zawodzinski, T.A., Jr.; Schiraldi, D.A. Evaluation of electrochemical performance for surface-modified carbons as catalyst support in Polymer Electrolyte Membrane (PEM) fuel cells. J. Power Sources 2007, 172, 530–541. [Google Scholar] [CrossRef]
- Guha, A.; Lu, W.; Zawodzinski, T.A., Jr.; Schiraldi, D.A. Surface-modified carbons as platinum catalyst supports for PEM fuel cells. Carbon 2007, 45, 1506–1517. [Google Scholar] [CrossRef]
- Meador, M.A.B.; Fabrizio, E.F.; Ilhan, F.; Dass, A.; Zhang, G.; Vassilaras, P.; Johnston, J.C.; Leventis, N. Crosslinking amine-modified silica aerogels with epoxies: Mechanically strong lightweight porous materials. Chem. Mater. 2005, 17, 1085–1098. [Google Scholar] [CrossRef]
- Fisher, A.E.; Pettigrew, K.A.; Rolison, D.R.; Stroud, R.H.; Long, J.W. Incorporation of homogeneous, nanoscale MnO2 within ultraporous carbon structures via self-limiting electroless deposition: Implications for electrochemical capacitors. Nano Lett. 2007, 7, 281–286. [Google Scholar] [CrossRef] [PubMed]
- Somlai, L.; Bandi, S.; Mathias, L.; Schiraldi, D.A. Facile processing of clays into organically modified aerogels. AIChE J. 2006, 52, 1162. [Google Scholar] [CrossRef]
- Bandi, S.; Schiraldi, D.A. Glass transition behavior of clay/poly(vinyl alcohol) composites. Macromolecules 2006, 39, 6537–6545. [Google Scholar] [CrossRef]
- Arndt, E.; Gawryla, M.D.; Schiraldi, D.A. Elastic, low density epoxy/clay aerogel composites. J. Mater. Chem. 2007, 17, 3525–3529. [Google Scholar] [CrossRef]
- Gawryla, M.D.; Liu, L.; Grunlan, J.; Schiraldi, D.A. pH tailoring electrical and mechanical behavior of polymer-clay-nanotube aerogels. Macromol. Rapid Commun. 2009, 30, 1669–1673. [Google Scholar] [CrossRef] [PubMed]
- Bandi, S.; Bell, M.; Schiraldi, D.A. Temperature-responsive clay aerogel polymer composites. Macromolecules 2005, 38, 9216–9220. [Google Scholar] [CrossRef]
- Chen, H.-B.; Chiou, B.-S.; Wang, Y.-Z.; Schiraldi, D.A. Biodegradable pectin/clay aerogels. ACS Appl. Mater. Interfaces 2013, 5, 1715–1721. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Schiraldi, D.A. Foam-like materials produced from milk and sodium montmorillonite clay using a freeze-drying process. Green Mater. 2013, 1, 11–15. [Google Scholar] [CrossRef]
- Chen, H.-B.; Wang, Y.-Z.; Sánchez-Soto, M.; Schiraldi, D.A. Low flammability, foam-like materials based on ammonium alginate and sodium montmorillonite clay. Polymer 2012, 53, 5825–5831. [Google Scholar] [CrossRef]
- Pojanavaraphan, T.; Magaraphan, R.; Chiou, B.-S.; Schiraldi, D.A. Development of biodegradable foamlike materials based on casein and sodium montmorillonite clay. Biomacromolecules 2010, 11, 2640–2646. [Google Scholar] [CrossRef] [PubMed]
- Okada, T.; Kato, T.; Yamaguchi, T.; Sakai, T.; Mishima, T. Layered clay aerogels by a freeze-drying process for a platinum-supported catalyst. Ind. Eng. Chem. Res. 2013, 52, 12018–12024. [Google Scholar] [CrossRef]
- Liu, H.W.; Huang, K.; Mi, J.D.; Jia, Y.; Huang, M.Y.; Jiang, Y.Y. Hydrogenation of nitrobenzene, cyclohexanone and octene catalyzed by silica-supported starch-l-glutamic acid–Fe complex. Polym. Adv. Technol. 2003, 14, 355–359. [Google Scholar] [CrossRef]
- Lu, P.; Teranishi, T.; Asakura, K.; Miyake, M.; Toshima, N.J. Polymer-protected Ni/Pd bimetallic nano-clusters: Preparation, characterization and catalysis for hydrogenation of nitrobenzene. Phys. Chem. B 1999, 103, 9673–9682. [Google Scholar] [CrossRef]
- Flores, R.; Lopez-Castillo, Z.K.; Kani, I.; Fackler, J.P., Jr.; Akgerman, A. Kinetics of the homogeneous catalytic hydrogenation of olefins in supercritical carbon dioxide using a fluoroacrylate copolymer grafted rhodium catalyst. Ind. Eng. Chem. Res. 2003, 42, 6720–6729. [Google Scholar] [CrossRef]
- Adlim, M.; Bakar, M.A.; Liew, K.Y.; Ismail, J. Synthesis of chitosan-stabilized platinum and palladium Na-noparticles and their hydrogenation activity. J. Mol. Catal. A Chem. 2004, 212, 141–149. [Google Scholar] [CrossRef]
- Liu, C.; Xu, Y.; Liao, S.; Yu, D. Synergic effect of polymer-supported bimetallic catalysts in the hydrogenation and isomerization of 1-octene. J. Mol. Catal. A Chem. 1999, 149, 119–123. [Google Scholar] [CrossRef]
- Bhattacharjee, S.; Bruening, M.L. Selective hydrogenation of monosubstituted alkenes by Pd nanoparticles embedded in polyelectrolyte films. Langmuir 2008, 24, 2916–2920. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Angelici, R.J. Combination catalysts consisting of a homogeneous catalyst tethered to a silica-supported palladium heterogeneous catalyst: Arene hydrogenation. J. Am. Chem. Soc. 1997, 119, 6937–6938. [Google Scholar] [CrossRef]
- Hansford, R.C. Platinum Group Metal on Silica-Alumina Hydrogenation Catalyst and Process. U.S. Patent 3,637,484, 25 January 1972. [Google Scholar]
- Collman, J.P.; Hegedus, L.S.; Norton, J.R.; Finke, R.G. Principles and Applications of Organotransition Metal Chemistry; University Science Books: Mill Valley, CA, USA, 1978; pp. 389–882. [Google Scholar]



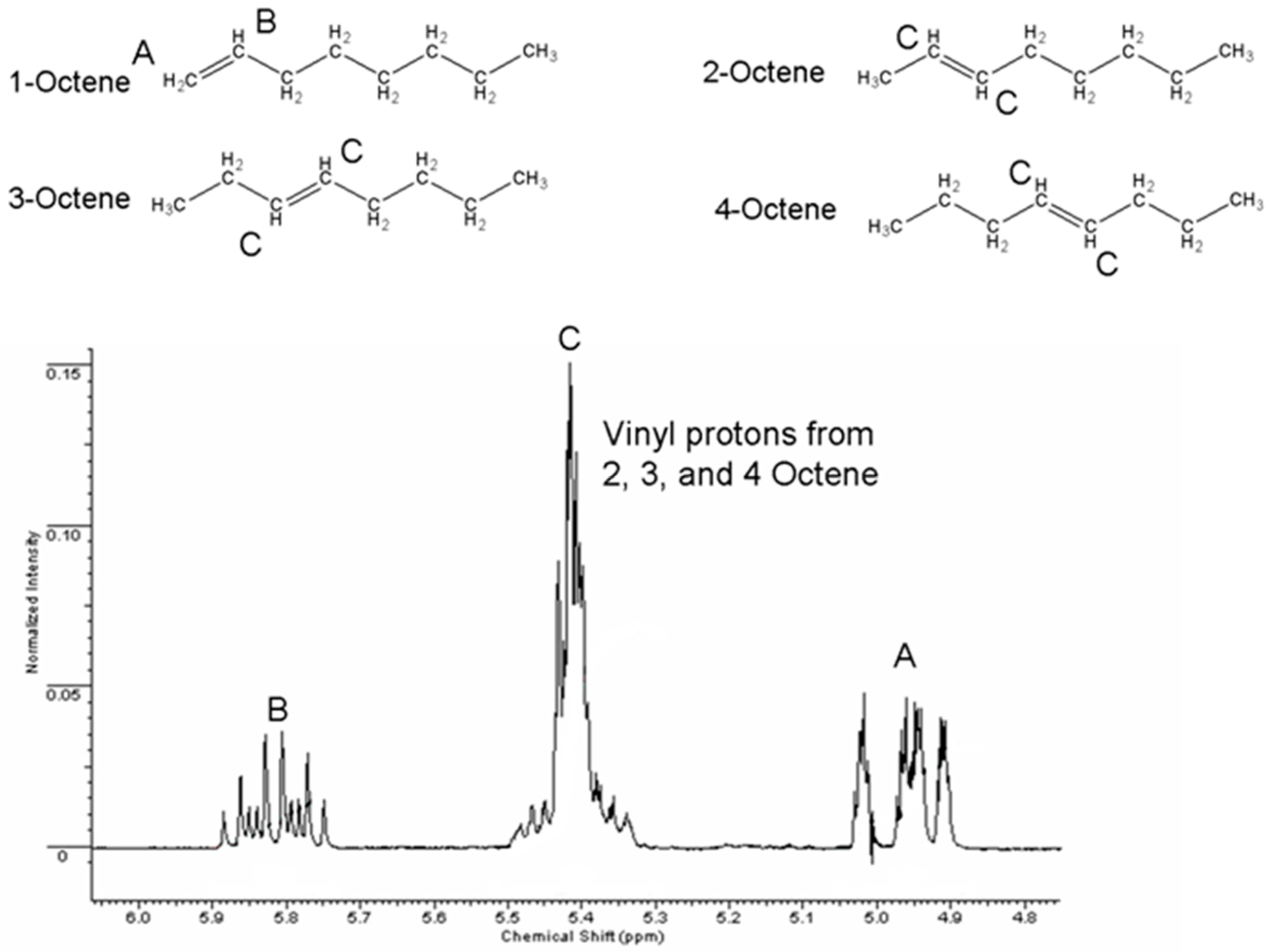
| Reaction Time (h) | Percentage of Total Composition | ||
|---|---|---|---|
| Unreacted 1-octene | 2-, 3-, and 4-octene | octane | |
| 0.25 | 3 | 50 | 47 |
| 0.5 | 4 | 5 | 91 |
| 1 | nd | Nd | 100 |
| 12 | nd | Nd | 100 |
| Sample | Amount of Pd-PGW (g) | Moles of Pd in Reaction | Moles of 1-Octene | Measured Conversion over 12 h | Mol Ratio of Substrate to Catalyst | 12 h Avg. TON (h−1) |
|---|---|---|---|---|---|---|
| Isomerization Powder | 0.38 | 1.1 × 10−3 | 0.13 | 97% | 120 | 10 |
| Isomerization Aerogel | 0.0828 | 2.4 × 10−4 | 0.064 | 25% | 270 | 6 |
| Hydrogenation Aerogel | 0.1876 | 5.5 × 10−4 | 0.072 | 100% | 300 | 11 * |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Griebel, J.J.; Gawryla, M.D.; Milliman, H.W.; Schiraldi, D.A. Clay Aerogel Supported Palladium Nanoparticles as Catalysts. Gels 2016, 2, 15. https://doi.org/10.3390/gels2020015
Griebel JJ, Gawryla MD, Milliman HW, Schiraldi DA. Clay Aerogel Supported Palladium Nanoparticles as Catalysts. Gels. 2016; 2(2):15. https://doi.org/10.3390/gels2020015
Chicago/Turabian StyleGriebel, Jared J., Matthew D. Gawryla, Henry W. Milliman, and David A. Schiraldi. 2016. "Clay Aerogel Supported Palladium Nanoparticles as Catalysts" Gels 2, no. 2: 15. https://doi.org/10.3390/gels2020015







