Low-Molecular-Weight Gelators as Base Materials for Ointments
Abstract
:1. Introduction
2. Ointments
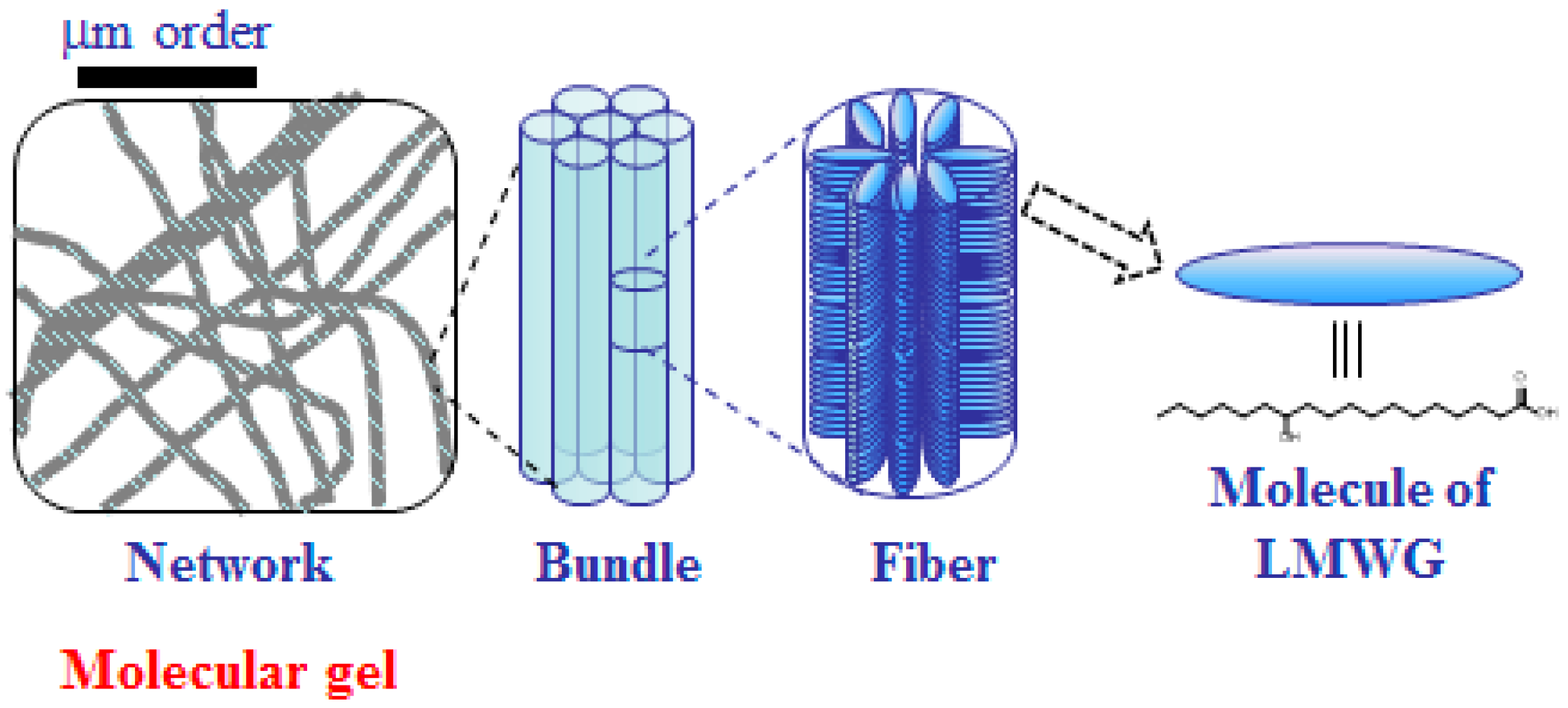
3. Molecular Gels
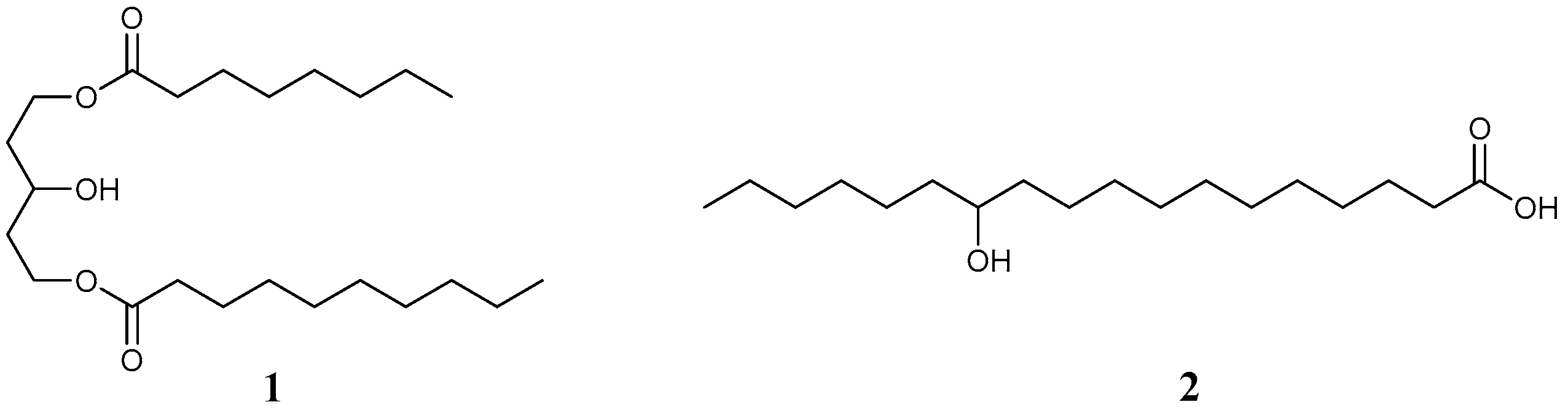
4. Studies Using Existing LMWGs
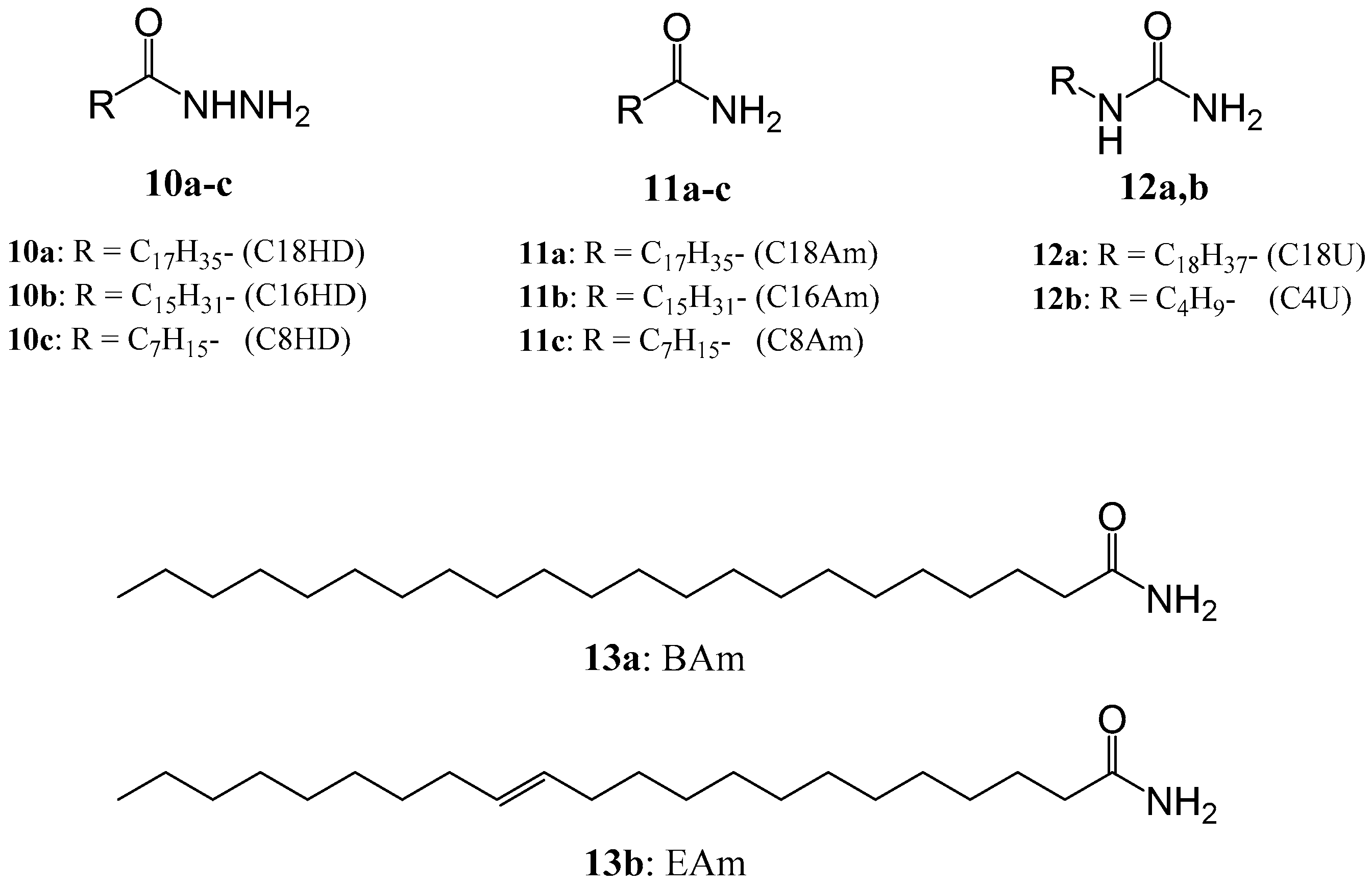
5. Studies Using New LMWGs
6. Conclusions
Acknowledgments
Conflicts of Interest
References
- Fox, L.P.; Merk, H.F.; Bickers, D.R. Dermatological Pharmacology. In Goodman & Gilman’s The Pharmacological Basis of Therapeutics, 11th ed.; Laurence, B., John, L., Keith, P., Eds.; The McGraw-Hill Companies Inc.: New York, NY, USA, 2005; pp. 1679–1706. [Google Scholar]
- Dayan, N. Delivery System Design in Topically Applied Formulations: An Overview. In Delivery System Handbook for Personal Care and Cosmetic Products, Technology, Applications, and Formulations; Rosen, M.R., Ed.; William Andrew, Inc.: New York, NY, USA, 2005; pp. 101–118. [Google Scholar]
- Sugibayashi, K.; Morimoto, Y. Transdermal Patches. In Gels Handbook, The Fundamentals; Osada, Y., Kajiwara, K., Fushimi, T., Irasa, O., Hirokawa, Y., Matsunaga, T., Shimomura, T., Wang, L., Ishida, H., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2001; Volume 3, Section 6; pp. 201–210. [Google Scholar]
- Inaki, T. Ointments for antiinflammatory drugs. In Gels Handbook, The Fundamentals; Osada, Y., Kajiwara, K., Fushimi, T., Irasa, O., Hirokawa, Y., Matsunaga, T., Shimomura, T., Wang, L., Ishida, H., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2001; Volume 3, Section 7; pp. 211–220. [Google Scholar]
- Weiss, R.G.; Terech, P. (Eds.) Molecular Gels: Materials With Self-assembled Fibrillar Networks; Springer: Dordrecht, The Netherlands, 2006.
- Fages, F. (Ed.) Low Molecular Mass Gelators Design, Self-Assembly, Function; Springer: Dordrecht, The Netherlands, 2005.
- Hanabusa, K.; Suzuki, M. Development of low-molecular-weight gelators and polymer-based gelators. Polym. J. 2014, 46, 776–782. [Google Scholar] [CrossRef]
- Du, X.-W.; Zhou, J.; Xu, B. Supramolecular Hydrogels Made of Basic Biological Building Blocks. Chem. Asian J. 2014, 9, 1446–1472. [Google Scholar] [CrossRef] [PubMed]
- Boekhoven, J.; Stupp, S.I. 25th Anniversary Article: Supramolecular Materials for Regenerative Medicine. Adv. Mater. 2014, 26, 1642–1659. [Google Scholar] [CrossRef] [PubMed]
- Babu, S.S.; Praveen, V.K.; Ajayaghosh, A. Functional π‑Gelators and Their Applications. Chem. Rev. 2014, 114, 1973–2129. [Google Scholar] [CrossRef] [PubMed]
- Kumar, K.; Steed, J.W. Supramolecular gel phase crystallization: Orthogonal self-assembly under non-equilibrium conditions. Chem. Soc. Rev. 2014, 43, 2080–2088. [Google Scholar] [CrossRef] [PubMed]
- Weiss, R.G. The Past, Present, and Future of Molecular Gels. What Is the Status of the Field, and Where Is It Going? J. Am. Chem. Soc. 2014, 136, 7519–7530. [Google Scholar] [CrossRef] [PubMed]
- Rogers, M.A.; Weiss, R.G. Systematic modifications of alkane-based molecular gelators and the consequences to the structures and properties of their gels. New J. Chem. 2015, 39, 785–799. [Google Scholar] [CrossRef]
- Cornwell, D.J.; Smith, D.K. Expanding the scope of gels–Combining polymers with low-molecular-weight gelators to yield modified self-assembling smart materials with high-tech applications. Mater. Horiz. 2015, 2, 279–293. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, X.-F.; Wang, T.-Y.; Liu, M.-H. Tuning Soft Nanostructures in Self-assembled Supramolecular Gels: From Morphology Control to Morphology-Dependent Functions. Small 2015, 11, 1025–1038. [Google Scholar] [CrossRef] [PubMed]
- Zurcher, D.M.; McNeil, A.J. Tools for Identifying Gelator Scaffolds and Solvents. J. Org. Chem. 2015, 80, 2473–2478. [Google Scholar] [CrossRef] [PubMed]
- Rest, C.; Kandanelli, R.; Fernández, G. Strategies to create hierarchical self-assembled structures via cooperative non-covalent interactions. Chem. Soc. Rev. 2015, 44, 2543–2572. [Google Scholar] [CrossRef] [PubMed]
- Lan, Y.; Corradini, M.G.; Weiss, R.G.; Raghavanc, S.R.; Rogers, M.A. To gel or not to gel: Correlating molecular gelation with solvent parameters. Chem. Soc. Rev. 2015, 44, 6035–6058. [Google Scholar] [CrossRef] [PubMed]
- Brambilla, D.; Luciani, P.; Leroux, J.-C. Breakthrough discoveries in drug delivery technologies: The next 30 years. J. Control. Release 2014, 190, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Murdana, S. Organogels in drug delivery. Expert Opin. Drug Deliv. 2005, 2, 489–505. [Google Scholar] [CrossRef] [PubMed]
- Rehmana, K.; Zulfakar, M.H. Recent advances in gel technologies for topical and transdermal drug delivery. Drug Dev. Ind. Pharm. 2014, 40, 433–440. [Google Scholar] [CrossRef] [PubMed]
- Skilling, K.J.; Citossi, F.; Bradshaw, T.D.; Ashford, M.; Kellama, B.; Marlow, M. Insights into low molecular mass organic gelators: A focus on drug delivery and tissue engineering applications. Soft Matter 2014, 10, 237–256. [Google Scholar] [CrossRef] [PubMed]
- Du, X.-W.; Zhou, J.; Shi, J.-F.; Xu, B. Supramolecular Hydrogelators and Hydrogels: From Soft Matter to Molecular Biomaterials. Chem. Rev. 2015, 115, 13165–13307. [Google Scholar] [CrossRef] [PubMed]
- Franz, T.J. Percutaneous Absorption: On the Relevance of in Vitro Data. J. Invest. Dermatol. 1975, 3, 190–195. [Google Scholar] [CrossRef]
- Ng, S.-F.; Rouse, J.J.; Sanderson, F.D.; Meidan, V.; Eccleston, G.M. Validation of a Static Franz Diffusion Cell System for In Vitro Permeation Studies. AAPS Pharm. Sci. Tech. 2010, 11, 1432–1441. [Google Scholar] [CrossRef] [PubMed]
- Grassi, M.; Grassi, G. Mathematical Modelling and Controlled Drug Delivery: Matrix Systems. Curr. Drug Deliv. 2005, 2, 97–116. [Google Scholar] [CrossRef] [PubMed]
- Raval, A.; Parikh, J.; Engineer, C. Mechanism and in Vitro Release Kinetic Study of Sirolimus from a Biodegradable Polymeric Matrix Coated Cardiovascular Stent. Ind. Eng. Chem. Res. 2011, 50, 9539–9549. [Google Scholar] [CrossRef]
- Higuchi, T. Rate of release of medicaments from ointment bases containing drugs in suspension. Pharm. Sci. 1961, 50, 874–875. [Google Scholar] [CrossRef]
- Peppas, N.A. Analysis of Fickian and non-Fickian drug release from polymers. Pharm. Acta Helv. 1985, 60, 110–111. [Google Scholar] [PubMed]
- Grijalvo, S.; Mayr, J.; Eritja, R.; Díaz, D.D. Biodegradable liposome-encapsulated hydrogels for biomedical applications: A marriage of convenience. Biomater. Sci. 2016. [Google Scholar] [CrossRef] [PubMed]
- Mewis, J. Thixotropy—A General Review. J. Non-Newtonian Fluid Mech. 1979, 6, 1–20. [Google Scholar] [CrossRef]
- Barnes, H.A. Thixotropy—A review. J. Non-Newtonian Fluid Mech. 1997, 70, 1–33. [Google Scholar] [CrossRef]
- Mewis, J.; Wagner, N.J. Thixotropy. Adv. Colloid Interface Sci. 2009, 147–148, 214–227. [Google Scholar] [CrossRef] [PubMed]
- Goodwin, J.W.; Hughes, R.W. Rheology for Chemists: An Introduction, 2nd ed.; RSC Publishing: Cambridge, UK, 2008. [Google Scholar]
- Yu, X.-D.; Chen, L.-M.; Zhang, M.-M.; Yi, T. Low-molecular-mass gels responding to ultrasound and mechanical stress: Towards self-healing materials. Chem. Soc. Rev. 2014, 43, 5346–5371. [Google Scholar] [CrossRef] [PubMed]
- Sobczuk, A.A.; Tsuchiya, Y.; Shiraki, T.; Tamaru, S.-I.; Shinkai, S. Creation of Chiral Thixotropic Gels through a Crown–Ammonium Interaction and their Application to a Memory-Erasing Recycle System. Chem. Eur. J. 2012, 18, 2832–2838. [Google Scholar] [CrossRef] [PubMed]
- Yan, N.; Xu, Z.-Y.; Diehn, K.K.; Raghavan, S.R.; Fang, Y.; Weiss, R.G. Pyrenyl-Linker-Glucono Gelators. Correlations of Gel Properties with Gelator Structures and Characterization of Solvent Effects. Langmuir 2013, 29, 793–805. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.-Y.; Peng, J.-X.; Yan, N.; Yu, H.; Zhang, S.-S.; Liu, K.-Q.; Fang, Y. Simple design but marvelous performances: Molecular gels of superior strength and self-healing properties. Soft Matter 2013, 9, 1091–1099. [Google Scholar] [CrossRef]
- Higashi, D.; Yoshida, M.; Yamanaka, M. Thixotropic Hydrogel Formation in Various Aqueous Solutions through Self-Assembly of an Amphiphilic Tris-Urea. Chem. Asian. J. 2013, 8, 2584–2587. [Google Scholar] [CrossRef] [PubMed]
- Penaloza, D.P., Jr.; Shundo, A.; Matsumoto, K.; Ohno, M.; Miyaji, K.; Goto, M.; Tanaka, K. Spatial heterogeneity in the sol–gel transition of a supramolecular system. Soft Matter 2013, 9, 5166–5172. [Google Scholar] [CrossRef]
- Sun, Z.-F.; Li, Z.-Y.; He, Y.-H.; Shen, R.-J.; Deng, L.; Yang, M.-H.; Liang, Y.-Z.; Zhang, Y. Ferrocenoyl Phenylalanine: A New Strategy Toward Supramolecular Hydrogels with Multistimuli Responsive Properties. J. Am. Chem. Soc. 2013, 135, 13379–13386. [Google Scholar] [CrossRef] [PubMed]
- Hoshizawa, H.; Minemura, Y.; Yoshikawa, K.; Suzuki, M.; Hanabusa, K. Thixotropic Hydrogelators Based on a Cyclo(dipeptide) Derivative. Langmuir 2013, 29, 14666–14673. [Google Scholar] [CrossRef] [PubMed]
- Baral, A.; Roy, S.; Dehsorkhi, A.; Hamley, I.W.; Mohapatra, S.; Ghosh, S.; Banerjee, A. Assembly of an Injectable Noncytotoxic Peptide-Based Hydrogelator for Sustained Release of Drugs. Langmuir 2014, 30, 929–936. [Google Scholar] [CrossRef] [PubMed]
- Ono, F.; Watanabe, H.; Shinkai, S. Structural optimization of super-gelators derived from naturally-occurring mannose and their morphological diversity. RSC Adv. 2014, 4, 25940–25947. [Google Scholar] [CrossRef]
- Schön, E.-M.; Marqués-López, E.; Herrera, R.P.; Alemán, C.; Díaz, D.D. Exploiting Molecular Self-Assembly: From Urea-Based Organocatalysts to Multifunctional Supramolecular Gels. Chem. Eur. J. 2014, 20, 10720–10731. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ohsedo, Y.; Oono, M.; Saruhashi, K.; Watanabe, H. N-Alkylamido-d-Glucamine-Based Gelators for the Generation of Thixotropic Gels. RSC Adv. 2014, 4, 48554–48558. [Google Scholar] [CrossRef]
- Suzuki, M.; Hayakawa, Y.; Hanabusa, K. Thixotropic Supramolecular Gel Based on l-Lysine Derivatives. Gels 2015, 1, 81–93. [Google Scholar] [CrossRef]
- Wu, Y.; Liu, K.-Q.; Chen, X.-L.; Chen, Y.-P.; Zhang, S.-F.; Peng, J.-X.; Fang, Y. A novel calix[4]arene-based dimeric-cholesteryl derivative: Synthesis, gelation and unusual properties. New J. Chem. 2015, 39, 639–649. [Google Scholar] [CrossRef]
- Bhattacharjee, S.; Samanta, S.K.; Moitra, P.; Pramoda, K.; Kumar, R.; Bhattacharya, S.; Rao, C.N.R. Nanocomposite Made of an Oligo(p-phenylenevinylene)-Based Trihybrid Thixotropic Metallo(organo)gel Comprising Nanoscale Metal–Organic Particles, Carbon Nanohorns, and Silver Nanoparticles. Chem. Eur. J. 2015, 21, 5467–5476. [Google Scholar] [CrossRef] [PubMed]
- Pénzesa, T.; Blazsób, G.; Aignera, Z.; Falkayb, G.; Erősa, I. Topical absorption of piroxicam from organogels—In vitro and in vivo correlations. Int. J. Pharm. 2005, 298, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Satapathy, D.; Biswas, D.; Behera, D.; Sagiri, S.S.; Pal, K.; Pramanik, K. Sunflower-Oil-Based Lecithin Organogels as Matrices for Controlled Drug Delivery. J. Appl. Polym. Sci. 2013, 129, 585–594. [Google Scholar] [CrossRef]
- Iwanaga, K.; Sumizawa, T.; Miyazaki, M.; Kakemi, M. Characterization of organogel as a novel oral controlled release formulation for lipophilic compounds. Int. J. Pharm. 2010, 388, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Iwanaga, K.; Mineo Kawai, M.; Miyazaki, M.; Kakemi, M. Application of organogels as oral controlled release formulations of hydrophilic drugs. Int. J. Pharm. 2012, 436, 869–872. [Google Scholar] [CrossRef] [PubMed]
- Bastiata, G.; Plourdea, F.; Motulskya, A.; Furtosb, A.; Dumontc, Y.; Quirionc, R.; Fuhrmanna, G.; Leroux, J.-C. Tyrosine-based rivastigmine-loaded organogels in the treatment of Alzheimer’s disease. Biomaterials 2010, 31, 6031–6038. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dufresne, M.-H.; Marouf, E.; Kränzlin, Y.; Gauthier, M.A.; Leroux, J.-C. Lipase Is Essential for the Study of in Vitro Release Kinetics from Organogels. Mol. Pharm. 2012, 9, 1803–1811. [Google Scholar] [CrossRef] [PubMed]
- Okesola, B.O.; Vieira, V.M.P.; Cornwell, D.J.; Whitelaw, N.K.; Smith, D.K. 1,3:2,4-Dibenzylidene-d-sorbitol (DBS) and its derivatives—Efficient, versatile and industrially-relevant low-molecular-weight gelators with over 100 years of history and a bright future. Soft Matter 2015, 11, 4768–4787. [Google Scholar] [CrossRef] [PubMed]
- Howe, E.J.; Okesola, B.O.; Smith, D.K. Self-assembled sorbitol-derived supramolecular hydrogels for the controlled encapsulation and release of active pharmaceutical ingredients. Chem. Commun. 2015, 51, 7451–7454. [Google Scholar] [CrossRef] [PubMed]
- Mallia, V.A.; George, M.; Blair, D.L.; Weiss, R.G. Robust Organogels from Nitrogen-Containing Derivatives of (R)-12-Hydroxystearic Acid as Gelators: Comparisons with Gels from Stearic Acid Derivatives. Langmuir 2009, 25, 8615–8625. [Google Scholar] [CrossRef] [PubMed]
- Toro-Vazquez, J.F.; Morales-Rueda, J.; Torres-Martínez, J.; Charó-Alonso, M.A.; Mallia, V.A.; Weiss, R.G. Cooling Rate Effects on the Microstructure, Solid Content, and Rheological Properties of Organogels of Amides Derived from Stearic and (R)‑12-Hydroxystearic Acid in Vegetable Oil. Langmuir 2013, 29, 7642–7645. [Google Scholar] [CrossRef] [PubMed]
- Lescanne, M.; Grondin, P.; d’Aléo, A.; Fages, F.; Pozzo, J.L.; Monval, O.M.; Reinheimer, P.; Colin, A. Thixotropic Organogels Based on a Simple N-Hydroxyalkyl Amide: Rheological and Aging Properties. Langmuir 2004, 20, 3032–3041. [Google Scholar] [CrossRef] [PubMed]
- Ohsedo, Y.; Miyamoto, M.; Watanabe, H.; Oono, M.; Tanaka, A. Alkylhydrazide Derivatives as New Organogelators and their Potential Ability to Gel Electrolytes. Bull. Chem. Soc. Jpn. 2013, 86, 671–673. [Google Scholar] [CrossRef]
- Ohsedo, Y.; Watanabe, H.; Oono, M.; Tanaka, A. Mixing Enhancement Effect of Low-Molecular-Weight Organogelators for Thixotropic Organogel Creation. Chem. Lett. 2013, 42, 363–365. [Google Scholar] [CrossRef]
- Hirst, A.R.; Smith, D.K. Two-Component Gel-Phase Materials—Highly Tunable Self-Assembling Systems. Chem. Eur. J. 2005, 11, 5496–5508. [Google Scholar] [CrossRef] [PubMed]
- Buerkle, L.E.; Rowan, S.J. Supramolecular gels formed from multi-component low molecular weight species. Chem. Soc. Rev. 2012, 41, 6089–6102. [Google Scholar] [CrossRef] [PubMed]
- Raeburn, J.; Adams, D.J. Multicomponent low molecular weight gelators. Chem. Commun. 2015, 51, 5170–5180. [Google Scholar] [CrossRef] [PubMed]
- Ohsedo, Y.; Watanabe, H.; Oono, M.; Tanaka, A. Improved Mechanical Properties of Alkylamide Organogels via a Mixing Enhancement Effect. RSC Adv. 2013, 3, 5803–5806. [Google Scholar] [CrossRef]
- Ohsedo, Y.; Oono, M.; Tanaka, A.; Watanabe, H. Mixing induced thixotropy of a two-component system of alkylurea organogelators having different Alkyl chains. New J. Chem. 2013, 37, 2250–2253. [Google Scholar]
- Ohsedo, Y.; Oono, M.; Saruhashi, K.; Watanabe, H. Onset of Mixing-Induced Thixotropy in Hydrogels by Mixing Two Homologues of Low-Molecular-Weight Hydrogelators. RSC Adv. 2014, 4, 43560–43563. [Google Scholar] [CrossRef]
- Ohsedo, Y.; Oono, M.; Saruhashi, K.; Watanabe, H.; Miyamoto, N. A New Composite Thixotropic Hydrogel Composed of a Low-Molecular-Weight Hydrogelator and a Nanosheet. RSC Adv. 2014, 4, 44837–44840. [Google Scholar] [CrossRef]
- Ohsedo, Y.; Taniguchi, T.; Oono, M.; Saruhashi, K.; Watanabe, H. Creation of Thixotropic Multicomponent Alkylamide Organogels Containing Non-volatile Oil as Potential Drug Release Host Materials. RSC Adv. 2014, 4, 35484–35488. [Google Scholar] [CrossRef]
- MSDS for n-octanamide 629-01-6, TCI AMERICA website. Available online: http://www.tcichemicals.com/eshop/en/us/commodity/O0238/ (accessed on 29 February 2016).
- Ohsedo, Y.; Taniguchi, T.; Oono, M.; Saruhashi, K.; Watanabe, H. Long-Chain Alkylamide-Derived Oil Gels: Mixing Induced Onset of Thixotropy and Application in Sustained Drug Release. New J. Chem. 2015, 39, 6482–6490. [Google Scholar] [CrossRef]



© 2016 by the author; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ohsedo, Y. Low-Molecular-Weight Gelators as Base Materials for Ointments. Gels 2016, 2, 13. https://doi.org/10.3390/gels2020013
Ohsedo Y. Low-Molecular-Weight Gelators as Base Materials for Ointments. Gels. 2016; 2(2):13. https://doi.org/10.3390/gels2020013
Chicago/Turabian StyleOhsedo, Yutaka. 2016. "Low-Molecular-Weight Gelators as Base Materials for Ointments" Gels 2, no. 2: 13. https://doi.org/10.3390/gels2020013




