Host-Specialist Dominated Ectomycorrhizal Communities of Pinus cembra are not Affected by Temperature Manipulation
Abstract
:1. Introduction
2. Material and Methods
2.1. Site Descriptions
2.2. Experimental Design
2.3. Soil Parameters
2.4. Soil Fungal Biomass
2.5. In-Growth Mesh Bags
2.6. Hyphal Length in Mesh Bags
2.7. Sampling and Processing of Ectomycorrhizae
2.8. Molecular Identification of Ectomycorrhiza
2.9. Ectomycorrhiza Inoculum Potential of the Soil
2.10. Sequence Analysis
2.11. Data Handling and Statistical Analysis
3. Results
3.1. Soil Temperature and Soil Moisture
3.2. Soil pH
3.3. Soil Organic Matter
| Measured Variable | W | K | CO | CM | N |
|---|---|---|---|---|---|
| May | |||||
| SOM 1 | 28.1 ± 6.1 a | 28.7 ± 6.7 a | 27.5 ± 7 a | 29.5 ± 4.5 a | 26.9 ± 2.2 a |
| SWC 2 | 43.6 ± 3.2 a | 47.0 ± 7.0 a, b | 50.5 ± 3.2 b | 51.8 ± 2.2 b | 48.3 ± 3.7 a, b |
| pH | 3.8 ± 0.1 a | 3.9 ± 0.2 a | 3.9 ± 0.2 a | 4.2 ± 0.2 b | 4.2 ± 0.0 b |
| October | |||||
| SOM 1 | 26.5 ± 3.4 a | 25.5 ± 5.0 a | 25.8 ± 4.8 a | 27.2 ± 4. 2 a | 22.1 ± 1.9 a |
| SWC 2 | 37.5 ± 5.1 a | 37.4 ± 7.3 a | 42.3 ± 4.3 a, b | 43.9 ± 6.4 a, b | 46.6 ± 9.6 b |
| pH | 3.9 ± 0.1 a | 4.0 ± 0.1 a | 4.0 ± 0.1 a | 4.0 ± 0.1 a | 4.0 ± 0.0 a |
3.4. Root Dry Weight
3.5. Soil Fungal Biomass
3.6. Hyphal Length for Estimation of Fungal Biomass in Mesh Bags
3.7. Ectomycorrhiza Root Tips and Their Degree of Mycorrhization
| Measured Variable | W | K | CO | CM | N |
|---|---|---|---|---|---|
| May | |||||
| Root dry weight 1 | 1.1 ± 1.8 a | 0.5 ± 0.4 a | 1.3 ± 1.6 a | 0.3 ± 0.3 a | 0.7 ± 0.5 a |
| EM root tips | 1828 ± 1919 | 1590 ± 1192 | 1374 ± 1209 | 797 ± 613 | 1440 ± 1493 |
| Mycorrhization 2 | 99.8 ± 0.5 a | 100 ± 0.1 a | 99.4 ± 1.8 a | 99.6 ± 0.9 a | 99.3 ± 0.9 a |
| Ergosterol 3 | 51.6 ± 16.3 a | 45.8 ± 18.1 a | 42.2 ± 11.1 a | 51.3 ± 18.3 a | 47.4 ± 8.4 a |
| October | |||||
| Root dry weight 1 | 1.2 ± 1.6 a | 0.8 ± 0.6 a | 1.0 ± 0.9 a | 0.8 ± 0.5 a | 0.7 ± 0.7 a |
| EM root tips | 2476 ± 1545 | 2000 ± 1282 | 2988 ± 4338 | 2006 ± 1037 | 1846 ± 1701 |
| Mycorrhization 2 | 99.6 ± 0.6 a | 99.0 ± 0.9 a | 99.0 ± 1.3 a | 99.6 ± 0.5 a | 98.2 ± 1.8 a |
| Ergosterol 3 | 34.7 ± 9. 4 a | 39.5 ± 10.3 a | 35.6 ± 13.9 a | 43.4 ± 10.5 a | 19.0 ± 4.3 b |
| Hyphal length | 2.3 ± 1.2 | 3.9 ± 2.2 | 9.8 ± 5.5 | 9.9 ± 7. 8 | 1.6 ± 1.2 |
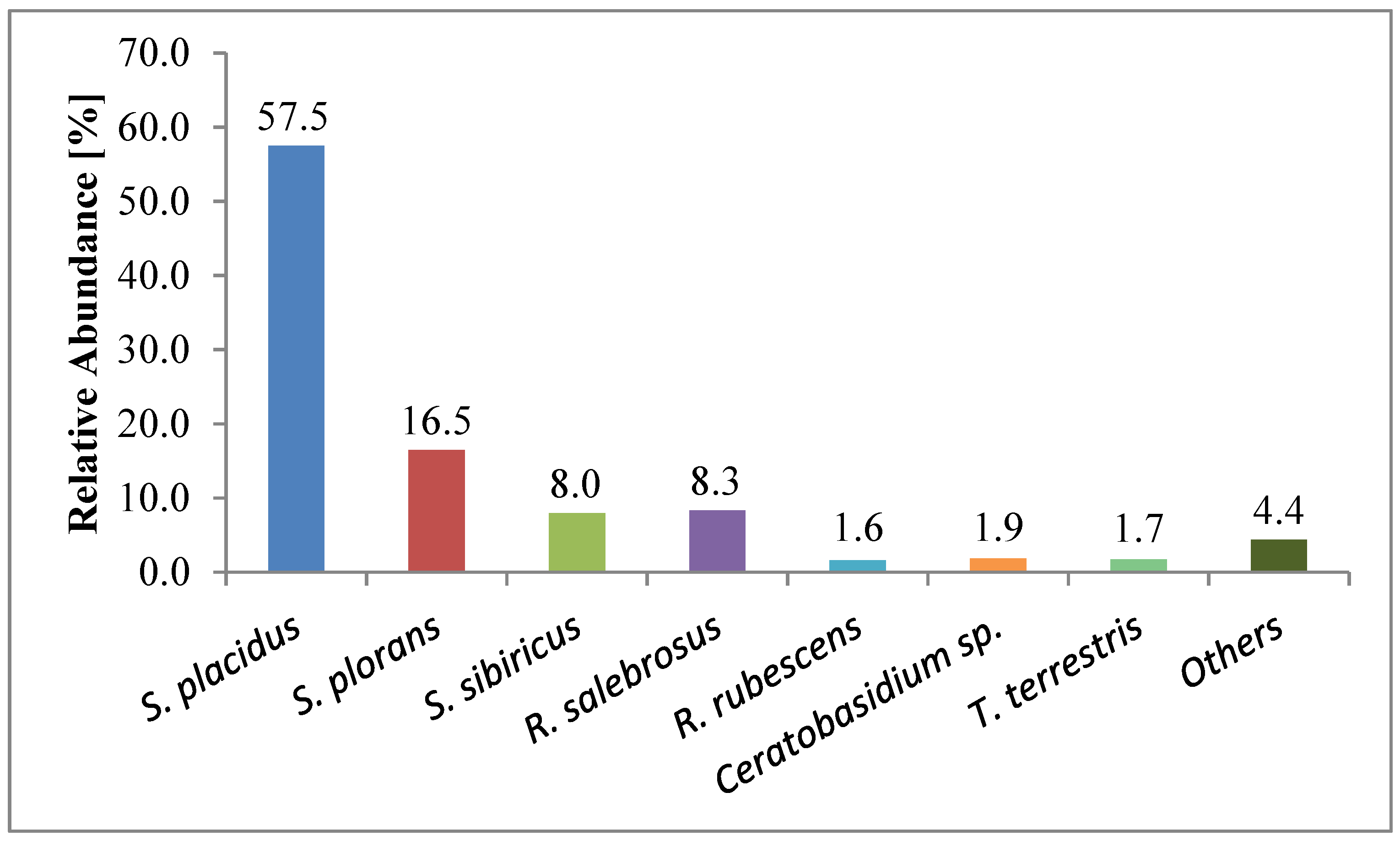
3.8. Ectomycorrhiza Communities of P. cembra
| Closest BLAST Match | Root Tips | Mesh Bags | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| W | K | CO | CM | N | W | K | CO | CM | N | |
| Suillus placidus | X | X | X | X | X | - | - | - | - | - |
| Rhizopogon salebrosus | X | X | X | X | X | X | X | X | - | X |
| Suillus plorans | X | X | X | X | X | X | X | X | X | X |
| Thelephora terrestris | - | X | X | X | - | - | X | X | X | - |
| Suillus sibiricus | X | - | X | X | X | - | - | X | - | - |
| Ceratobasidium sp. | X | - | X | - | X | - | - | - | - | - |
| Phialocephala fortinii 1 | - | - | X | X | X | - | - | - | - | - |
| Amphinema byssoides | - | - | X | X | - | - | - | - | X | - |
| Cortinarius anomalus | X | - | X | - | - | - | - | - | - | - |
| Xerocomus ferrugineus | X | - | - | - | - | - | - | - | - | - |
| Lactarius deterrimus | - | - | - | - | X | - | - | - | - | - |
| Wilcoxina sp. | - | - | - | X | - | - | - | - | - | - |
| Articulospora tetracladia 1 | X | - | - | X | X | - | - | - | - | - |
| Phialocephala fortinii 2 | - | - | - | - | X | - | - | - | - | - |
| Helotiales sp. 4 | - | - | X | - | - | - | - | - | - | - |
| Rhizopogon rubescens | - | - | X | - | - | - | - | - | - | - |
| Articulospora tetracladia 2 | - | X | - | - | X | - | - | - | - | - |
| Helotiales sp. 1 | X | - | - | - | - | - | - | - | - | - |
| Lactarius rufus | - | - | X | - | - | - | - | - | - | - |
| Meliniomyces bicolor | - | - | - | - | - | - | - | - | - | X |
| Phialocephala fortinii 3 | - | - | - | - | X | - | - | - | - | - |
| Helotiales sp. 3 | - | - | - | - | - | X | - | - | - | - |
| Rhizoscyphus ericae | - | - | - | - | - | X | - | - | - | - |
| Sistotrema sp. | - | - | - | - | - | - | - | - | X | - |
| Sebacinales sp. | - | - | - | - | - | - | - | - | X | - |
| Suillus sp. | - | - | - | - | - | - | - | - | X | - |
| Sebacina sp. | - | - | - | - | - | - | - | X | - | - |
| Glomeromycetes sp. | - | - | - | - | - | - | - | - | - | X |
| Phialocephala fortinii 3 | - | - | - | - | X | - | - | - | - | - |
| Plot | S (n = 4) | E (n = 4) | H (n = 4) | D’ (n = 4) | Indicator Species |
|---|---|---|---|---|---|
| W | 4.75 ± 1.89 | 0.60 ± 0.17 | 0.85 ± 0.37 | 0.45 ± 0.18 | - |
| K | 3.25 ± 0.50 | 0.61 ± 0.32 | 0.71 ± 0.34 | 0.41 ± 0.22 | - |
| CO | 5.50 ± 1.73 | 0.61 ± 0.04 | 1.02 ± 0.15 | 0.53 ± 0.04 | - |
| CM | 5.00 ± 1.41 | 0.56 ± 0.15 | 0.90 ± 0.33 | 0.45 ± 0.18 | - |
| N | 6.25 ± 1.50 | 0.70 ± 0.14 | 1.25 ± 0.13 | 0.63 ± 0.08 | S. sibiricus (p < 0.03) |
| Season | 4.95 ± 1.67 | 0.62 ± 0.17 | 0.95 ± 0.31 | 0.50 ± 0.16 | - |

3.9. The Ectomycorrhiza Inoculum Potential
3.10. Effect of Soil Temperature Manipulation
3.11. Effect of Nitrogen Fertilization
| Plot | CM | N |
|---|---|---|
| May | ||
| C tot 1 | 16.1 ± 3.3 a | 14.8 ± 1.1 a |
| N tot 2 | 0.82 ± 0.13 a | 0.85 ± 0.06 a |
| C:N | 19.6 ± 1.4 a | 17.4 ± 1.2 b |
| SOM | 29.5 ± 4.5 a | 26.9 ± 2.2 a |
| October | ||
| C tot 1 | 13.4 ± 2.7 a | 10.5 ± 0.6 b |
| N tot 2 | 0.70 ± 0.11 a | 0.64 ± 0.04 a |
| C:N | 19.1 ± 2.0 a | 16.3 ± 0.6 b |
| SOM 3 | 27.2 ± 4.2 a | 22.1 ±1.9 b |
4. Discussion
4.1. The mycobionts of P. cembra in Haggen
4.2. The Inoculum of Ectomycorrhizae in the Soil
4.3. Suitability and Sustainability of Ectomycorrhiza Partners of P. cembra
4.4. Beneficial Properties of Suillus Host Specialists
4.5. Influence of Soil Temperature Manipulation on P. cembra Ectomycorrhiza
4.6. The Effect of Nitrogen Fertilization on P. cembra Ectomycorrhiza
5. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Casalegno, S.; Amatulli, G.; Camia, A.; Nelson, A.; Pekkarinen, A. Vulnerability of Pinus cembra L. In the Alps and the Carpathian mountains under present and future climates. For. Ecol Manag. 2010, 259, 750–761. [Google Scholar]
- Ulber, M.; Gugerli, F.; Bozic, G. Swiss stone pine (Pinus cembra). In Technical Guidelines for Genetic Conservation and Use; European Forest Genetic Resources Programme (EUFORGEN): Rome, Italy, 2004. [Google Scholar]
- Apetrei, C.L.; Tuchilus, C.; Aprotosoaie, A.C.; Oprea, A.; Malterud, K.E.; Miron, A. Chemical, antioxidant and antimicrobial investigations of Pinus cembra L. Bark and needles. Molecules 2011, 16, 7773–7788. [Google Scholar] [CrossRef] [PubMed]
- Rossi, S.; Deslauriers, A.; Anfodillo, T.; Carraro, V. Evidence of threshold temperatures for xylogenesis in conifers at high altitudes. Oecologia 2007, 152, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Oberhuber, W. Influence of climate on radial growth of Pinus cembra within the alpine timberline ecotone. Tree Physiol. 2004, 24, 291–301. [Google Scholar] [CrossRef] [PubMed]
- Carrer, M.; Soraruf, L.; Lingua, E. Convergent space-time tree regeneration patterns along an elevation gradient at high altitude in the Alps. For. Ecol. Manag. 2013, 304, 1–9. [Google Scholar] [CrossRef]
- Keller, G. Mykorrhiza-Untersuchungen bei der Zirbe (Pinus cembra L.) unter besonderer Berücksichtigung chromatographischer Merkmale (in German). Cent. Forstwes. 1989, 106, 129–147. [Google Scholar]
- Bacher, M.; Zoell, M.; Peintner, U. Ectomycorrhizal status of Larix decidua-, Picea abies- and Pinus cembra-nursery plants in South Tyrol. For. Obs. 2010, 5, 3–30. [Google Scholar]
- Simard, S.W.; Beiler, K.J.; Bingham, M.A.; Deslippe, J.R.; Philip, L.J.; Teste, F.P. Mycorrhizal networks: Mechanisms, ecology and modelling. Fungal Biol. Rev. 2012, 26, 39–60. [Google Scholar] [CrossRef]
- Moser, M. Die künstliche Mykorrhizaimpfung an Forstpflanzen (in German). Forstwirtsch. Cent. 1958, 77, 32–40. [Google Scholar] [CrossRef]
- Heumader, J. Cultivation of cembran pine plants for high-elevation afforestations. In Proceedings of the International Workshop on Subalpine Stone Pines and Their Environment: The Status of Our Knowledge, St. Moritz, Switzerland, 5–11 September 1992.
- Kranabetter, J. Ectomycorrhizal community effects on hybrid spruce seedling growth and nutrition in clearcuts. Can. J. Bot. 2004, 82, 983–991. [Google Scholar] [CrossRef]
- Selosse, M.-A.; Baudoin, E.; Vandenkoornhuyse, P. Symbiotic microorganisms, a key for ecological success and protection of plants. Comptes Rendus Biol. 2004, 327, 639–648. [Google Scholar] [CrossRef]
- Gassner, M. Beiträge zur Siedlungs- und Wirtschaftsgeschichte des inneren Sellraintales (in German). Veröff. Mus. Ferd. 1925, 4, 79. [Google Scholar]
- Leys, E.; Neuwinger, I.; Heumader, J.; Stolz, W. Bericht über Versuchsflächen bei Hochlagen (in German). Cent. Gesamte Forstwes. 1970, 87, 117–124. [Google Scholar]
- Neuwinger, I.; Czell, A. Standortuntersuchungen in subalpinen Aufforstungsgebieten. 1. Teil: Böden in den tiroler Zentralalpen (in German). Sonderdr. Forstw. Cbl. 1959, 78, 327–372. [Google Scholar] [CrossRef]
- Schiechtl, H.M. Zur Frage der Wiederaufforstung von Sonnenhängen in den Hochlagen der Innenalpen (in German). Allg. Forstztg. 1970, 81, 312–314. [Google Scholar]
- Neuwinger, I. Standortuntersuchungen am sonnberg im sellrainer obertal, tirol (in German); Forstlichen Bundesversuchsanstalt: Vienna, Austria, 1972. Available online: http://www.zobodat.at/pdf/Mitteilungen-forstlichen-Bundes-Versuchsanstalt_96_1972_0177-0207.pdf (accessed on 30 April 2015).
- Göbl, F. Die Zirbenmykorrhiza in Pflanzgärten. Cent. Gesamte Forstwes. 1963, 80, 20–30. [Google Scholar]
- Göbl, F.; Ladurner, H. Mykorrhizen und Pilze der Hochlagenaufforstung Haggen (in German); Forstlichen Bundesversuchsanstalt: Vienna, Austria, 2000. Available online: https://bfw.ac.at/publ/neu/mitteilungen173/kapitel1-3.pdf (accessed on 30 April 2015).
- Ohga, S.; Wood, D.A. Efficacy of ectomycorrhizal basidiomycetes on Japanese larch seedlings assessed by ergosterol assay. Mycologia 2000, 92, 394–398. [Google Scholar] [CrossRef]
- Piola, F.; Rohr, R.; von Aderkas, P. Controlled mycorrhizal initiation as a means to improve root development in somatic embryo plantlets of hybrid larch (Larix x eurolepis). Physiol. Plant. 1995, 95, 575–580. [Google Scholar] [CrossRef]
- Visser, S. Ectomycorrhizal fungal succession in jack pine stands following wildfire. New Phytol. 1995, 129, 389–401. [Google Scholar] [CrossRef]
- Rudawska, M.; Leski, T.; Trocha, L.K.; Gornowicz, R. Ectomycorrhizal status of norway spruce seedlings from bare-root forest nurseries. For. Ecol. Manag. 2006, 236, 375–384. [Google Scholar] [CrossRef]
- Keller, G. Mykosoziologische Studie über die Mykorrhizapilze der Zirbe. Artenspektrum und Sukzession in der hoch-subalpinen Stufe der tiroler Zentralalpen (in German). Ber. Forst. Bundes-Vers. 1997, 98, 80. [Google Scholar]
- Gardes, M.; Bruns, T.D. Community structure of ectomycorrhizal fungi in a Pinus muricata forest: Above- and below-ground views. Can. J. Bot. 1996, 74, 1572–1583. [Google Scholar] [CrossRef]
- ICCP. Summary for Policymarkers. In Climate Change 2007 Impacts, Adaptation and Vulnerability. Contribution of Working Group II to the Forth Assessment Report of the Intergovernmental Panel on Climate Change; Parry, M.L., Canziani, O.F., Palutikof, J.P., van der Linden, P.J., Hanson, C.E., Eds.; Cambridge University Press: Cambridge, UK, 2007; pp. 7–22. [Google Scholar]
- Kuhnert, R.; Oberkofler, I.; Peintner, U. Fungal growth and biomass development is boosted by plants in snow-covered soil. Microb. Ecol. 2012, 64, 79–90. [Google Scholar] [CrossRef] [PubMed]
- Wallander, H.; Nilsson, L.O.; Hagerberg, D.; Baath, E. Estimation of the biomass and seasonal growth of external mycelium of ectomycorrhizal fungi in the field. New Phytol. 2001, 151, 753–760. [Google Scholar] [CrossRef]
- Kilian, W.; Mueller, F. Neue Wuchsgebietsgliederung—Folgen fuer Herkunftsbezeichnung forstlichen Vermehrungsgutes (in German). Oesterreichische Forstztg. 1994, 4, 13–15. [Google Scholar]
- Schlichtling, E.; Stahr, K. Bodenkundliches Praktikum; Blackwall Wissenschafts-Verlag Berlin, Wien: Berlin, Germany, 1995. [Google Scholar]
- Spath, M.; Insam, H.; Peintner, U.; Kelderer, M.; Kuhnert, R.; Franke-Whittle, I.H. Linking soil biotic and abiotic factors to apple replant disease: A greenhouse approach. J. Phytopathol. 2014, 163, 287–299. [Google Scholar] [CrossRef]
- Hanssen, J.F.; Thingstad, T.F.; Goksøyr, J. Evaluation of hyphal lengths and fungal biomass in soil by a membrane filter technique. Oikos 1974, 25, 102–107. [Google Scholar] [CrossRef]
- Pfabel, C.; Eckhardt, K.U.; Baum, C.; Struck, C.; Frey, P.; Weih, M. Impact of ectomycorrhizal colonization and rust infection on the secondary metabolism of poplar (Populus trichocarpa x deltoides). Tree Physiol. 2012, 32, 1357–1364. [Google Scholar] [CrossRef] [PubMed]
- Agerer, R. Colour Atlas of Ectomycorrhizae; Einhorn-Verlag: Schwäbisch Gmünd, Germany, 1987–2002; Volume 1–12. [Google Scholar]
- Agerer, R. Exploration types of ectomycorrhizae—A proposal to classify ectomycorrhizal mycelial systems according to their patterns of differentiation and putative ecological importance. Mycorrhiza 2001, 11, 107–114. [Google Scholar] [CrossRef]
- Iotti, M.; Zambonelli, A. A quick and precise technique for identifying ectomycorrhizas by PCR. Mycol. Res. 2006, 110, 60–65. [Google Scholar] [CrossRef] [PubMed]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and Direct Sequencing of Fungal Ribosomal RNA Genes for Phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Michael, A.I., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press, Inc.: New York, NY, USA, 1990; pp. 315–322. [Google Scholar]
- Gardes, M.; Bruns, T.D. ITS primers with enhanced specificity for Basidiomycetes—Application to the identification of mycorrhizae and rusts. Mol. Ecol. 1993, 2, 113–118. [Google Scholar] [CrossRef] [PubMed]
- NCBI—Standard Nucleotide BLAST. Available online: http://blast.ncbi.nlm.nih.gov/Blast.cgi?PROGRAM=blastn&PAGE_TYPE=BlastSearch&LINK_LOC=blasthome (accessed on 3 March 2015).
- UNITE—Unified System for the DNA Based Fungal Species Linked to the Classification. Available online: https://unite.ut.ee/index.php (accessed on 3 March 2015).
- Koljalg, U.; Larsson, K.-H.; Abarenkov, K.; Nilsson, R.H.; Alexander, I.J.; Eberhardt, U.; Erland, S.; Hoiland, K.; Kjoller, R.; Larsson, E.; et al. UNITE: A database providing web-based methods for the molecular identification of ectomycorrhizal fungi. New Phytol. 2005, 166, 1063–1068. [Google Scholar] [CrossRef] [PubMed]
- McCune, B.; Mefford, M.J. PC-ORD. Multivariate Analysis of Ecological Data. Version 6; MjM Software: Gleneden Beach, OR, USA, 2011. [Google Scholar]
- Schmid, V.; Göbl, F.; Peintner, U. ITS primers for the identification of three Suillus spp. Specific to Pinus cembra. In Proceedings of the Abstracts of the 5th International Conference on Mycorrhiza, Granada, Spain, 23–27 July 2006; p. 143.
- Krpata, D.; Muhlmann, O.; Kuhnert, R.; Ladurner, H.; Gobl, F.; Peintner, U. High diversity of ectomycorrhizal fungi associated with Arctostaphylos uva-ursi in subalpine and alpine zones: Potential inoculum for afforestation. For. Ecol. Manag. 2007, 250, 167–175. [Google Scholar] [CrossRef]
- Peter, M.; Ayer, F.; Egli, S.; Honegger, R. Above- and below-ground community structure of ectomycorrhizal fungi in three norway spruce (Picea abies) stands in Switzerland. Can. J. Bot. 2001, 79, 1134–1151. [Google Scholar] [CrossRef]
- Bacher, M. Ektomykorrhizapartner der europäischen Lärche (Larix decidua) und der Zirbe (Pinus cembra) in Südtirols Hochlagen und Veränderung des Mykorrhizabesatzes von Setzlingen (in German). Dissertation, University of Innsbruck, 12 April 2010. [Google Scholar]
- Mohatt, K.R.; Cripps, C.L.; Lavin, M. Ectomycorrhizal fungi of whitebark pine (a tree in peril) revealed by sporocarps and molecular analysis of mycorrhizae from treeline forests in the Greater Yellowstone Ecosystem. Bot. Bot. 2008, 86, 14–25. [Google Scholar] [CrossRef]
- Kipfer, T.; Egli, S.; Ghazoul, J.; Moser, B.; Wohlgemuth, T. Susceptibility of ectomycorrhizal fungi to soil heating. Fungal Biol. 2010, 114, 467–472. [Google Scholar] [CrossRef] [PubMed]
- Hasselquist, N.; Germino, M.J.; McGonigle, T.; Smith, W.K. Variability of Cenococcum colonization and its ecophysiological significance for young conifers at alpine-treeline. New Phytol. 2005, 165, 867–873. [Google Scholar] [CrossRef] [PubMed]
- Collier, F.A.; Bidartondo, M.I. Waiting for fungi: The ectomycorrhizal invasion of lowland heathlands. J. Ecol. 2009, 97, 950–963. [Google Scholar] [CrossRef] [Green Version]
- Ashkannejhad, S.; Horton, T.R. Ectomycorrhizal ecology under primary succession on coastal sand dunes: Interactions involving Pinus contorta, suilloid fungi and deer. New Phytol. 2006, 169, 345–354. [Google Scholar] [CrossRef] [PubMed]
- Moser, M. Die Bedeutung der Mykorrhiza bei Aufforstungen unter besonderer Berücksichtigung von Hochlagen (in German). In Mykorrhiza. Internat. Symposium; Gustav Fischer Verlag: Jena, Germany, 1960; pp. 407–424. [Google Scholar]
- Mejstrik, V.K.; Hadac, E. Mycorrhizas of Arctostaphylos uva-ursi. Pedobiologia 1975, 15, 336–342. [Google Scholar]
- Kretzer, A.; Li, Y.; Szaro, T.; Bruns, T.D. Internal transribed spacer sequencs from 38 recognized species of Suillus sensu lato: Phylogentic and taxonomic implications. Mycologia 1996, 88, 776–785. [Google Scholar] [CrossRef]
- Wu, Q.X.; Mueller, G.M.; Lutzoni, F.M.; Huang, Y.Q.; Guo, S.Y. Phylogenetic and biogeographic relationships of Eastern Asian and Eastern North American disjunct Suillus species (fungi) as inferred from nuclear ribosomal RNA ITS sequences. Mol. Phylogenet. Evol. 2000, 17, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Keller, G. Utilization of inorganic and organic nitrogen sources by high-subalpine ectomycorrhizal fungi of Pinus cembra in pure culture. Mycol. Res. 1996, 100, 989–998. [Google Scholar] [CrossRef]
- Pennanen, T.; Heiskanen, J.; Korkama, T. Dynamics of ectomycorrhizal fungi and growth of norway spruce seedlings after planting on a mounded forest clearcut. For. Ecol. Manag. 2005, 213, 243–252. [Google Scholar] [CrossRef]
- Chapman, W.K.; Paul, L. Evidence that northern pioneering pines with tuberculate mycorrhizae are unaffected by varying soil nitrogen levels. Microb. Ecol. 2012, 64, 964–972. [Google Scholar] [CrossRef] [PubMed]
- Paul, L.R.; Chapman, B.K.; Chanway, C.P. Nitrogen Fixation Associated with Suillus tomentosus Tuberculate Ectomycorrhizae on Pinus contorta var. latifolia. Ann. Bot. 2007, 99, 1101–1109. [Google Scholar] [CrossRef]
- Schindlbacher, A.; Rodler, A.; Kuffner, M.; Kitzler, B.; Sessitsch, A.; Zechmeister-Boltenstern, S. Experimental warming effects on the microbial community of a temperate mountain forest soil. Soil Biol. Biochem. 2011, 43, 1417–1425. [Google Scholar] [CrossRef] [PubMed]
- Zentralanstalt für Meteorologie und Geodynamik (ZAMG) (in German). Available online: http://www.zamg.ac.at/ (accessed on 3 March 2015).
- Högberg, M.N.; Yarwood, S.A.; Myrold, D.D. Fungal but not bacterial soil communities recover after termination of decadal nitrogen additions to boreal forest. Soil Biol. Biochem. 2014, 72, 35–43. [Google Scholar] [CrossRef]
- Parrent, J.L.; Vilgalys, R. Biomass and compositional responses of ectomycorrhizal fungal hyphae to elevated CO2 and nitrogen fertilization. New Phytol. 2007, 176, 164–174. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, L.O.; Wallander, H. Production of external mycelium by ectomycorrhizal fungi in a norway spruce forest was reduced in response to nitrogen fertilization. New Phytol. 2003, 158, 409–416. [Google Scholar] [CrossRef]
- Clemmensen, K.E.; Bahr, A.; Ovaskainen, O.; Dahlberg, A.; Ekblad, A.; Wallander, H.; Stenlid, J.; Finlay, R.D.; Wardle, D.A.; Lindahl, B.D. Roots and associated fungi drive long-term carbon sequestration in boreal forest. Science 2013, 339, 1615–1618. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors. Lcensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rainer, G.; Kuhnert, R.; Unterholzer, M.; Dresch, P.; Gruber, A.; Peintner, U. Host-Specialist Dominated Ectomycorrhizal Communities of Pinus cembra are not Affected by Temperature Manipulation. J. Fungi 2015, 1, 55-75. https://doi.org/10.3390/jof1010055
Rainer G, Kuhnert R, Unterholzer M, Dresch P, Gruber A, Peintner U. Host-Specialist Dominated Ectomycorrhizal Communities of Pinus cembra are not Affected by Temperature Manipulation. Journal of Fungi. 2015; 1(1):55-75. https://doi.org/10.3390/jof1010055
Chicago/Turabian StyleRainer, Georg, Regina Kuhnert, Mara Unterholzer, Philipp Dresch, Andreas Gruber, and Ursula Peintner. 2015. "Host-Specialist Dominated Ectomycorrhizal Communities of Pinus cembra are not Affected by Temperature Manipulation" Journal of Fungi 1, no. 1: 55-75. https://doi.org/10.3390/jof1010055
APA StyleRainer, G., Kuhnert, R., Unterholzer, M., Dresch, P., Gruber, A., & Peintner, U. (2015). Host-Specialist Dominated Ectomycorrhizal Communities of Pinus cembra are not Affected by Temperature Manipulation. Journal of Fungi, 1(1), 55-75. https://doi.org/10.3390/jof1010055






