Dynamic Experiments for Bioprocess Parameter Optimization with Extreme Halophilic Archaea
Abstract
:1. Introduction
1.1. Halophiles
1.2. Bioreactor Technology with Extreme Halophiles
1.3. Optimal Process Parameters for Extreme Halophilic Archaea
1.4. Dynamic Experiments for Process Parameter Optimization
1.5. Goal and Structure
2. Experimental Section
2.1. Strains and Cultivation Procedures
2.2. Defined Medium Composition
2.3. Bioreactor Setup, Cultivation Conditions
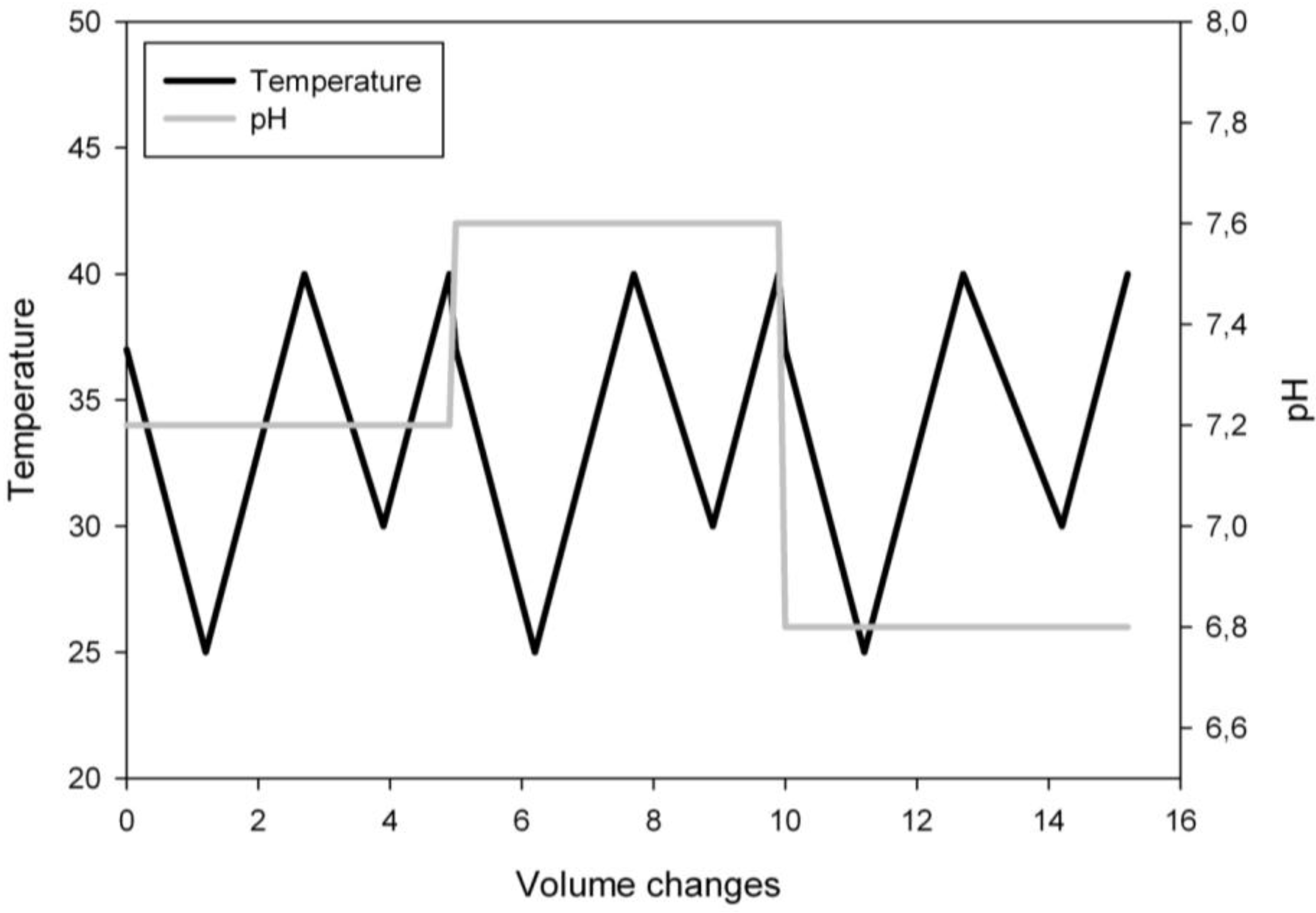
2.4. Experimental Design
2.5. Off-Line Analytics
2.6. Data Evaluation
3. Results and Discussion
3.1. Ramp Design
3.2. Analytics: The Necessity for Off-Line Data
3.3. Ramp Down
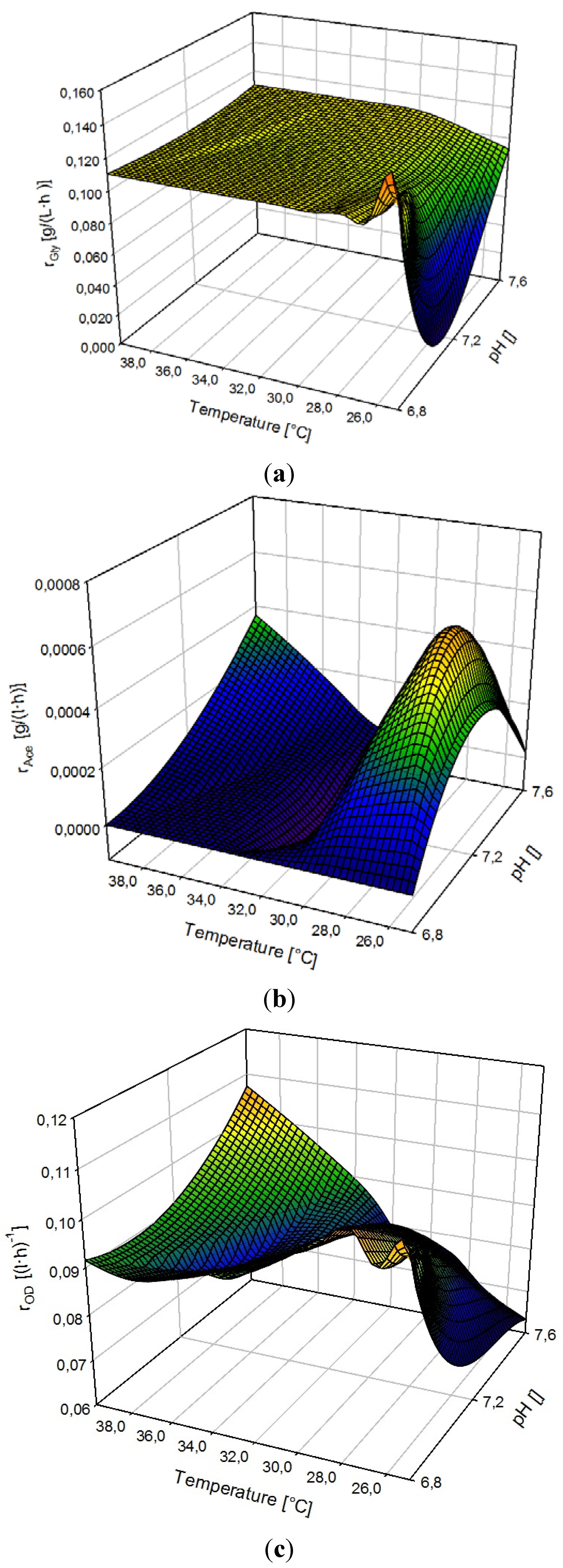
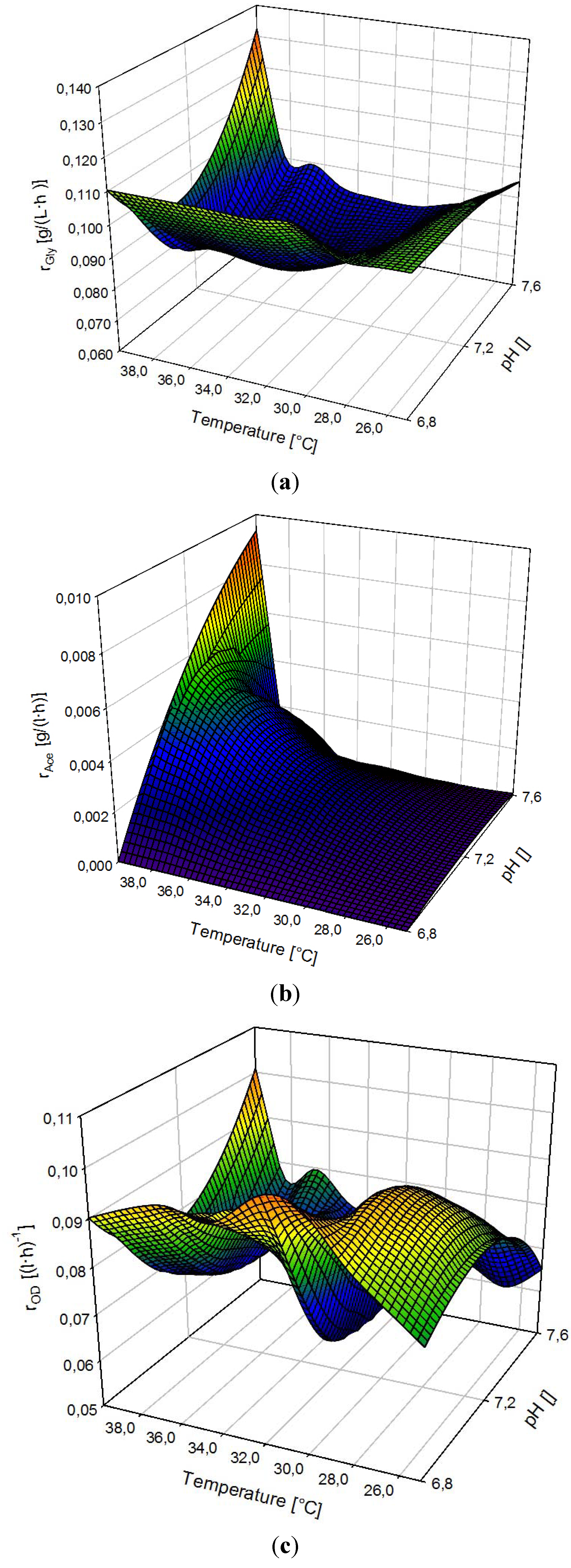
3.4. Ramp Up
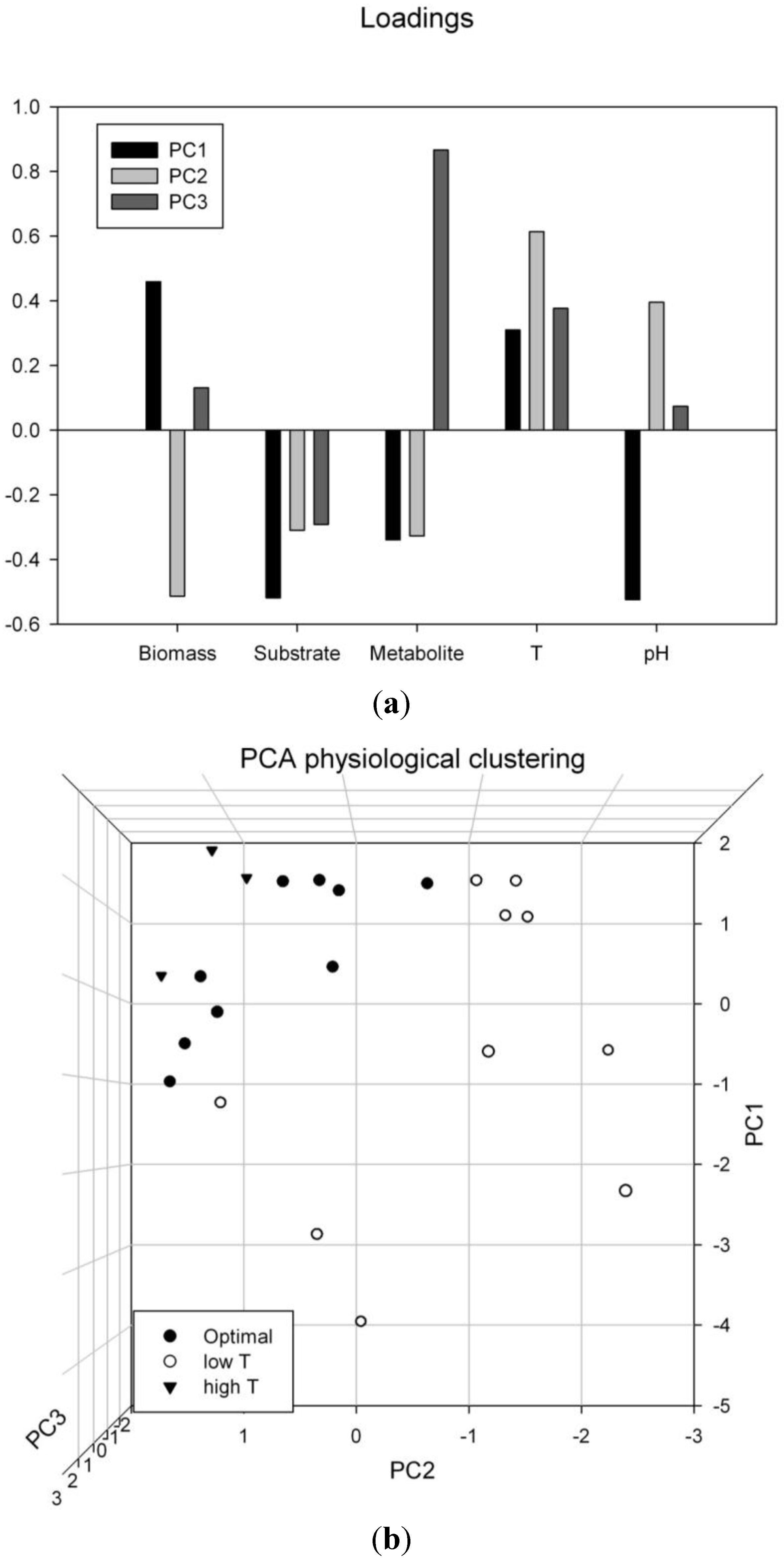
3.5. Multivariate Data Analysis: Supporting the Findings of Temperature and pH Optima
3.6. Verification of the Dynamic Experimental Results
| dynamic conditions | steady state | |||||
|---|---|---|---|---|---|---|
| Experiment | down 1 | down 2 | up 1 | up 2 | continuous culture | batch culture |
| T (°C) | 37–36 | 38–36 | 38–39 | 36–37 | 37 | 37 |
| pH | 7.2 | 7.2 | 7.2 | 7.2 | 7.2 | 7.2 |
| D = µ (h−1) | 0.030 ± 0.005 | 0.035 ± 0.003 | µmax | |||
| rGly (g/L/h) | 0.104 | 0.071 | 0.076 | 0.057 | 0.120 | 0.108 |
| rAce (g/L/h) | 0.003 | 0.008 | 0.005 | 0.004 | 0.002 | 0.002 |
| rOD (g/L/h) | 0.080 | 0.052 | 0.067 | 0.049 | 0.070 | 0.083 |
| qS (g/g/h) | 0.056 ± 0.005 | 0.050 ± 0.005 | 0.051 ± 0.005 | 0.053 ± 0.005 | 0.047 ± 0.005 | 0.054 ± 0.005 |
3.7. Data Repeatability and Reproducibility
4. Conclusions
Conflicts of Interest
References
- Oren, A. Industrial and environmental applications of halophilic microorganisms. Environ. Technol. 2010, 31, 825–834. [Google Scholar] [CrossRef]
- Zhuang, X.; Han, Z.; Bai, Z.; Zhuang, G.; Shim, H. Progress in decontamination by halophilic microorganisms in saline wastewater and soil. Environ. Pollut. 2010, 158, 1119–1126. [Google Scholar] [CrossRef]
- Pendashteh, A.R.; Fakhru’l-Razi, A.; Chuah, T.G.; Dayang Radiah, A.B.; Madaeni, S.S.; Zurina, Z.A. Biological treatment of produced water in a sequencing batch reactor by a consortium of isolated halophilic microorganisms. Environ. Technol. 2010, 31, 1229–1239. [Google Scholar] [CrossRef]
- Li, W.-G.; Ma, F.; Wei, L.; Zhang, D.-W.; Liu, F.; Wang, G.-Y.; Su, J.-F. Isolation and identification of moderately halophilic bacterium and enhanced treatment of hypersaline tanning wastewater by the bacterium. JSCUT 2008, 36, 89–94. [Google Scholar]
- Cyplik, P.; Czaczyk, K.; Piotrowska-Cyplik, A.; Marecik, R.; Grajek, W. Removal of nitrates from brine using Haloferax mediterranei Archeon. Environ. Prot. Eng. 2010, 36, 5–16. [Google Scholar]
- Rodriguez-Valera, F.; Garcia Lillo, J.A.; Anton, J.; Meseguer, I. Biopolymer Production by Haloferax mediterranei. In General and Applied Aspects of Halophilic Microorganisms; Springer: New York, NY, USA, 1991; pp. 373–380. [Google Scholar]
- Rodriguez-Saiz, M.; Sanchez-Porro, C.; Fuente, J.L.; Mellado, E.; Barredo, J.L. Engineering the halophilic bacterium Halomonas elongata to produce β-carotene. Appl. Microbiol. Biotechnol. 2007, 77, 637–643. [Google Scholar] [CrossRef]
- Oren, A. Molecular ecology of extremely halophilic Archaea and Bacteria. FEMS Microbiol. Ecol. 2002, 39, 1–7. [Google Scholar] [CrossRef]
- Fendrihan, S.; Legat, A.; Pfaffenhuemer, M.; Gruber, C.; Weidler, G.; Gerbl, F.; Stan-Lotter, H. Extremely halophilic archaea and the issue of long-term microbial survival. Rev. Environ. Sci. Biotechnol. 2006, 5, 203–218. [Google Scholar] [CrossRef]
- Kivistoe, A.T.; Karp, M.T. Halophilic anaerobic fermentative bacteria. J. Biotechnol. 2011, 152, 114–124. [Google Scholar] [CrossRef]
- Kapdan, I.K.; Erten, B. Anaerobic treatment of saline wastewater by Halanaerobium lacusrosei. Process Biochem. 2007, 42, 449–453. [Google Scholar] [CrossRef]
- Martínez-Espinosa, R.M.; Zafrilla, B.; Camacho, M.; Bonete, M.J. Nitrate and nitrite removal from salted water by Haloferax mediterranei. Biocatal. Biotransfor. 2007, 25, 295–300. [Google Scholar] [CrossRef]
- Oren, A. Uptake and turnover of acetate in hypersaline environments. FEMS Microbiol. Ecol. 1995, 18, 75–84. [Google Scholar] [CrossRef]
- Brasen, C.; Schonheit, P. Mechanisms of acetate formation and acetate activation in halophilic archaea. Arch. Microbiol. 2001, 175, 360–368. [Google Scholar] [CrossRef]
- Oren, A.; Gurevich, P. Diversity of lactate metabolism in halophilic archaea. Can. J. Microbiol. 1995, 41, 302–307. [Google Scholar] [CrossRef]
- Anton, J.; Meseguer, I.; Rodriguez-Valera, F. Production of an extracellular polysaccharide by Haloferax mediterranei. Appl. Environ. Microbiol. 1988, 54, 2381–2386. [Google Scholar]
- Oren, A. The role of glycerol in the nutrition of halophilic archaeal communities: A study of respiratory electron transport. FEMS Microbiol. Ecol. 1995, 16, 281–290. [Google Scholar] [CrossRef]
- Cuadros-Orellana, S.; Pohlschroeder, M.; Durrant, L.R. Isolation and characterization of halophilic archaea able to grow in aromatic compounds. Int. Biodeterior. Biodegrad. 2006, 57, 151–154. [Google Scholar] [CrossRef]
- Siebers, B.; Schönheit, P. Unusual pathways and enzymes of central carbohydrate metabolism in Archaea. Curr. Opin. Microbiol. 2005, 8, 695–705. [Google Scholar] [CrossRef]
- Sato, T.; Atomi, H. Novel metabolic pathways in Archaea. Curr. Opin. Microbiol. 2011, 14, 307–314. [Google Scholar] [CrossRef]
- Falb, M.; Müller, K.; Königsmaier, L.; Oberwinkler, T.; Horn, P.; von Gronau, S.; Gonzalez, O.; Pfeiffer, F.; Bornberg-Bauer, E.; Oesterhelt, D. Metabolism of halophilic archaea. Extremophiles 2008, 12, 177–196. [Google Scholar] [CrossRef]
- Hawkes, F.R.; Dinsdale, R.; Hawkes, D.L.; Hussy, I. Sustainable fermentative hydrogen production: Challenges for process optimisation. Int. J. Hydrogen Energ. 2002, 27, 1339–1347. [Google Scholar] [CrossRef]
- Herwig, C. Process analytical technology in biotechnology. Chem. Ing. Tech. 2010, 82, 405–414. [Google Scholar] [CrossRef]
- Sagmeister, P.; Wechselberger, P.; Herwig, C. Information processing: Rate-based investigation of cell physiological changes along design space development. PDA J. Pharm. Sci. Technol. 2012, 66, 526–541. [Google Scholar] [CrossRef]
- Rittmann, S.; Seifert, A.; Herwig, C. Quantitative analysis of media dilution rate effects on Methanothermobacter marburgensis grown in continuous culture on H2 and CO2. Biomass Bioenerg. 2012, 36, 293–301. [Google Scholar] [CrossRef]
- Lorantfy, B.; Seyer, B.; Herwig, C. Stoichiometric and kinetic analysis of extreme halophilic Archaea on various substrates in a corrosion resistant bioreactor. New Biotechnol. 2013, in press. [Google Scholar]
- Cyplik, P.; Grajek, W.; Marecik, R.; Króliczak, P.; Dembczyński, R. Application of a membrane bioreactor to denitrification of brine. Desalination 2007, 207, 134–143. [Google Scholar] [CrossRef]
- Fang, C.-J.; Ku, K.-L.; Lee, M.-H.; Su, N.-W. Influence of nutritive factors on C50 carotenoids production by Haloferax mediterranei ATCC 33500 with two-stage cultivation. Bioresour. Technol. 2010, 101, 6487–6493. [Google Scholar] [CrossRef]
- Bowers, K.J.; Wiegel, J. Temperature and pH optima of extremely halophilic archaea: A mini-review. Extremophiles 2011, 15, 119–128. [Google Scholar] [CrossRef]
- Robinson, J.L.; Pyzyna, B.; Atrasz, R.G.; Henderson, C.A.; Morrill, K.L; Burd, A.M.; DeSoucy, E.; Fogleman, R.E., III; Naylor, J.B.; Steele, S.M.; Elliott, D.R.; Leyva, K.J.; Shand, R.F. Growth kinetics of extremely halophilic Archaea (family Halobacteriaceae) as revealed by Arrhenius plots. J. Bacteriol. 2005, 187, 923–929. [Google Scholar] [CrossRef]
- Losen, M.; Froehlich, B.; Pohl, M.; Buechs, J. Effect of oxygen limitation and medium composition on Escherichia coli fermentation in shake-flask cultures. Biotechnol. Prog. 2004, 20, 1062–1068. [Google Scholar] [CrossRef]
- Vazquez, M.; Martin, A.M. Optimization of Phaffia rhodozyma continuous culture through response surface methodology. Biotechnol. Bioeng. 1998, 57, 314–320. [Google Scholar] [CrossRef]
- Mateles, R.I.; Battat, E. Continuous culture used for media optimization. Appl. Environ. Microbiol. 1974, 28, 901–905. [Google Scholar]
- Spadiut, O.; Rittmann, S.; Dietzsch, C.; Herwig, C. Dynamic process conditions in bioprocess development. Eng. Life Sci. 2013, 13, 88–101. [Google Scholar] [CrossRef]
- Shahhosseini, S. Simulation and optimisation of PHB production in fed-batch culture of Ralstonia eutropha. Process Biochem. 2004, 9, 936–969. [Google Scholar]
- Wechselberger, P.; Sagmeister, P.; Herwig, C. Model-based analysis on the extractability of information from data in dynamic fed-batch experiments. Biotechnol. Progr. 2013, 29, 285–296. [Google Scholar] [CrossRef]
- Dietzsch, C.; Spadiut, O.; Herwig, C. A dynamic method based on the specific substrate uptake rate to set up a feeding strategy for Pichia pastoris. Microbial. Cell Factories 2011, 10, 14. [Google Scholar] [CrossRef]
- Garcia, L.J.; Rodriguez-Valera, F. Effects of culture conditions on poly(beta-hydroxybutyric acid) production by Haloferax mediterranei. Appl. Environ. Microbiol. 1990, 56, 2517–2521. [Google Scholar]
- Oren, A.; Gurevich, P. Production of D-lactate, acetate, and pyruvate from glycerol in communities of halophilic archaea in the Dead Sea and in saltern crystallizer ponds. FEMS Microbiol. Ecol. 1994, 14, 147–155. [Google Scholar]
- Sonnleitner, B.; Kaeppeli, O. Growth of Saccharomyces cerevisiae is controlled by its limited respiratory capacity: Formulation and verification of a hypothesis. Biotechnol. Bioeng. 1986, 28, 927–937. [Google Scholar] [CrossRef]
- Wechselberger, P.; Herwig, C. Model-based analysis on the relationship of signal quality to real-time extraction of information in bioprocesses. Biotechnol. Prog. 2012, 28, 265–275. [Google Scholar] [CrossRef]
- Romesburg, H.C. Cluster Analysis for Researchers; Lulu Press: Raleigh, NC, USA, 2004. [Google Scholar]
- Lorantfy, B.; Jazini, M.; Herwig, C. Investigation of the physiological response to oxygen limited process conditions of Pichia pastoris Mut+ strain using a two-compartment scale-down system. J. Biosci. Bioeng. 2013, 116, 371–379. [Google Scholar] [CrossRef]
- Ren, H.; Yuan, J. Model-based specific growth rate control for Pichia pastoris to improve recombinant protein production. J. Chem. Technol. Biot. 2005, 80, 1268–1272. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lorantfy, B.; Seyer, B.; Herwig, C. Dynamic Experiments for Bioprocess Parameter Optimization with Extreme Halophilic Archaea. Bioengineering 2014, 1, 1-17. https://doi.org/10.3390/bioengineering1010001
Lorantfy B, Seyer B, Herwig C. Dynamic Experiments for Bioprocess Parameter Optimization with Extreme Halophilic Archaea. Bioengineering. 2014; 1(1):1-17. https://doi.org/10.3390/bioengineering1010001
Chicago/Turabian StyleLorantfy, Bettina, Bernhard Seyer, and Christoph Herwig. 2014. "Dynamic Experiments for Bioprocess Parameter Optimization with Extreme Halophilic Archaea" Bioengineering 1, no. 1: 1-17. https://doi.org/10.3390/bioengineering1010001





