High-Temperature Wide Thermal Hysteresis of an Iron(II) Dinuclear Double Helicate
Abstract
:1. Introduction
2. Results and Discussion
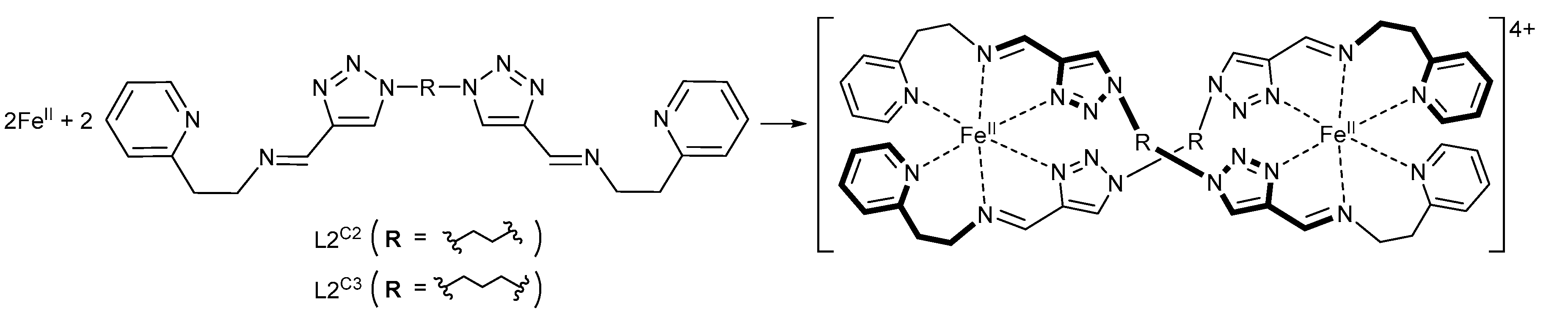
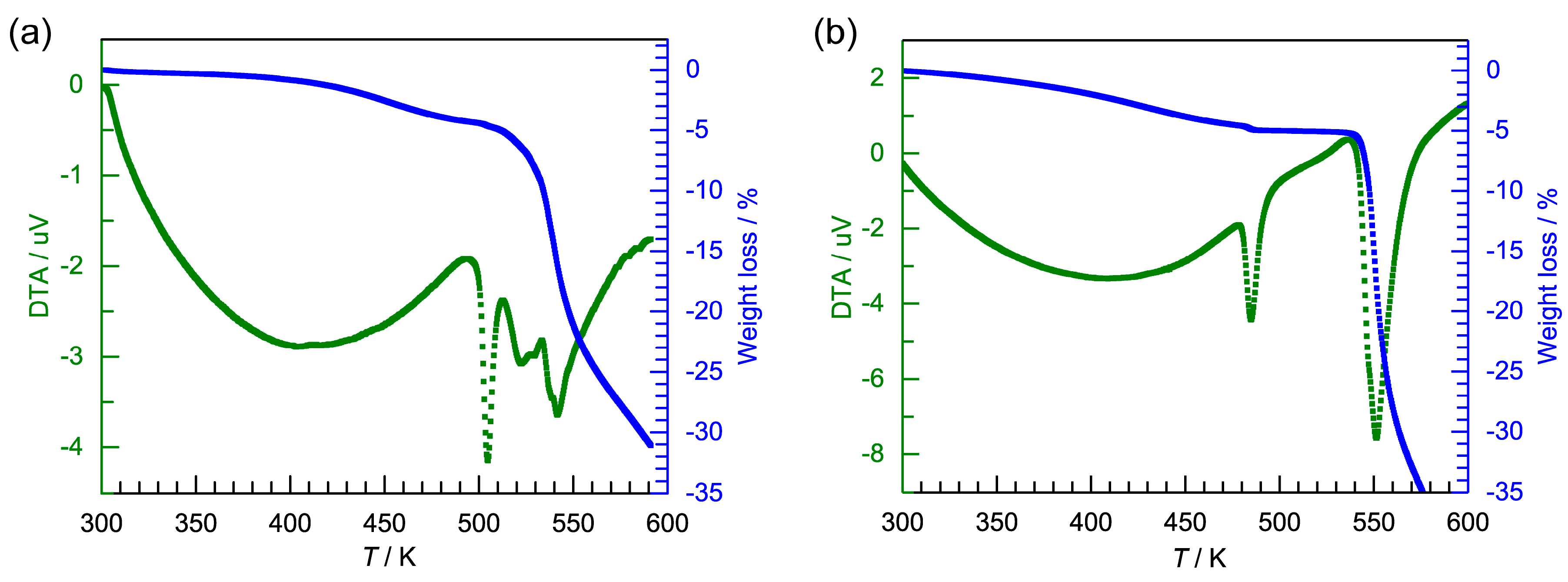
2.1. Synthesis and Characterization of [FeII2(L2C3)2](PF6)4·1.5H2O·1.5MeCN (1·PF6) and [FeII2(L2C3)2](AsF6)4·3H2O·MeCN (1·AsF6)
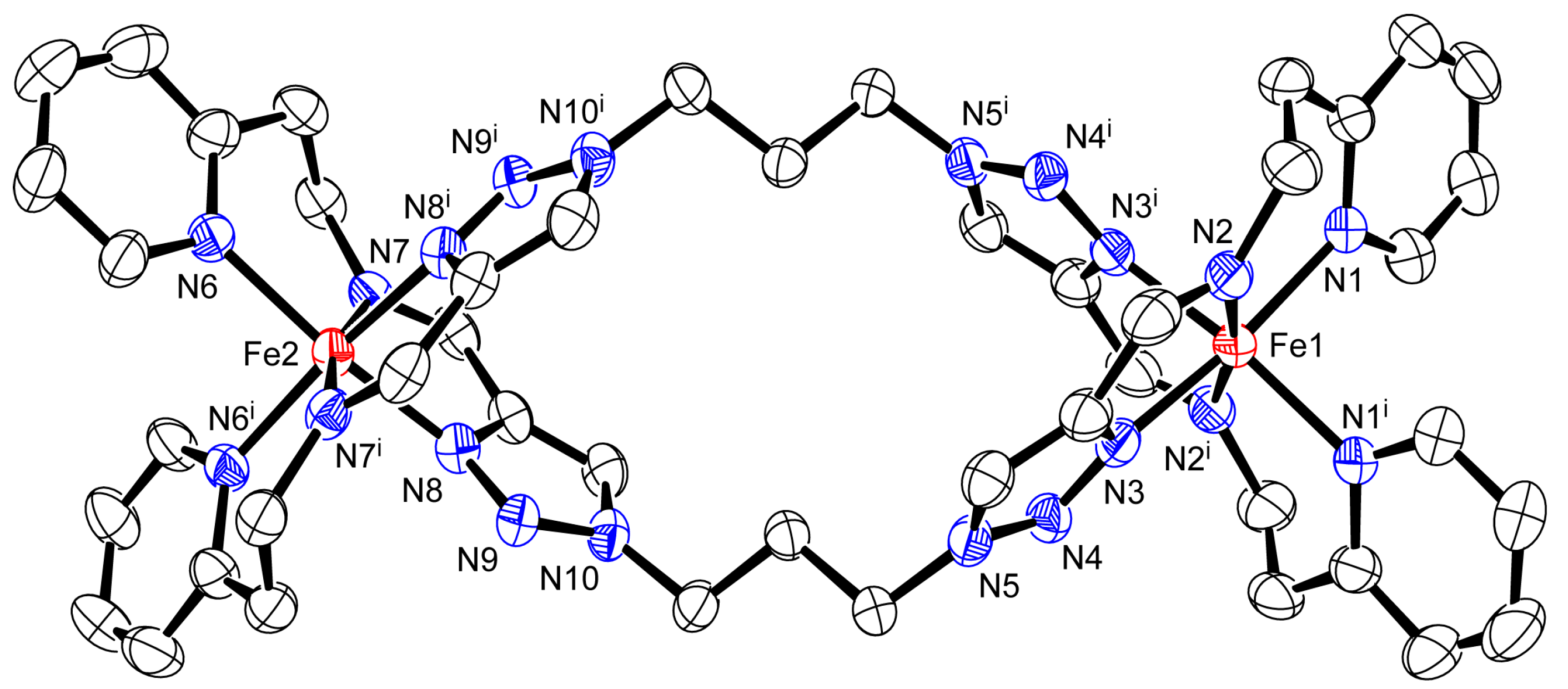
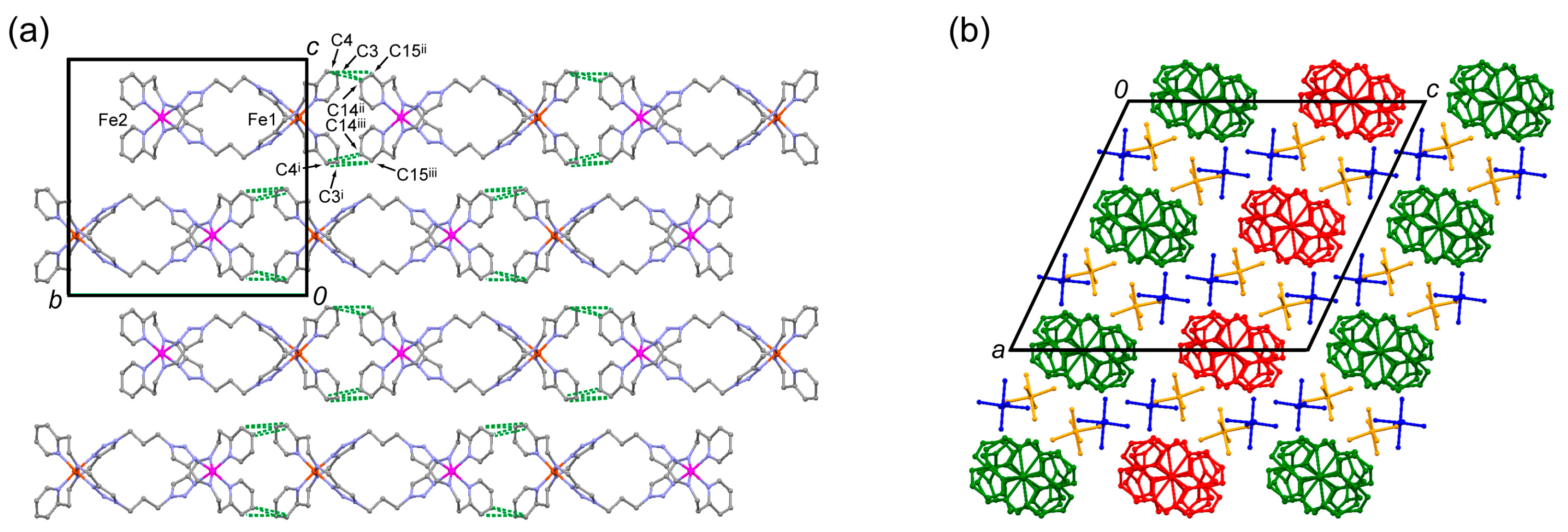
2.2. Crystal Structure of 1·PF6 and 1·AsF6
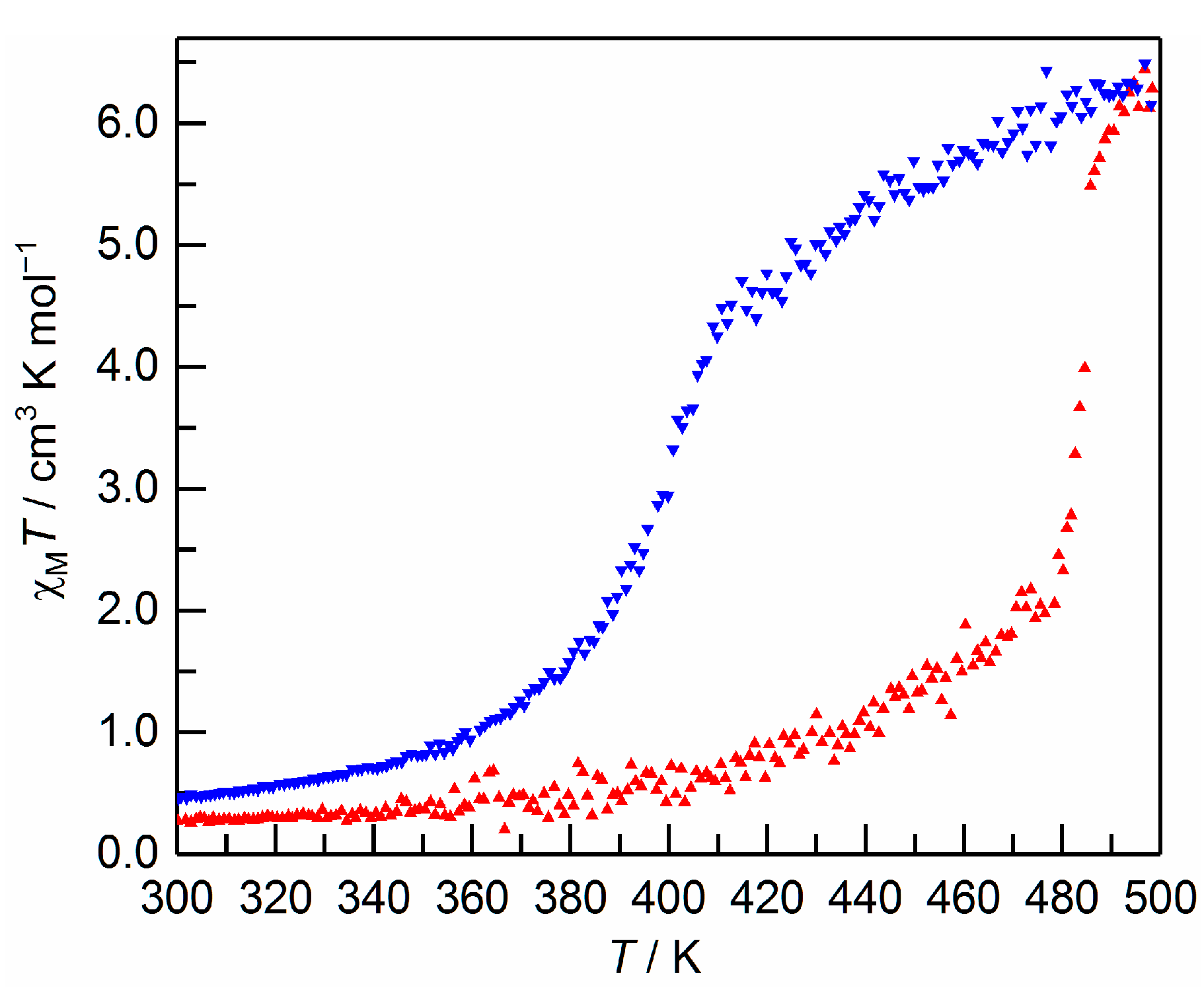
2.3. Magnetic Property of 1·AsF6
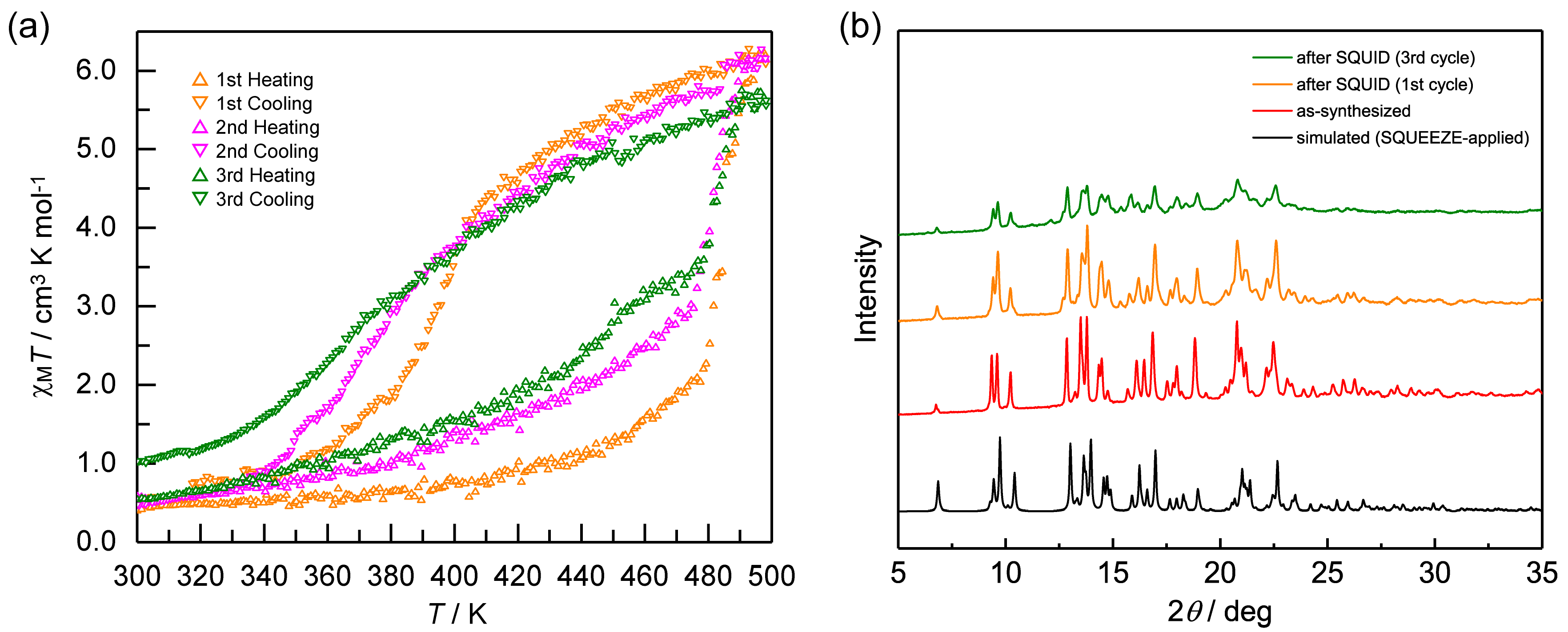
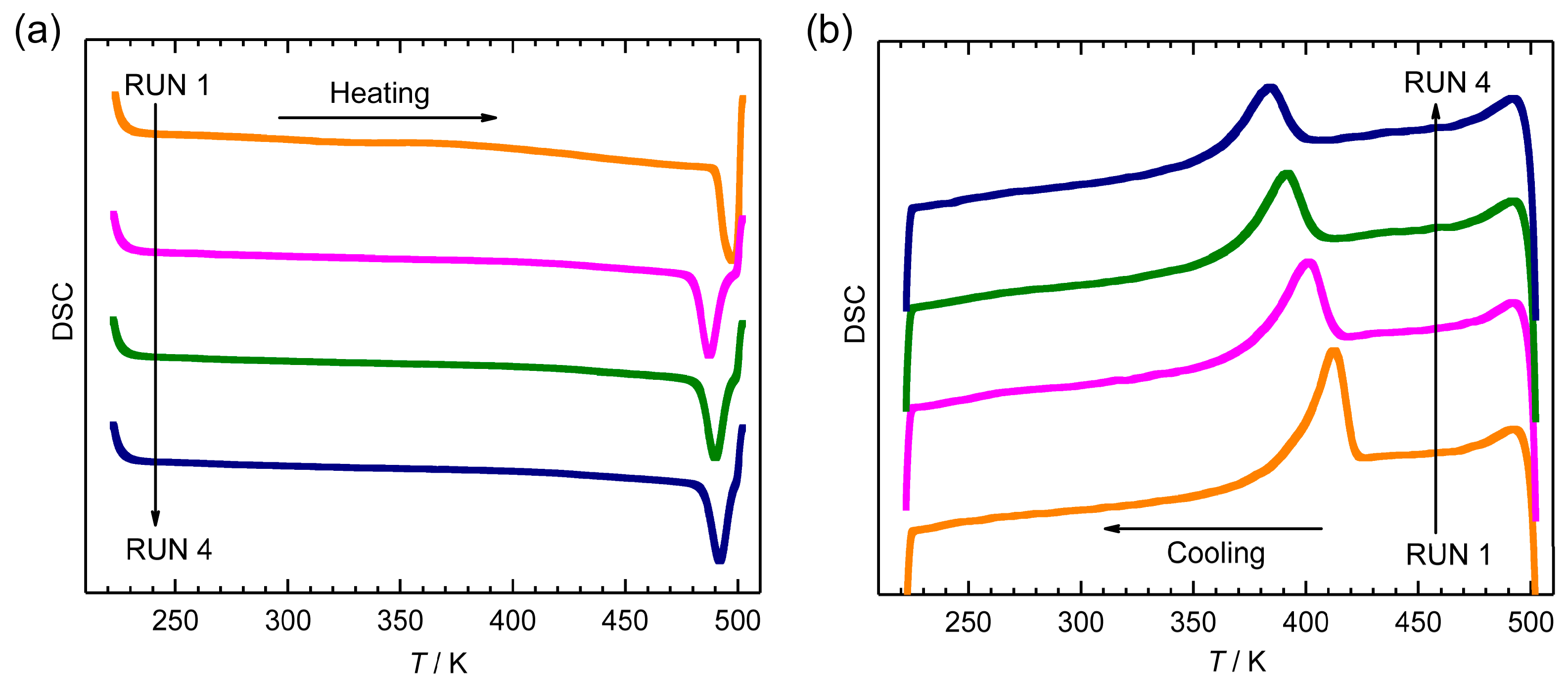
2.4. Reproducibility of Hysteretic SCO of 1·AsF6
3. Materials and Methods
3.1. Synthesis of FeII Complexes
3.1.1. General
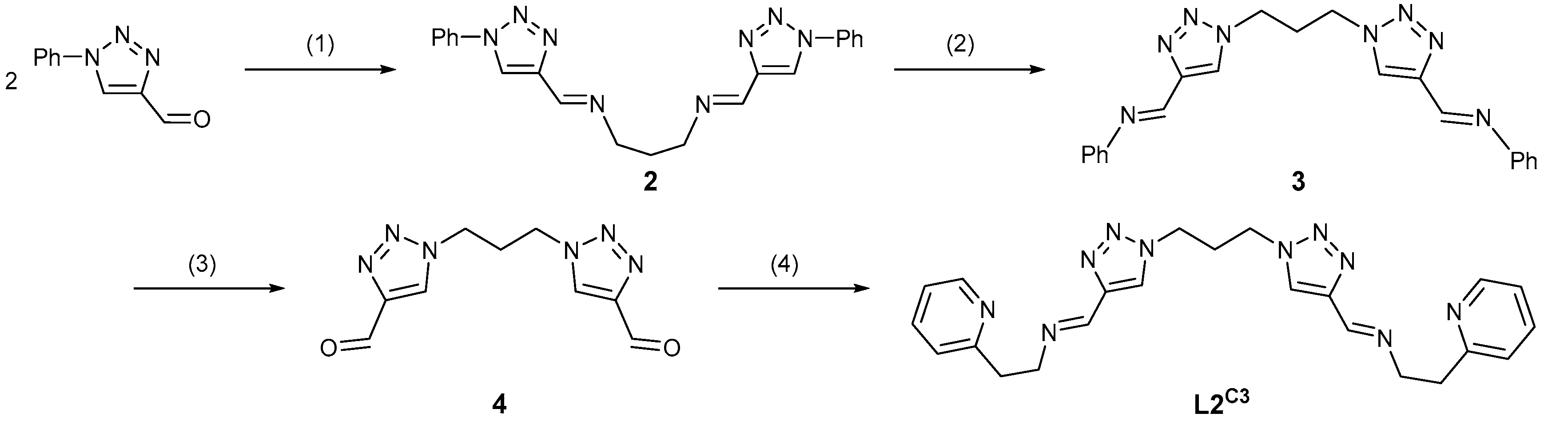
3.1.2. Preparation of 1,1′-(propane-1,3-diyl)bis(1H-1,2,3-triazole-4-carbaldehyde) (4)
3.1.3. Synthesis of the Bis-Tridentate Ligand L2C3 = 1,1′-[propane-1,3-diylbis(1H-1,2,3-triazole-1,4-diyl)]bis{N-[2-(pyridin-2-yl)ethyl]methanimine}
3.1.4. Preparation of [FeII2(L2C3)2](PF6)4·1.5H2O·1.5MeCN (1·PF6)
3.1.5. Preparation of [FeII2(L2C3)2](AsF6)4·3H2O·MeCN (1·AsF6)
3.2. Physical Measurements
3.3. Crystallographic Data Collection and Structure Analyses
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gütlich, P.; Goodwin, H.A. (Eds.) Spin Crossover in Transition Metal Compounds I-III; Topics in Current Chemistry; Springer: Berlin, Germany, 2004; Volume 233–235. [Google Scholar]
- Halcrow, M.A. (Ed.) Spin-Crossover Materials–Properties and Applications; John Wiley & Sons: Chichester, UK, 2013. [Google Scholar]
- Gütlich, P.; Gaspar, A.B.; Garcia, Y. Spin state switching in iron coordination compounds. Beilstein J. Org. Chem. 2013, 9, 342–391. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, D.M.; Peters, J.C. Solution and Solid-State Spin-Crossover Behavior in a Pseudotetrahedral d7 Ion. J. Am. Chem. Soc. 2003, 125, 11162–11163. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Lord, R.L.; Noll, B.C.; Baik, M.-H.; Schulz, C.E.; Scheidt, W.R. Cyanide: A Strong-Field Ligand for Ferrohemes and Hemoproteins? Angew. Chem. Int. Ed. 2008, 47, 10144–10146. [Google Scholar]
- Scepaniak, J.J.; Harris, T.D.; Vogel, C.S.; Sutter, J.; Meyer, K.; Smith, J.M. Spin Crossover in a Four-Coordinate Iron(II) Complex. J. Am. Chem. Soc. 2011, 133, 3824–3827. [Google Scholar] [PubMed]
- Mossin, S.; Tran, B.L.; Adhikari, D.; Pink, M.; Heinemann, F.W.; Sutter, J.; Szilagyi, R.K.; Meyer, K.; Mindiola, D.J. A Mononuclear Fe(III) Single Molecule Magnet with a 3/2↔5/2 Spin Crossover. J. Am. Chem. Soc. 2012, 134, 13651–13661. [Google Scholar] [PubMed]
- Weber, B.; Bauer, W.; Obel, J. An Iron(II) Spin-Crossover Complex with a 70 K Wide Thermal Hysteresis Loop. Angew. Chem. Int. Ed. 2008, 47, 10098–10101. [Google Scholar] [CrossRef] [PubMed]
- Weber, B.; Bauer, W.; Pfaffender, T.; Dîrtu, M.M.; Naik, A.D.; Rotaru, A.; Garcia, Y. Influence of Hydrogen Bonding on the Hysteresis Width in Iron(II) Spin-Crossover Complexes. Eur. J. Inorg. Chem. 2011, 2011, 3193–3206. [Google Scholar]
- Zhao, X.-H.; Zhang, S.-L.; Shao, D.; Wang, X.-Y. Spin Crossover in [Fe(2-Picolylamine)3]2+ Adjusted by Organosulfonate Anions. Inorg. Chem. 2015, 54, 7857–7867. [Google Scholar] [CrossRef] [PubMed]
- Hayami, S.; Gu, Z.-Z.; Yoshiki, H.; Fujishima, A.; Sato, O. Iron (III) spin-crossover compounds with a wide apparent thermal hysteresis around room temperature. J. Am. Chem. Soc. 2001, 123, 11644–11650. [Google Scholar] [PubMed]
- Schäfer, B.; Rajnák, C.; Šalitroš, I.; Fuhr, O.; Klar, D.; Schmitz-Antoniak, C.; Weschke, E.; Wende, H.; Ruben, M. Room temperature switching of a neutral molecular iron(II) complex. Chem. Commun. 2013, 49, 10986–10988. [Google Scholar] [CrossRef] [PubMed]
- Iasco, O.; Rivière, E.; Guillot, R.; Cointe, M.B.-L.; Meunier, J.-F.; Bousseksou, A.; Boillot, M.-L. FeII(pap-5NO2)2 and FeII(qsal-5NO2)2 Schiff-Base Spin-Crossover Complexes: A Rare Example with Photomagnetism and Room-Temperature Bistability. Inorg. Chem. 2015, 54, 1791–1799. [Google Scholar] [CrossRef] [PubMed]
- Kröber, J.; Codjovi, E.; Kahn, O.; Grolière, F.; Jay, C. A Spin Transition System with a Thermal Hysteresis at Room Temperature. J. Am. Chem. Soc. 1993, 115, 9810–9811. [Google Scholar] [CrossRef]
- Kahn, O.; Martinez, C.J. Spin-Transition Polymers: From Molecular Materials toward Memory Devices. Science 1998, 279, 44–48. [Google Scholar] [CrossRef]
- Muñoz-Lara, F.J.; Gaspar, A.B.; Aravena, D.; Ruiz, D.; Muñoz, M.C.; Ohba, M.; Ohtani, R.; Kitagawa, S.; Real, J.A. Enhanced bistability by guest inclusion in Fe(II) spin crossover porous coordination polymers. Chem. Commun. 2012, 48, 4686–4688. [Google Scholar] [CrossRef] [PubMed]
- Lochenie, C.; Bauer, W.; Railliet, A.P.; Schlamp, S.; Garcia, Y.; Weber, B. Large Thermal Hysteresis for Iron(II) Spin Crossover Complexes with N-(Pyrid-4-yl)isonicotinamide. Inorg. Chem. 2014, 53, 11563–11572. [Google Scholar] [CrossRef] [PubMed]
- Dîrtu, M.M.; Naik, A.D.; Rotaru, A.; Spinu, L.; Poelman, D.; Garcia, Y. FeII Spin Transition Materials Including an Amino–Ester 1,2,4-Triazole Derivative, Operating at, below, and above Room Temperature. Inorg. Chem. 2016, 55, 4278–4295. [Google Scholar] [CrossRef] [PubMed]
- Murray, K.S. Advances in Polynuclear Iron(II), Iron(III) and Cobalt(II) Spin-Crossover Compounds. Eur. J. Inorg. Chem. 2008, 3101–3121. [Google Scholar] [CrossRef]
- Nihei, M.; Sekine, Y.; Suganami, N.; Nakazawa, K.; Nakao, A.; Nakao, H.; Murakami, Y.; Oshio, H. Controlled Intramolecular Electron Transfers in Cyanide-Bridged Molecular Squares by Chemical Modifications and External Stimuli. J. Am. Chem. Soc. 2011, 133, 3592–3600. [Google Scholar] [CrossRef] [PubMed]
- Hietsoi, O.; Dunk, P.W.; Stout, H.D.; Arroyave, A.; Kovnir, K.; Irons, R.E.; Kassenova, N.; Erkasov, R.; Achim, C.; Shatruk, M. Spin Crossover in Tetranuclear Fe(II) Complexes, {[(tpma)Fe(μ-CN)]4}X4 (X = ClO4−, BF4−). Inorg. Chem. 2014, 53, 13070–13077. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, T.; Newton, G.N.; Shiga, T.; Hayami, S.; Matsui, Y.; Okamoto, H.; Kumai, R.; Murakami, Y.; Oshio, H. Programmable spin-state switching in a mixed-valence spin-crossover iron grid. Nat. Commun. 2014, 5, 3865. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.-Q.; Wang, Y.-T.; Cui, A.-L.; Kou, H.-Z. Toward Higher Nuclearity: Tetranuclear Cobalt(II) Metallogrid Exhibiting Spin Crossover. Inorg. Chem. 2014, 53, 2613–2618. [Google Scholar] [CrossRef] [PubMed]
- Steinert, M.; Schneider, B.; Dechert, S.; Demeshko, S.; Meyer, F. Spin-State Versatility in a Series of Fe4 [2 × 2] Grid Complexes: Effects of Counteranions, Lattice Solvent, and Intramolecular Cooperativity. Inorg. Chem. 2016, 55, 2363–2373. [Google Scholar] [CrossRef] [PubMed]
- Charbonnière, L.J.; Williams, A.F.; Piguet, C.; Bernardinelli, G.; Rivara-Minten, E. Structural, Magnetic, and Electrochemical Properties of Dinuclear Triple Helices: Comparison with Their Mononuclear Analogues. Chem. Eur. J. 1998, 4, 485–493. [Google Scholar] [CrossRef]
- Ksenofontov, V.; Gaspar, A.B.; Niel, V.; Reiman, S.; Real, J.A.; Gütlich, P. On the Nature of the Plateau in Two-Step Dinuclear Spin-Crossover Complexes. Chem. Eur. J. 2004, 10, 1291–1298. [Google Scholar] [CrossRef] [PubMed]
- Weber, B.; Kaps, E.S.; Obel, J.; Achterhold, K.; Parak, F.G. Synthesis and Characterization of a Dinuclear Iron(II) Spin Crossover Complex with Wide Hysteresis. Inorg. Chem. 2008, 47, 10779–10787. [Google Scholar] [CrossRef] [PubMed]
- Min, K.S.; Swierczek, K.; DiPasquale, A.G.; Rheingold, A.L.; Reiff, W.M.; Arif, A.M.; Miller, J.S. A dinuclear iron(II) complex, [(TPyA)FeII(THBQ2−)FeII(TPyA)](BF4)2 [TPyA = tris(2-pyridylmethyl)amine; THBQ2− = 2,3,5,6-tetrahydroxy-1,4-benzoquinonate] exhibiting both spin crossover with hysteresis and ferromagnetic exchange. Chem. Commun. 2008, 317–319. [Google Scholar] [CrossRef]
- Sunatsuki, Y.; Kawamoto, R.; Fujita, K.; Maruyama, H.; Suzuki, T.; Ishida, H.; Kojima, M.; Iijima, S.; Matsumoto, N. Structures and Spin States of Bis(tridentate)-Type Mononuclear and Triple Helicate Dinuclear Iron(II) Complexes of Imidazole-4-carbaldehyde azine. Inorg. Chem. 2009, 48, 8784–8795. [Google Scholar] [CrossRef] [PubMed]
- Sunatsuki, Y.; Kawamoto, R.; Fujita, K.; Maruyama, H.; Suzuki, T.; Ishida, H.; Kojima, M.; Iijima, S.; Matsumoto, N. Structures and spin states of mono- and dinuclear iron(II) complexes of imidazole-4-carbaldehyde azine and its derivatives. Coord. Chem. Rev. 2010, 254, 1871–1881. [Google Scholar] [CrossRef]
- Archer, R.J.; Hawes, C.S.; Jameson, G.N.L.; Mckee, V.; Moubaraki, B.; Chilton, N.F.; Murray, K.S.; Schmitt, W.; Kruger, P.E. Partial spin crossover behaviour in a dinuclear iron(II) triple helicate. Dalton Trans. 2011, 40, 12368–12373. [Google Scholar] [CrossRef] [PubMed]
- Schneider, C.J.; Cashion, J.D.; Chilton, N.F.; Etrillard, C.; Fuentealba, M.; Howard, J.A.K.; Létard, J.-F.; Milsmann, C.; Moubaraki, B.; Sparkes, H.A.; et al. Spin Crossover in a 3,5-Bis(2-pyridyl)-1,2,4-triazolate-Bridged Dinuclear Iron(II) Complex [{Fe(NCBH3)(py)}2-(μ-L1)2]—Powder versus Single Crystal Study. Eur. J. Inorg. Chem. 2013, 2013, 850–864. [Google Scholar] [CrossRef]
- Kulmaczewski, R.; Olguín, J.; Kitchen, J.A.; Feltham, H.L.C.; Jameson, G.N.L.; Tallon, J.L.; Brooker, S. Remarkable Scan Rate Dependence for a Highly Constrained Dinuclear Iron(II) Spin Crossover Complex with a Wide Thermal Hysteresis Loop. J. Am. Chem. Soc. 2014, 136, 878–881. [Google Scholar] [CrossRef] [PubMed]
- Hagiwara, H.; Tanaka, T.; Hora, S. Synthesis, structure, and spin crossover above room temperature of a mononuclear and related dinuclear double helicate iron(II) complexes. Dalton Trans. 2016, 45, 17132–17140. [Google Scholar] [CrossRef] [PubMed]
- Halcrow, M.A. Spin-crossover Compounds with Wide Thermal Hysteresis. Chem. Lett. 2014, 43, 1178–1188. [Google Scholar] [CrossRef]
- Brooker, S. Spin crossover with thermal hysteresis: Practicalities and lessons learnt. Chem. Soc. Rev. 2015, 44, 2880–2892. [Google Scholar] [CrossRef] [PubMed]
- Fujinami, T.; Nishi, K.; Hamada, D.; Murakami, K.; Matsumoto, N.; Iijima, S.; Kojima, M.; Sunatsuki, Y. Scan Rate Dependent Spin Crossover Iron(II) Complex with Two Different Relaxations and Thermal Hysteresis fac-[FeII(HLn-Pr)3]Cl·PF6 (HLn-Pr = 2-Methylimidazol-4-yl-methylideneamino-n-propyl). Inorg. Chem. 2015, 54, 7291–7300. [Google Scholar] [CrossRef] [PubMed]
- Bao, X.; Guo, P.-H.; Liu, W.; Tucek, J.; Zhang, W.-X.; Leng, J.-D.; Chen, X.-M.; Gural’skiy, I.; Salmon, L.; Bousseksou, A.; et al. Remarkably high-temperature spin transition exhibited by new 2D metal–organic frameworks. Chem. Sci. 2012, 3, 1629–1633. [Google Scholar] [CrossRef]
- Liu, W.; Bao, X.; Li, J.-Y.; Qin, Y.-L.; Chen, Y.-C.; Ni, Z.-P.; Tong, M.-L. High-Temperature Spin Crossover in Two Solvent-Free Coordination Polymers with Unusual High Thermal Stability. Inorg. Chem. 2015, 54, 3006–3011. [Google Scholar] [CrossRef] [PubMed]
- Rostovtsev, V.V.; Green, L.G.; Fokin, V.V.; Sharpless, K.B. A Stepwise Huisgen Cycloaddition Process: Copper(I)-Catalyzed Regioselective “Ligation” of Azides and Terminal Alkynes. Angew. Chem. Int. Ed. 2002, 41, 2596–2599. [Google Scholar] [CrossRef]
- Tornøe, C.W.; Christensen, C.; Meldal, M. Peptidotriazoles on Solid Phase: [1,2,3]-Triazoles by Regiospecific Copper(I)-Catalyzed 1,3-Dipolar Cycloadditions of Terminal Alkynes to Azides. J. Org. Chem. 2002, 67, 3057–3064. [Google Scholar] [CrossRef] [PubMed]
- Pathigoolla, A.; Pola, R.P.; Sureshan, K.M. A versatile solvent-free azide–alkyne click reaction catalyzed by in situ generated copper nanoparticles. Appl. Catal. A: Gen. 2013, 453, 151–158. [Google Scholar] [CrossRef]
- Schiff, H. Mittheilungen aus dem Universitätslaboratorium in Pisa: Eine neue Reihe organischer Basen. Justus Liebigs Ann. Chem. 1864, 131, 118–119. [Google Scholar] [CrossRef]
- L’abbé, G.; Bruynseels, M. Replacement of aryl by alkyl in 1-substituted 1H-1,2,3-triazole-4-carbaldehydes. J. Chem. Soc. Perkin Trans. 1 1990, 1492–1493. [Google Scholar] [CrossRef]
- L’abbé, G.; Bruynseels, M.; Delbeke, P.; Toppet, S. Molecular rearrangements of 4-iminomethyl-1,2,3-triazoles. Replacement of 1-aryl substituents in 1H-1,2,3-triazole-4-carbaldehydes. J. Heterocycl. Chem. 1990, 27, 2021–2027. [Google Scholar] [CrossRef]
- Hagiwara, H.; Minoura, R.; Okada, S.; Sunatsuki, Y. Synthesis, Structure, and Magnetic Property of a New Mononuclear Iron (II) Spin Crossover Complex with a Tripodal Ligand Containing Three 1, 2, 3-Triazole Groups. Chem. Lett. 2014, 43, 950–952. [Google Scholar] [CrossRef]
- Hagiwara, H.; Okada, S. A polymorphism-dependent T1/2 shift of 100 K in a hysteretic spin-crossover complex related to differences in intermolecular weak CH···X hydrogen bonds (X = S vs. S and N). Chem. Commun. 2016, 52, 815–818. [Google Scholar] [CrossRef] [PubMed]
- Schneider, C.J.; Moubaraki, B.; Cashion, J.D.; Turner, D.R.; Leita, B.A.; Batten, S.R.; Murray, K.S. Spin crossover in di-, tri- and tetranuclear, mixed-ligand tris(pyrazolyl)methane iron(II) complexes. Dalton Trans. 2011, 40, 6939–6951. [Google Scholar] [CrossRef] [PubMed]
- Nakamoto, K. Infrared and Raman Spectra of Inorganic and Coordination Compounds, 6th ed.; John Wiley & Sons: Hoboken, NJ, USA, 2009. [Google Scholar]
- Yamada, M.; Hagiwara, H.; Torigoe, H.; Matsumoto, N.; Kojima, M.; Dahan, F.; Tuchagues, J.-P.; Re, N.; Iijima, S. A Variety of Spin-Crossover Behaviors Depending on the Counter Anion: Two-Dimensional Complexes Constructed by NH···Cl− Hydrogen Bonds, [FeIIH3LMe]Cl·X (X = PF6−, AsF6−, SbF6−, CF3SO3−; H3LMe = Tris[2-{[(2-methylimidazol-4-yl)methylidene]amino}ethyl]amine). Chem. Eur. J. 2006, 12, 4536–4549. [Google Scholar] [CrossRef] [PubMed]
- Spek, A.L. PLATON SQUEEZE: A tool for the calculation of the disordered solvent contribution to the calculated structure factors. Acta Crystallogr. Sect. C Struct. Chem. 2015, 71, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Marchivie, M.; Guionneau, P.; Létard, J.-F.; Chasseau, D. Photo-induced spin-transition: The role of the iron(II) environment distortion. Acta Crystallogr. Sect. B 2005, 61, 25–28. [Google Scholar] [CrossRef] [PubMed]
- Llunell, M.; Casanova, D.; Cirera, J.; Alemany, P.; Alvarez, S. SHAPE2.1. Program for Calculating Continuous Shape Measures of Polyhedral Structures; Universitat de Barcelona: Barcelona, Spain, 2013. [Google Scholar]
- Guionneau, P.; Marchivie, M.; Bravic, G.; Létard, J.-F.; Chasseau, D. Structural aspects of spin crossover. Examples of the [FeIILn(NCS)2] complexes. Top. Curr. Chem. 2004, 234, 97–128. [Google Scholar]
- Kahn, O. Molecular Magnetism; VCH: Weinheim, Germany, 1993. [Google Scholar]
- Burla, M.C.; Caliandro, R.; Carrozzini, B.; Cascarano, G.L.; Cuocci, C.; Giacovazzo, C.; Mallamo, M.; Mazzone, A.; Polidori, G. Crystal structure determination and refinement via SIR2014. J. Appl. Cryst. 2015, 48, 306–309. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Cryst. 2015, C71, 3–8. [Google Scholar]
- Kabuto, C.; Akine, S.; Nemoto, T.; Kwon, E. Release of Software (Yadokari-XG 2009) for Crystal Structure Analyses. J. Cryst. Soc. Jpn. 2009, 51, 218–224. [Google Scholar] [CrossRef]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. OLEX2: A complete structure solution, refinement and analysis program. J. Appl. Cryst. 2009, 42, 339–341. [Google Scholar] [CrossRef]

| [FeII2L2C32](PF6)4 1.5H2O·1.5MeCN (1·PF6) a | [FeII2L2C32](AsF6)4·3H2O·MeCN (1·AsF6) a | |
|---|---|---|
| Formula | C49H59.5N21.5O1.5P4F24Fe2 | C48H61N21O3As4F24Fe2 |
| Formula weight | 1665.26 | 1847.55 |
| Crystal system | monoclinic | monoclinic |
| Space group | C2/c (No. 15) | C2/c (No.15) |
| a/Å | 19.049(6) | 19.410(3) |
| b/Å | 19.056(5) | 19.034(3) |
| c/Å | 20.600(9) | 20.772(5) |
| β/deg. | 115.832(3) | 115.723(2) |
| V/Å3 | 6731(4) | 6914(2) |
| Z | 4 | 4 |
| dcalcd./g·cm−3 | 1.643 | 1.775 |
| μ (Mo Kα)/mm−1 | 0.649 | 2.441 |
| R1 b (I > 2sigma(I)) | 0.0879 | 0.0520 |
| wR2 c (I > 2sigma(I)) | 0.2141 | 0.1167 |
| R1 b (all data) | 0.1054 | 0.0626 |
| wR2 c (all data) | 0.2286 | 0.1236 |
| u c | 0.0791 | 0.0425 |
| v c | 27.4133 | 16.3374 |
| S | 1.142 | 1.112 |
| CCDC number | 1544125 | 1544126 |
| Fe1 Site | 1·PF6 | 1·AsF6 | Fe2 Site | 1·PF6 | 1·AsF6 |
|---|---|---|---|---|---|
| Fe1–N Bond Lengths (Å) | Fe2–N Bond Lengths (Å) | ||||
| Fe1–N1 | 1.998(4) | 2.007(3) | Fe2–N6 | 2.033(4) | 2.007(3) |
| Fe1–N2 | 1.990(4) | 1.970(4) | Fe2–N7 | 1.911(4) | 1.962(3) |
| Fe1–N3 | 1.931(4) | 1.954(3) | Fe2–N8 | 1.957(4) | 1.945(3) |
| Fe1–N1 i | 1.998(4) | 2.007(3) | Fe2–N6 i | 2.034(4) | 2.007(3) |
| Fe1–N2 i | 1.990(5) | 1.970(4) | Fe2–N7 i | 1.911(4) | 1.962(3) |
| Fe1–N3 i | 1.931(4) | 1.954(3) | Fe2–N8 i | 1.957(4) | 1.945(3) |
| Average Fe1–N | 1.973 | 1.977 | Average Fe2–N | 1.967 | 1.971 |
| N–Fe1–N Bond Angles (°) | N–Fe2–N Bond Angles (°) | ||||
| N1–Fe1–N2 | 89.1(2) | 91.35(15) | N6–Fe2–N7 | 93.38(17) | 92.63(12) |
| N1–Fe1–N3 | 171.26(19) | 170.93(14) | N6–Fe2–N8 | 172.89(15) | 172.40(12) |
| N1–Fe1–N1 i | 91.4(2) | 92.67(18) | N6–Fe2–N6 i | 93.9(2) | 92.05(17) |
| N1–Fe1–N2 i | 98.00(19) | 95.70(14) | N6–Fe2–N7 i | 95.50(16) | 96.18(12) |
| N1–Fe1–N3 i | 92.58(16) | 91.47(12) | N6–Fe2–N8 i | 89.72(17) | 91.84(12) |
| N2–Fe1–N3 | 82.65(18) | 80.20(13) | N7–Fe2–N8 | 80.17(17) | 80.47(12) |
| N2–Fe1–N1 i | 98.00(19) | 95.69(15) | N7–Fe2–N6 i | 95.50(16) | 96.17(12) |
| N2–Fe1–N2 i | 169.9(3) | 169.79(18) | N7–Fe2–N7 i | 167.0(3) | 167.32(16) |
| N2–Fe1–N3 i | 89.86(17) | 92.27(13) | N7–Fe2–N8 i | 90.38(16) | 90.16(12) |
| N3–Fe1–N1 i | 92.58(16) | 91.47(12) | N8–Fe2–N6 i | 89.72(17) | 91.84(12) |
| N3–Fe1–N2 i | 89.85(17) | 92.27(13) | N8–Fe2–N7 i | 90.38(16) | 90.16(12) |
| N3-Fe1–N3 i | 84.6(2) | 85.61(17) | N8–Fe2–N8 i | 87.3(3) | 85.07(17) |
| N1 i–Fe1–N2 i | 89.1(2) | 91.36(15) | N6 i–Fe2–N7 i | 93.38(17) | 92.63(12) |
| N1 i–Fe1–N3 i | 171.26(19) | 170.93(14) | N6 i–Fe2–N8 i | 172.89(15) | 172.41(12) |
| N2 i–Fe1–N3 i | 82.65(18) | 80.20(14) | N7 i–Fe2–N8 i | 80.17(17) | 80.47(12) |
| Σ (°) [54] | 44.8 | 48.2 | 45.3 | 47.7 | |
| Θ (°) [52] | 113.9 | 120.1 | 106.9 | 108.1 | |
| CShMs c [53] | 0.594 | 0.616 | 0.636 | 0.590 | |
| Octahedral volume (Å3) | 10.132 | 10.183 | 10.029 | 10.098 | |
| 1·PF6 | 1·AsF6 | |
|---|---|---|
| Cg1 a···Cg2 b,ii | 4.139(4) | 4.123(3) |
| C3···C14 ii | 3.582(13) | 3.610(9) |
| C3···C15 ii | 3.403(12) | 3.423(8) |
| C4···C14 ii | 3.432(11) | 3.421(7) |
| C4···C15 ii | 3.711(11) | 3.703(6) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hora, S.; Hagiwara, H. High-Temperature Wide Thermal Hysteresis of an Iron(II) Dinuclear Double Helicate. Inorganics 2017, 5, 49. https://doi.org/10.3390/inorganics5030049
Hora S, Hagiwara H. High-Temperature Wide Thermal Hysteresis of an Iron(II) Dinuclear Double Helicate. Inorganics. 2017; 5(3):49. https://doi.org/10.3390/inorganics5030049
Chicago/Turabian StyleHora, Shiori, and Hiroaki Hagiwara. 2017. "High-Temperature Wide Thermal Hysteresis of an Iron(II) Dinuclear Double Helicate" Inorganics 5, no. 3: 49. https://doi.org/10.3390/inorganics5030049






