Continuous Production of IF-WS2 Nanoparticles by a Rotary Process
Abstract
:1. Introduction
2. Results and Discussion
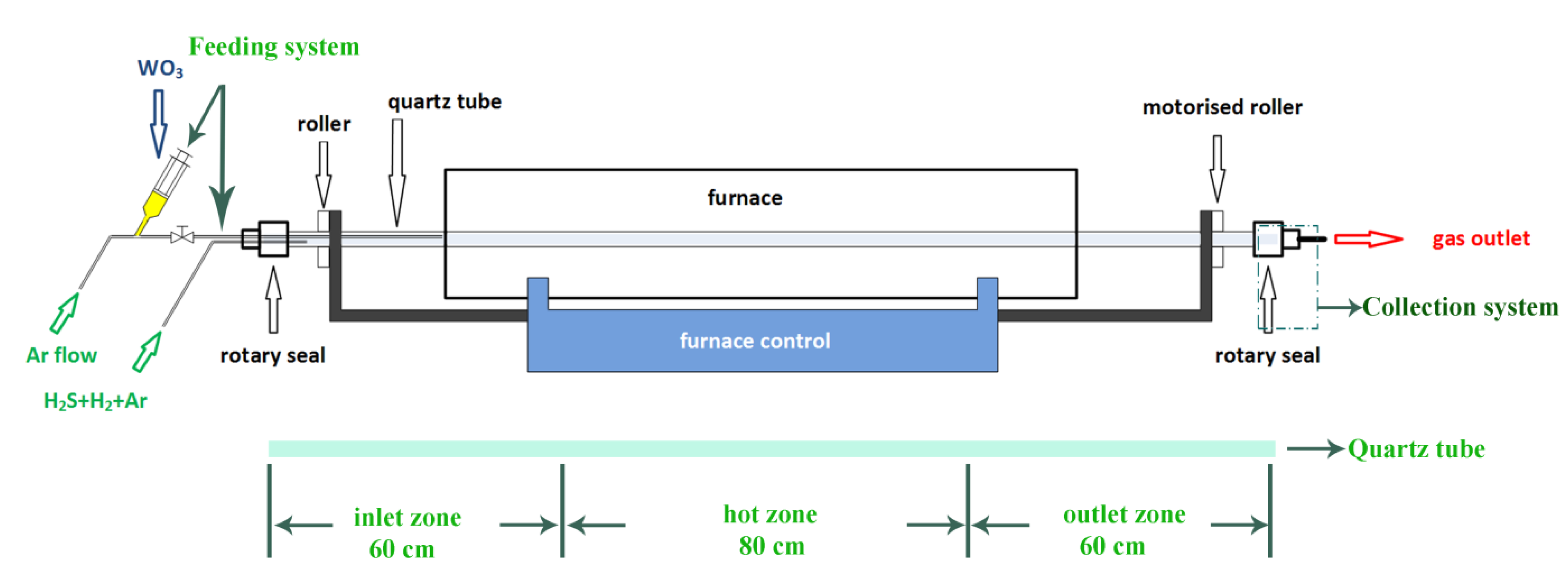
2.1. Design and Modification of the Rotary Furnace
| Temperature | Gas supply | Feeding level | Speed range | Inclination angle |
|---|---|---|---|---|
| 0–1200 °C | Ar, H2S, H2 | Up to 100 g* | 0–350 rpm | −5 to 5° |
2.2. IF-WS2 Synthesis by Different Methods
2.2.1. WO3 and S Reaction under H2


2.2.2. APT as Precursor and H2S as Reaction Gas


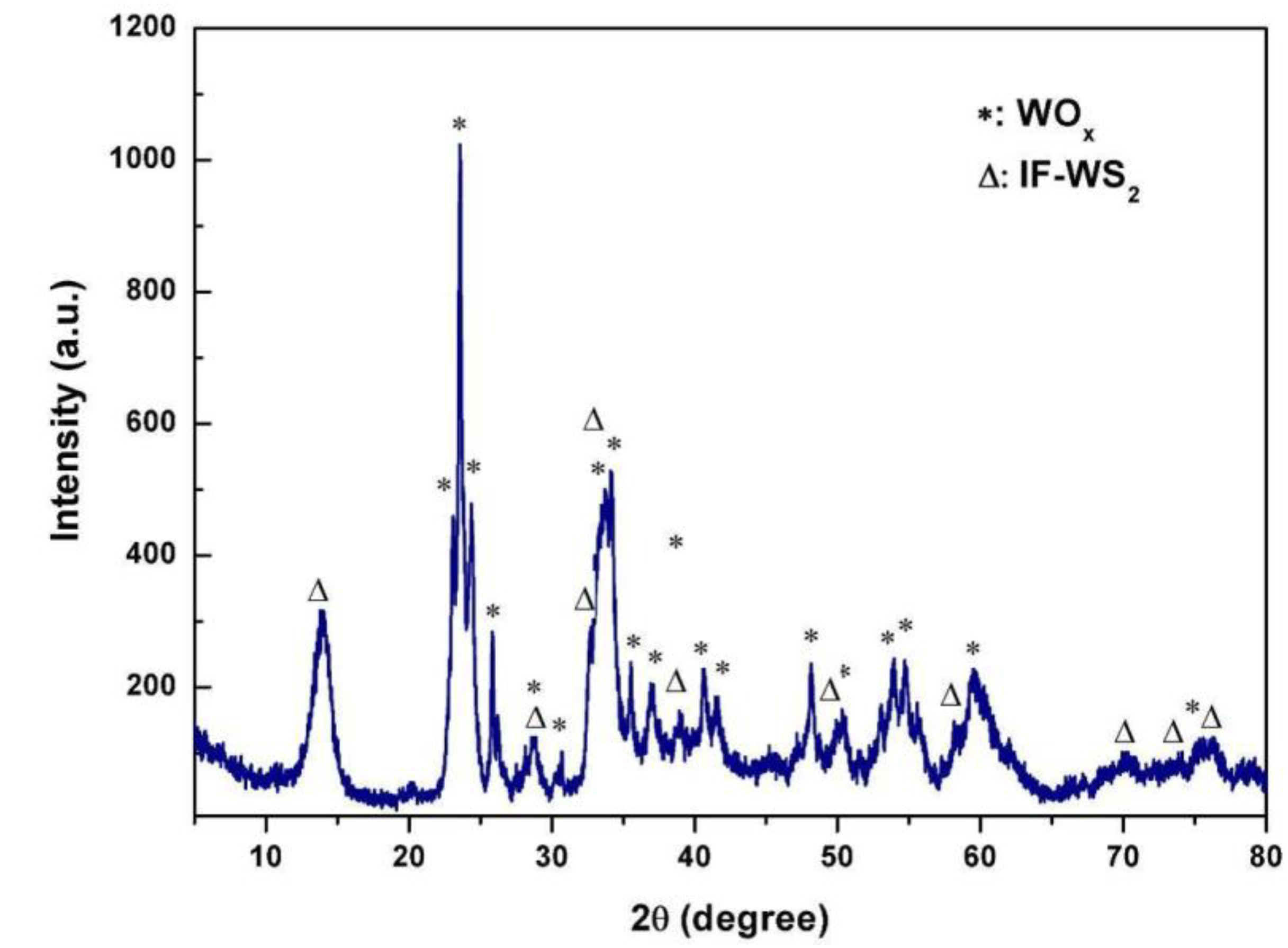
2.2.3. WO3 and H2S Synthesis of WS2 Nanomaterials

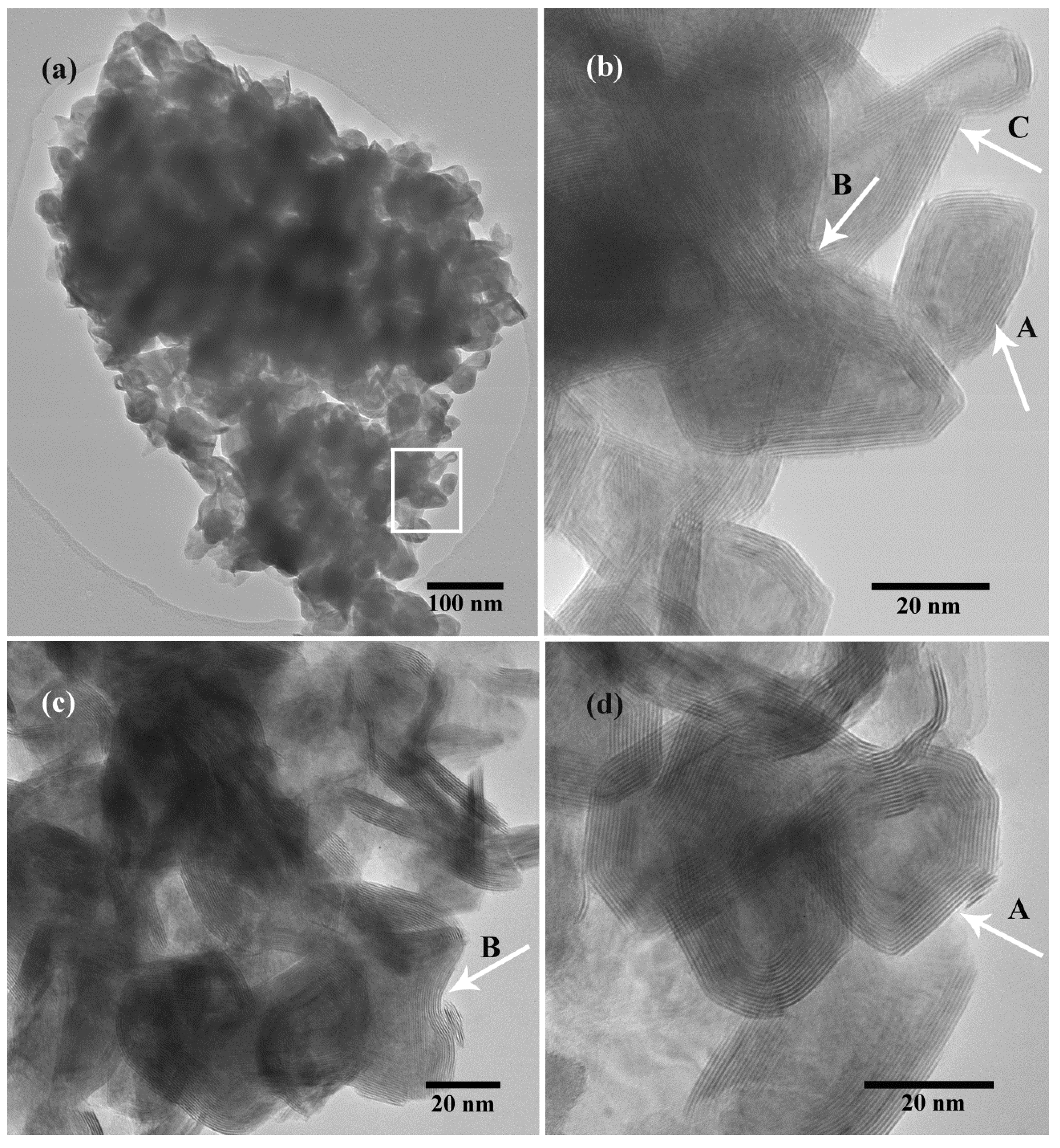
2.3. Optimization of IF-WS2 Synthesis from WO3 Precursor and H2S Gas
2.3.1. Reaction Time
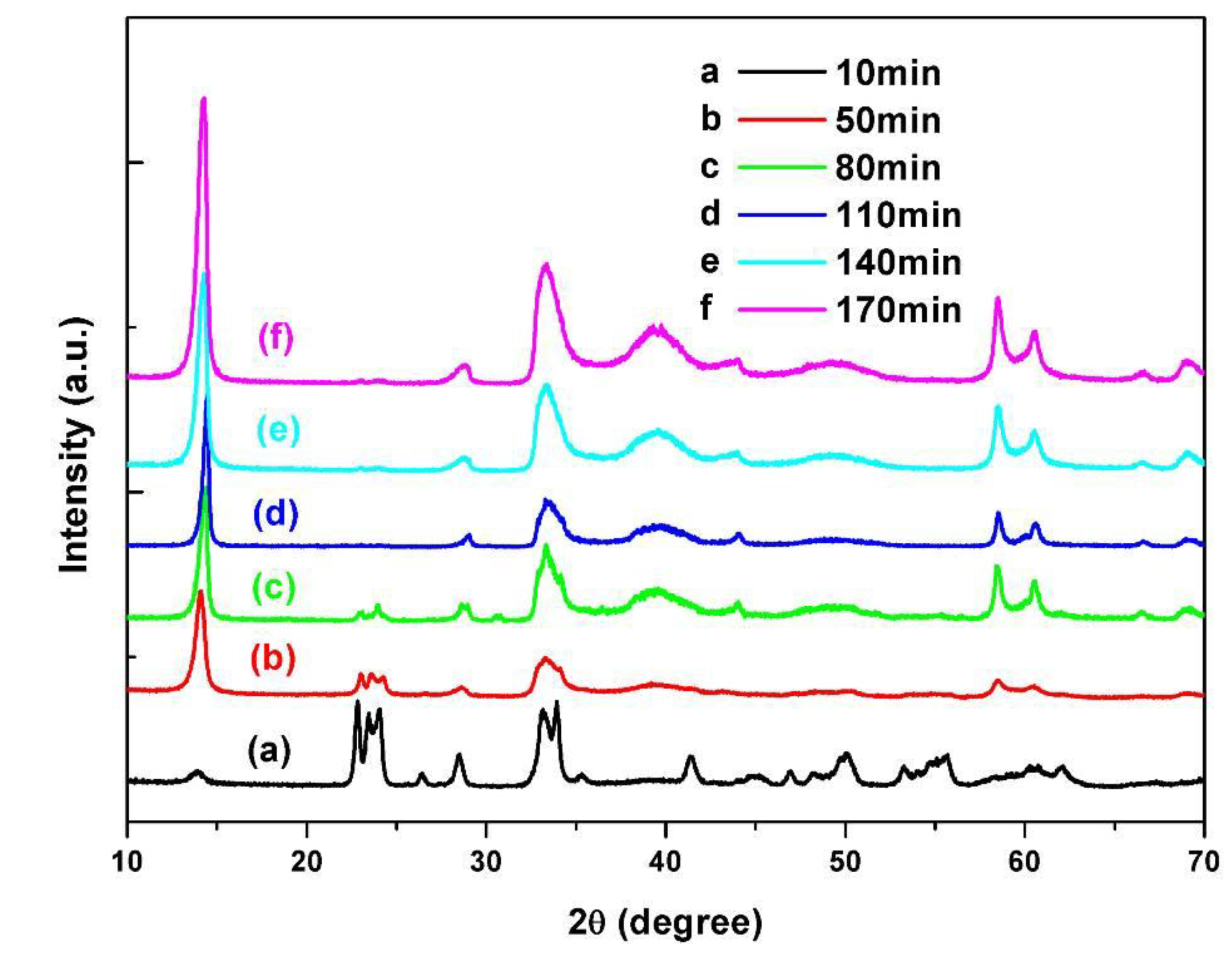
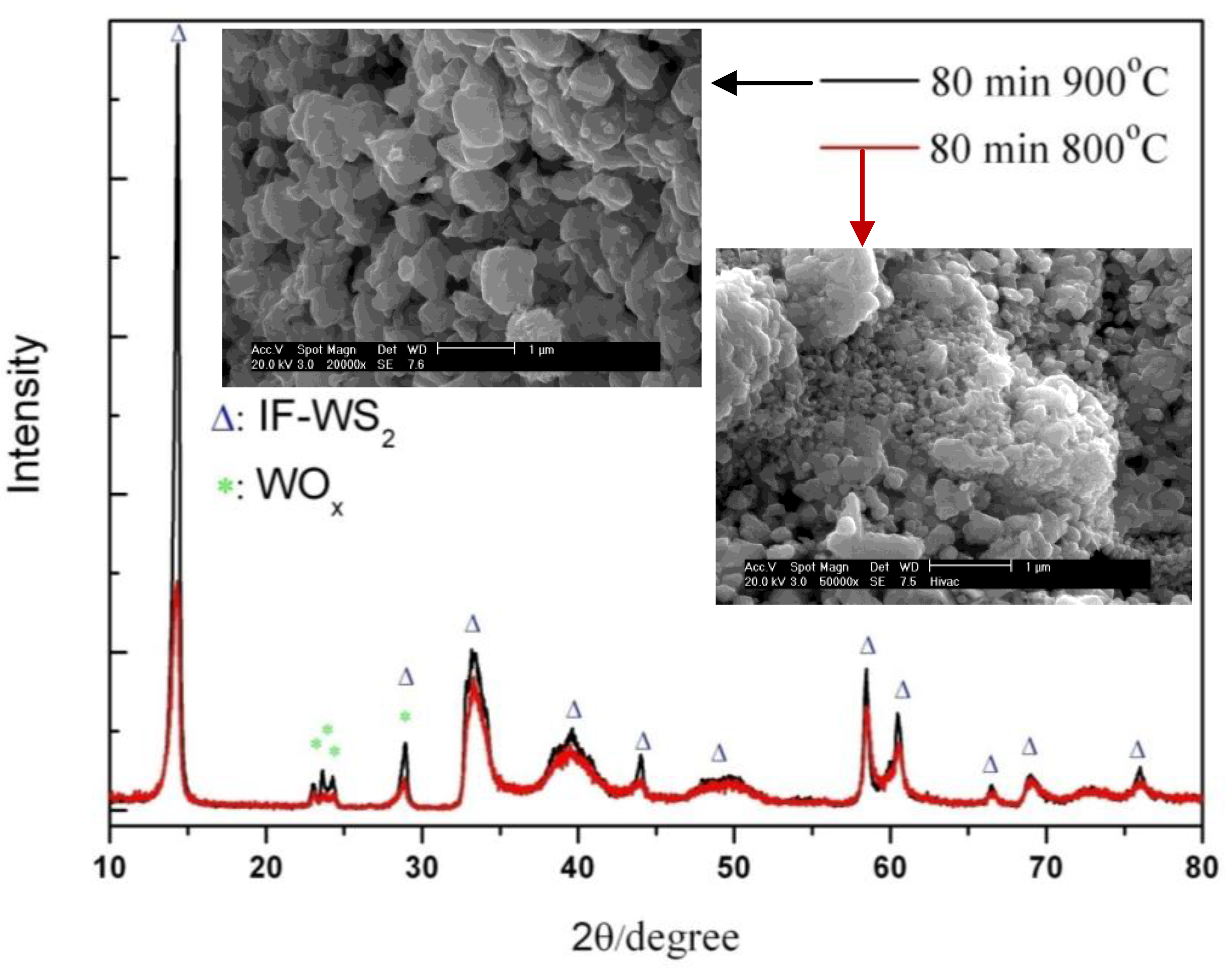
2.3.2. Reaction Temperatures

2.3.3. Influence of H2

2.4. Further Refinement and Modification
2.4.1. IF-WS2 Synthesis Using a New Baffled Quartz Tube


2.4.2. A Continuous Feeding System

3. Experimental Section
3.1. Materials
3.2. Experimental Set-Up
3.3. IF-WS2 Synthesis by Different Methods
3.3.1. Synthesis of IF-WS2 Nanoparticles from Mixed WO3 and S Powder with H2
3.3.2. Synthesis of IF-WS2 Nanoparticles from APT Precursor with H2S Gas
| Precursor put in (g) | Gas feed Rate (H2:H2S:Ar) (mL/min) | Reaction time (min) | Reaction temperature (°C) | |
|---|---|---|---|---|
| S1 | 6 | 80:0:30 | 60 | 800 |
| S2 | 12 | 80:0:30 | 120 | 800 |
| AHS1 | 6 | 0:12:100 | 120 | 800 |
| W1 | 6 | 0:12:100 | 90 | 800 |
| H1 | 6 | 8:12:100 | 90 | 800 |
| B1 | 18 | 10:30:160 | 120 | 800 |
| B2 | 18 | 20:30:100 | 120 | 800 |
| B3 | 18 | 20:30:100 10:30:100 | 120 | 800 |
| FB1 | 50 | 50:50:100 0: 25:100 | 180 | 800 |
| FB2 | 100 | 50:50:100 0:25:100 | 300 | 800 |
3.3.3. Synthesis of IF-WS2 Nanoparticles WO3 and H2S Gas
| WO3 Precursor (g) | Gas feed rate (H2S:Ar) (mL/min) | Reaction time (min) | Reaction temperature (°C) | |
|---|---|---|---|---|
| A | 6 | 12:100 | 10 | 800 |
| B | 6 | 12:100 | 50 | 800 |
| C | 6 | 12:100 | 80 | 800 |
| D | 6 | 12:100 | 110 | 800 |
| E | 6 | 12:100 | 140 | 800 |
| F | 6 | 12:100 | 170 | 800 |
| G | 6 | 12:100 | 80 | 900 |
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tenne, R.; Margulis, L.; Genut, M.; Hodes, G. Polyhedral and cylindrical structures of tungsten disulphide. Nature 1992, 360, 444–446. [Google Scholar] [CrossRef]
- Margulis, L.; Salitra, G.; Tenne, R.; Talianker, M. Nested fullerene-like structures. Nature 1993, 365, 113–114. [Google Scholar]
- Feldman, Y.; Wasserman, E.; Srolovitz, D.J.; Tenne, R. High rate, gas phase growth of MoS2 nested inorganic fullerenes and nanotubes. Science 1995, 267, 222–225. [Google Scholar] [CrossRef]
- Chopra, N.G.; Luyken, R.J.; Cherrey, K.; Crespi, V.H.; Cohen, M.L.; Louie, S.G.; Zettl, A. Boron nitride nanotubes. Science 1995, 269, 966–967. [Google Scholar] [CrossRef]
- Rosenfeld, H.Y.; Grunbaum, E.; Tenne, R.; Sloan, J.; Hutchison, J.L. Cage structures and nanotubes of NiCl2. Nature 1998, 395, 336. [Google Scholar] [CrossRef]
- Vollath, D.; Szabo, D.V. Synthesis of nanocrystalline MoS2 and WS2 in a microwave plasma. Mater. Lett. 1998, 35, 236–244. [Google Scholar] [CrossRef]
- Mackie, E.B.; Galvan, D.H.; Adem, E.; Talapatra, S.; Yang, G.L.; Migone, A.D. Production of WS2 nanotubes by an activation method. Adv. Mater. 2000, 12, 495–498. [Google Scholar] [CrossRef]
- Remskar, M.; Skraba, Z.; Regula, M.; Ballif, C.; Sanjine’s, R.; Levy, F. New crystal structures of WS2: microtubes, ribbons, and ropes. Adv. Mater. 1998, 10, 246–249. [Google Scholar] [CrossRef]
- Li, Y.D.; Li, X.L.; He, R.R.; Zhu, J.; Deng, Z.X. Artificial lamellar mesostructures to WS2 nanotubes. J. Am. Chem. Soc. 2002, 124, 1411–1416. [Google Scholar] [CrossRef]
- Li, X.L.; Ge, J.P.; Li, Y.D. Atmospheric pressure chemical vapor deposition: An alternative route to large-scale MoS2 and WS2 inorganic fullerene-like nanostructures and nanoflowers. Chem. A Eur. J. 2004, 10, 6163–6171. [Google Scholar] [CrossRef]
- Margolin, A.; Deepak, F.L.; Popovitz-Biro, R.; Bar-Sada, M.; Feldman, Y.; Tenne, R. Fullerene-like WS2 nanoparticles and nanotubes by the vapor-phase synthesis of WCln and H2S. Nanotechnology 2008, 19. [Google Scholar] [CrossRef]
- Seifert, G.; Terrones, H.; Terrones, M.; Jungnickel, G.; Frauenheim, T. Structure and electronic properties of MoS2 nanotubes. Phys. Rev. Lett. 2000, 85, 146–149. [Google Scholar] [CrossRef]
- Dominko, R.; Arcon, D.; Mrzel, A.; Zorko, A.; Cevc, P.; Venturini, P.; Gaberscek, M.; Remskar, M.; Mihailovic, D. Dichalcogenide nanotube electrodes for Li-Ion batteries. Adv. Mater. 2002, 14, 1531–1534. [Google Scholar] [CrossRef]
- Kaplan-Ashiri, I.; Tenne, R. Mechanical properties of WS2 nanotubes. J. Cluster Sci. 2007, 18, 549–563. [Google Scholar] [CrossRef]
- Rapoport, L.; Feldman, Y.; Homyonfer, M.; Cohen, H.; Sloan, J.; Hutchison, J.L.; Tenne, R. Inorganic fullerene-like material as additives to lubricants: Structure–function relationship. Wear 1999, 225–229, 975–982. [Google Scholar] [CrossRef]
- Kaplan-Ashiri, I.; Cohen, S.R.; Gartsman, K.; Ivanovskaya, V.; Heine, T.; Seifert, G.; Wiesel, I.; Wagner, H.D.; Tenne, R. On the mechanical behavior of WS2 nanotubes under axial tension and compression. Proc. Natl. Acad. Sci. 2006, 103, 523–528. [Google Scholar] [CrossRef]
- Rapoport, L.; Bilik, Y.; Feldman, Y.; Homyonfer, M.; Cohen, S.R.; Tenne, R. Hollow nanoparticles of WS2 as potential solid-state lubricants. Nature 1997, 387, 791–793. [Google Scholar] [CrossRef]
- Hou, X.H.; Shan, C.X.; Choy, K.L. Microstructures and tribological properties of PEEK-based nanocomposite coatings incorporating inorganic fullerene-like nanoparticles. Surf. Coat. Technol. 2008, 202, 2287–2291. [Google Scholar] [CrossRef]
- Brown, S.; Musfeldt, J.L.; Mihut, I.; Betts, J.B.; Migliori, A.; Zak, A.; Tenne, R. Bulk vs. nanoscale WS2: Finite size effects and solid-state lubrication. Nano Lett. 2007, 7, 2365–2369. [Google Scholar] [CrossRef]
- Frey, G.L.; Elani, S.; Homyonfer, M.; Feldman, Y.; Tenne, R. Optical-absorption spectra of inorganic fullerene like MS2 (M = Mo, W). Phys. Rev. B 1998, 57, 6666–6671. [Google Scholar] [CrossRef]
- Zhu, Y.Q.; Sekine, T.; Li, Y.H.; Wang, W.X.; Fay, M.W.; Edwards, H.; Brown, P.D.; Fleischer, N.; Tenne, R. WS2 and MoS2 inorganic fullerenes—super shock absorbers at very high pressures. Adv. Mater. 2005, 17, 1500–1503. [Google Scholar] [CrossRef]
- Zhu, Y.Q.; Sekine, T.; Li, Y.H.; Fay, M.W.; Zhao, Y.M.; Poa, C.H.P.; Wang, W.X.; Roe, M.J.; Brown, P.D.; Fleischer, N.; et al. Shock-absorbing and failure mechanisms of WS2 and MoS2 nanoparticles with fullerene-like structures under shock wave pressure. J. Am. Chem. Soc. 2005, 127, 16263–16272. [Google Scholar] [CrossRef]
- Zhu, Y.Q.; Sekine, T.; Brigatti, K.S.; Firth, S.; Tenne, R.; Rosentsveig, R.; Kroto, H.W.; Walton, D.R.M. Shock-wave resistance of WS2 nanotubes. J. Am. Chem. Soc. 2003, 125, 1329–1333. [Google Scholar] [CrossRef]
- Hogg, P.J. Composites in Armor. Science 2006, 314, 1100–1101. [Google Scholar] [CrossRef]
- Feldman, Y.; Zak, A.; Popovitz-Biro, R.; Tenne, R. New reactor for production of tungsten disulfide hollow onion-like (inorganic fullerene-like) nanoparticles. Solid State Sci. 2000, 2, 663–672. [Google Scholar] [CrossRef]
- Feldman, Y.; Frey, G.L.; Homyonfer, M.; Lyakhovitskaya, V.; Margulis, L.; Cohen, H.; Hodes, G.; Hutchison, J.L.; Tenne, R. Bulk synthesis of inorganic fullerene-like MS2 (M = Mo, W) from the respective trioxides and the reaction mechanism. J. Am. Chem. Soc. 1996, 118, 5362–5367. [Google Scholar] [CrossRef]
- Rothschild, A.; Sloan, J.; Tenne, R. Growth of WS2 nanotubes phases. J. Am. Chem. Soc. 2000, 122, 5169–5179. [Google Scholar] [CrossRef]
- Dai, J.J.; Cui, H.P.; Grace, J.R. Biomass feeding for thermochemical reactors. Prog. Energy Combust. Sci. 2012, 38, 716–736. [Google Scholar] [CrossRef]
- Wilson, D.H.; Dunnington, D.L. Plan ahead to avoid feeding problems. Chem. Eng. N. Y. 1991, 98, 72–81. [Google Scholar]
- Hao, X.H.; Guo, L.J.; Mao, X.; Zhang, X.M.; Chen, X.J. Hydrogen production from glucose used as a model compound of biomass gasified in supercritical water. Int. J. Hydrogen Energy 2003, 28, 55–64. [Google Scholar]
- Deepak, F.L.; Tenne, R. Gas-phase synthesis of inorganic fullerene-like, structures and inorganic nanotubes. Cent. Eur. J. Chem. 2008, 6, 373–389. [Google Scholar] [CrossRef]
- Rosentsveig, R.; Margolin, A.; Feldman, Y.; Popovitz-Biro, R.; Tenne, R. WS2 nanotube bundles and foils. Chem. Mater. 2002, 14, 471–474. [Google Scholar] [CrossRef]
- Margolin, A.; Rosentsveig, R.; Albu-Yaron, A.; Popovitz-Biro, R.; Tenne, R. Study of the growth mechanism of WS2 nanotubes produced by a fluidized bed reactor. J. Mater. Chem. 2004, 14, 617–624. [Google Scholar] [CrossRef]
- Alaei, M.; Rashidi, A.; Mahjoub, A. Two suitable methods for the preparation of inorganic fullerene-like (IF) WS2 nanoparticles. Iran. J. Chem. Chem. Eng. 2009, 28, 91–98. [Google Scholar]
- Wiesel, I.; Arbel, H.; Albu-Yaron, A.; Popovitz-Biro, R.; Gordon, J.M.; Feuermann, D.; Tenne, R. Synthesis of WS2 and MoS2 fullerene-like nanoparticles from solid precursors. Nano Res. 2009, 2, 416–424. [Google Scholar] [CrossRef]
- Zhu, Y.Q.; Hsu, W.K.; Terrones, H.; Grobert, N.; Chang, B.H.; Terrones, M.; Wei, B.Q.; Kroto, H.W.; Walton, D.R.M.; Boothroyd, C.B.; et al. Morphology, structure and growth of WS2 nanotubes. J. Mater. Chem. 2000, 10, 2570–2577. [Google Scholar] [CrossRef]
- Li, Y.H.; Zhao, Y.M.; Ma, R.Z.; Zhu, Y.Q.; Fisher, N.; Jin, Y.Z.; Zhang, X.P. Novel route to WOx nanorods and WS2 nanotubes from WS2 inorganic fullerenes. J. Phys. Chem. B 2006, 110, 18191–18195. [Google Scholar]
- Zhu, Y.Q.; Hu, W.B.; Hsu, W.K.; Terrones, M.; Grobert, N.; Hare, J.P.; Kroto, H.W.; Walton, D.R.M.; Terrones, H. Tungsten oxide tree-like structures. Chem. Phys. Lett. 1999, 309, 327–334. [Google Scholar] [CrossRef]
- Zak, A.; Sallacan-Ecker, L.; Margolin, A.; Feldman, Y.; Popovitz-Biro, R.; Albu-Yaron, A.; Genut, M.; Tenne, R. Scaling up of the WS2 nanotubes synthesis. Fullerenes Nanotubes Carbon Nanostruct. 2011, 19, 18–26. [Google Scholar]
- Zak, A.; Sallacan-Ecker, L.; Margolin, A.; Genut, M.; Tenne, R. Insight into the growth mechanism of WS2 nanotubes in the scaled-up fluidized-bed reactor. Nano 2009, 4, 91–98. [Google Scholar] [CrossRef]
- Finnie, G.J.; Kruyt, N.P.; Ye, M.; Zeilstra, C.; Kuipers, J.A.M. Longitudinal and transverse mixing in rotary kilns: A discrete element method approach. Chem. Eng. Sci. 2005, 60, 4083–4091. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Xu, F.; Wang, N.; Chang, H.; Xia, Y.; Zhu, Y. Continuous Production of IF-WS2 Nanoparticles by a Rotary Process. Inorganics 2014, 2, 313-333. https://doi.org/10.3390/inorganics2020313
Xu F, Wang N, Chang H, Xia Y, Zhu Y. Continuous Production of IF-WS2 Nanoparticles by a Rotary Process. Inorganics. 2014; 2(2):313-333. https://doi.org/10.3390/inorganics2020313
Chicago/Turabian StyleXu, Fang, Nannan Wang, Hong Chang, Yongde Xia, and Yanqiu Zhu. 2014. "Continuous Production of IF-WS2 Nanoparticles by a Rotary Process" Inorganics 2, no. 2: 313-333. https://doi.org/10.3390/inorganics2020313





