Microwave-Assisted Synthesis of Boron-Modified TiO2 Nanocrystals
Abstract
:1. Introduction
2. Results and Discussion
2.1. TEM
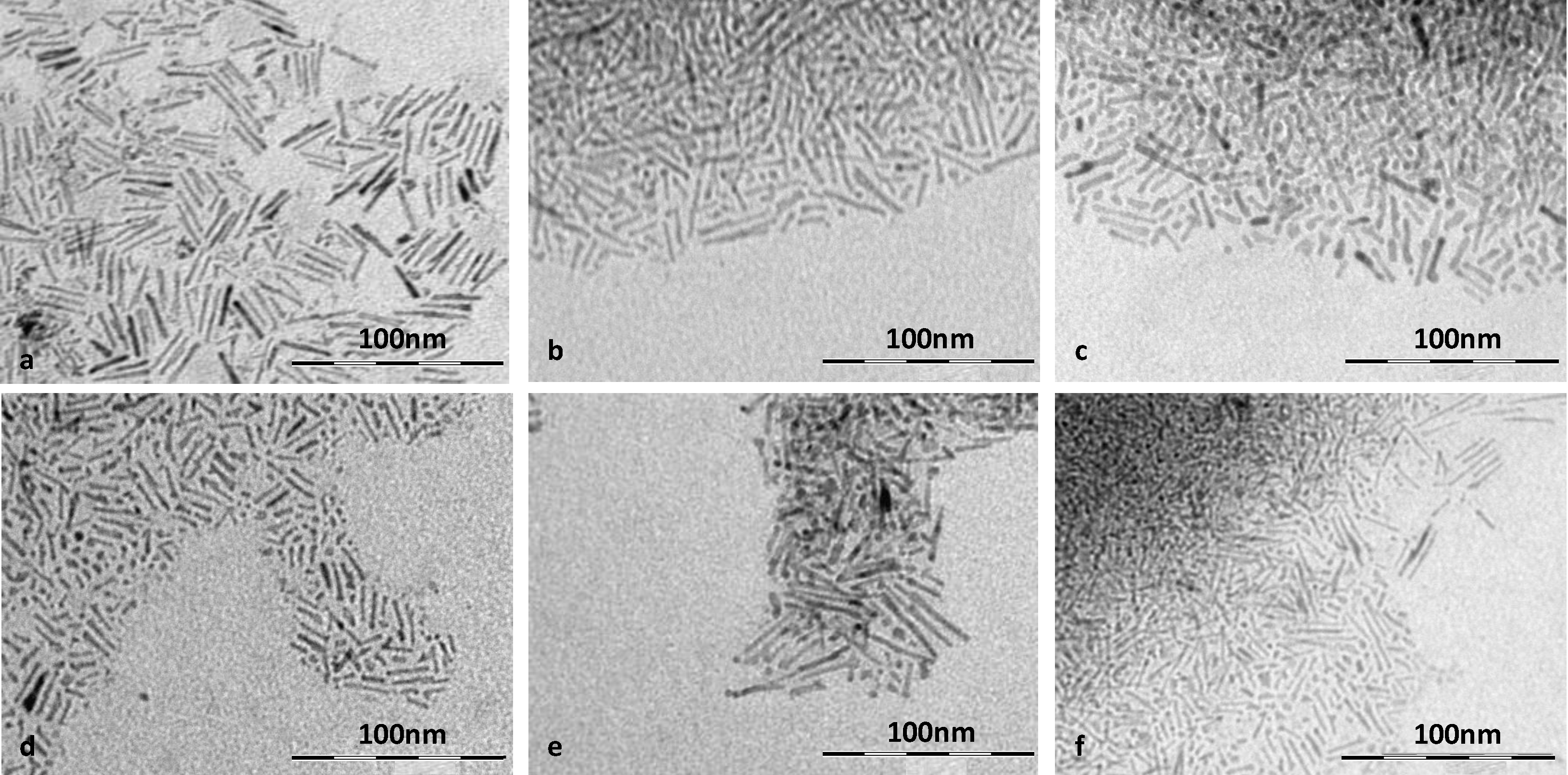
| Sample | TTIP:BA a | TTIP:OA a | B:Ti b | Mean Rod Length (nm) ± σ c | Rod Diameter (nm) ± Δd d | Aspect Ratio ± Δ e |
|---|---|---|---|---|---|---|
| TOB1 | 1:1 | 1:6 | 0.0108 | 27.0 ± 8.6 | 3.5 ± 0.6 | 7.7 ± 0.5 |
| TOB2 | 1:1 | 1:8 | 0.0240 | 18.75 ± 9.5 | 3.3 ± 0.4 | 5.7 ± 0.6 |
| TOB3 | 1:1 | 1:12 | 0.0245 | 13.6 ± 6.5 | 3.7 ± 0.5 | 3.6 ± 0.6 |
| TOB4 | 1:2 | 1:6 | 0.1138 | 14.2 ± 7.2 | 3.6 ± 0.6 | 3.9 ± 0.7 |
| TOB5 | 1:2 | 1:8 | 0.0987 | 16.5 ± 9.2 | 3.3 ± 0.4 | 5.0 ± 0.7 |
| TOB6 | 1:2 | 1:12 | 0.0641 | 22.7 ± 10.6 | 3.1 ± 0.8 | 7.3 ± 0.7 |
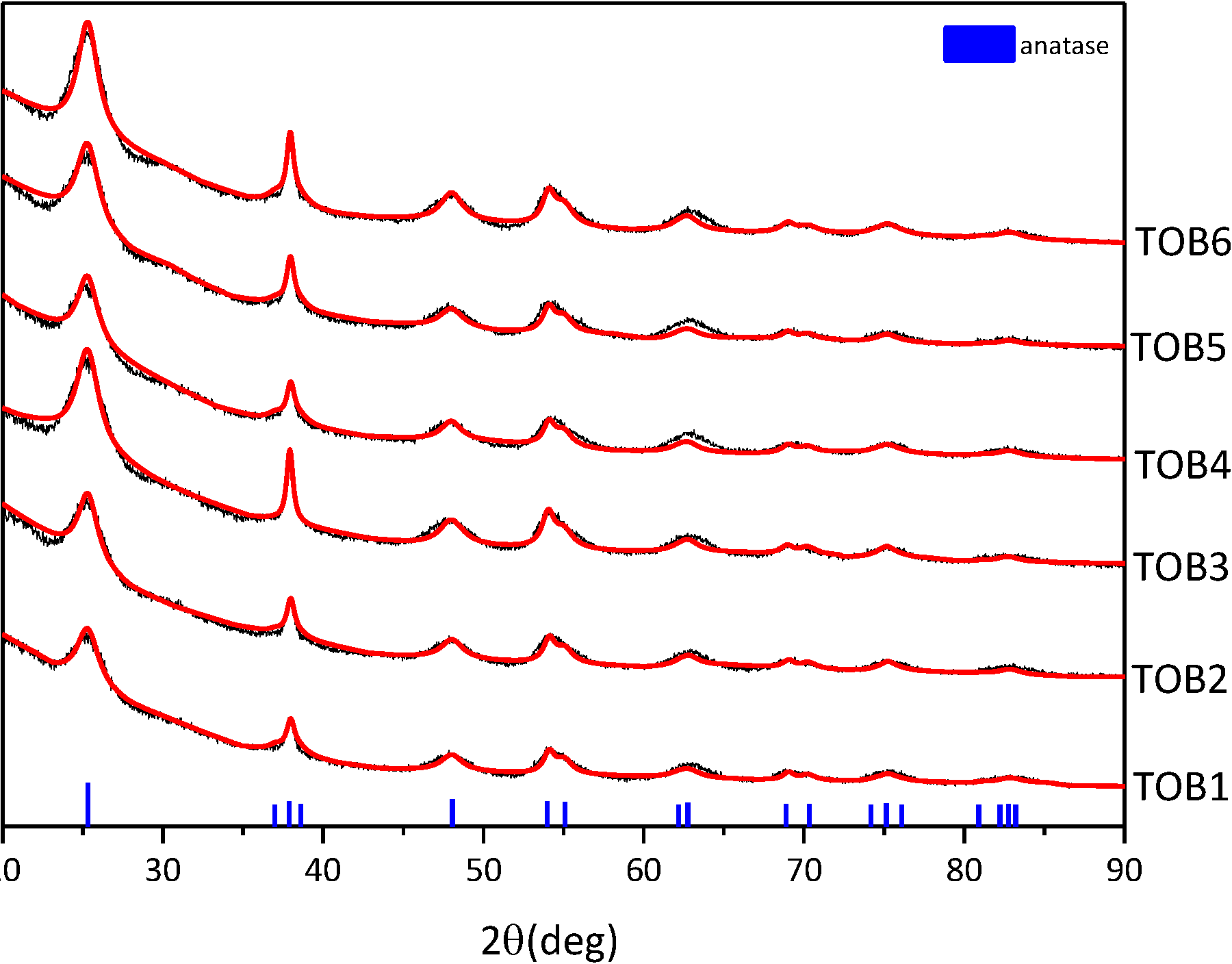
2.2. XRD
| Sample | Apparent Size Ratio | Ti a | O a | 004 b | 200 b | Eg(1) c | Eg(3) c |
|---|---|---|---|---|---|---|---|
| TOB1 | 2.5 | 0.97 | 2.04 | 97.67 | 35.22 | 150.6 | 652.9 |
| TOB2 | 2.7 | 0.99 | 2.59 | 101.41 | 38.63 | 151.1 | 652.6 |
| TOB3 | 4.2 | 0.95 | 2.34 | 136.56 | 35.10 | 152.1 | 654.2 |
| TOB4 | 2.5 | 1.09 | 1.88 | 102.43 | 37.19 | 150.7 | 651.2 |
| TOB5 | 3.1 | 0.89 | 2.04 | 106.75 | 34.45 | 152.0 | 656.6 |
| TOB6 | 3.1 | 1.02 | 2.23 | 114.73 | 36.61 | 151.5 | 652.0 |
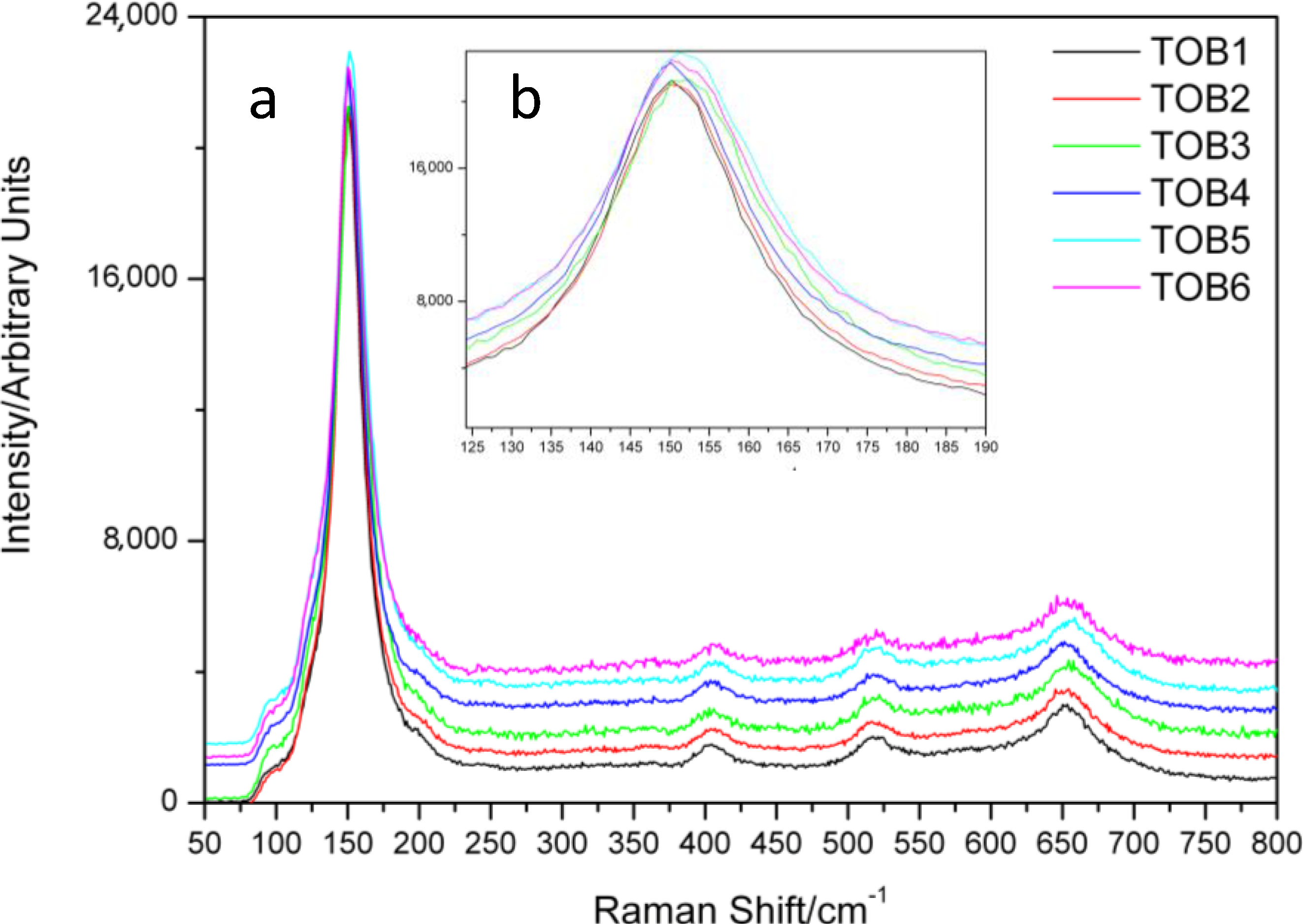
2.3. Micro Raman
2.4. FTIR
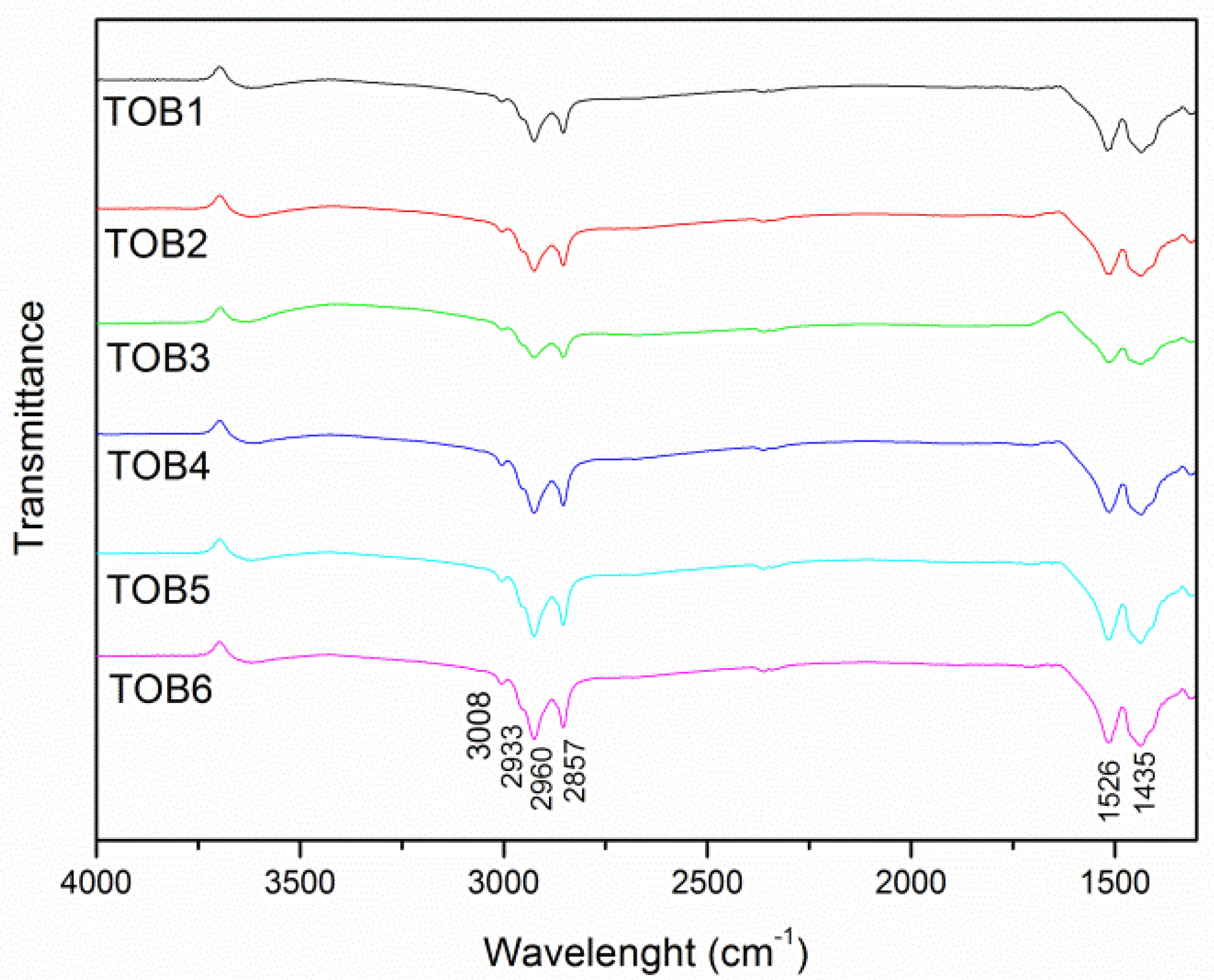
2.5. BET
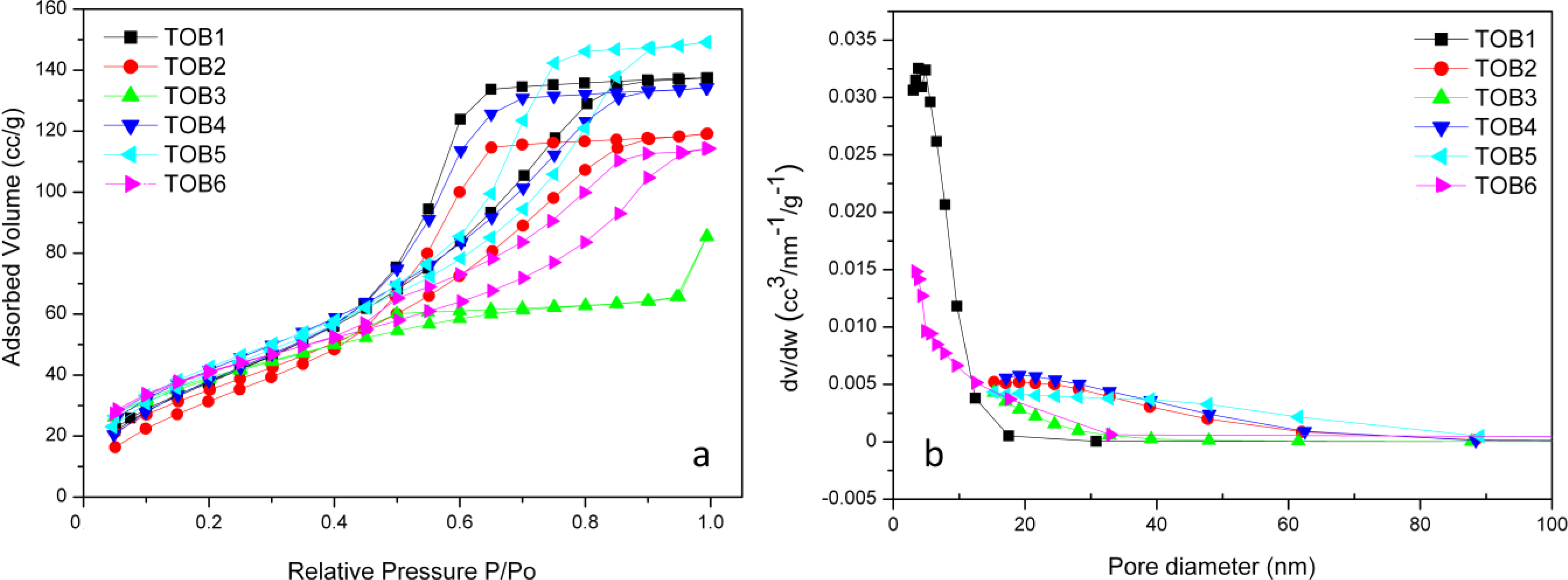
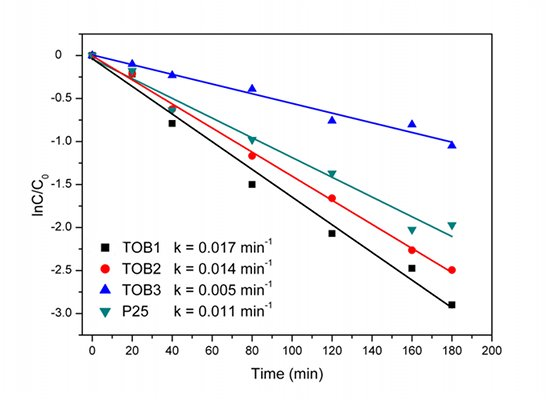
2.6. Photocatalysis
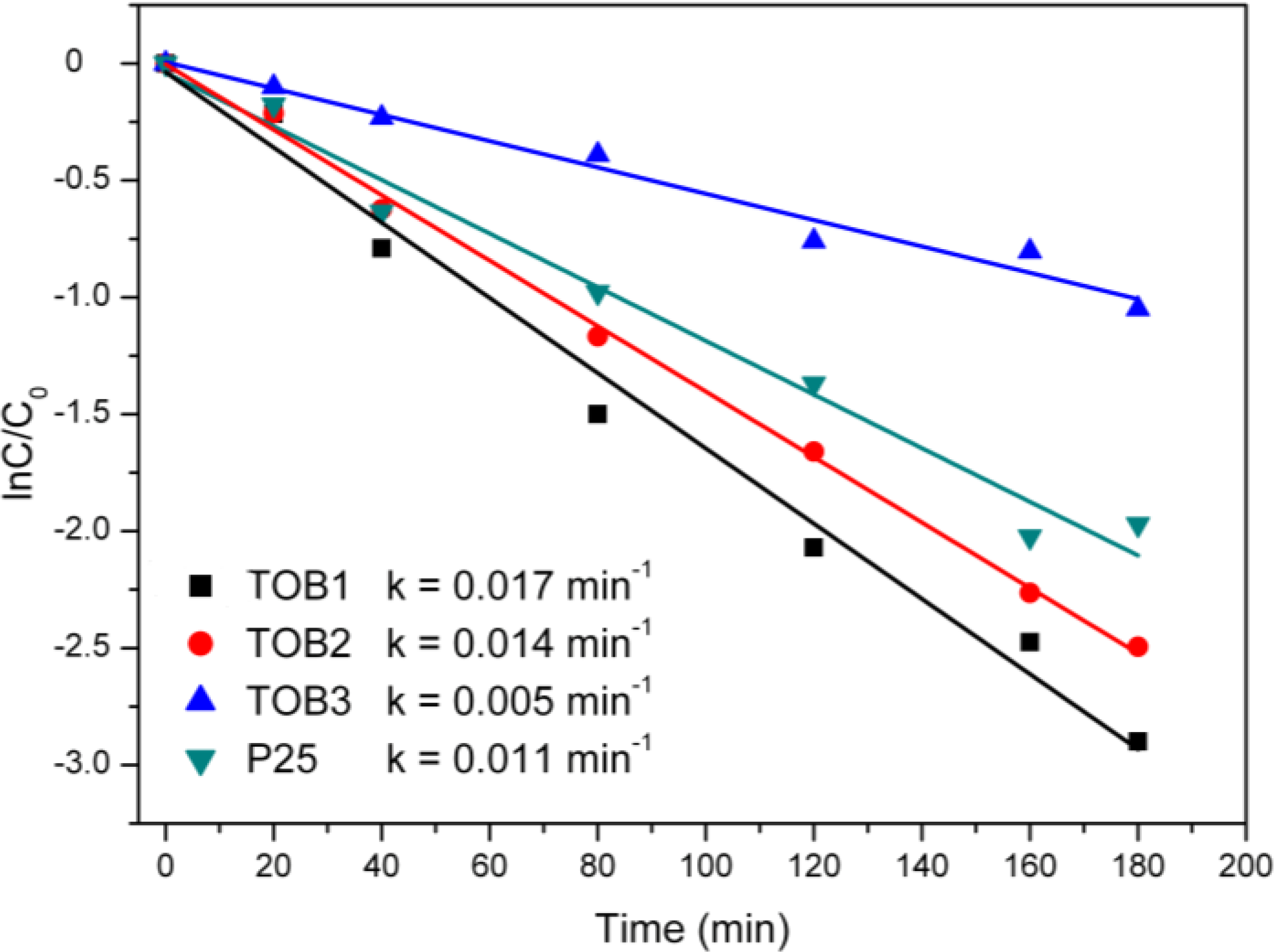
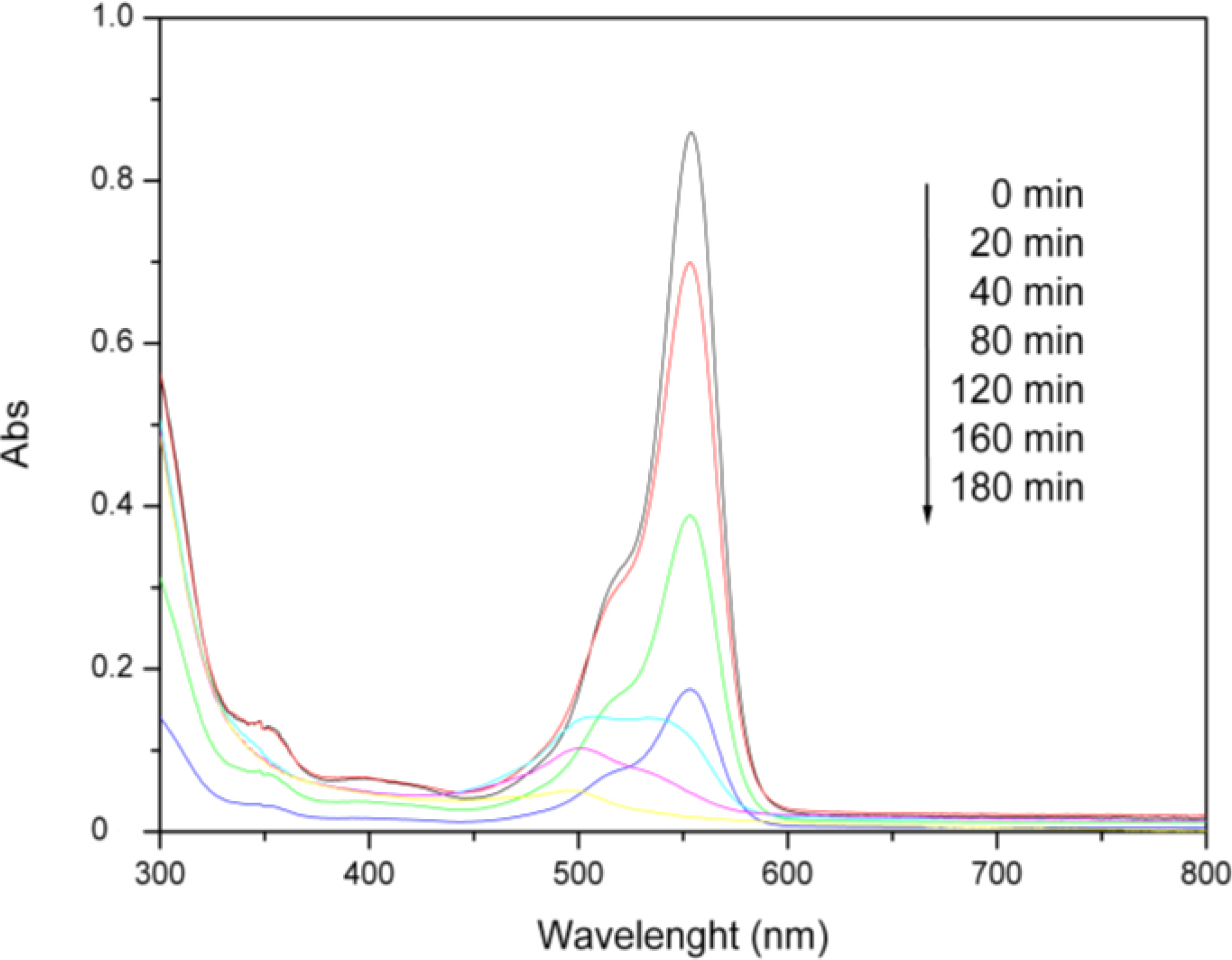
3. Experimental Section
3.1. Synthesis
3.2. Removal of Capping Layer from the TiO2 Samples
3.3. Samples Characterization
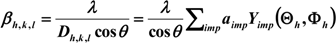
3.4. Photocatalytic Activity Measurement
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References and Notes
- Chen, X.; Mao, S.S. Titanium Dioxide Nanomaterials: Synthesis, Properties, Modifications, and Applications. Chem. Rev. 2007, 107, 2891–2959. [Google Scholar] [CrossRef]
- O’Regan, B.; Gratzel, M. A low-cost, high-efficiency solar cell based on dye-sensitized colloidal TiO2 films. Nature 1991, 353, 737–740. [Google Scholar] [CrossRef]
- Iuchi, K.; Ohko, Y.; Tatsuma, T.; Fujishima, A. Cathode-Separated TiO2 Photocatalysts Applicable to a Photochromic Device Responsive to Backside Illumination. Chem. Mater. 2004, 16, 1165–1167. [Google Scholar] [CrossRef]
- Wagemaker, M.; van de Krol, R.; Kentgens, A.P.M.; van Well, A.A.; Mulder, F.M. Two Phase Morphology Limits Lithium Diffusion in TiO2 (Anatase): A 7Li MAS NMR Study. J. Am. Chem. Soc. 2001, 123, 11454–11461. [Google Scholar]
- Kamat, P.V. Photophysical, Photochemical and Photocatalytic Aspects of Metal Nanoparticles. J. Phys. Chem. B 2002, 106, 7729–7744. [Google Scholar] [CrossRef]
- Gombac, V.; de Rogatis, L.; Gasparotto, A.; Vicario, G.; Montini, T.; Barreca, D.; Balducci, G.; Fornasiero, P.; Tondello, E.; Graziani, M. TiO2 nanopowders doped with boron and nitrogen for photocatalytic applications. Chem. Phys. 2007, 339, 111–123. [Google Scholar] [CrossRef]
- Ling, Q.; Sun, J.; Zhou, Q. Preparation and characterization of visible-light-driven titania photocatalyst co-doped with boron and nitrogen. Appl. Surf. Sci. 2008, 254, 3236–3241. [Google Scholar] [CrossRef]
- Liu, G.; Zhao, Y.; Sun, C.; Li, F.; Lu, G.Q.; Cheng, H.-M. Synergistic Effects of B/N Doping on the Visible-Light Photocatalytic Activity of Mesoporous TiO2. Angew. Chem. Int. Ed. 2008, 47, 4516–4520. [Google Scholar] [CrossRef]
- Umebayashi, T.; Yamaki, T.; Itoh, H.; Asai, K. Band gap narrowing of titanium dioxide by sulfur doping. Appl. Phys. Lett. 2002, 81, 454–456. [Google Scholar] [CrossRef]
- Wang, H.; Lewis, J.P. Effects of dopant states on photoactivity in carbon-doped TiO2. J. Phys. Condens. Matter 2005, 17, L209–L213. [Google Scholar] [CrossRef]
- Asahi, R.; Morikawa, T.; Ohwaki, T.; Aoki, K.; Taga, Y. Visible-Light Photocatalysis in Nitrogen-Doped Titanium Oxides. Science 2001, 293, 269–271. [Google Scholar] [CrossRef]
- Chen, D.; Yang, D.; Wang, Q.; Jiang, Z.Y. Effects of boron doping on photocatalytic activity and microstructure of titanium dioxide nanoparticles. Ind. Eng. Chem. Res. 2006, 45, 4110–4116. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, Q. Preparation and characterization of titania photocatalyst co-doped with boron, nickel and cerium. Mater. Lett. 2008, 62, 2589–2592. [Google Scholar] [CrossRef]
- Zaleska, A.; Sobczak, J.W.; Grabowska, E.; Hupka, J. Preparation and photocatalytic activity of boron-modified TiO2 under UV and visible light. Appl. Catal. B 2008, 78, 92–100. [Google Scholar] [CrossRef]
- De Marco, L.; Manca, M.; Giannuzzi, R.; Malara, F.; Melcarne, G.; Ciccarella, G.; Zama, I.; Cingolani, R.; Gigli, G. Novel Preparation Method of TiO2-Nanorod-Based Photoelectrodes for Dye-Sensitized Solar Cells with Improved Light-Harvesting Efficiency. J. Phys. Chem. C 2010, 114, 4228–4236. [Google Scholar] [CrossRef]
- Ciccarella, G.; Martelli, C.; Matteucci, F.; Zama, I. Process for the Preparation of Titanium Dioxide having Nanometric Dimensions and Controlled Shape. Patent WO2011006659, 20 January 2011. [Google Scholar]
- Melcarne, G.; de Marco, L.; Carlino, E.; Martina, F.; Manca, M.; Cingolani, R.; Gigli, G.; Ciccarella, G. Surfactant-free synthesis of pure anatase TiO2 nanorods suitable for dye-sensitized solar cells. J. Mater. Chem. 2010, 20, 7248–7254. [Google Scholar] [CrossRef]
- Xu, H.; Picca, R.A.; de Marco, L.; Carlucci, C.; Scrascia, A.; Papadia, P.; Scremin, B.F.; Carlino, E.; Giannini, C.; Malitesta, C.; et al. Nonhydrolytic Route to Boron-Doped TiO2 Nanocrystals. Eur. J. Inorg. Chem. 2013, 3, 364–374. [Google Scholar]
- Chen, Z.; Li, W.K.; Zeng, W.J.; Li, M.S.; Xiang, J.H.; Zho, Z.H.; Huang, J.L. Microwave hydrothermal synthesis of nanocrystalline rutile. Mater. Lett. 2008, 62, 4343–4344. [Google Scholar] [CrossRef]
- Monti, D.; Ponrouch, A.; Estruga, M.; Palacín, M.R.; Ayllón, J.A.; Roig, A. Microwaves as a synthetic route for preparing electrochemically active TiO2 nanoparticles. J. Mater. Res. 2013, 28, 340–347. [Google Scholar] [CrossRef]
- Yoon, S.; Lee, E.-S.; Manthiram, A. Microwave-Solvothermal Synthesis of Various Polymorphs of Nanostructured TiO2 in Different Alcohol Media and Their Lithium Ion Storage Properties. Inorg. Chem. 2012, 51, 3505–3512. [Google Scholar] [CrossRef]
- Komarneni, S.; Roy, R.; Li, Q.H. Microwave-hydrothermal synthesis of ceramic powders. Mater. Res. Bull. 1992, 27, 1393–1405. [Google Scholar] [CrossRef]
- Komarneni, S.; Rajha, R.K.; Katsuki, H. Microwave-hydrothermal processing of titanium dioxide. Mater. Chem. Phys. 1999, 61, 50–54. [Google Scholar] [CrossRef]
- Carlucci, C.; Xu, H.; Scremin, B.F.; Giannini, C.; Altamura, D.; Carlino, E.; Videtta, V.; Conciauro, F.; Gigli, G.; Ciccarella, G. Selective synthesis of TiO2 nanocrystals with morphology control with the microwave-solvothermal method. CrystEngComm 2014, 16, 1817–1824. [Google Scholar] [CrossRef]
- Xu, H.; Carlucci, C.; Scremin, B.F.; Giannini, C.; Sibillano, T.; Scrascia, A.; Capodilupo, A.L.; Gigli, G.; Ciccarella, G. Synthesis of Ultrafine Anatase Titanium Dioxide (TiO2) Nanocrystals by the Microwave-Solvothermal Method. J. Nanoeng. Nanomanuf. 2014, 4, 28–32. [Google Scholar] [CrossRef]
- Ohsaka, T.; Izumi, F.; Fujiki, Y. Raman Spectrum of Anatase, TiO2. J. Raman Spectrosc. 1978, 7, 321–324. [Google Scholar] [CrossRef]
- Scremin, B.F.; Belviso, M.R.; Altamura, D.; Giannini, C.; Cozzoli, P.D. Comparative Raman Study of Organic-Free and Surfactant-Capped Rod-Shaped Anatase TiO2 Nanocrystals. Sci. Adv. Mater. 2014, 6, 923–932. [Google Scholar] [CrossRef]
- Georgescu, D.; Baia, L.; Ersen, O.; Baia, M.; Simon, S. Experimental assessment of the phonon confinement in TiO2 anatase nanocrystallites by Raman spectroscopy. J. Raman Spectrosc. 2012, 43, 876–883. [Google Scholar] [CrossRef]
- Thistlethwaite, P.J.; Hook, M.S. Diffuse Reflectance Fourier Transform Infrared Study of the Adsorption of Oleate/Oleic Acid onto Titania. Langmuir 2000, 16, 4993–4998. [Google Scholar] [CrossRef]
- Nara, M.; Torii, H.; Tasumi, M. Correlation between the Vibrational Frequencies of the Carboxylate Group and the Types of Its Coordination to a Metal Ion: An ab Initio Molecular Orbital Study. J. Phys. Chem. 1996, 100, 19812–19817. [Google Scholar] [CrossRef]
- Sing, K.S.W.; Everett, D.H.; Haul, R.A.W.; Moscou, L.; Pierotti, R.A.; Roquerol, J.; Siemieniewska, T. Reporting Physisorption Data for Gas/Solid Systems with Special Reference to the Determination of Surface Area and Porosity. Pure Appl. Chem. 1985, 57, 603–619. [Google Scholar]
- Barka, N.; Qourzal, S.; Assabbane, A.; Nounah, A.; Ichou, Y.A. Factors influencing the photocatalytic degradation of Rhodamine B by TiO2 coated non-woven paper. J. Photochem. Photobiol. A 2008, 195, 346–351. [Google Scholar] [CrossRef]
- Chen, F.; Zhao, J.; Hidaka, H. Highly selective deethylation of rhodamine B: Adsorption and photooxidation pathways of the dye on the TiO2/SiO2 composite photocatalyst. Int. J. Photoenergy 2003, 5, 209–217. [Google Scholar] [CrossRef]
- Li, X.; Ye, J. Photocatalytic Degradation of Rhodamine B over Pb3Nb4O13/Fumed SiO2 Composite under Visible Light Irradiation. J. Phys. Chem. C 2007, 111, 13109–13116. [Google Scholar] [CrossRef]
- Vig, J.R. UV/ozone cleaning of surfaces. J. Vac. Sci. Technol. A 1985, 3, 1027–1034. [Google Scholar] [CrossRef]
- Refinement of powder (Rietveld) and single-crystal diffraction data.
- Jarvinen, M. Application of symmetrized harmonics expansion to correction of the preferred orientation effect. J. Appl. Crystallogr. 1993, 26, 525–531. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Carlucci, C.; Scremin, B.F.; Sibillano, T.; Giannini, C.; Filippo, E.; Perulli, P.; Capodilupo, A.L.; Corrente, G.A.; Ciccarella, G. Microwave-Assisted Synthesis of Boron-Modified TiO2 Nanocrystals. Inorganics 2014, 2, 264-277. https://doi.org/10.3390/inorganics2020264
Carlucci C, Scremin BF, Sibillano T, Giannini C, Filippo E, Perulli P, Capodilupo AL, Corrente GA, Ciccarella G. Microwave-Assisted Synthesis of Boron-Modified TiO2 Nanocrystals. Inorganics. 2014; 2(2):264-277. https://doi.org/10.3390/inorganics2020264
Chicago/Turabian StyleCarlucci, Claudia, Barbara Federica Scremin, Teresa Sibillano, Cinzia Giannini, Emanuela Filippo, Patrizia Perulli, Agostina Lina Capodilupo, Giuseppina Anna Corrente, and Giuseppe Ciccarella. 2014. "Microwave-Assisted Synthesis of Boron-Modified TiO2 Nanocrystals" Inorganics 2, no. 2: 264-277. https://doi.org/10.3390/inorganics2020264
APA StyleCarlucci, C., Scremin, B. F., Sibillano, T., Giannini, C., Filippo, E., Perulli, P., Capodilupo, A. L., Corrente, G. A., & Ciccarella, G. (2014). Microwave-Assisted Synthesis of Boron-Modified TiO2 Nanocrystals. Inorganics, 2(2), 264-277. https://doi.org/10.3390/inorganics2020264