Single- to Triple-Wall WS2 Nanotubes Obtained by High-Power Plasma Ablation of WS2 Multiwall Nanotubes
Abstract
:1. Introduction
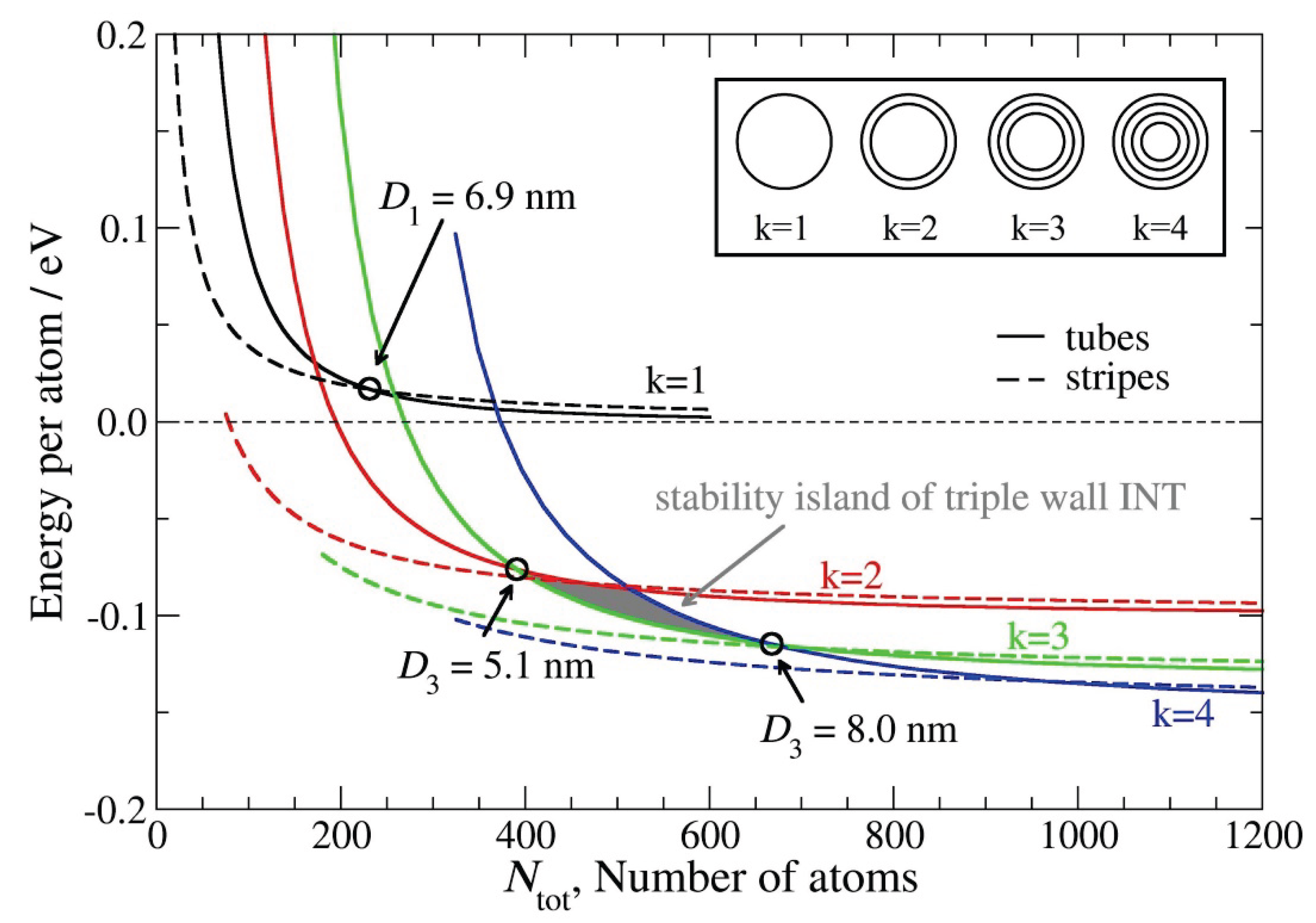
2. Results and Discussion
2.1. Scanning and Transmission Electron Microscopy Analysis



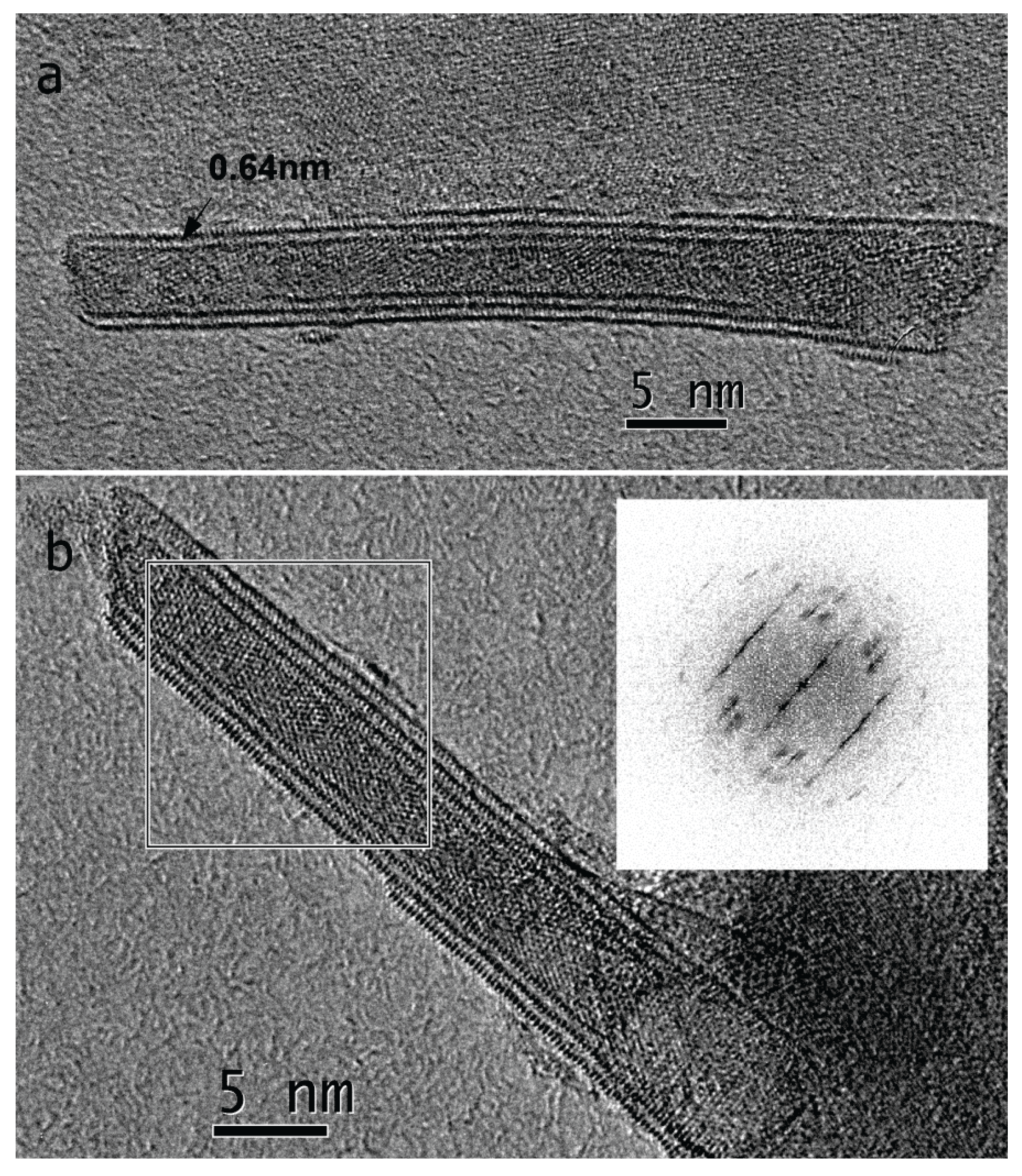
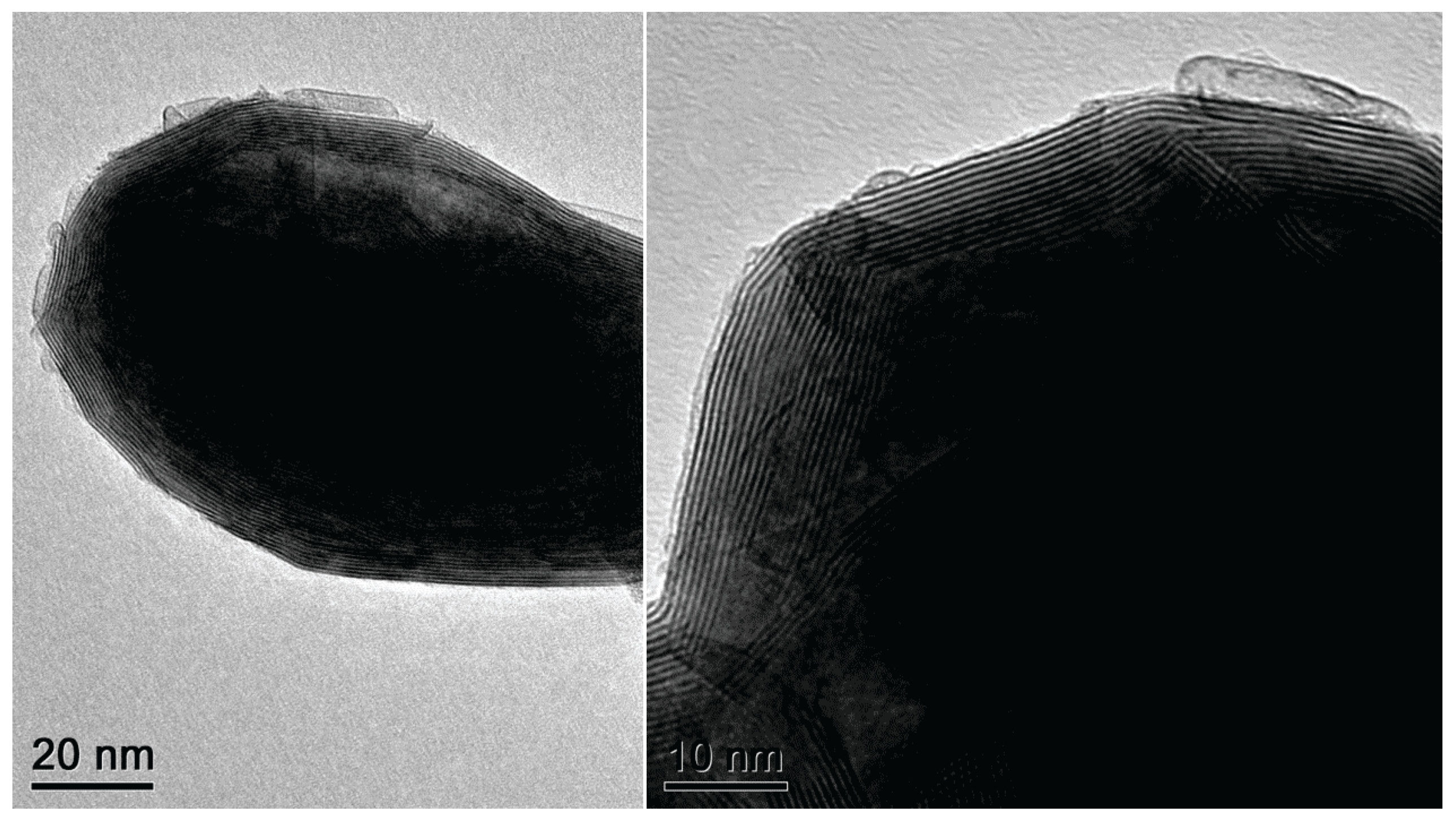
2.2. Growth Mechanism

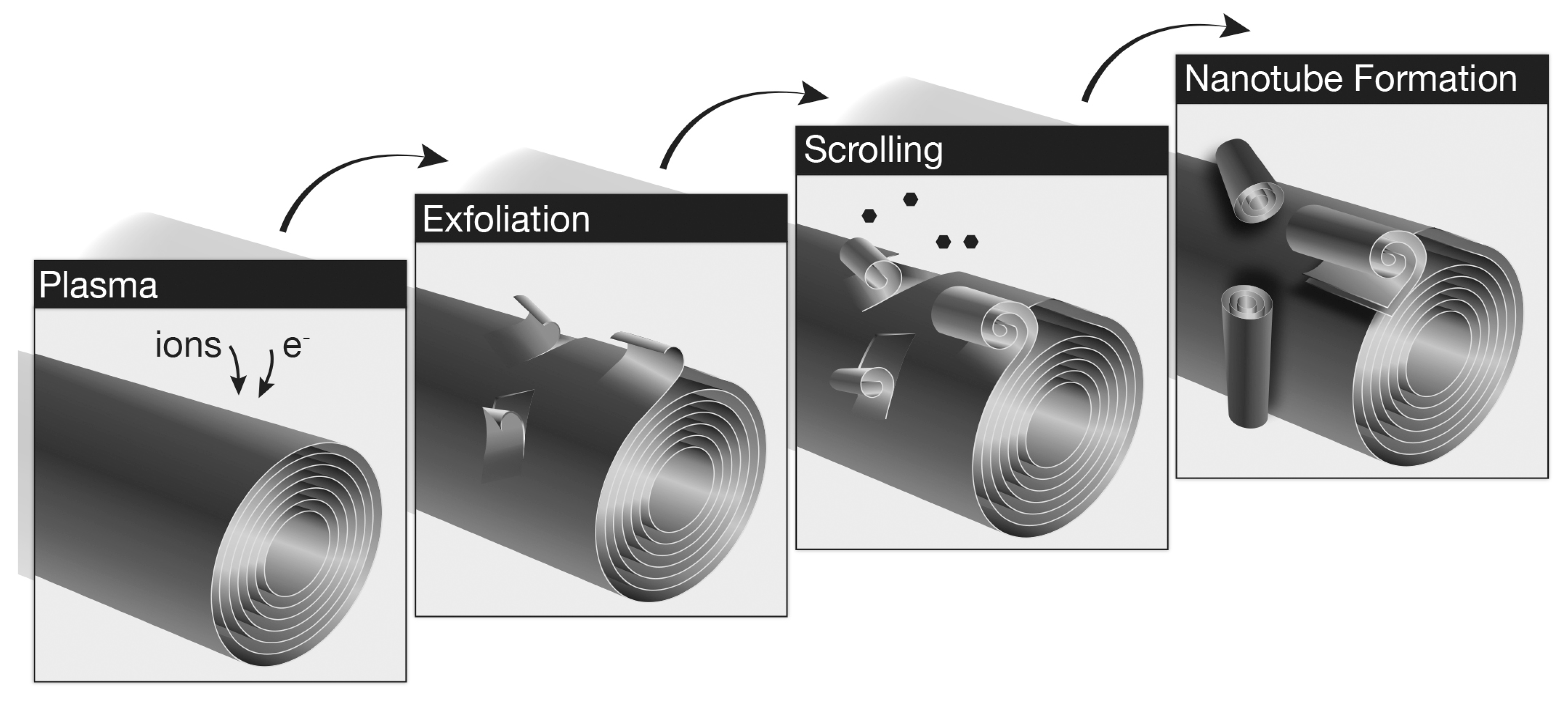

3. Experimental Section
3.1. Plasma Treatment
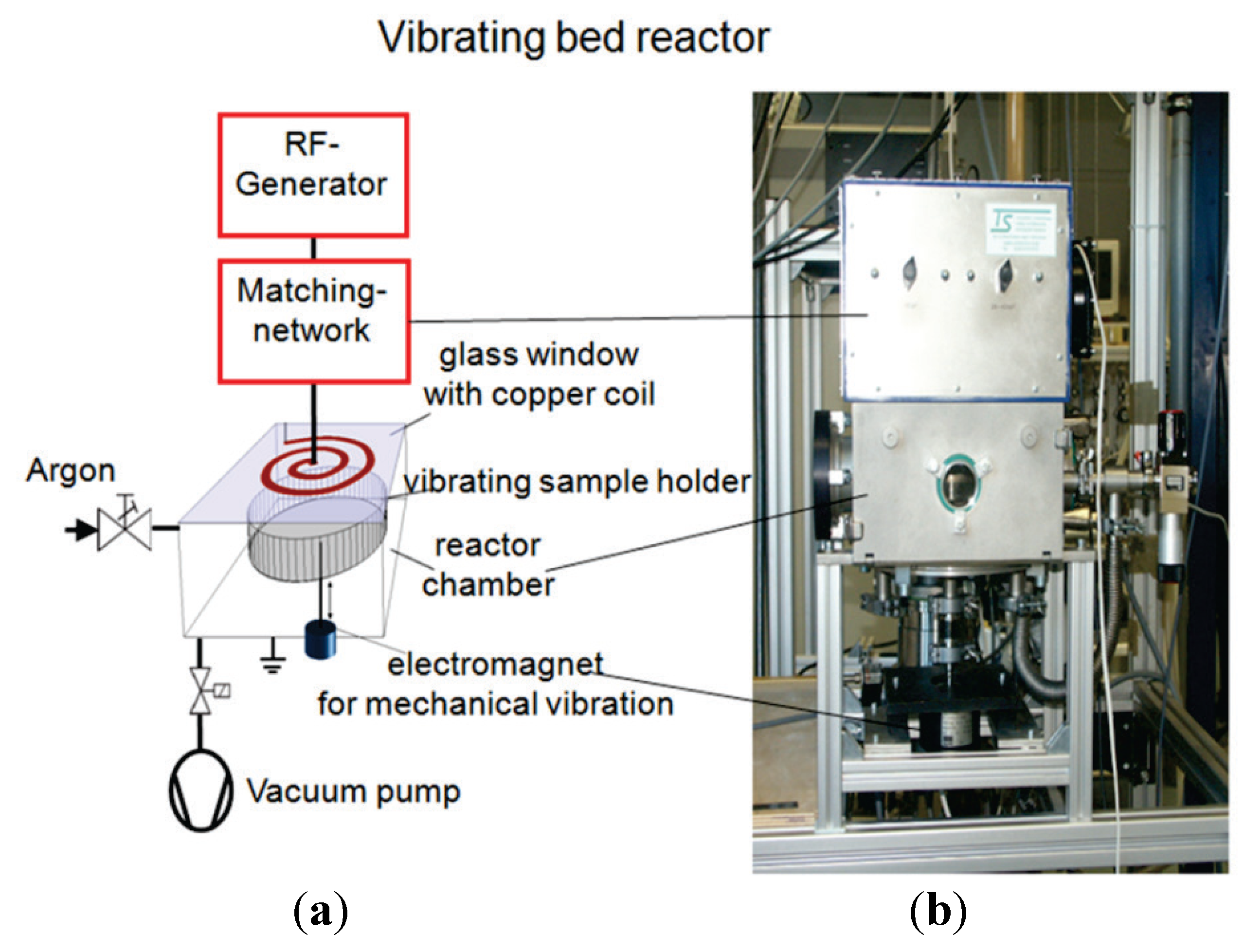
3.2. Electron Microscopy
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interests
References
- Tenne, R.; Margulis, L.; Genut, M.; Hodes, G. Polyhedral and cylindrical structures of tungsten disulphide. Nature 1992, 360, 444–446. [Google Scholar] [CrossRef]
- Zak, A.; Sallacan-Ecker, L.; Margolin, A.; Genut, M.; Tenne, R. Insight into the growth mechanism of WS2 nanotubes in the scaled-up fluidized-bed reactor. Nano 2009, 4, 91–98. [Google Scholar] [CrossRef]
- Chopra, N.G.; Luyken, J.; Cherry, K.; Crespi, V.H.; Cohen, M.L.; Louie, S.G.; Zettl, A. Boron Nitride Nanotubes. Science 1995, 269, 966–967. [Google Scholar] [CrossRef]
- Feldman, Y.; Wasserman, E.; Srolovitz, D.J.; Tenne, R. High-rate, gas-phase growth of MoS2 nested inorganic fullerenes and nanotubes. Science 1995, 267, 222–225. [Google Scholar] [CrossRef]
- Remskar, M.; Skraba, Z.; Cleton, F.; Sanjines, R.; Levy, F. MoS2 as microtubes. Appl. Phys. Lett. 1996, 69, 351–353. [Google Scholar] [CrossRef]
- Rubio, A.; Corkill, J.F.; Cohen, M.L. Theory of graphitic boron nitride nanotubes. Phys. Rev. B 1994, 49, 5081–5084. [Google Scholar] [CrossRef]
- Seifert, G.; Terrones, H.; Terrones, M.; Jungnickel, G.; Frauenheim, T. Structure and electronic properties of MoS2 nanotubes. Phys. Rev. Lett. 2000, 85, 146–149. [Google Scholar] [CrossRef]
- Linnolahti, M.; Pakkanen, T.A. Quantum chemical treatment of large nanotubes via use of line group symmetry: Structural preferences of magnesium dichloride nanotubes. J. Phys. Chem. B 2006, 110, 4675–4678. [Google Scholar] [CrossRef]
- Seifert, G.; Köhler, T.; Tenne, R. Stability of metal chalcogenide nanotubes. J. Phys. Chem. B 2002, 106, 2497–2501. [Google Scholar] [CrossRef]
- Therese, H.A.; Li, J.; Kolb, U.; Tremel, W. Facile large scale synthesis of WS2 nanotubes from WO3 nanorods prepared by a hydrothermal route. Solid State Sci. 2005, 7, 67–72. [Google Scholar] [CrossRef]
- Li, Y.D.; Li, X.L.; He, R.R.; Zhu, J.; Deng, Z.X. Artificial lamellar mesostructures to WS2 nanotubes. J. Am. Chem. Soc. 2002, 124, 1411–1416. [Google Scholar] [CrossRef]
- Sen, R.; Govindaraj, A.; Suenaga, K.; Suzuki, S.; Kataura, H.; Iijima, S.; Achiba, Y. Encapsulated and hollow closed-cage structures of WS2 and MoS2 prepared by laser ablation at 450–1050 °C. Chem. Phys. Lett. 2001, 340, 242–248. [Google Scholar] [CrossRef]
- Margulis, L.; Salitra, G.; Tenne, R.; Talianker, M. Nested fullerene-like structures. Nature 1993, 365, 113–114. [Google Scholar]
- Tenne, R. Doped and Heteroatom-Containing Fullerene-like Structures and Nanotubes. Adv. Mater. 1995, 7, 965–995. [Google Scholar] [CrossRef]
- Singh, D.M.D.J.; Pradeep, T.; Bhattacharjee, J.; Waghmare, U.V. Novel cage clusters of MoS(2) in the gas phase. J. Phys. Chem. A 2005, 109, 7339–7342. [Google Scholar]
- Parilla, P.A.; Dillon, A.C.; Jones, K.M.; Riker, G.; Schulz, D.L.; Ginley, D.S.; Heben, M.J. The first true inorganic fullerenes? Nature 1999, 397, 114. [Google Scholar] [CrossRef]
- Enyashin, A.N.; Gemming, S.; Bar-Sadan, M.; Popovitz-Biro, R.; Hong, S.Y.; Prior, Y.; Tenne, R.; Seifert, G. Structure and stability of molybdenum sulfide fullerenes. Angew. Chem. Int. Ed. 2007, 46, 623–627. [Google Scholar] [CrossRef]
- Enyashin, A.N.; Bar-Sadan, M.; Sloan, J.; Houben, L.; Seifert, G. Nanoseashells and nanooctahedra of MoS2: Routes to inorganic fullerenes. Chem. Mater. 2009, 21, 5627–5636. [Google Scholar] [CrossRef]
- Albu-Yaron, A.; Levy, M.; Tenne, R.; Popovitz-Biro, R.; Weidenbach, M.; Bar-Sadan, M.; Houben, L.; Enyashin, A.N.; Seifert, G.; Feuermann, D.; et al. MoS2 hybrid nanostructures: from octahedral to quasi-spherical shells within individual nanoparticles. Angew. Chem. Int. Ed. 2011, 123, 1850–1854. [Google Scholar] [CrossRef]
- Alexandrou, I.; Sano, N.; Burrows, A.; Meyer, R.R.; Wang, H.; Kirkland, A.I.; Kiely, C.J.; Amaratunga, G.A.J. Structural investigation of MoS2 core–shell nanoparticles formed by an arc discharge in water. Nanotechnology 2003, 14, 913–917. [Google Scholar] [CrossRef]
- Gordon, J.M.; Katz, E.A.; Feuermann, D.; Albu-Yaron, A.; Levy, M.; Tenne, R. Singular MoS2, SiO2 and Si nanostructures—synthesis by solar ablation. J. Mater. Chem. 2003, 18, 458–462. [Google Scholar]
- Journet, C.; Maser, W.K.; Bernier, P.; Loiseau, A.; de-la Chapelle, M.L.; Lefrant, S.; Deniard, P.; Lee, R.; Fischer, J.E. Large-scale production of single-walled carbon nanotubes by the electric-arc technique. Nature 1997, 388, 756–758. [Google Scholar] [CrossRef]
- Qin, L.-C.; Zhao, X.; Hirahara, K.; Miyamoto, Y.; Ando, Y.; Iijima, S. Materials science: The smallest carbon nanotube. Nature 2000, 408, 50. [Google Scholar]
- Wang, N.; Tang, Z.K.; Li, G.D.; Chen, J.S. Materials science: Single-walled 4 Å carbon nanotube arrays. Nature 2000, 408, 50–51. [Google Scholar]
- Hernandez, E.; Goze, C.; Bernier, P.; Rubio, A. Elastic properties of Cand BxCyNz composite nanotubes. Phys. Rev. Lett. 1998, 80, 4502–4505. [Google Scholar] [CrossRef]
- Lorenz, T.; Teich, D.; Joswig, J.-O.; Seifert, G. Theoretical study of the mechanical behavior of individual TiS2 and MoS2 nanotubes. J. Phys. Chem. C 2012, 116, 11714–11721. [Google Scholar] [CrossRef]
- Koshland, D.E. Stereochemistry and the mechanism of enzymatic reactions. Biolog. Rev. 1953, 28, 416–436. [Google Scholar] [CrossRef]
- Herrmann, I.; Brüser, V.; Fiechter, S.; Kersten, H.; Bogdanoff, P. Electrocatalysts for oxygen reduction prepared by plasma treatment of carbon-supported cobalt tetramethoxyphenylporphyrin. J. Electrochem. Soc. 2005, 152, A2179–A2185. [Google Scholar] [CrossRef]
- Meichsner, J.; Schmidt, M.; Schneider, R.; Wagner, H.E. Nonthermal Plasma Chemistry and Physics; CRC Press: Boca Raton, FL, USA, 2013; Section 3.8.7; pp. 120–121. [Google Scholar]
- Savastenko, N.A.; Müller, S.; Anklam, K.; Brüser, M.; Quade, A.; Walter, C.; Brüser, V. Effect of plasma treatment on the properties of Fe-based electrocatalysts. Surf. Coat. Technol. 2011, 205, 439–442. [Google Scholar] [CrossRef]
- Kortshagen, U. Nonthermal plasma synthesis of semiconductor nanocrystals. J. Phys. D Appl. Phys. 2009, 42, 113001. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Brüser, V.; Popovitz-Biro, R.; Albu-Yaron, A.; Lorenz, T.; Seifert, G.; Tenne, R.; Zak, A. Single- to Triple-Wall WS2 Nanotubes Obtained by High-Power Plasma Ablation of WS2 Multiwall Nanotubes. Inorganics 2014, 2, 177-190. https://doi.org/10.3390/inorganics2020177
Brüser V, Popovitz-Biro R, Albu-Yaron A, Lorenz T, Seifert G, Tenne R, Zak A. Single- to Triple-Wall WS2 Nanotubes Obtained by High-Power Plasma Ablation of WS2 Multiwall Nanotubes. Inorganics. 2014; 2(2):177-190. https://doi.org/10.3390/inorganics2020177
Chicago/Turabian StyleBrüser, Volker, Ronit Popovitz-Biro, Ana Albu-Yaron, Tommy Lorenz, Gotthard Seifert, Reshef Tenne, and Alla Zak. 2014. "Single- to Triple-Wall WS2 Nanotubes Obtained by High-Power Plasma Ablation of WS2 Multiwall Nanotubes" Inorganics 2, no. 2: 177-190. https://doi.org/10.3390/inorganics2020177






