Rewiring of Developing Spinal Nociceptive Circuits by Neonatal Injury and Its Implications for Pediatric Chronic Pain
Abstract
:1. Introduction
2. Spinal Mechanisms Contributing to Central Sensitization
3. Neonatal Injury “Primes” Developing Nociceptive Circuits in the CNS
4. Neonatal Tissue Damage Shapes Synaptic Integration in Adult Spinal Projection Neurons
5. Long-Term Potentiation at Sensory Synapses onto Spinal Projection Neurons
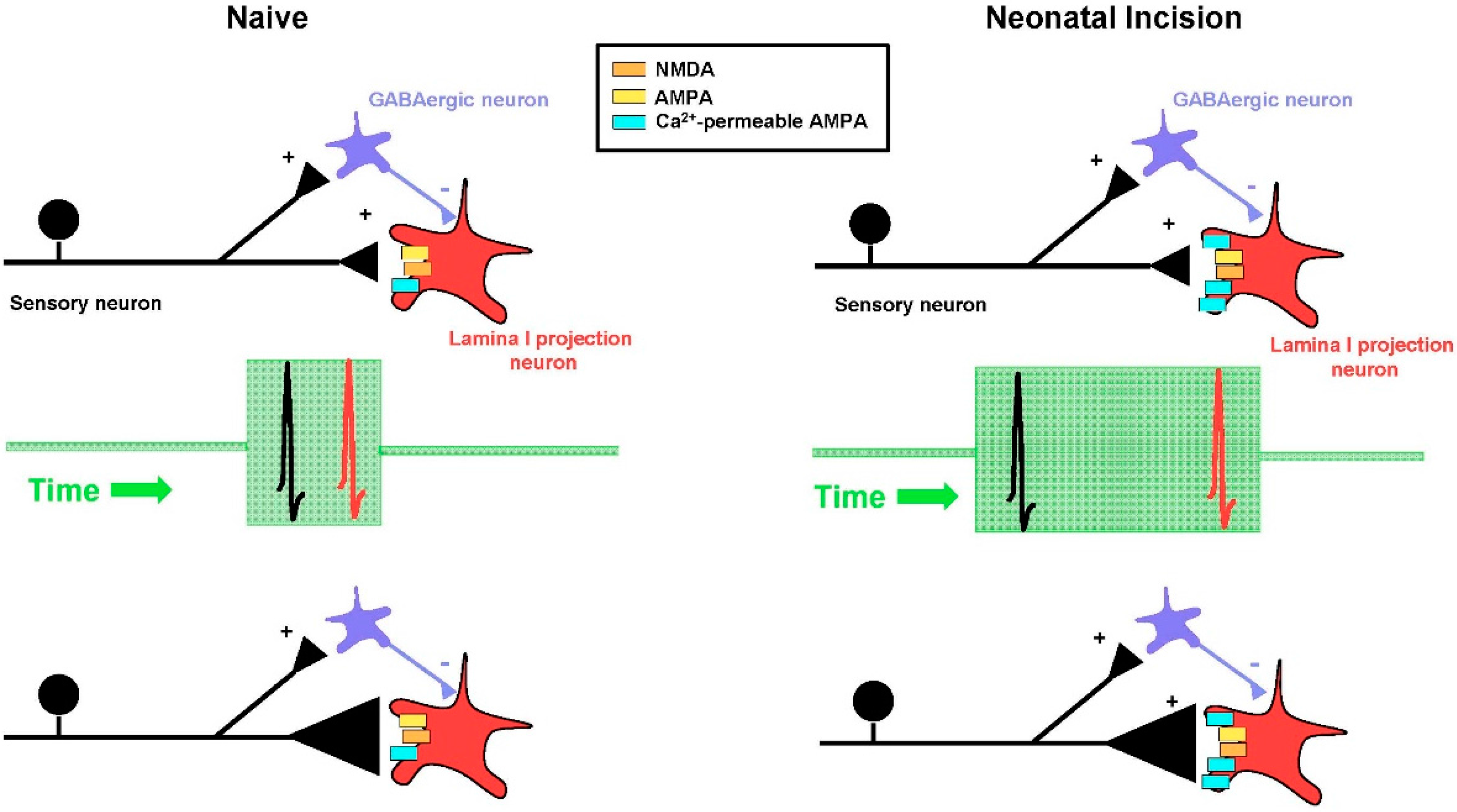
6. Neonatal Injury Relaxes the Timing Rules Governing Spike Timing-Dependent LTP in Adult Spinal Pain Circuits
7. Future Directions
Acknowledgments
Conflicts of Interest
References
- Todd, A.J. Neuronal circuitry for pain processing in the dorsal horn. Nat. Rev. Neurosci. 2010, 11, 823–836. [Google Scholar] [CrossRef] [PubMed]
- Sherman, S.E.; Loomis, C.W. Morphine insensitive allodynia is produced by intrathecal strychnine in the lightly anesthetized rat. Pain 1994, 56, 17–29. [Google Scholar] [CrossRef]
- Sivilotti, L.; Woolf, C.J. The contribution of GABAA and glycine receptors to central sensitization: Disinhibition and touch-evoked allodynia in the spinal cord. J. Neurophysiol. 1994, 72, 169–179. [Google Scholar] [PubMed]
- Yaksh, T.L. Behavioral and autonomic correlates of the tactile evoked allodynia produced by spinal glycine inhibition: Effects of modulatory receptor systems and excitatory amino acid antagonists. Pain 1989, 37, 111–123. [Google Scholar] [CrossRef]
- Torsney, C.; MacDermott, A.B. Disinhibition opens the gate to pathological pain signaling in superficial neurokinin 1 receptor-expressing neurons in rat spinal cord. J. Neurosci. 2006, 26, 1833–1843. [Google Scholar] [CrossRef] [PubMed]
- Peirs, C.; Williams, S.P.; Zhao, X.; Walsh, C.E.; Gedeon, J.Y.; Cagle, N.E.; Goldring, A.C.; Hioki, H.; Liu, Z.; Marell, P.S.; et al. Dorsal Horn Circuits for Persistent Mechanical Pain. Neuron 2015, 87, 797–812. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Dong, H.; Gao, Y.; Gong, Y.; Ren, Y.; Gu, N.; Zhou, S.; Xia, N.; Sun, Y.Y.; Ji, R.R.; et al. A feed-forward spinal cord glycinergic neural circuit gates mechanical allodynia. J. Clin. Investig. 2013, 123, 4050–4062. [Google Scholar] [CrossRef] [PubMed]
- Petitjean, H.; Pawlowski, S.A.; Fraine, S.L.; Sharif, B.; Hamad, D.; Fatima, T.; Berg, J.; Brown, C.M.; Jan, L.Y.; Ribeiro-da-Silva, A.; et al. Dorsal Horn Parvalbumin Neurons Are Gate-Keepers of Touch-Evoked Pain after Nerve Injury. Cell Rep. 2015, 13, 1246–1257. [Google Scholar] [CrossRef] [PubMed]
- Duan, B.; Cheng, L.; Bourane, S.; Britz, O.; Padilla, C.; Garcia-Campmany, L.; Krashes, M.; Knowlton, W.; Velasquez, T.; Ren, X.; et al. Identification of spinal circuits transmitting and gating mechanical pain. Cell 2014, 159, 1417–1432. [Google Scholar] [CrossRef] [PubMed]
- Woolf, C.J. Central sensitization: Implications for the diagnosis and treatment of pain. Pain 2011, 152 (Suppl. 3), S2–S15. [Google Scholar] [CrossRef] [PubMed]
- Latremoliere, A.; Woolf, C.J. Central sensitization: A generator of pain hypersensitivity by central neural plasticity. J. Pain 2009, 10, 895–926. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, S.; Lippross, S.; Neuhuber, W.L.; Zeilhofer, H.U. PGE(2) selectively blocks inhibitory glycinergic neurotransmission onto rat superficial dorsal horn neurons. Nat. Neurosci. 2002, 5, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Foster, E.; Wildner, H.; Tudeau, L.; Haueter, S.; Ralvenius, W.T.; Jegen, M.; Johannssen, H.; Hosli, L.; Haenraets, K.; Ghanem, A.; et al. Targeted ablation, silencing, and activation establish glycinergic dorsal horn neurons as key components of a spinal gate for pain and itch. Neuron 2015, 85, 1289–1304. [Google Scholar] [CrossRef] [PubMed]
- Muller, F.; Heinke, B.; Sandkühler, J. Reduction of glycine receptor-mediated miniature inhibitory postsynaptic currents in rat spinal lamina I neurons after peripheral inflammation. Neuroscience 2003, 122, 799–805. [Google Scholar] [CrossRef] [PubMed]
- Harvey, R.J.; Depner, U.B.; Wassle, H.; Ahmadi, S.; Heindl, C.; Reinold, H.; Smart, T.G.; Harvey, K.; Schutz, B.; Bo-Salem, O.M.; et al. GlyR alpha3: An essential target for spinal PGE2-mediated inflammatory pain sensitization. Science 2004, 304, 884–887. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Acuna, M.A.; Yevenes, G.E.; Ralvenius, W.T.; Benke, D.; Di, L.A.; Lara, C.O.; Munoz, B.; Burgos, C.F.; Moraga-Cid, G.; Corringer, P.J.; et al. Phosphorylation state-dependent modulation of spinal glycine receptors alleviates inflammatory pain. J. Clin. Investig. 2016, 126, 2547–2560. [Google Scholar] [CrossRef] [PubMed]
- Malmberg, A.B.; Chen, C.; Tonegawa, S.; Basbaum, A.I. Preserved acute pain and reduced neuropathic pain in mice lacking PKCgamma. Science 1997, 278, 279–283. [Google Scholar] [CrossRef] [PubMed]
- Doyon, N.; Vinay, L.; Prescott, S.A.; De Koninck, Y. Chloride Regulation: A Dynamic Equilibrium Crucial for Synaptic Inhibition. Neuron 2016, 89, 1157–1172. [Google Scholar] [CrossRef] [PubMed]
- Ben-Ari, Y. Excitatory actions of gaba during development: The nature of the nurture. Nat. Rev. Neurosci. 2002, 3, 728–739. [Google Scholar] [CrossRef] [PubMed]
- Rohrbough, J.; Spitzer, N.C. Regulation of intracellular Cl− levels by Na(+)-dependent Cl− cotransport distinguishes depolarizing from hyperpolarizing GABAA receptor-mediated responses in spinal neurons. J. Neurosci. 1996, 16, 82–91. [Google Scholar] [PubMed]
- Ehrlich, I.; Lohrke, S.; Friauf, E. Shift from depolarizing to hyperpolarizing glycine action in rat auditory neurones is due to age-dependent Cl− regulation. J. Physiol. 1999, 520 Pt 1, 121–137. [Google Scholar] [CrossRef] [PubMed]
- Rivera, C.; Voipio, J.; Payne, J.A.; Ruusuvuori, E.; Lahtinen, H.; Lamsa, K.; Pirvola, U.; Saarma, M.; Kaila, K. The K+/Cl− co-transporter KCC2 renders GABA hyperpolarizing during neuronal maturation. Nature 1999, 397, 251–255. [Google Scholar] [PubMed]
- Coull, J.A.M.; Beggs, S.; Boudreau, D.; Boivin, D.; Tsuda, M.; Inoue, K.; Gravel, C.; Salter, M.W.; De Koninck, Y. BDNF from microglia causes the shift in neuronal anion gradient underlying neuropathic pain. Nature 2005, 438, 1017–1021. [Google Scholar] [CrossRef] [PubMed]
- Coull, J.A.; Boudreau, D.; Bachand, K.; Prescott, S.A.; Nault, F.; Sik, A.; De Koninck, P.; De Koninck, Y. Trans-synaptic shift in anion gradient in spinal lamina I neurons as a mechanism of neuropathic pain. Nature 2003, 424, 938–942. [Google Scholar] [CrossRef] [PubMed]
- Hubner, C.A.; Stein, V.; Hermans-Borgmeyer, I.; Meyer, T.; Ballanyi, K.; Jentsch, T.J. Disruption of KCC2 reveals an essential role of K-Cl cotransport already in early synaptic inhibition. Neuron 2001, 30, 515–524. [Google Scholar] [CrossRef]
- Cordero-Erausquin, M.; Coull, J.A.; Boudreau, D.; Rolland, M.; De Koninck, Y. Differential maturation of GABA action and anion reversal potential in spinal lamina I neurons: Impact of chloride extrusion capacity. J. Neurosci. 2005, 25, 9613–9623. [Google Scholar] [CrossRef] [PubMed]
- Baccei, M.L.; Fitzgerald, M. Development of GABAergic and glycinergic transmission in the neonatal rat dorsal horn. J. Neurosci. 2004, 24, 4749–4757. [Google Scholar] [CrossRef] [PubMed]
- Schwaller, F.; Fitzgerald, M. The consequences of pain in early life: Injury-induced plasticity in developing pain pathways. Eur. J. Neurosci. 2014, 39, 344–352. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Baccei, M.L. Excitatory synapses in the rat superficial dorsal horn are strengthened following peripheral inflammation during early postnatal development. Pain 2009, 143, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Baccei, M.L. Neonatal tissue damage facilitates nociceptive synaptic input to the developing superficial dorsal horn via NGF-dependent mechanisms. Pain 2011, 152, 1846–1855. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Walker, S.M.; Fitzgerald, M.; Baccei, M.L. Activity-dependent modulation of glutamatergic signaling in the developing rat dorsal horn by early tissue injury. J. Neurophysiol. 2009, 102, 2208–2219. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Xie, W.; Zhang, J.M.; Baccei, M.L. Peripheral nerve injury sensitizes neonatal dorsal horn neurons to tumor necrosis factor-alpha. Mol. Pain 2009, 5, 10. [Google Scholar] [CrossRef] [PubMed]
- McKelvey, R.; Berta, T.; Old, E.; Ji, R.R.; Fitzgerald, M. Neuropathic pain is constitutively suppressed in early life by anti-inflammatory neuroimmune regulation. J. Neurosci. 2015, 35, 457–466. [Google Scholar] [CrossRef] [PubMed]
- Vega-Avelaira, D.; McKelvey, R.; Hathway, G.; Fitzgerald, M. The emergence of adolescent onset pain hypersensitivity following neonatal nerve injury. Mol. Pain 2012, 8, 30. [Google Scholar] [CrossRef] [PubMed]
- Hermann, C.; Hohmeister, J.; Demirakca, S.; Zohsel, K.; Flor, H. Long-term alteration of pain sensitivity in school-aged children with early pain experiences. Pain 2006, 125, 278–285. [Google Scholar] [CrossRef] [PubMed]
- Hohmeister, J.; Kroll, A.; Wollgarten-Hadamek, I.; Zohsel, K.; Demirakca, S.; Flor, H.; Hermann, C. Cerebral processing of pain in school-aged children with neonatal nociceptive input: An exploratory fMRI study. Pain 2010, 150, 257–267. [Google Scholar] [CrossRef] [PubMed]
- Walker, S.M.; Franck, L.S.; Fitzgerald, M.; Myles, J.; Stocks, J.; Marlow, N. Long-term impact of neonatal intensive care and surgery on somatosensory perception in children born extremely preterm. Pain 2009, 141, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Newnham, C.A.; Inder, T.E.; Milgrom, J. Measuring preterm cumulative stressors within the NICU: The Neonatal Infant Stressor Scale. Early Hum. Dev. 2009, 85, 549–555. [Google Scholar] [CrossRef] [PubMed]
- Hohmann, A.G.; Neely, M.H.; Pina, J.; Nackley, A.G. Neonatal chronic hind paw inflammation alters sensitization to intradermal capsaicin in adult rats: A behavioral and immunocytochemical study. J. Pain 2005, 6, 798–808. [Google Scholar] [CrossRef] [PubMed]
- Chu, Y.C.; Chan, K.H.; Tsou, M.Y.; Lin, S.M.; Hsieh, Y.C.; Tao, Y.X. Mechanical pain hypersensitivity after incisional surgery is enhanced in rats subjected to neonatal peripheral inflammation: Effects of N-methyl-d-aspartate receptor antagonists. Anesthesiology 2007, 106, 1204–1212. [Google Scholar] [CrossRef] [PubMed]
- Ren, K.; Anseloni, V.; Zou, S.P.; Wade, E.B.; Novikova, S.I.; Ennis, M.; Traub, R.J.; Gold, M.S.; Dubner, R.; Lidow, M.S. Characterization of basal and re-inflammation-associated long-term alteration in pain responsivity following short-lasting neonatal local inflammatory insult. Pain 2004, 110, 588–596. [Google Scholar] [CrossRef] [PubMed]
- Walker, S.M.; Tochiki, K.K.; Fitzgerald, M. Hindpaw incision in early life increases the hyperalgesic response to repeat surgical injury: Critical period and dependence on initial afferent activity. Pain 2009, 147, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Beggs, S.; Currie, G.; Salter, M.W.; Fitzgerald, M.; Walker, S.M. Priming of adult pain responses by neonatal pain experience: Maintenance by central neuroimmune activity. Brain 2012, 135 Pt 2, 404–417. [Google Scholar] [CrossRef] [PubMed]
- Ren, K.; Novikova, S.I.; He, F.; Dubner, R.; Lidow, M.S. Neonatal local noxious insult affects gene expression in the spinal dorsal horn of adult rats. Mol. Pain 2005, 1, 27. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Blankenship, M.L.; Baccei, M.L. Deficits in glycinergic inhibition within adult spinal nociceptive circuits after neonatal tissue damage. Pain 2013, 154, 1129–1139. [Google Scholar] [CrossRef] [PubMed]
- Farrant, M.; Nusser, Z. Variations on an inhibitory theme: Phasic and tonic activation of GABA(A) receptors. Nat. Rev. Neurosci. 2005, 6, 215–229. [Google Scholar] [CrossRef] [PubMed]
- Takazawa, T.; MacDermott, A.B. Glycinergic and GABAergic tonic inhibition fine tune inhibitory control in regionally distinct subpopulations of dorsal horn neurons. J. Physiol. 2010, 588 Pt 14, 2571–2587. [Google Scholar] [CrossRef] [PubMed]
- Bice, T.N.; Beal, J.A. Quantitative and neurogenic analysis of neurons with supraspinal projections in the superficial dorsal horn of the rat lumbar spinal cord. J. Comp. Neurol. 1997, 388, 565–574. [Google Scholar] [CrossRef]
- Bice, T.N.; Beal, J.A. Quantitative and neurogenic analysis of the total population and subpopulations of neurons defined by axon projection in the superficial dorsal horn of the rat lumbar spinal cord. J. Comp. Neurol. 1997, 388, 550–564. [Google Scholar] [CrossRef]
- Dahlhaus, A.; Ruscheweyh, R.; Sandkühler, J. Synaptic input of rat spinal lamina I projection and unidentified neurones in vitro. J. Physiol. 2005, 566 Pt 2, 355–368. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, H.; Heinke, B.; Ruscheweyh, R.; Sandkühler, J. Synaptic plasticity in spinal lamina I projection neurons that mediate hyperalgesia. Science 2003, 299, 1237–1240. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Kritzer, E.; Craig, P.E.; Baccei, M.L. Aberrant synaptic integration in adult lamina I projection neurons following neonatal tissue damage. J. Neurosci. 2015, 35, 2438–2451. [Google Scholar] [CrossRef] [PubMed]
- Nakatsuka, T.; Chen, M.; Takeda, D.; King, C.; Ling, J.; Xing, H.; Ataka, T.; Vierck, C.; Yezierski, R.; Gu, J.G. Substance P-driven feed-forward inhibitory activity in the mammalian spinal cord. Mol. Pain 2005, 1, 20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, J.; Baccei, M.L. Neonatal tissue injury reduces the intrinsic excitability of adult mouse superficial dorsal horn neurons. Neuroscience 2014, 256, 392–402. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, H.; Stark, J.; Fischer, H.; Wagner, M.; Drdla, R.; Jager, T.; Sandkühler, J. Synaptic amplifier of inflammatory pain in the spinal dorsal horn. Science 2006, 312, 1659–1662. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Baccei, M.L. Developmental regulation of membrane excitability in rat spinal lamina I projection neurons. J. Neurophysiol. 2012, 107, 2604–2614. [Google Scholar] [CrossRef] [PubMed]
- Ruscheweyh, R.; Ikeda, H.; Heinke, B.; Sandkühler, J. Distinctive membrane and discharge properties of rat spinal lamina I projection neurones in vitro. J. Physiol. 2004, 555 Pt 2, 527–543. [Google Scholar] [CrossRef] [PubMed]
- Ruscheweyh, R.; Wilder-Smith, O.; Drdla, R.; Liu, X.G.; Sandkühler, J. Long-term potentiation in spinal nociceptive pathways as a novel target for pain therapy. Mol. Pain 2011, 7, 20. [Google Scholar] [CrossRef] [PubMed]
- Sandkühler, J.; Liu, X. Induction of long-term potentiation at spinal synapses by noxious stimulation or nerve injury. Eur. J. Neurosci. 1998, 10, 2476–2480. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.J.; Ren, W.J.; Zhong, Y.; Yang, T.; Wei, X.H.; Xin, W.J.; Liu, C.C.; Zhou, L.H.; Li, Y.Y.; Liu, X.G. Limited BDNF contributes to the failure of injury to skin afferents to produce a neuropathic pain condition. Pain 2010, 148, 148–157. [Google Scholar] [CrossRef] [PubMed]
- Hathway, G.J.; Vega-Avelaira, D.; Moss, A.; Ingram, R.; Fitzgerald, M. Brief, low frequency stimulation of rat peripheral C-fibres evokes prolonged microglial-induced central sensitization in adults but not in neonates. Pain 2009, 144, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.C.; Zhang, Y.Q.; Zhao, Z.Q. Involvement of nitric oxide in long-term potentiation of spinal nociceptive responses in rats. Neuroreport 2005, 16, 1197–1201. [Google Scholar] [CrossRef] [PubMed]
- Klein, T.; Magerl, W.; Hopf, H.C.; Sandkühler, J.; Treede, R.D. Perceptual correlates of nociceptive long-term potentiation and long-term depression in humans. J Neurosci. 2004, 24, 964–971. [Google Scholar] [CrossRef] [PubMed]
- Klein, T.; Magerl, W.; Nickel, U.; Hopf, H.C.; Sandkühler, J.; Treede, R.D. Effects of the NMDA-receptor antagonist ketamine on perceptual correlates of long-term potentiation within the nociceptive system. Neuropharmacology 2007, 52, 655–661. [Google Scholar] [CrossRef] [PubMed]
- Schilder, A.; Magerl, W.; Hoheisel, U.; Klein, T.; Treede, R.D. Electrical high-frequency stimulation of the human thoracolumbar fascia evokes long-term potentiation-like pain amplification. Pain 2016. [Google Scholar] [CrossRef] [PubMed]
- Dan, Y.; Poo, M.M. Spike timing-dependent plasticity of neural circuits. Neuron 2004, 44, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Larsen, R.S.; Rao, D.; Manis, P.B.; Philpot, B.D. STDP in the Developing Sensory Neocortex. Front. Synaptic. Neurosci. 2010, 2, 9. [Google Scholar] [CrossRef] [PubMed]
- Feldman, D.E. The spike-timing dependence of plasticity. Neuron 2012, 75, 556–571. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Baccei, M.L. Neonatal Tissue Damage Promotes Spike Timing-Dependent Synaptic Long-Term Potentiation in Adult Spinal Projection Neurons. J. Neurosci. 2016, 36, 5405–5416. [Google Scholar] [CrossRef] [PubMed]
- Mayer, M.L.; Westbrook, G.L.; Guthrie, P.B. Voltage-dependent block by Mg2+ of NMDA responses in spinal cord neurones. Nature 1984, 309, 261–263. [Google Scholar] [CrossRef] [PubMed]
- Koester, H.J.; Sakmann, B. Calcium dynamics in single spines during coincident pre- and postsynaptic activity depend on relative timing of back-propagating action potentials and subthreshold excitatory postsynaptic potentials. Proc. Natl. Acad. Sci. USA 1998, 95, 9596–9601. [Google Scholar] [CrossRef] [PubMed]
- Cho, K.; Aggleton, J.P.; Brown, M.W.; Bashir, Z.I. An experimental test of the role of postsynaptic calcium levels in determining synaptic strength using perirhinal cortex of rat. J. Physiol. 2001, 532 Pt 2, 459–466. [Google Scholar] [CrossRef] [PubMed]
- Cormier, R.J.; Greenwood, A.C.; Connor, J.A. Bidirectional synaptic plasticity correlated with the magnitude of dendritic calcium transients above a threshold. J. Neurophysiol. 2001, 85, 399–406. [Google Scholar] [PubMed]
- Yang, S.N.; Tang, Y.G.; Zucker, R.S. Selective induction of LTP and LTD by postsynaptic [Ca2+]i elevation. J. Neurophysiol. 1999, 81, 781–787. [Google Scholar] [PubMed]
- Hollmann, M.; Hartley, M.; Heinemann, S. Ca2+ permeability of KA-AMPA-gated glutamate receptor channels depends on subunit composition. Science 1991, 252, 851–853. [Google Scholar] [CrossRef] [PubMed]
- Washburn, M.S.; Numberger, M.; Zhang, S.; Dingledine, R. Differential dependence on GluR2 expression of three characteristic features of AMPA receptors. J. Neurosci. 1997, 17, 9393–9406. [Google Scholar] [PubMed]
- Youn, D.H.; Royle, G.; Kolaj, M.; Vissel, B.; Randic, M. Enhanced LTP of primary afferent neurotransmission in AMPA receptor GluR2-deficient mice. Pain 2008, 136, 158–167. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, B.; Ahmadi, S.; Heppenstall, P.A.; Lewin, G.R.; Schott, C.; Borchardt, T.; Seeburg, P.H.; Zeilhofer, H.U.; Sprengel, R.; Kuner, R. The AMPA receptor subunits GluR-A and GluR-B reciprocally modulate spinal synaptic plasticity and inflammatory pain. Neuron 2004, 44, 637–650. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Dissing-Olesen, L.; MacVicar, B.A.; Stevens, B. Microglia: Dynamic Mediators of Synapse Development and Plasticity. Trends Immunol. 2015, 36, 605–613. [Google Scholar] [CrossRef] [PubMed]
- Clark, A.K.; Gruber-Schoffnegger, D.; Drdla-Schutting, R.; Gerhold, K.J.; Malcangio, M.; Sandkühler, J. Selective activation of microglia facilitates synaptic strength. J. Neurosci. 2015, 35, 4552–4570. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the author; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baccei, M.L. Rewiring of Developing Spinal Nociceptive Circuits by Neonatal Injury and Its Implications for Pediatric Chronic Pain. Children 2016, 3, 16. https://doi.org/10.3390/children3030016
Baccei ML. Rewiring of Developing Spinal Nociceptive Circuits by Neonatal Injury and Its Implications for Pediatric Chronic Pain. Children. 2016; 3(3):16. https://doi.org/10.3390/children3030016
Chicago/Turabian StyleBaccei, Mark L. 2016. "Rewiring of Developing Spinal Nociceptive Circuits by Neonatal Injury and Its Implications for Pediatric Chronic Pain" Children 3, no. 3: 16. https://doi.org/10.3390/children3030016




