A Laboratory Assessment of Factors That Affect Bacterial Adhesion to Contact Lenses
Abstract
:1. Introduction
2. Experimental Section
| Proprietary name | ACUVUE® 2 | ACUVUE® OASYS™ |
|---|---|---|
| United States Adopted Name (USAN) | etafilcon A | senofilcon A |
| Manufacturer | Johnson & Johnson | Johnson & Johnson |
| Water content (%) | 58 | 38 |
| Oxygen Permeability (Dk) | 21 | 103 |
| Centre thickness (mm) - 3.00 Ds | 0.08 | 0.07 |
| Oxygen Transmissibility (Dk/t) at 35 °C | 25 | 147 |
| FDA group | IV | I |
| Surface treatment | None | No surface treatment. Internal wetting agent (PVP) that also coats the surface |
| Principal monomers | HEMA + MA | mPDMS + DMA + HEMA + siloxane macromer + PVP + TEGDMA |
2.1. Bacterial Strains
2.2. Assay Media
2.3. Incubation Period
2.4. Inoculum Size
2.5. Adhesion Conditions
2.6. Statistics
3. Results and Discussion
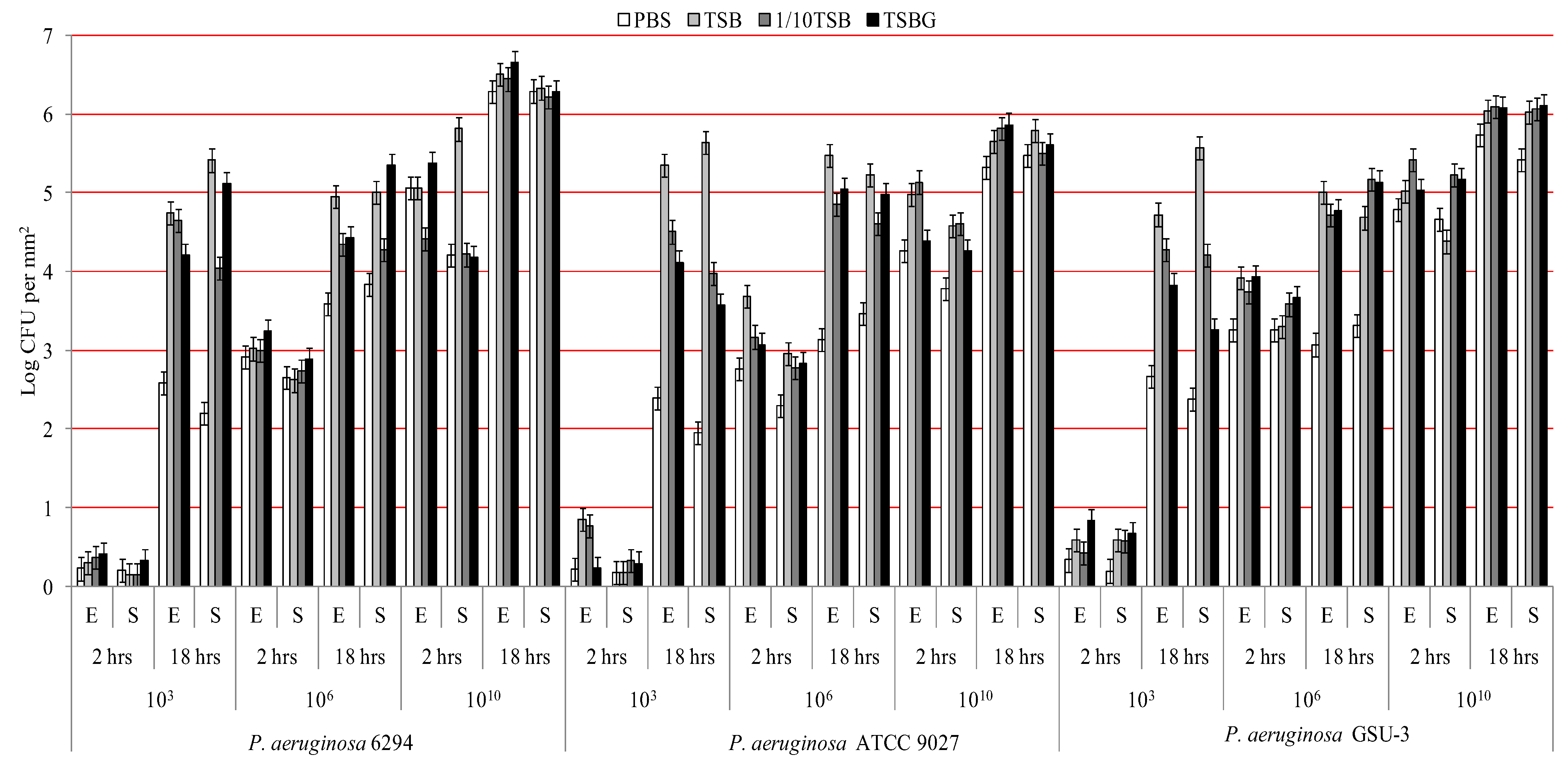

| Influencing factor for bacterial adhesion | Partial Eta squared | |
|---|---|---|
| P. aeruginosa | S. aureus | |
| Inoculum size | 0.75 | 0.43 |
| Incubation period | 0.64 | 0.37 |
| Assay media | 0.19 | 0.54 |
| Type of lens | 0.01 | 0.01 |
| Bacterial strains | 0.02 | 0.00 |
| Bacteria* | Inoculum size (CFU mL−1) | Media | Incubation time (hours) | etafilcon A | senofilcon A | ||||
| Estimated mean (CFU mm-2) | 95% Confidence Interval (CFU mm−2) | Estimated mean (CFU mm−2) | 95% Confidence Interval (CFU mm−2) | ||||||
| Lower Bound | Upper Bound | Lower Bound | Upper Bound | ||||||
| P aeruginosa 6294 | 1010 | 1/10 TSB | 2 | 4.4 | 4.3 | 4.6 | 4.2 | 4.1 | 4.4 |
| 106 | 1/10 TSB | 18 | 4.3 | 4.2 | 4.5 | 4.3 | 4.1 | 4.4 | |
| 106 | PBS | 18 | 3.6 | 3.4 | 3.7 | 3.8 | 3.7 | 4.0 | |
| 103 | 1/10 TSB | 18 | 4.7 | 4.5 | 4.8 | 4.0 | 3.9 | 4.2 | |
| P. aeruginosa ATCC 9027 | 1010 | TSBG | 2 | 4.4 | 4.2 | 4.5 | 4.3 | 4.1 | 4.4 |
| 1010 | PBS | 2 | 4.3 | 4.1 | 4.4 | 3.8 | 3.6 | 3.9 | |
| 106 | 1/10 TSB | 18 | 4.9 | 4.7 | 5.0 | 4.6 | 4.5 | 4.8 | |
| 106 | PBS | 18 | 3.1 | 3.0 | 3.3 | 3.5 | 3.3 | 3.6 | |
| 103 | 1/10 TSB | 18 | 4.5 | 4.4 | 4.7 | 4.0 | 3.8 | 4.1 | |
| 103 | TSBG | 18 | 4.1 | 4.0 | 4.3 | 3.6 | 3.4 | 3.7 | |
| P. aeruginosa GSU-3 | 1010 | PBS | 2 | 4.8 | 4.6 | 4.9 | 4.7 | 4.5 | 4.8 |
| 106 | 1/10 TSB | 2 | 3.7 | 3.6 | 3.9 | 3.6 | 3.4 | 3.7 | |
| 106 | TSBG | 2 | 3.9 | 3.8 | 4.1 | 3.7 | 3.5 | 3.8 | |
| 106 | PBS | 2 | 3.3 | 3.1 | 3.4 | 3.3 | 3.1 | 3.4 | |
| 106 | PBS | 18 | 3.1 | 2.9 | 3.2 | 3.3 | 3.2 | 3.5 | |
| 106 | TSB | 2 | 3.9 | 3.8 | 4.1 | 3.3 | 3.2 | 3.4 | |
| 106 | 1/10 TSB | 18 | 4.3 | 4.1 | 4.4 | 4.2 | 4.1 | 4.4 | |
| 103 | TSBG | 18 | 3.8 | 3.7 | 4.0 | 3.3 | 3.1 | 3.4 | |
| S. aureus 31 | 1010 | 1/10 TSB | 2 | 3.7 | 3.5 | 3.8 | 3.3 | 3.2 | 3.5 |
| 1010 | TSBG | 2 | 3.5 | 3.3 | 3.6 | 3.3 | 3.2 | 3.5 | |
| 1010 | TSB | 2 | 4.2 | 4.1 | 4.4 | 3.5 | 3.4 | 3.7 | |
| 1010 | TSB | 2 | 4.2 | 4.1 | 4.4 | 3.5 | 3.4 | 3.7 | |
| 106 | 1/10 TSB | 18 | 4.5 | 4.4 | 4.7 | 4.4 | 4.3 | 4.6 | |
| S. aureus 38 | 1010 | 1/10 TSB | 18 | 4.3 | 4.2 | 4.5 | 4.2 | 4.1 | 4.4 |
| 1010 | TSBG | 18 | 4.3 | 4.2 | 4.5 | 4.2 | 4.1 | 4.4 | |
| 1010 | TSB | 2 | 3.3 | 3.2 | 3.4 | 3.2 | 3.0 | 3.3 | |
| 106 | 1/10 TSB | 18 | 4.2 | 4.1 | 4.4 | 4.4 | 4.3 | 4.5 | |
| 103 | 1/10 TSB | 18 | 4.1 | 4.0 | 4.3 | 3.9 | 3.8 | 4.0 | |
| 103 | TSB | 18 | 4.4 | 4.3 | 4.6 | 4.1 | 4.0 | 4.3 | |
| 103 | TSBG | 18 | 4.0 | 3.8 | 4.1 | 3.6 | 3.5 | 3.8 | |
Conflict of Interest
References
- Green, M.; Apel, A.; Stapleton, F. Risk factors and causative organisms in microbial keratitis. Cornea 2008, 27, 22–27. [Google Scholar] [CrossRef]
- Holden, B.A.; La Hood, D.; Grant, T.; Newton-Howes, J.; Baleriola-Lucas, C.; Willcox, M.D.; Sweeney, D.F. Gram-negative bacteria can induce contact lens related acute red eye (CLARE) responses. CLAO J. 1996, 22, 47–52. [Google Scholar]
- Wu, P.; Stapleton, F.; Willcox, M.D. The causes of and cures for contact lens-induced peripheral ulcer. Eye Contact Lens 2003, 29, S63–S66. [Google Scholar]
- Willcox, M.; Sharma, S.; Naduvilath, T.J.; Sankaridurg, P.R.; Gopinathan, U.; Holden, B.A. External ocular surface and lens microbiota in contact lens wearers with corneal infiltrates during extended wear of hydrogel lenses. Eye Contact Lens 2011, 37, 90–95. [Google Scholar] [CrossRef]
- Otri, A.M.; Fares, U.; Al-Aqaba, M.A.; Miri, A.; Faraj, L.A.; Said, D.G.; Maharajan, S.; Dua, H.S. Profile of sight-threatening infectious keratitis: A prospective study. Acta ophthalmologica 2012. [Google Scholar]
- Tuli, S.S.; Iyer, S.A.; Driebe, W.T., Jr. Fungal keratitis and contact lenses: An old enemy unrecognized or a new nemesis on the block? Eye Contact Lens 2007, 33, 415–417. [Google Scholar] [CrossRef]
- Yoder, J.S.; Verani, J.; Heidman, N.; Hoppe-Bauer, J.; Alfonso, E.C.; Miller, D.; Jones, D.B.; Bruckner, D.; Langston, R.; Jeng, B.H.; et al. Acanthamoeba keratitis: the persistence of cases following a multistate outbreak. Ophthalmic Epidemiol. 2012, 19, 221–225. [Google Scholar] [CrossRef]
- Keay, L.; Edwards, K.; Naduvilath, T.; Forde, K.; Stapleton, F. Factors affecting the morbidity of contact lens-related microbial keratitis: A population study. Invest. Ophthalmol. Vis. Sci. 2006, 47, 4302–4308. [Google Scholar] [CrossRef] [Green Version]
- Dutta, D.; Cole, N.; Willcox, M. Factors influencing bacterial adhesion to contact lenses. Mol. Vis. 2012, 18, 14–21. [Google Scholar] [CrossRef]
- Giraldez, M.J.; Resua, C.G.; Lira, M.; Oliveira, M.E.; Magarinos, B.; Toranzo, A.E.; Yebra-Pimentel, E. Contact lens hydrophobicity and roughness effects on bacterial adhesion. Optom. Vis. Sci. 2010, 87, E426–E431. [Google Scholar]
- Willcox, M.D.P.; Hume, E.B.H.; Aliwarga, Y.; Kumar, N.; Cole, N. A novel cationic-peptide coating for the prevention of microbial colonization on contact lenses. J. Appl. Microbiol. 2008, 105, 1817–1825. [Google Scholar] [CrossRef]
- Garcia-Saenz, M.C.; Arias-Puente, A.; Fresnadillo-Martinez, M.J.; Paredes-Garcia, B. Adherence of two strains of Staphylococcus epidermidis to contact lenses. Cornea 2002, 21, 511–515. [Google Scholar] [CrossRef]
- Vermeltfoort, P.B.; Rustema-Abbing, M.; de Vries, J.; Bruinsma, G.M.; Busscher, H.J.; van der Linden, M.L.; Hooymans, J.M.; van der Mei, H.C. Influence of day and night wear on surface properties of silicone hydrogel contact lenses and bacterial adhesion. Cornea 2006, 25, 516–523. [Google Scholar] [CrossRef]
- Mathews, S.M.; Spallholz, J.E.; Grimson, M.J.; Dubielzig, R.R.; Gray, T.; Reid, T.W. Prevention of bacterial colonization of contact lenses with covalently attached selenium and effects on the rabbit cornea. Cornea 2006, 25, 806–814. [Google Scholar]
- Kodjikian, L.; Casoli-Bergeron, E.; Malet, F.; Janin-Manificat, H.; Freney, J.; Burillon, C.; Colin, J.; Steghens, J.P. Bacterial adhesion to conventional hydrogel and new silicone-hydrogel contact lens materials. Graefes Arch. Clin. Exp. Ophthalmol. 2008, 246, 267–273. [Google Scholar] [CrossRef]
- Williams, T.J.; Schneider, R.P.; Willcox, M.D. The effect of protein-coated contact lenses on the adhesion and viability of gram negative bacteria. Curr. Eye Res. 2003, 27, 227–235. [Google Scholar] [CrossRef]
- George, M.; Ahearn, D.; Pierce, G.; Gabriel, M. Interactions of Pseudomonas aeruginosa and Staphylococcus epidermidis in adhesion to a hydrogel. Eye Contact Lens 2003, 29, S105–S109; Discussion S115–S118, S192–S194. [Google Scholar]
- Dutta, D.; Cole, N.; Kumar, N.; Willcox, M.D. Broad spectrum antimicrobial activity of melimine covalently bound to contact lenses. Invest. Ophthalmol. Vis. Sci 2013, 54, 175–182. [Google Scholar] [CrossRef]
- Borazjani, R. Relative primary adhesion of Pseudomonas aeruginosa, Serratia marcescens and Staphylococcus aureus to HEMA-type contact lenses and an extended wear silicone hydrogel contact lens of high oxygen permeability. Cont. Lens Anterior Eye 2004, 27, 3–8. [Google Scholar] [CrossRef]
- Bandara, B.M.K.; Sankaridurg, P.R.; Willcox, M.D.P. Non-steroidal anti inflammatory agents decrease bacterial colonisation of contact lenses and prevent adhesion to human corneal epithelial cells. Curr. Eye Res. 2004, 29, 245–251. [Google Scholar] [CrossRef]
- Willcox, M.D.; Hume, E.B.; Vijay, A.K.; Petcavich, R. Ability of silver-impregnated contact lenses to control microbial growth colonisation. J. Optom. 2010, 3, 143–148. [Google Scholar] [CrossRef]
- Randler, C.; Matthes, R.; McBain, A.; Giese, B.; Fraunholz, M.; Sietmann, R.; Kohlmann, T.; Hubner, N.; Kramer, A. A three-phase in-vitro system for studying Pseudomonas aeruginosa adhesion and biofilm formation upon hydrogel contact lenses. BMC Microbiol. 2010, 9, 12. [Google Scholar]
- McLaughlin-Borlace, L.; Stapleton, F.; Matheson, M.; Dart, J.K. Bacterial biofilm on contact lenses and lens storage cases in wearers with microbial keratitis. J. Appl. Microbiol. 1998, 84, 827–838. [Google Scholar]
- Efron, N.; Morgan, P.B.; Woods, C.A. Survey of contact lens prescribing to infants, children, and teenagers. Optom. Vis. Sci. 2011, 88, 461–468. [Google Scholar] [CrossRef]
- Fleiszig, S.M.J.; WienerKronish, J.P.; Miyazaki, H.; Vallas, V.; Mostov, K.E.; Kanada, D.; Sawa, T.; Yen, T.S.B.; Frank, D.W. Pseudomonas aeruginosa-mediated cytotoxicity and invasion correlate with distinct genotypes at the loci encoding exoenzyme S. Infec. Immunity 1997, 65, 579–586. [Google Scholar]
- Schubert, T.L.; Hume, E.B.; Willcox, M.D. Staphylococcus aureus ocular isolates from symptomatic adverse events: antibiotic resistance and similarity of bacteria causing adverse events. Clin. Exp. Optom. 2008, 91, 148–155. [Google Scholar] [CrossRef]
- Lakkis, C.; Fleiszig, S.M. Resistance of Pseudomonas aeruginosa isolates to hydrogel contact lens disinfection correlates with cytotoxic activity. J. Clin. Microbiol. 2001, 39, 1477–1486. [Google Scholar]
- Parment, P.A.; Gabriel, M.; Bruse, G.W.; Stegall, S.; Ahearn, D.G. Adherence of Serratia marcescens, Serratia liquefaciens, Pseudomonas aeruginosa and Staphylococcus epidermidis to blood transfusion bags (CPD-SAGMAN sets). Scand. J. Infec. Dis. 1993, 25, 721–724. [Google Scholar] [CrossRef]
- Henriques, M.; Sousa, C.; Lira, M.; Elisabete, M.; Oliveira, R.; Azeredo, J. Adhesion of Pseudomonas aeruginosa and Staphylococcus epidermidis to silicone-hydrogel contact lenses. Optom. Vis. Sci 2005, 82, 446–450. [Google Scholar] [CrossRef] [Green Version]
- Subbaraman, L.N.; Borazjani, R.; Zhu, H.; Zhao, Z.; Jones, L.; Willcox, M.D. Influence of protein deposition on bacterial adhesion to contact lenses. Optom. Vis. Sci. 2011, 88, 959–966. [Google Scholar] [CrossRef]
- Miller, M.J.; Ahearn, D.G. Adherence of Pseudomonas aeruginosa to hydrophilic contact lenses and other substrata. J. Clin. Microbiol. 1987, 25, 1392–1397. [Google Scholar]
- Klotz, S.A.; Butrus, S.I.; Misra, R.P.; Osato, M.S. The contribution of bacterial surface hydrophobicity to the process of adherence of Pseudomonas aeruginosa to hydrophilic contact lenses. Curr. Eye Res. 1989, 8, 195–202. [Google Scholar]
- Vijay, A.K.; Zhu, H.; Ozkan, J.; Wu, D.; Masoudi, S.; Bandara, R.; Borazjani, R.N.; Willcox, M.D. Bacterial adhesion to unworn and worn silicone hydrogel lenses. Optom. Vis. Sci. 2012, 89, 1095–1106. [Google Scholar] [CrossRef]
- Zhang, S.; Borazjani, R.N.; Salamone, J.C.; Ahearn, D.G.; Crow, S.A., Jr.; Pierce, G.E. In vitro deposition of lysozyme on etafilcon A and balafilcon A hydrogel contact lenses: Effects on adhesion and survival of Pseudomonas aeruginosa and Staphylococcus aureus. Cont. Lens Anterior Eye 2005, 28, 113–119. [Google Scholar] [CrossRef]
- Tran, V.B.; Fleiszig, S.M.; Evans, D.J.; Radke, C.J. Dynamics of flagellum- and pilus-mediated association of Pseudomonas aeruginosa with contact lens surfaces. Appl. Environ. Microbiol. 2011, 77, 3644–3652. [Google Scholar] [CrossRef]
- Bruinsma, G.M.; van der Mei, H.C.; Busscher, H.J. Bacterial adhesion to surface hydrophilic and hydrophobic contact lenses. Biomaterials 2001, 22, 3217–3224. [Google Scholar] [CrossRef]
- Andrews, C.S.; Denyer, S.P.; Hall, B.; Hanlon, G.W.; Lloyd, A.W. A comparison of the use of an ATP-based bioluminescent assay and image analysis for the assessment of bacterial adhesion to standard HEMA and biomimetic soft contact lenses. Biomaterials 2001, 22, 3225–3233. [Google Scholar] [CrossRef]
- Szczotka-Flynn, L.B.; Imamura, Y.; Chandra, J.; Yu, C.; Mukherjee, P.K.; Pearlman, E.; Ghannoum, M.A. Increased resistance of contact lens-related bacterial biofilms to antimicrobial activity of soft contact lens care solutions. Cornea 2009, 28, 918–926. [Google Scholar] [CrossRef]
- Donlan, R.M.; Costerton, J.W. Biofilms: Survival mechanisms of clinically relevant microorganisms. Clin. Microbiol. Rev. 2002, 15, 167–193. [Google Scholar] [CrossRef]
- Evans, D.J.; Allison, D.G.; Brown, M.R.; Gilbert, P. Effect of growth-rate on resistance of gram-negative biofilms to cetrimide. J. Antimicrob. Chemother. 1990, 26, 473–478. [Google Scholar] [CrossRef]
- Stapleton, F.; Dart, J.; Matheson, M.; Woodward, E. Bacterial adherence and glycocalyx formation on unworn hydrogel lenses. J. Brit. Contact Lens Assoc. 1993, 16, 113–116. [Google Scholar] [CrossRef]
- Williams, T.J.; Willcox, M.D.; Schneider, R.P. Interactions of bacteria with contact lenses: The effect of soluble protein and carbohydrate on bacterial adhesion to contact lenses. Optom. Vis. Sci. 1998, 75, 266–271. [Google Scholar] [CrossRef]
- Miller, M.J.; Wilson, L.A.; Ahearn, D.G. Effects of protein, mucin, and human tears on adherence of Pseudomonas aeruginosa to hydrophilic contact lenses. J. Clin. Microbiol. 1988, 26, 513–517. [Google Scholar]
- Willcox, M.D.; Carnt, N.; Diec, J.; Naduvilath, T.; Evans, V.; Stapleton, F.; Iskandar, S.; Harmis, N.; de la Jara, P.L.; Holden, B.A. Contact lens case contamination during daily wear of silicone hydrogels. Optom. Vis. Sci. 2010, 87, 456–464. [Google Scholar]
- Dantam, J.; Zhu, H.; Willcox, M.; Ozkan, J.; Naduvilath, T.; Thomas, V.; Stapleton, F. In vivo assessment of antimicrobial efficacy of silver-impregnated contact lens storage cases. Invest. Ophthalmol. Vis. Sci. 2012, 53, 1641–1648. [Google Scholar] [CrossRef]
- Pens, C.J.; da Costa, M.; Fadanelli, C.; Caumo, K.; Rott, M. Acanthamoeba spp. and bacterial contamination in contact lens storage cases and the relationship to user profiles. Parasitol. Res. 2008, 103, 1241–1245. [Google Scholar] [CrossRef]
- Fleiszig, S.M.; Efron, N. Microbial flora in eyes of current and former contact lens wearers. J. Clin. Microbiol. 1992, 30, 1156–1161. [Google Scholar]
- Donzis, P.B.; Mondino, B.J.; Weissman, B.A.; Bruckner, D.A. Microbial contamination of contact lens care systems. Am. J. Ophthalmol. 1987, 104, 325–333. [Google Scholar]
- Wilson, L.A.; Sawant, A.D.; Simmons, R.B.; Ahearn, D.G. Microbial contamination of contact lens storage cases and solutions. Am. J. Ophthalmol. 1990, 110, 193–198. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Dutta, D.; Willcox, M.D. A Laboratory Assessment of Factors That Affect Bacterial Adhesion to Contact Lenses. Biology 2013, 2, 1268-1281. https://doi.org/10.3390/biology2041268
Dutta D, Willcox MD. A Laboratory Assessment of Factors That Affect Bacterial Adhesion to Contact Lenses. Biology. 2013; 2(4):1268-1281. https://doi.org/10.3390/biology2041268
Chicago/Turabian StyleDutta, Debarun, and Mark DP Willcox. 2013. "A Laboratory Assessment of Factors That Affect Bacterial Adhesion to Contact Lenses" Biology 2, no. 4: 1268-1281. https://doi.org/10.3390/biology2041268





