The Surprising Role of Amyloid Fibrils in HIV Infection
Abstract
:1. Introduction
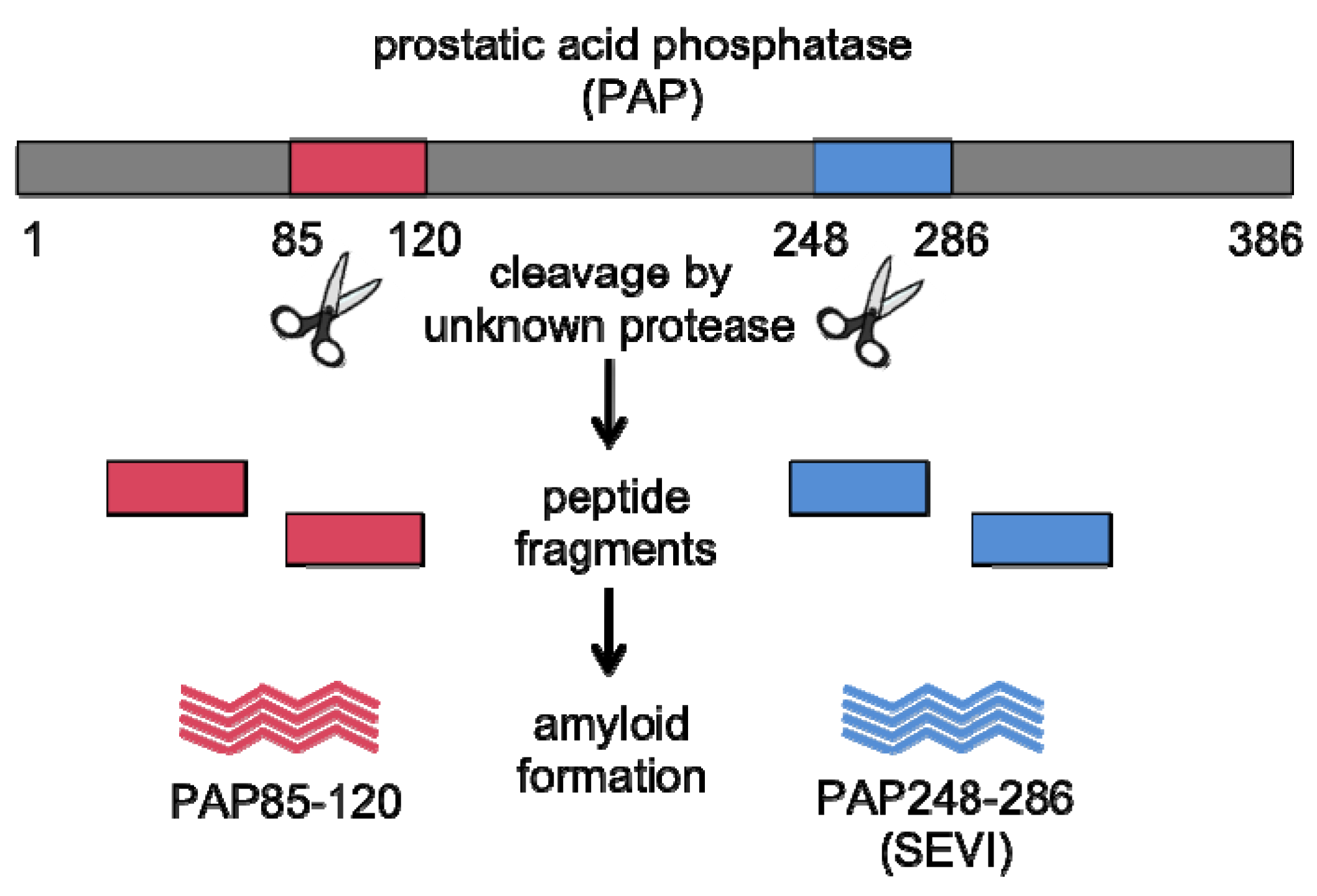
2. How Does SEVI Augment HIV Infection?
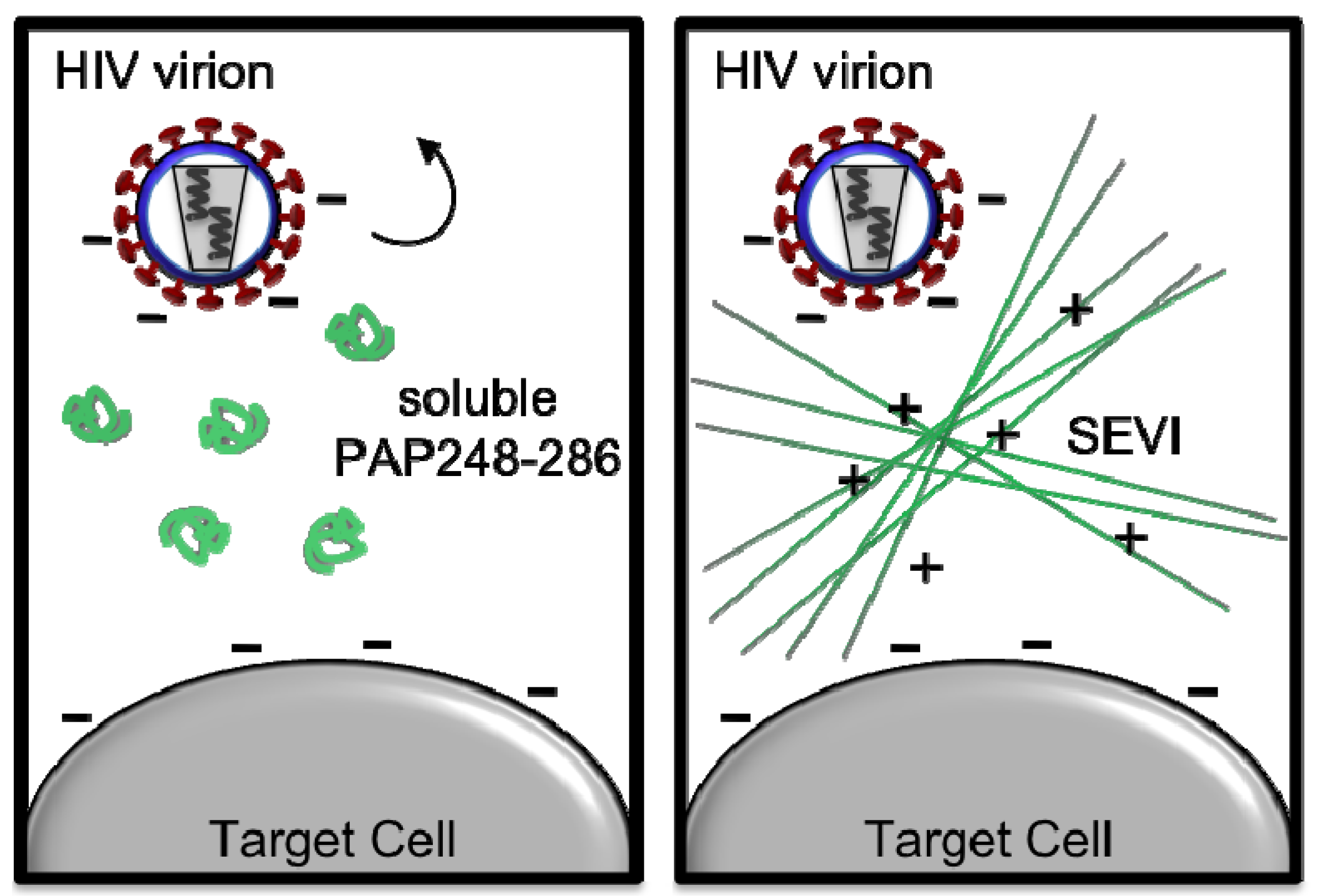
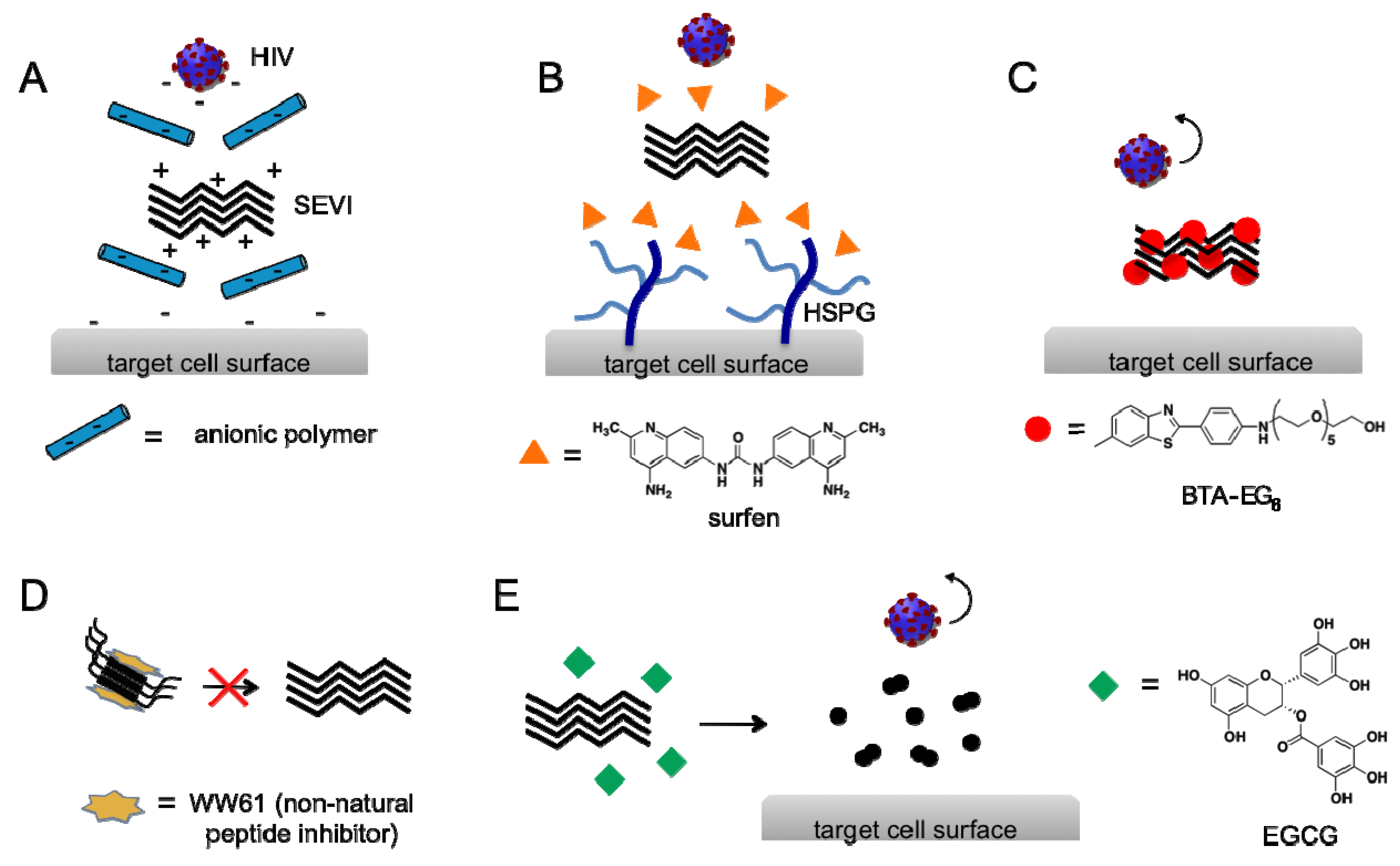
3. Current Strategies to Counteract SEVI-Mediated Enhancement of HIV Infection
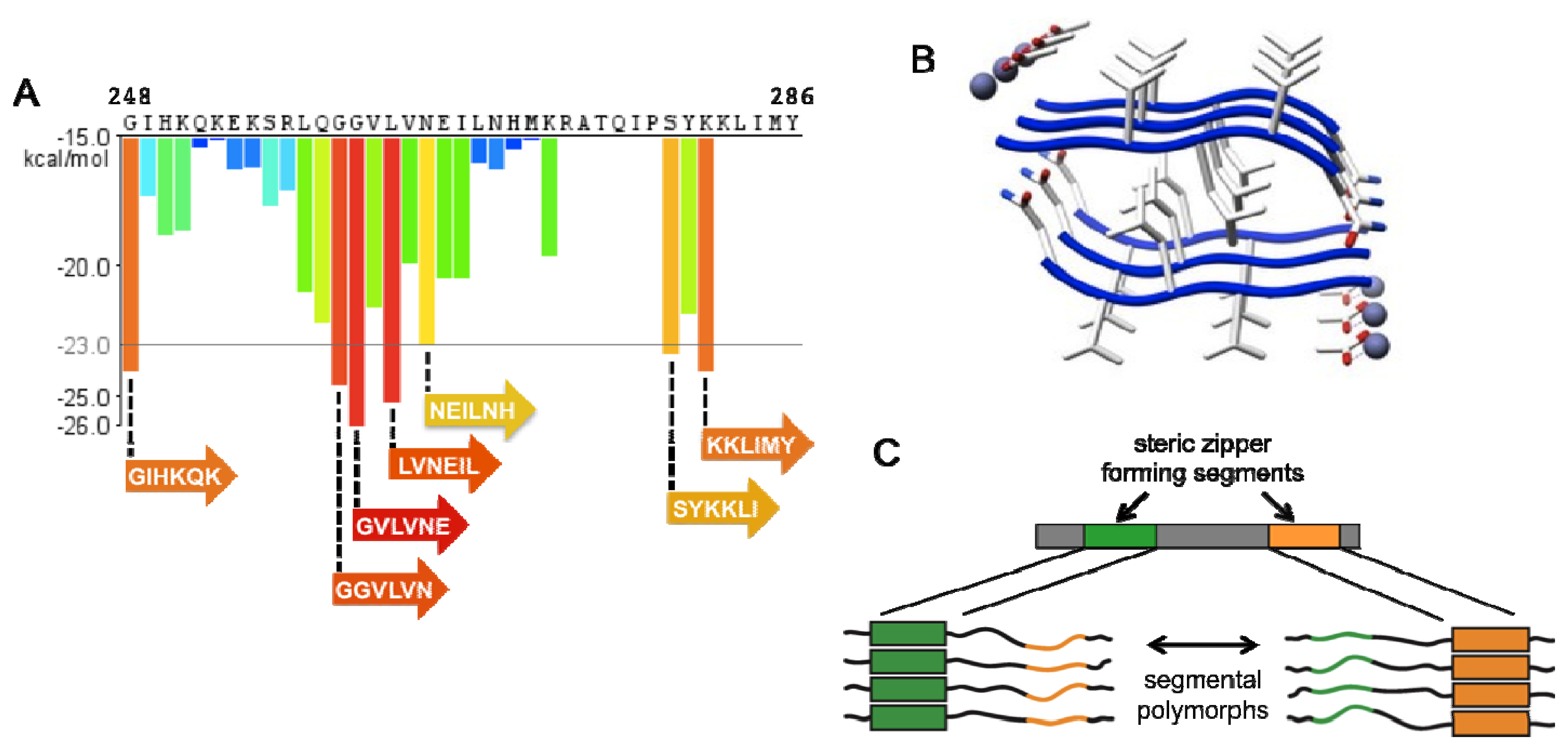
4. Amyloid Strains
5. PAP85-120 and SEM Amyloid Fibrils
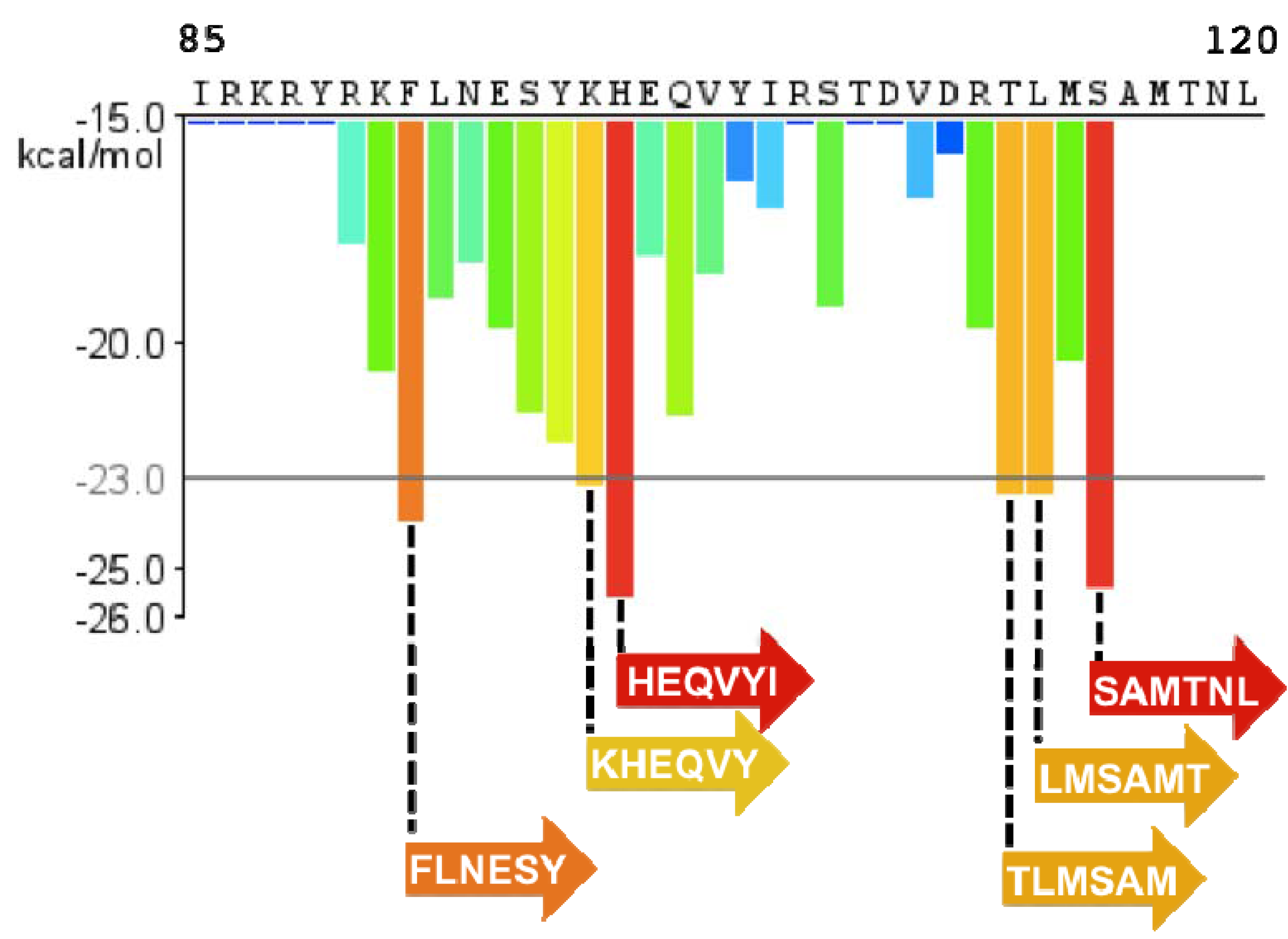
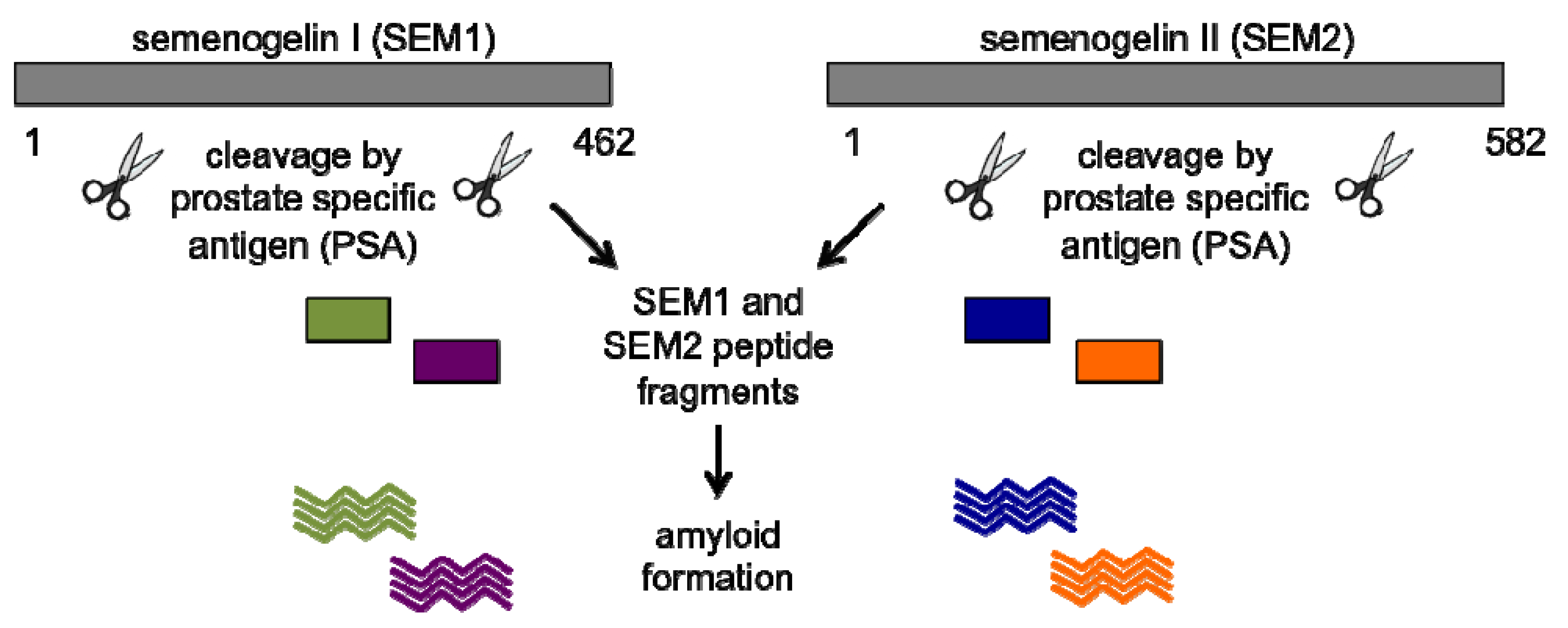
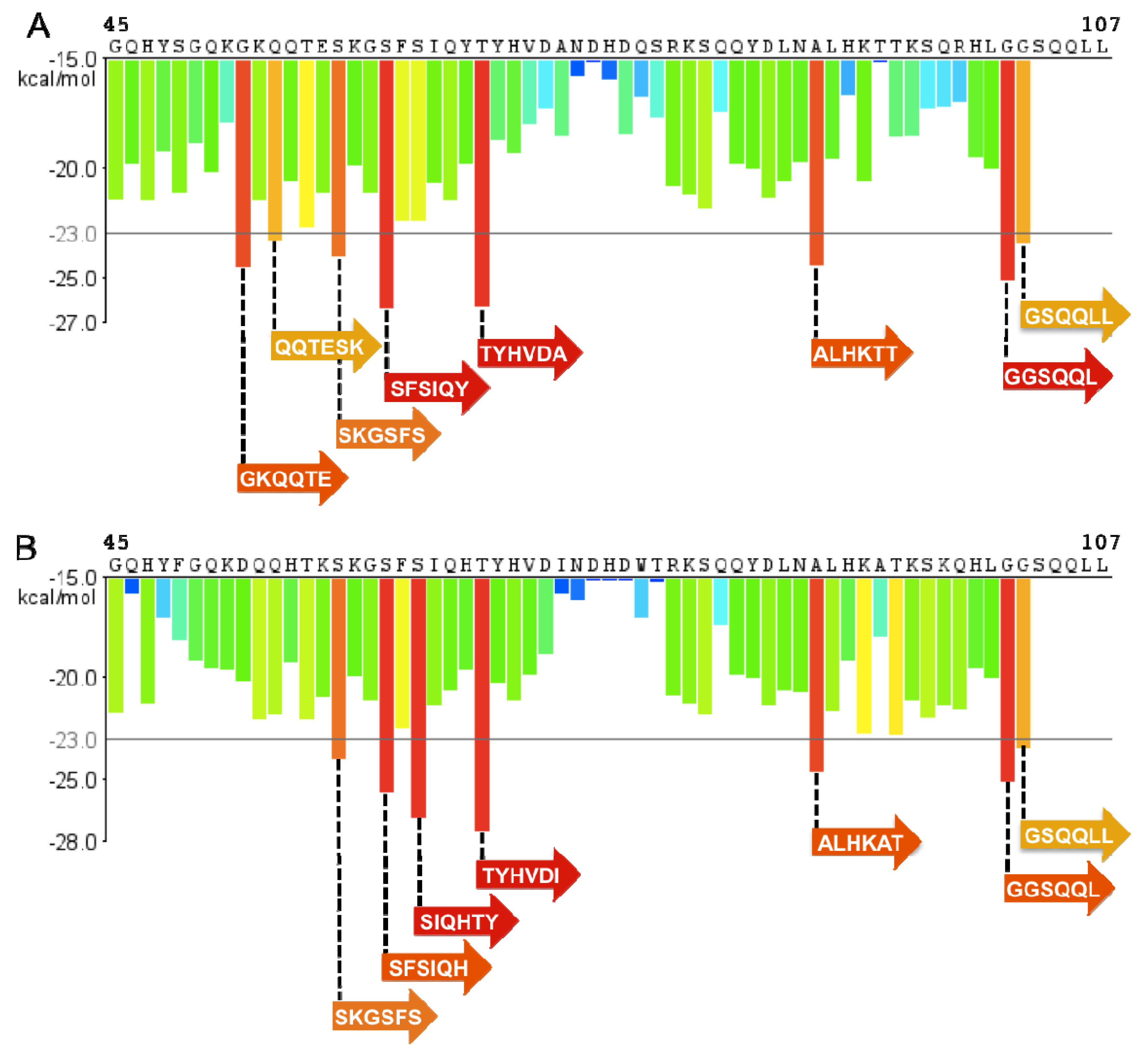
6. A Physiological Role for Semen-Derived Amyloid Fibrils?
7. Looking to the Future
8. Concluding Remarks
Acknowledgements
References
- Ascher, M.S.; Sheppard, H.W.; Winkelstein, W., Jr.; Vittinghoff, E. Aetiology of AIDS. Lancet 1993, 341, 1223. [Google Scholar]
- Schechter, M.T.; Craib, K.J.; Montaner, J.S.; Lee, T.N.; O’Shaughnessy, M.V.; Gelmon, K.A. Aetiology of AIDS. Lancet 1993, 341, 1222–1223. [Google Scholar]
- Weiss, R.A. How does HIV cause AIDS? Science 1993, 260, 1273–1279. [Google Scholar]
- Weiss, R.A.; Jaffe, H.W. Duesberg, HIV and AIDS. Nature 1990, 345, 659–660. [Google Scholar] [CrossRef]
- UNAIDS, Global Report: UNAIDS Report on the Global AIDS Epidemic 2010; UNAIDS: Geneva, Switzerland, 2010; pp. 1–359.
- Royce, R.A.; Sena, A.; Cates, W., Jr.; Cohen, M.S. Sexual transmission of HIV. N. Engl. J. Med. 1997, 336, 1072–1078. [Google Scholar] [CrossRef]
- Gray, R.H.; Wawer, M.J.; Brookmeyer, R.; Sewankambo, N.K.; Serwadda, D.; Wabwire-Mangen, F.; Lutalo, T.; Li, X.; van Cott, T.; Quinn, T.C. Probability of HIV-1 transmission per coital act in monogamous, heterosexual, HIV-1-discordant couples in Rakai, Uganda. Lancet 2001, 357, 1149–1153. [Google Scholar]
- Pilcher, C.D.; Tien, H.C.; Eron, J.J., Jr.; Vernazza, P.L.; Leu, S.Y.; Stewart, P.W.; Goh, L.E.; Cohen, M.S. Brief but efficient: Acute HIV infection and the sexual transmission of HIV. J. Infect. Dis. 2004, 189, 1785–1792. [Google Scholar] [CrossRef]
- Dimitrov, D.S.; Willey, R.L.; Sato, H.; Chang, L.J.; Blumenthal, R.; Martin, M.A. Quantitation of human immunodeficiency virus type 1 infection kinetics. J. Virol. 1993, 67, 2182–2190. [Google Scholar]
- Rusert, P.; Fischer, M.; Joos, B.; Leemann, C.; Kuster, H.; Flepp, M.; Bonhoeffer, S.; Gunthard, H.F.; Trkola, A. Quantification of infectious HIV-1 plasma viral load using a boosted in vitro infection protocol. Virology 2004, 326, 113–129. [Google Scholar] [CrossRef]
- Chuck, A.S.; Clarke, M.F.; Palsson, B.O. Retroviral infection is limited by Brownian motion. Hum. Gen. Ther. 1996, 7, 1527–1534. [Google Scholar] [CrossRef]
- Eckert, D.M.; Kim, P.S. Mechanisms of viral membrane fusion and its inhibition. Annu. Rev. Biochem. 2001, 70, 777–810. [Google Scholar] [CrossRef]
- Orloff, G.M.; Orloff, S.L.; Kennedy, M.S.; Maddon, P.J.; McDougal, J.S. Penetration of CD4 T cells by HIV-1. The CD4 receptor does not internalize with HIV, and CD4-related signal transduction events are not required for entry. J. Immunol. 1991, 146, 2578–2587. [Google Scholar]
- Galvin, S.R.; Cohen, M.S. The role of sexually transmitted diseases in HIV transmission. Nat. Rev. Microbiol. 2004, 2, 33–42. [Google Scholar] [CrossRef]
- Kim, K.A.; Yolamanova, M.; Zirafi, O.; Roan, N.R.; Staendker, L.; Forssmann, W.G.; Burgener, A.; Dejucq-rainsford, N.; Hahn, B.H.; Shaw, G.M.; Greene, W.C.; Kirchhoff, F.; Münch, J. Semen-mediated enhancement of HIV infection is donor-dependent and correlates with the levels of SEVI. Retrovirology 2010, 7, 55. [Google Scholar] [Green Version]
- Munch, J.; Rucker, E.; Standker, L.; Adermann, K.; Goffinet, C.; Schindler, M.; Wildum, S.; Chinnadurai, R.; Rajan, D.; Specht, A.; Gimenez-Gallego, G.; Sanchez, P.C.; Fowler, D.M.; Koulov, A.; Kelly, J.W.; Mothes, W.; Grivel, J.C.; Margolis, L.; Keppler, O.T.; Forssmann, W.G.; Kirchhoff, F. Semen-derived amyloid fibrils drastically enhance HIV infection. Cell 2007, 131, 1059–1071. [Google Scholar] [CrossRef]
- Olsen, J.S.; Brown, C.; Capule, C.C.; Rubinshtein, M.; Doran, T.M.; Srivastava, R.K.; Feng, C.; Nilsson, B.L.; Yang, J.; Dewhurst, S. Amyloid binding small molecules efficiently block SEVI and semen mediated enhancement of HIV-1 infection. J. Biol. Chem. 2010, 285, 35488–35496. [Google Scholar]
- Roan, N.R.; Greene, W.C. A seminal finding for understanding HIV transmission. Cell 2007, 131, 1044–1046. [Google Scholar] [CrossRef]
- Roan, N.R.; Munch, J.; Arhel, N.; Mothes, W.; Neidleman, J.; Kobayashi, A.; Smith-McCune, K.; Kirchhoff, F.; Greene, W.C. The cationic properties of SEVI underlie its ability to enhance human immunodeficiency virus infection. J. Virol. 2009, 83, 73–80. [Google Scholar]
- Ronnberg, L.; Vihko, P.; Sajanti, E.; Vihko, R. Clomiphene citrate administration to normogonadotropic subfertile men: Blood hormone changes and activation of acid phosphatase in seminal fluid. Int. J. Androl. 1981, 4, 372–378. [Google Scholar] [CrossRef]
- Wurm, M.; Schambach, A.; Lindemann, D.; Hanenberg, H.; Standker, L.; Forssmann, W.G.; Blasczyk, R.; Horn, P.A. The influence of semen-derived enhancer of virus infection on the efficiency of retroviral gene transfer. J. Gene Med. 2010, 12, 137–146. [Google Scholar]
- Cushman, M.; Johnson, B.S.; King, O.D.; Gitler, A.D.; Shorter, J. Prion-like disorders: Blurring the divide between transmissibility and infectivity. J. Cell Sci. 2010, 123, 1191–1201. [Google Scholar] [CrossRef]
- Wojtowicz, W.M.; Farzan, M.; Joyal, J.L.; Carter, K.; Babcock, G.J.; Israel, D.I.; Sodroski, J.; Mirzabekov, T. Stimulation of enveloped virus infection by beta-amyloid fibrils. J. Biol. Chem. 2002, 277, 35019–35024. [Google Scholar]
- Caughey, B.; Lansbury, P.T. Protofibrils, pores, fibrils, and neurodegeneration: Separating the responsible protein aggregates from the innocent bystanders. Annu. Rev. Neurosci. 2003, 26, 267–298. [Google Scholar] [CrossRef]
- Morimoto, R.I. Stress, aging, and neurodegenerative disease. N. Engl. J. Med. 2006, 355, 2254–2255. [Google Scholar] [CrossRef]
- Skovronsky, D.M.; Lee, V.M.; Trojanowski, J.Q. Neurodegenerative diseases: New concepts of pathogenesis and their therapeutic implications. Annu. Rev. Pathol. 2006, 1, 151–170. [Google Scholar] [CrossRef]
- Dobson, C.M. Protein folding and misfolding. Nature 2003, 426, 884–890. [Google Scholar] [CrossRef]
- Fandrich, M.; Fletcher, M.A.; Dobson, C.M. Amyloid fibrils from muscle myoglobin. Nature 2001, 410, 165–166. [Google Scholar]
- Fowler, D.M.; Koulov, A.V.; Alory-Jost, C.; Marks, M.S.; Balch, W.E.; Kelly, J.W. Functional amyloid formation within mammalian tissue. PLoS Biol. 2006, 4, e6. [Google Scholar] [CrossRef]
- Fowler, D.M.; Koulov, A.V.; Balch, W.E.; Kelly, J.W. Functional amyloid—From bacteria to humans. Trends Biochem. Sci. 2007, 32, 217–224. [Google Scholar] [CrossRef]
- Si, K.; Choi, Y.B.; White-Grindley, E.; Majumdar, A.; Kandel, E.R. Aplysia CPEB can form prion-like multimers in sensory neurons that contribute to long-term facilitation. Cell 2010, 140, 421–435. [Google Scholar] [CrossRef]
- Watt, B.; van Niel, G.; Fowler, D.M.; Hurbain, I.; Luk, K.C.; Stayrook, S.E.; Lemmon, M.A.; Raposo, G.; Shorter, J.; Kelly, J.W.; Marks, M.S. N-terminal domains elicit formation of functional Pmel17 amyloid fibrils. J. Biol. Chem. 2009, 284, 35543–35555. [Google Scholar]
- Shorter, J.; Lindquist, S. Prions as adaptive conduits of memory and inheritance. Nat. Rev. Genet. 2005, 6, 435–450. [Google Scholar] [CrossRef]
- True, H.L.; Lindquist, S.L. A yeast prion provides a mechanism for genetic variation and phenotypic diversity. Nature 2000, 407, 477–483. [Google Scholar]
- Halfmann, R.; Jarosz, D.F.; Jones, S.K.; Chang, A.; Lancaster, A.K.; Lindquist, S. Prions are a common mechanism for phenotypic inheritance in wild yeasts. Nature 2012, 482, 363–368. [Google Scholar]
- Suzuki, G.; Shimazu, N.; Tanaka, M. A yeast prion, Mod5, promotes acquired drug resistance and cell survival under environmental stress. Science 2012, 336, 355–359. [Google Scholar] [CrossRef]
- Barnhart, M.M.; Chapman, M.R. Curli biogenesis and function. Annu. Rev. Microbiol. 2006, 60, 131–147. [Google Scholar] [CrossRef]
- Eanes, E.D.; Glenner, G.G. X-ray diffraction studies on amyloid filaments. J. Histochem. Cytochem. 1968, 16, 673–677. [Google Scholar] [CrossRef]
- Nelson, R.; Eisenberg, D. Structural models of Amyloid–Like fibrils. Adv. Protein Chem. 2006, 73, 235–282. [Google Scholar] [CrossRef]
- Sunde, M.; Blake, C. The structure of amyloid fibrils by electron microscopy and X-ray diffraction. Adv. Protein Chem. 1997, 50, 123–159. [Google Scholar] [CrossRef]
- Roan, N.R.; Sowinski, S.; Munch, J.; Kirchhoff, F.; Greene, W.C. Aminoquinoline surfen inhibits the action of SEVI (semen-derived enhancer of viral infection). J. Biol. Chem. 2010, 285, 1861–1869. [Google Scholar]
- Arcasoy, S.M.; Latoche, J.D.; Gondor, M.; Pitt, B.R.; Pilewski, J.M. Polycations increase the efficiency of adenovirus-mediated gene transfer to epithelial and endothelial cells in vitro. Gene Ther. 1997, 4, 32–38. [Google Scholar]
- Davis, H.E.; Morgan, J.R.; Yarmush, M.L. Polybrene increases retrovirus gene transfer efficiency by enhancing receptor-independent virus adsorption on target cell membranes. Biophys. Chem. 2002, 97, 159–172. [Google Scholar] [CrossRef]
- Brender, J.R.; Nanga, R.P.; Popovych, N.; Soong, R.; Macdonald, P.M.; Ramamoorthy, A. The amyloidogenic SEVI precursor, PAP248–286, is highly unfolded in solution despite an underlying helical tendency. Biochim. Biophys. Acta 2011, 1808, 1161–1169. [Google Scholar] [CrossRef]
- Nanga, R.P.R.; Brender, J.R.; Vivekanandan, S.; Popovych, N.; Ramamoorthy, A. NMR structure in a membrane environment reveals putative amyloidogenic regions of the SEVI precursor peptide PAP248-286. J. Am. Chem. Soc. 2009, 131, 17972–17979. [Google Scholar]
- Sievers, S.A.; Karanicolas, J.; Chang, H.W.; Zhao, A.; Jiang, L.; Zirafi, O.; Stevens, J.T.; Munch, J.; Baker, D.; Eisenberg, D. Structure-based design of non-natural amino-acid inhibitors of amyloid fibril formation. Nature 2011, 475, 96–100. [Google Scholar]
- Tompa, P. Structural disorder in amyloid fibrils: Its implication in dynamic interactions of proteins. FEBS J. 2009, 276, 5406–5415. [Google Scholar] [CrossRef]
- Arnold, F.; Schnell, J.; Zirafi, O.; Sturzel, C.; Meier, C.; Weil, T.; Standker, L.; Forssmann, W.G.; Roan, N.R.; Greene, W.C.; Kirchhoff, F.; Munch, J. Naturally occurring fragments from two distinct regions of the prostatic acid phosphatase form amyloidogenic enhancers of HIV infection. J. Virol. 2012, 86, 1244–1249. [Google Scholar]
- Roan, N.R.; Muller, J.A.; Liu, H.; Chu, S.; Arnold, F.; Sturzel, C.M.; Walther, P.; Dong, M.; Witkowska, H.E.; Kirchhoff, F.; Munch, J.; Greene, W.C. Peptides released by physiological cleavage of semen coagulum proteins form amyloids that enhance HIV infection. Cell Host Microbe 2011, 10, 541–550. [Google Scholar] [CrossRef]
- Cutler, B.; Justman, J. Vaginal microbicides and the prevention of HIV transmission. Lancet Infect. Dis. 2008, 8, 685–697. [Google Scholar] [CrossRef]
- Klasse, P.J.; Shattock, R.; Moore, J.P. Antiretroviral drug-based microbicides to prevent HIV-1 sexual transmission. Annu. Rev. Med. 2008, 59, 455–471. [Google Scholar] [CrossRef]
- Garg, A.B.; Nuttall, J.; Romano, J. The future of HIV microbicides: Challenges and opportunities. Antivir. Chem. Chemother. 2009, 19, 143–150. [Google Scholar]
- Clavel, F.; Hance, A.J. HIV drug resistance. N. Engl. J. Med. 2004, 350, 1023–1035. [Google Scholar]
- Callahan, L.N.; Phelan, M.; Mallinson, M.; Norcross, M.A. Dextran sulfate blocks antibody binding to the principal neutralizing domain of human immunodeficiency virus type 1 without Interfering with gpl20-CD4 interactions. J. Virol. 1991, 65, 1543–1550. [Google Scholar]
- Rider, C.C. The potential for heparin and its derivatives in the therapy and prevention of HIV-1 infection. Glycoconj. J. 1997, 14, 639–642. [Google Scholar] [CrossRef]
- Witvrouw, M.; de Clercq, E. Sulfated polysaccharides extracted from sea algae as potential antiviral drugs. Gen. Pharmacol. 1997, 29, 497–511. [Google Scholar] [CrossRef]
- Luscher-Mattli, M. Polyanions—A lost chance in the fight against HIV and other virus diseases? Antivir. Chem. Chemother. 2000, 11, 249–259. [Google Scholar]
- van de Wijgert, J.H.; Shattock, R.J. Vaginal microbicides: Moving ahead after an unexpected setback. Aids 2007, 21, 2369–2376. [Google Scholar] [CrossRef]
- Lanza, T.J.; Durette, P.L.; Rollins, T.; Siciliano, S.; Cianciarulo, D.N.; Kobayashi, S.V.; Caldwell, C.G.; Springer, M.S.; Hagmann, W.K. Substituted 4,6-diaminoquinolines as inhibitors of C5a receptor binding. J. Med. Chem. 1992, 35, 252–258. [Google Scholar]
- Schuksz, M.; Fuster, M.M.; Brown, J.R.; Crawford, B.E.; Ditto, D.P.; Lawrence, R.; Glass, C.A.; Wang, L.; Tor, Y.; Esko, J.D. Surfen, a small molecule antagonist of heparan sulfate. Proc. Natl. Acad. Sci. USA 2008, 105, 13075–13080. [Google Scholar]
- Capule, C.C.; Brown, C.; Olsen, J.S.; Dewhurst, S.; Yang, J. Oligovalent amyloid-binding agents reduce SEVI-mediated enhancement of HIV-1 infection. J. Am. Chem. Soc. 2012, 134, 905–908. [Google Scholar]
- Olzscha, H.; Schermann, S.M.; Woerner, A.C.; Pinkert, S.; Hecht, M.H.; Tartaglia, G.G.; Vendruscolo, M.; Hayer-Hartl, M.; Hartl, F.U.; Vabulas, R.M. Amyloid-like aggregates sequester numerous metastable proteins with essential cellular functions. Cell 2011, 144, 67–78. [Google Scholar] [CrossRef]
- Alavez, S.; Vantipalli, M.C.; Zucker, D.J.; Klang, I.M.; Lithgow, G.J. Amyloid-binding compounds maintain protein homeostasis during ageing and extend lifespan. Nature 2011, 472, 226–229. [Google Scholar]
- Sawaya, M.R.; Sambashivan, S.; Nelson, R.; Ivanova, M.I.; Sievers, S.A.; Apostol, M.I.; Thompson, M.J.; Balbirnie, M.; Wiltzius, J.J.; McFarlane, H.T.; Madsen, A.O.; Riekel, C.; Eisenberg, D. Atomic structures of amyloid cross-beta spines reveal varied steric zippers. Nature 2007, 447, 453–457. [Google Scholar]
- Goldschmidt, L.; Teng, P.K.; Riek, R.; Eisenberg, D. Identifying the amylome, proteins capable of forming amyloid-like fibrils. Proc. Natl. Acad. Sci. USA 2010, 107, 3487–3492. [Google Scholar]
- Moravcevic, K.; Mendrola, J.M.; Schmitz, K.R.; Wang, Y.H.; Slochower, D.; Janmey, P.A.; Lemmon, M.A. Kinase associated-1 domains drive MARK/PAR1 kinases to membrane targets by binding acidic phospholipids. Cell 2010, 143, 966–977. [Google Scholar] [CrossRef]
- Scheibel, T.; Parthasarathy, R.; Sawicki, G.; Lin, X.M.; Jaeger, H.; Lindquist, S.L. Conducting nanowires built by controlled self-assembly of amyloid fibers and selective metal deposition. Proc. Natl. Acad. Sci. USA 2003, 100, 4527–4532. [Google Scholar]
- Smith, J.F.; Knowles, T.P.; Dobson, C.M.; Macphee, C.E.; Welland, M.E. Characterization of the nanoscale properties of individual amyloid fibrils. Proc. Natl. Acad. Sci. USA 2006, 103, 15806–15811. [Google Scholar]
- Shorter, J. Hsp104: A weapon to combat diverse neurodegenerative disorders. Neurosignals 2008, 16, 63–74. [Google Scholar] [CrossRef]
- Roberts, B.E.; Shorter, J. Escaping amyloid fate. Nat. Struct. Mol. Biol. 2008, 15, 544–546. [Google Scholar] [CrossRef]
- Ehrnhoefer, D.E.; Bieschke, J.; Boeddrich, A.; Herbst, M.; Masino, L.; Lurz, R.; Engemann, S.; Pastore, A.; Wanker, E.E. EGCG redirects amyloidogenic polypeptides into unstructured, off-pathway oligomers. Nat. Struct. Mol. Biol. 2008, 15, 558–566. [Google Scholar]
- Ehrnhoefer, D.E.; Duennwald, M.; Markovic, P.; Wacker, J.L.; Engemann, S.; Roark, M.; Legleiter, J.; Marsh, J.L.; Thompson, L.M.; Lindquist, S.; Muchowski, P.J.; Wanker, E.E. Green tea (−)-epigallocatechin-gallate modulates early events in huntingtin misfolding and reduces toxicity in Huntington’s disease models. Hum. Mol. Genet. 2006, 15, 2743–2751. [Google Scholar] [CrossRef]
- Masuda, M.; Suzuki, N.; Taniguchi, S.; Oikawa, T.; Nonaka, T.; Iwatsubo, T.; Hisanaga, S.; Goedert, M.; Hasegawa, M. Small molecule inhibitors of alpha-synuclein filament assembly. Biochemistry 2006, 45, 6085–6094. [Google Scholar]
- Meng, F.; Abedini, A.; Plesner, A.; Verchere, C.B.; Raleigh, D.P. The flavanol (−)-epigallocatechin 3-gallate inhibits amyloid formation by islet amyloid polypeptide, disaggregates amyloid fibrils, and protects cultured cells against IAPP-induced toxicity. Biochemistry 2010, 49, 8127–8133. [Google Scholar]
- Bieschke, J.; Russ, J.; Friedrich, R.P.; Ehrnhoefer, D.E.; Wobst, H.; Neugebauer, K.; Wanker, E.E. EGCG remodels mature alpha-synuclein and amyloid-beta fibrils and reduces cellular toxicity. Proc. Natl. Acad. Sci. USA 2010, 107, 7710–7715. [Google Scholar]
- Roberts, B.E.; Duennwald, M.L.; Wang, H.; Chung, C.; Lopreiato, N.P.; Sweeny, E.A.; Knight, M.N.; Shorter, J. A synergistic small-molecule combination directly eradicates diverse prion strain structures. Nat. Chem. Biol. 2009, 5, 936–946. [Google Scholar] [CrossRef]
- Hauber, I.; Hohenberg, H.; Holstermann, B.; Hunstein, W.; Hauber, J. The main green tea polyphenol epigallocatechin-3-gallate counteracts semen-mediated enhancement of HIV infection. Proc. Natl. Acad. Sci. USA 2009, 106, 9033–9038. [Google Scholar]
- Popovych, N.; Brender, J.R.; Soong, R.; Vivekanandan, S.; Hartman, K.; Basrur, V.; Macdonald, P.M.; Ramamoorthy, A. Site specific interaction of the polyphenol EGCG with the SEVI amyloid precursor peptide PAP(248–286). J. Phys. Chem. B 2012, 116, 3650–3658. [Google Scholar]
- Zhu, Q.Y.; Zhang, A.; Tsang, D.; Huang, Y.; Chen, Z.Y. Stability of green tea catechins. J. Agric. Food Chem. 1997, 45, 4624–4628. [Google Scholar] [CrossRef]
- Hartjen, P.; Frerk, S.; Hauber, I.; Matzat, V.; Thomssen, A.; Holstermann, B.; Hohenberg, H.; Schulze, W.; Schulze Zur Wiesch, J.; van Lunzen, J. Assessment of the range of the HIV-1 infectivity enhancing effect of individual human semen specimen and the range of inhibition by EGCG. AIDS Res. Ther. 2012, 9, 2. [Google Scholar] [CrossRef]
- Kawai, K.; Tsuno, N.H.; Kitayama, J.; Okaji, Y.; Yazawa, K.; Asakage, M.; Hori, N.; Watanabe, T.; Takahashi, K.; Nagawa, H. Epigallocatechin gallate, the main component of tea polyphenol, binds to CD4 and interferes with gp120 binding. J. Allergy Clin. Immunol. 2003, 112, 951–957. [Google Scholar] [CrossRef]
- Nance, C.L.; Siwak, E.B.; Shearer, W.T. Preclinical development of the green tea catechin, epigallocatechin gallate, as an HIV-1 therapy. J. Allergy Clin. Immunol. 2009, 123, 459–465. [Google Scholar] [CrossRef]
- Williamson, M.P.; McCormick, T.G.; Nance, C.L.; Shearer, W.T. Epigallocatechin gallate, the main polyphenol in green tea, binds to the T-cell receptor, CD4: Potential for HIV-1 therapy. J. Allergy Clin. Immunol. 2006, 118, 1369–1374. [Google Scholar] [CrossRef]
- Isaacs, C.E.; Wen, G.Y.; Xu, W.; Jia, J.H.; Rohan, L.; Corbo, C.; di Maggio, V.; Jenkins, E.C., Jr.; Hillier, S. Epigallocatechin gallate inactivates clinical isolates of herpes simplex virus. Antimicrob. Agents Chemother. 2008, 52, 962–970. [Google Scholar]
- Ciesek, S.; von Hahn, T.; Colpitts, C.C.; Schang, L.M.; Friesland, M.; Steinmann, J.; Manns, M.P.; Ott, M.; Wedemeyer, H.; Meuleman, P.; Pietschmann, T.; Steinmann, E. The green tea polyphenol, epigallocatechin-3-gallate, inhibits hepatitis C virus entry. Hepatology 2011, 54, 1947–1955. [Google Scholar]
- Goldsbury, C.S.; Cooper, G.J.; Goldie, K.N.; Muller, S.A.; Saafi, E.L.; Gruijters, W.T.; Misur, M.P.; Engel, A.; Aebi, U.; Kistler, J. Polymorphic fibrillar assembly of human amylin. J. Struct. Biol. 1997, 119, 17–27. [Google Scholar] [CrossRef]
- Petkova, A.T.; Leapman, R.D.; Guo, Z.; Yau, W.M.; Mattson, M.P.; Tycko, R. Self-propagating, molecular-level polymorphism in Alzheimer’s beta-amyloid fibrils. Science 2005, 307, 262–265. [Google Scholar] [CrossRef]
- Tanaka, M.; Chien, P.; Naber, N.; Cooke, R.; Weissman, J.S. Conformational variations in an infectious protein determine prion strain differences. Nature 2004, 428, 323–328. [Google Scholar]
- Wiltzius, J.J.; Landau, M.; Nelson, R.; Sawaya, M.R.; Apostol, M.I.; Goldschmidt, L.; Soriaga, A.B.; Cascio, D.; Rajashankar, K.; Eisenberg, D. Molecular mechanisms for protein-encoded inheritance. Nat. Struct. Mol. Biol. 2009, 16, 973–978. [Google Scholar]
- Shorter, J. Emergence and natural selection of drug-resistant prions. Mol. Biosyst. 2010, 6, 1115–1130. [Google Scholar] [CrossRef]
- Duennwald, M.L.; Shorter, J. Countering amyloid polymorphism and drug resistance with minimal drug cocktails. Prion 2010, 4, 244–251. [Google Scholar] [CrossRef]
- Wang, H.; Duennwald, M.L.; Roberts, B.E.; Rozeboom, L.M.; Zhang, Y.L.; Steele, A.D.; Krishnan, R.; Su, L.J.; Griffin, D.; Mukhopadhyay, S.; Hennessy, E.J.; Weigele, P.; Blanchard, B.J.; King, J.; Deniz, A.A.; Buchwald, S.L.; Ingram, V.M.; Lindquist, S.; Shorter, J. Direct and selective elimination of specific prions and amyloids by 4,5-dianilinophthalimide and analogs. Proc. Natl. Acad. Sci. USA 2008, 105, 7159–7164. [Google Scholar]
- Weissmann, C. Mutation and selection of prions. PLoS Pathogens. 2012, 8, e1002582. [Google Scholar] [CrossRef]
- de Lamirande, E. Semenogelin, the main protein of the human semen coagulum, regulates sperm function. Semin. Thromb. Hemost. 2007, 33, 60–68. [Google Scholar] [CrossRef]
- Robert, M.; Gagnon, C. Semenogelin I: A coagulum forming, multifunctional seminal vesicle protein. Cell. Mol. Life Sci. 1999, 55, 944–960. [Google Scholar] [CrossRef]
- Robert, M.; Gagnon, C. Purification and characterization of the active precursor of a human sperm motility inhibitor secreted by the seminal vesicles: Identity with semenogelin. Biol. Reprod. 1996, 55, 813–821. [Google Scholar] [CrossRef]
- Robert, M.; Gibbs, B.F.; Jacobson, E.; Gagnon, C. Characterization of prostate-specific antigen proteolytic activity on its major physiological substrate, the sperm motility inhibitor precursor/semenogelin I. Biochemistry 1997, 36, 3811–3819. [Google Scholar]
- Maji, S.K.; Perrin, M.H.; Sawaya, M.R.; Jessberger, S.; Vadodaria, K.; Rissman, R.A.; Singru, P.S.; Nilsson, K.P.; Simon, R.; Schubert, D.; Eisenberg, D.; Rivier, J.; Sawchenko, P.; Vale, W.; Riek, R. Functional amyloids as natural storage of peptide hormones in pituitary secretory granules. Science 2009, 325, 328–332. [Google Scholar]
- Berson, J.F.; Theos, A.C.; Harper, D.C.; Tenza, D.; Raposo, G.; Marks, M.S. Proprotein convertase cleavage liberates a fibrillogenic fragment of a resident glycoprotein to initiate melanosome biogenesis. J. Cell Biol. 2003, 161, 521–533. [Google Scholar] [CrossRef]
- Olsen, J.S.; Dimaio, J.T.M.; Doran, T.M.; Brown, C.; Nilsson, B.L.; Dewhurst, S. Seminal plasma accelerates SEVI fibril formation by the PAP[248–286] peptide. J. Biol. Chem. 2012. [Google Scholar]
- Hartjen, P.; Frerk, S.; Hauber, I.; Matzat, V.; Thomssen, A.; Holstermann, B.; Hohenberg, H.; Schulze, W.; Schulze Zur Wiesch, J.; van Lunzen, J. Assessment of the range of the HIV-1 infectivity enhancing effect of individual human semen specimen and the range of inhibition by EGCG. AIDS Res. Ther. 2012, 9, 2. [Google Scholar] [CrossRef]
- Doncel, G.F. Exploiting common targets in human fertilization and HIV infection: Development of novel contraceptive microbicides. Hum. Reprod. Update 2006, 12, 103–117. [Google Scholar] [CrossRef]
- Doncel, G.; Mauck, C. Vaginal microbicides: A novel approach to preventing sexual transmission of HIV. Curr. HIV/AIDS Rep. 2004, 1, 25–32. [Google Scholar] [CrossRef]
- Jones, R. Interaction of zona pellucida glycoproteins, sulphated carbohydrates and synthetic polymers with proacrosin, the putative egg-binding protein from mammalian spermatozoa. Development 1991, 111, 1155–1163. [Google Scholar]
- Oehninger, S.; Clark, G.F.; Acosta, A.A.; Hodgen, G.D. Nature of the inhibitory effect of complex saccharide moieties on the tight binding of human spermatozoa to the human zona pellucida. Fertil.Steril. 1991, 55, 165–169. [Google Scholar]
- Cohen, F.S.; Melikyan, G.B. The Energetics of Membrane Fusion from Binding, through Hemifusion, Pore Formation, and Pore Enlargement. J. Membr. Biol. 2004, 199, 1–14. [Google Scholar] [CrossRef]
- Evans, J.P.; Florman, H.M. The state of the union: The cell biology of fertilization. Nat. Cell Biol. 2002, 4, s57–s63. [Google Scholar] [CrossRef]
- Herbein, G.; Gras, G.; Khan, K.A.; Abbas, W. Macrophage signaling in HIV-1 infection. Retrovirology 2010, 7, 34. [Google Scholar] [CrossRef] [Green Version]
- Salicioni, A.M.; Platt, M.D.; Wertheimer, E.V.; Arcelay, E.; Allaire, A.; Sosnik, J.; Visconti, P.E. Signalling pathways involved in sperm capacitation. Soc. Reprod. Fertil. Suppl. 2007, 65, 245–259. [Google Scholar]
- Liu, Z.; Foote, R.H. Bull sperm motility and membrane integrity in media varying in osmolality. J. Dairy Sci. 1998, 81, 1868–1873. [Google Scholar] [CrossRef]
- Bjartell, A.; Malm, J.; Möller, C.; Gunnarsson, M.; Lundwall, A.; Lilja, H. Distribution and Tissue Expression of Semenogelin I and II in Man as Demonstrated by In Situ Hybridization and Immunocytochemistry. J. Androl. 1996, 17, 17–26. [Google Scholar]
- Herr, J.C.; Summers, T.A.; McGee, R.S.; Sutherland, W.M.; Sigman, M.; Evans, R.J. Characterization of a monoclonal antibody to a conserved epitope on human seminal vesicle-specific peptides: A novel probe/marker system for semen identification. Biol. Reprod. 1986, 35, 773–784. [Google Scholar] [CrossRef]
- Tessier, P.M.; Lindquist, S. Prion recognition elements govern nucleation, strain specificity and species barriers. Nature 2007, 447, 556–561. [Google Scholar]
- Perchiacca, J.M.; Ladiwala, A.R.; Bhattacharya, M.; Tessier, P.M. Structure-based design of conformation- and sequence-specific antibodies against amyloid beta. Proc. Natl. Acad. Sci. USA 2012, 109, 84–89. [Google Scholar]
- Dewhurst, S. Transmission-blocking vaccine for HIV-1. Available online: http://projectreporter.nih.gov/project_info_description.cfm?aid=7914361&icde=0 (access on 18 May 2012).
- Martellini, J.A.; Cole, A.L.; Svoboda, P.; Stuchlik, O.; Chen, L.M.; Chai, K.X.; Gangrade, B.K.; Sorensen, O.E.; Pohl, J.; Cole, A.M. HIV-1 enhancing effect of prostatic acid phosphatase peptides is reduced in human seminal plasma. PloS One 2011, 6, e16285. [Google Scholar]
- Balandya, E.; Sheth, S.; Sanders, K.; Wieland-Alter, W.; Lahey, T. Semen protects CD4+ target cells from HIV infection but promotes the preferential transmission of R5 tropic HIV. J. Immunol. 2010, 185, 7596–7604. [Google Scholar] [CrossRef]
- Sabatte, J.; Ceballos, A.; Raiden, S.; Vermeulen, M.; Nahmod, K.; Maggini, J.; Salamone, G.; Salomon, H.; Amigorena, S.; Geffner, J. Human seminal plasma abrogates the capture and transmission of human immunodeficiency virus type 1 to CD4+ T cells mediated by DC-SIGN. J. Virol. 2007, 81, 13723–13734. [Google Scholar]
- Vashist, S.; Cushman, M.; Shorter, J. Applying Hsp104 to protein-misfolding disorders. Biochem. Cell. Biol. 2010, 88, 1–13. [Google Scholar]
- Lo Bianco, C.; Shorter, J.; Regulier, E.; Lashuel, H.; Iwatsubo, T.; Lindquist, S.; Aebischer, P. Hsp104 antagonizes alpha-synuclein aggregation and reduces dopaminergic degeneration in a rat model of Parkinson disease. J. Clin. Invest. 2008, 118, 3087–3097. [Google Scholar] [CrossRef]
- Shorter, J.; Lindquist, S. Hsp104 catalyzes formation and elimination of self-replicating Sup35 prion conformers. Science 2004, 304, 1793–1797. [Google Scholar] [CrossRef]
- Wojtowicz, W.M.; Farzan, M.; Joyal, J.L.; Carter, K.; Babcock, G.J.; Israel, D.I.; Sodroski, J.; Mirzabekov, T. Stimulation of enveloped virus infection by beta-amyloid fibrils. J. Biol. Chem. 2002, 277, 35019–35024. [Google Scholar]
- Padian, N.S.; McCoy, S.I.; Manian, S.; Wilson, D.; Schwartlander, B.; Bertozzi, S.M. Evaluation of large-scale combination HIV prevention programs: Essential issues. J. Acquir. Immune Defic. Syndr. 2011, 58, e23–e28. [Google Scholar] [CrossRef]
- Green, E.C.; Halperin, D.T.; Nantulya, V.; Hogle, J.A. Uganda’s HIV prevention success: The role of sexual behavior change and the national response. AIDS Behav. 2006, 10, 335–346, discussion 347–350.. [Google Scholar] [CrossRef]
- Gregson, S.; Gonese, E.; Hallett, T.B.; Taruberekera, N.; Hargrove, J.W.; Lopman, B.; Corbett, E.L.; Dorrington, R.; Dube, S.; Dehne, K.; Mugurungi, O. HIV decline in Zimbabwe due to reductions in risky sex? Evidence from a comprehensive epidemiological review. Int. J. Epidemiol. 2010, 39, 1311–1323. [Google Scholar] [CrossRef]
- Halperin, D.T.; Mugurungi, O.; Hallett, T.B.; Muchini, B.; Campbell, B.; Magure, T.; Benedikt, C.; Gregson, S. A surprising prevention success: Why did the HIV epidemic decline in Zimbabwe? PLoS Med. 2011, 8, e1000414. [Google Scholar]
- Abdool Karim, Q.; Abdool Karim, S.S.; Frohlich, J.A.; Grobler, A.C.; Baxter, C.; Mansoor, L.E.; Kharsany, A.B.; Sibeko, S.; Mlisana, K.P.; Omar, Z.; Gengiah, T.N.; Maarschalk, S.; Arulappan, N.; Mlotshwa, M.; Morris, L.; Taylor, D. Effectiveness and safety of tenofovir gel, an antiretroviral microbicide, for the prevention of HIV infection in women. Science 2010, 329, 1168–1174. [Google Scholar]
- Grant, R.M.; Lama, J.R.; Anderson, P.L.; McMahan, V.; Liu, A.Y.; Vargas, L.; Goicochea, P.; Casapia, M.; Guanira-Carranza, J.V.; Ramirez-Cardich, M.E.; Montoya-Herrera, O.; Fernandez, T.; Veloso, V.G.; Buchbinder, S.P.; Chariyalertsak, S.; Schechter, M.; Bekker, L.G.; Mayer, K.H.; Kallas, E.G.; Amico, K.R.; Mulligan, K.; Bushman, L.R.; Hance, R.J.; Ganoza, C.; Defechereux, P.; Postle, B.; Wang, F.; McConnell, J.J.; Zheng, J.H.; Lee, J.; Rooney, J.F.; Jaffe, H.S.; Martinez, A.I.; Burns, D.N.; Glidden, D.V. Preexposure chemoprophylaxis for HIV prevention in men who have sex with men. N. Engl. J. Med. 2010, 363, 2587–2599. [Google Scholar]
- Celum, C.; Baeten, J.M. Tenofovir-based pre-exposure prophylaxis for HIV prevention: Evolving evidence. Curr. Opin. Infect. Dis. 2012, 25, 51–57. [Google Scholar] [CrossRef]
- Das, A.T.; Berkhout, B. HIV-1 evolution: Frustrating therapies, but disclosing molecular mechanisms. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2010, 365, 1965–1973. [Google Scholar] [CrossRef]
- Iwasa, Y.; Michor, F.; Nowak, M.A. Evolutionary dynamics of escape from biomedical intervention. Proc. Biol. Sci. 2003, 270, 2573–2578. [Google Scholar] [CrossRef]
- Iwasa, Y.; Michor, F.; Nowak, M.A. Evolutionary dynamics of invasion and escape. J. Theor. Biol. 2004, 226, 205–214. [Google Scholar] [CrossRef]
- Hogg, R.; Lima, V.; Sterne, J.A.; Grabar, S.; Battegay, M.; Bonarek, M.; D’Arminio Montforte, A.; Esteve, A.; Gill, M.J.; Harris, R.; Justice, A.; Hayden, A.; Lampe, F.; Mocroft, A.; Mugavero, M.J.; Staszewski, S.; Wasmuth, J.C.; van Sighem, A.; Kitahata, M.; Guest, J.; Egger, M.; May, M. Life expectancy of individuals on combination antiretroviral therapy in high-income countries: A collaborative analysis of 14 cohort studies. Lancet 2008, 372, 293–299. [Google Scholar]
- Staszewski, S.; Morales-Ramirez, J.; Tashima, K.T.; Rachlis, A.; Skiest, D.; Stanford, J.; Stryker, R.; Johnson, P.; Labriola, D.F.; Farina, D.; Manion, D.J.; Ruiz, N.M. Efavirenz plus zidovudine and lamivudine, efavirenz plus indinavir, and indinavir plus zidovudine and lamivudine in the treatment of HIV-1 infection in adults. Study 006 Team. N. Engl. J. Med. 1999, 341, 1865–1873. [Google Scholar]
- Kurth, A.E.; Celum, C.; Baeten, J.M.; Vermund, S.H.; Wasserheit, J.N. Combination HIV prevention: Significance, challenges, and opportunities. Curr. HIV/AIDS Rep. 2011, 8, 62–72. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Castellano, L.M.; Shorter, J. The Surprising Role of Amyloid Fibrils in HIV Infection. Biology 2012, 1, 58-80. https://doi.org/10.3390/biology1010058
Castellano LM, Shorter J. The Surprising Role of Amyloid Fibrils in HIV Infection. Biology. 2012; 1(1):58-80. https://doi.org/10.3390/biology1010058
Chicago/Turabian StyleCastellano, Laura M., and James Shorter. 2012. "The Surprising Role of Amyloid Fibrils in HIV Infection" Biology 1, no. 1: 58-80. https://doi.org/10.3390/biology1010058



