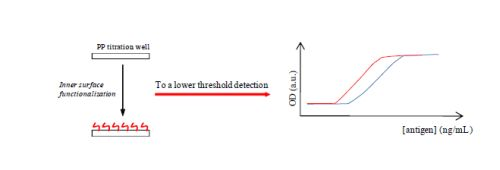
A New Approach for Detection Improvement of the Creutzfeldt-Jakob Disorder through a Specific Surface Chemistry Applied onto Titration Well
Abstract
:1. Introduction
2. Experimental Section
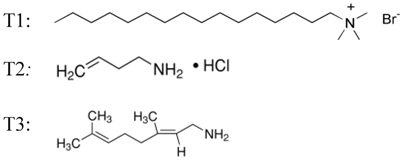
2.1. Materials
2.2. Cold Plasma Treatment
2.3. Amphiphilic Molecular Coating
2.4. Surface Characterization
2.5. ELISA Titration
2.5.1. Recombinant Human Prion Protein (PrPrechum) Sandwich ELISA Protocol
2.5.2. PrP-DVE Sandwich ELISA Protocol
3. Results and Discussion
3.1. ELISA Titration of Recombinant Human Prion Protein (PrPrechum)
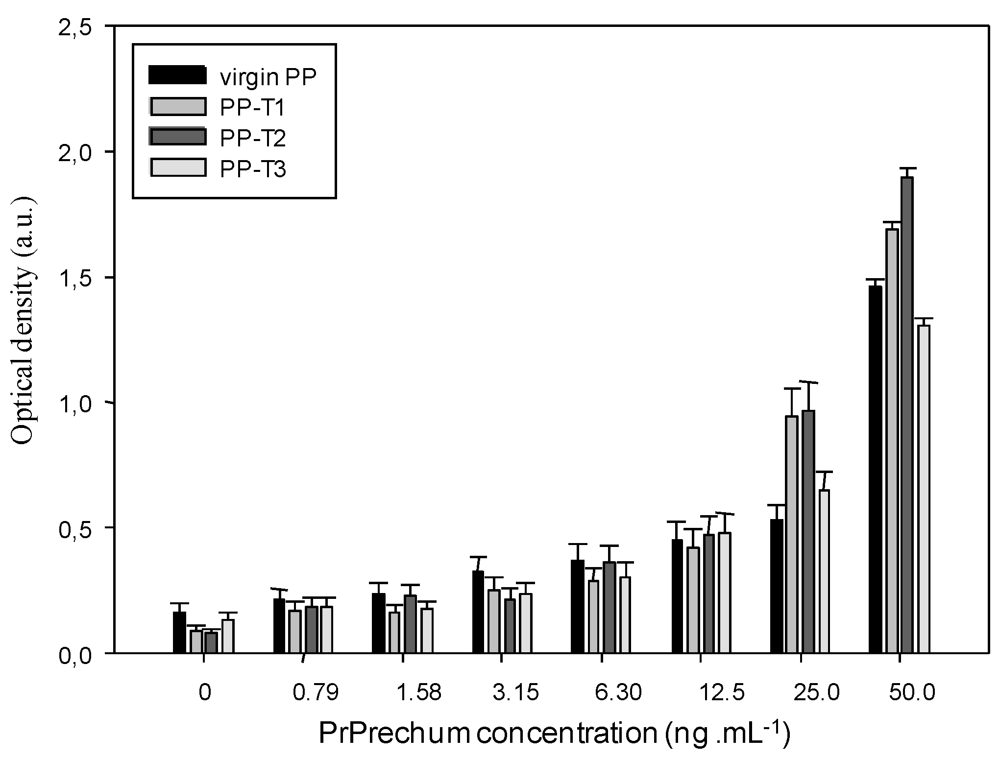
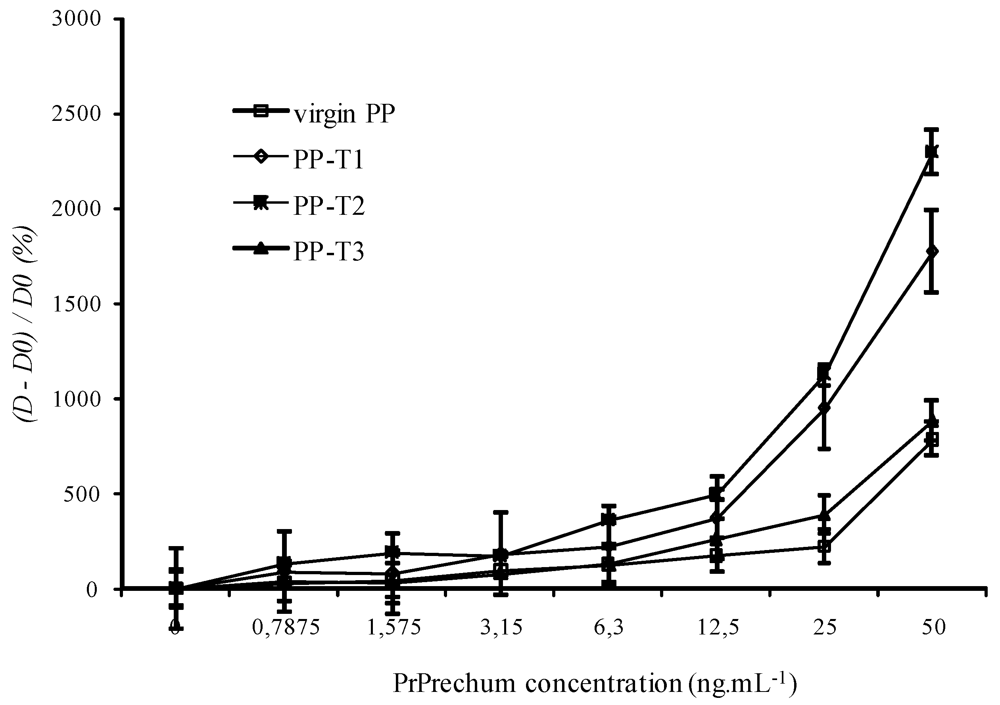
3.2. Aging of New Biofunctional Wells

3.3. ELISA Titration of Native Prion Protein (Pr-DVE)
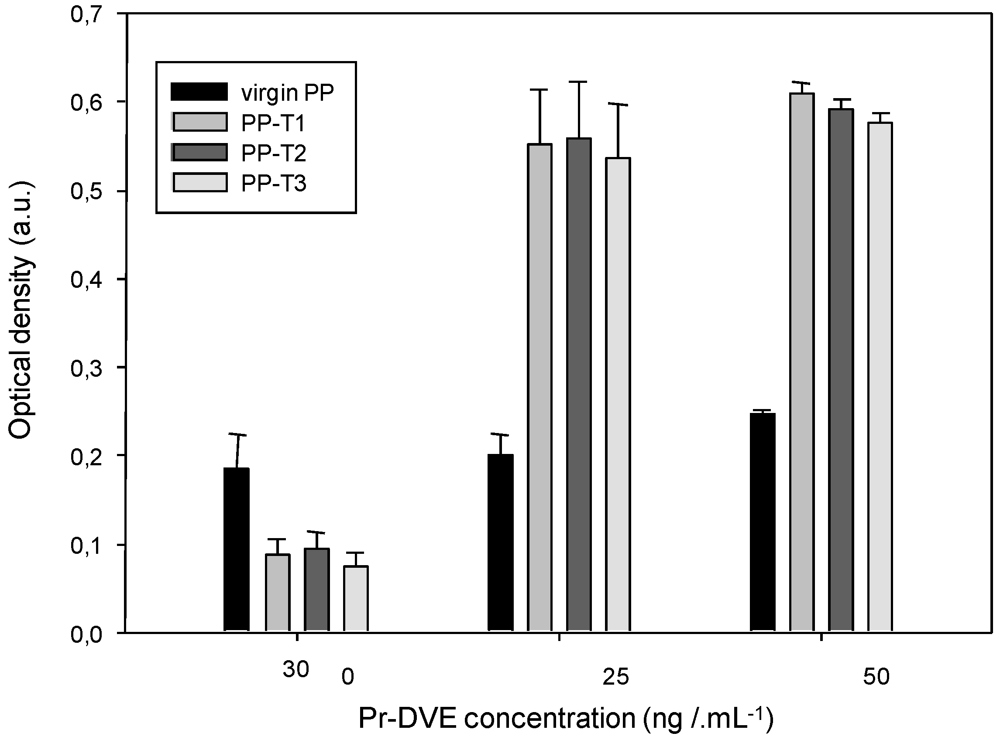

| Aging duration (month) | (Pr-DVE) (ng·mL–1) | PP-T1 | PP-T2 | PP-T3 |
|---|---|---|---|---|
| 0 | 0 | 0.088 ± 0.018 | 0.094 ± 0.020 | 0.074 ± 0.016 |
| 50 | 0.610 ± 0.026 | 0.591 ± 0.0114 | 0.576 ± 0.011 | |
| 8 | 0 | 0.128 ± 0.027 | 0.103 ± 0.022 | 0.146 ± 0.031 |
| 50 | 0.696 ± 0.013 | 0.512 ± 0.010 | 0.376 ± 0.007 |
3.4. Specificity of the ELISA Titration of PrPrechum and Pr-DVE
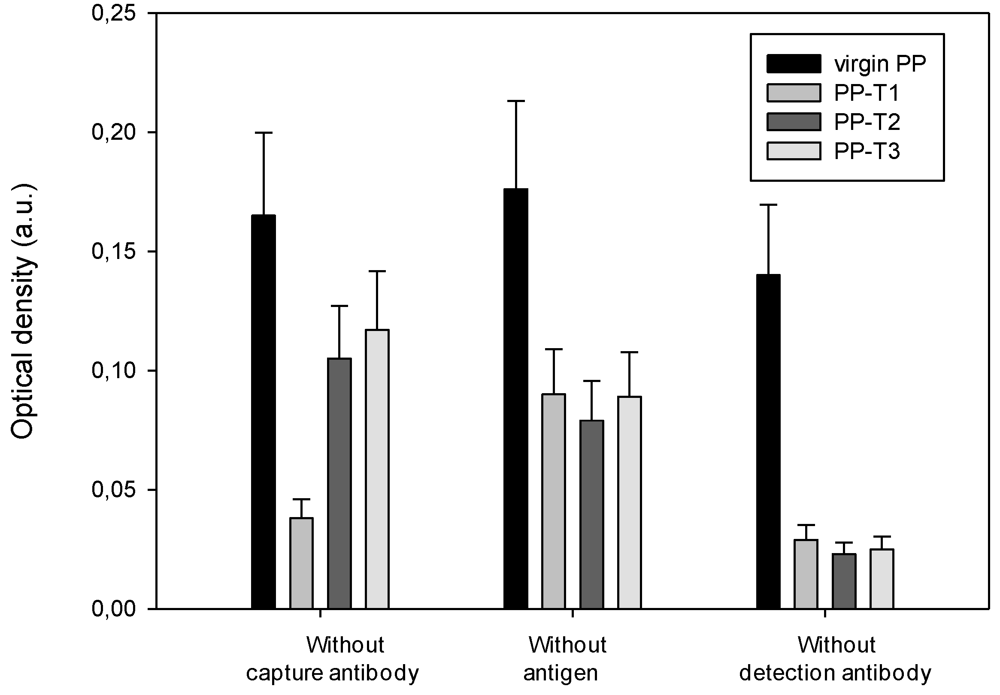

4. Conclusions
Acknowledgments
References
- Prusiner, S.B.; Hadlow, W.J.; Eklund, C.M.; Race, R.E. Sedimentation properties of the scrapie agent. PNAS 1977, 74, 4656–4660. [Google Scholar] [CrossRef]
- Bockman, J.M.; Kingsbury, D.T.; McKinley, M.P.; Bendheim, P.E.; Prusiner, S.B. Creutzfeldt-Jakob disease prion proteins in human brains. J. Med. 1985, 312, 73–78. [Google Scholar]
- Prusiner, S.B. Novel proteinaceous infectious particles cause scrapie. Science 1982, 216, 136–144. [Google Scholar]
- Prusiner, S.B. Prions. PNAS 1998, 95, 13363–13383. [Google Scholar]
- Oki, R.K.; Nwanebu, F.C. Prion and prion diseases. J. Clin. Exp. Microbiol. 2009, 9, 38–52. [Google Scholar]
- McKinley, M.P.; Masiarz, M.R.; Prusiner, S.B. Reversible chemical modification of the scrapie agent. Science 1981, 214, 1259–1261. [Google Scholar]
- Pan, K.M.; Baldwin, M.; Nguyen, J.; Gasset, M.; Serban, A.; Groth, D.; Mehlhorn, I.; Huang, Z.; Fletterick, R.J.; Cohen, F.E. Conversion of alpha-helices into beta-sheets features in the formation of the scrapie prion proteins. PNAS 1993, 90, 10962–10966. [Google Scholar]
- Horvath, V.; Kovacs, A.; Menyhard, D.K. Conformational studies on the prion protein 11-122 fragment. J. Mol. Struct. 2007, 804, 9–15. [Google Scholar]
- Beranger, F.; Crozet, C.; Goldsborough, A.; Lehmann, S. Treholose impairs aggregation of PrP-sc molecules and protects prion-infected cells against oxidative damage. Biochem. Biophys. Res. Comm. 2008, 374, 44–48. [Google Scholar]
- Welch, W.J.; Gambetti, P. Chaperoning brain diseases. Nature 1998, 392, 23–24. [Google Scholar] [CrossRef]
- Malaga-Trillo, E.; Solis, G.P.; Schrock, Y.; Geiss, C.; Luncz, L.; Thomanetz, V.; Stuermer, C.A. Regulation of embryonic cell adhesion by the prion protein. PLoS Biol. 2009, 7, 576–590. [Google Scholar]
- Lawson, V.A.; Collins, S.J.; Masters, C.L.; Hill, A.F. Prion protein glycosylation. J. Neurochem. 2005, 93, 793–801. [Google Scholar] [CrossRef]
- Castilla, J.P.; Saà, P.; Soto, C. Detection of prion in blood. Nat. Med. 2005, 11, 982–988. [Google Scholar]
- Cooper, J.K.; Ladhani, K.; Minor, P. Comparison of candidate vCJD in vitro diagnostic assays using identical sample sets. Vox Sang. 2012, 102, 100–109. [Google Scholar] [CrossRef]
- King, D.J.; Safar, J.G.; Legname, G.; Prusiner, S.B. Thioaptamer interactions with prion proteins: Sequence-specific and non-specific binding sites. J. Mol. Biol. 2007, 369, 1001–1014. [Google Scholar] [CrossRef]
- Sayer, N.M.; Cubin, M.; Rhie, A.; Bullock, M.; Tahiri-Alaoui, A.; James, W. Structural determinants of conformationally selective; prion-binding aptamers. J. Biol. Chem. 2004, 279, 13102–13109. [Google Scholar]
- Bieschke, J.; Giese, A.; Schulz-Schaeffer, W.; Zerr, I.; Poser, S.; Eigen, M. Ultrasensitive detection of pathological prion protein aggregates by dual-color scanning for intensely fluorescent targets. PNAS 2000, 97, 5468–5473. [Google Scholar] [CrossRef]
- Lane, A.; Stanley, C.J.; Dealler, S.; Wilson, S.M. Polymeric ligands with specificity for aggregated prion proteins. Clin. Chem. 2003, 49, 1774–1775. [Google Scholar] [CrossRef]
- Safar, J.; Cohen, F.; Prusiner, S.B. Quantitative traits of prion strains are enciphered in the conformation of the prion protein. Arch. Virol. Suppl. 2000, 16, 227–235. [Google Scholar]
- Ingrosso, L.; Vetrugno, V.; Cardone, F.; Pocchiari, M. Molecular diagnostics of transmissible spongiform encephalopathies. Trends Mol. Med. 2002, 8, 273–280. [Google Scholar] [CrossRef]
- Saborio, G.; Permanne, B.; Soto, C. Sensitive detection of pathological prion protein by cyclic amplification of protein misfolding. Nature 2001, 411, 810–813. [Google Scholar] [CrossRef]
- Yakovleva, O.; Janiak, A.; McKenzie, C.; McShane, L.; Brown, P.; Cervenakova, L. Effect of protease treatment on plasma infectivity in variant Creutzfeldt-Jakob disease mice. Transfusion 2004, 44, 1700–1705. [Google Scholar] [CrossRef]
- Gofflot, S.; El Moualij, B.; Zorzi, D.; Melen, L.; Roels, S.; Quatpers, D. Immuno-quantitative polymerase chain reaction for detection and quantitation of prion protein. Immunoassay Immunochem. 2005, 25, 241–258. [Google Scholar] [CrossRef]
- Zorzi, W.; El Moualij, B.; Zorzi, D.; Heinen, E.; Melen, L. Immuno PCR en Temps Réel Utilisant un ADN-Chimère Comme Marqueur d'Amplification. European Patent 1232283, 2001. [Google Scholar]
- Gofflot, S.; Deprez, M.; El Moualij, B.; Osman, A.; Thonnart, J.; Hougrand, O. Immunoquantitative PCR for prion protein detection in sporadic Creutzfeldt-Jakob disease. Clin. Chem. 2005, 51, 1605–1611. [Google Scholar] [CrossRef]
- Dupiereux, I.; Zorzi, W.; Lins, L.; Brasseur, R.; Colson, P.; Heinen, E.; El Moualij, B. Interaction of the 106-126 prion peptide with lipid membranes and potential implication for neurotoxicity. Biochem. Biophys. Res. Comm. 2005, 331, 894–901. [Google Scholar] [CrossRef]
- Mille, C.; Debarnot, D.; Zorzi, W.; El Moualij, B.; Coudreuse, A.; Legeay, G.; Quadrio, I.; Perret Liaudet, A.; Poncin-Epaillard, F. Increasing the detection limit of the Parkinson disorder through a specific surface chemistry applied onto inner surface of the titration well. J. Funct. Biomater. 2012, 3, 298–312. [Google Scholar] [CrossRef]
- Vrlinic, T.; Mille, C.; Debarnot, D.; Poncin-Epaillard, F. Oxygen atom density in capacitively coupled RF oxygen plasma. Vacuum 2009, 83, 792–796. [Google Scholar] [CrossRef]
- Mille, C.; Debarnot, D.; Zorzi, W.; El Moualij, B.; Coudreuse, A.; Legeay, G.; Quadrio, I.; Perret Liaudet, A.; Poncin-Epaillard, F. Study of the adhesion of neurodegenerative proteins on plasma-modified and coated polypropylene surfaces. J. Biomat. Sci. Polym. E 2011. [Google Scholar] [CrossRef]
- De Gennes, P.G. Wetting: statics and dynamics. Rev. Mod. Phys. 1985, 57, 827–863. [Google Scholar] [CrossRef]
- Cognard, J. Science et Technologies du Collage; Presses Polytechniques et Universitaire Romandes: Lausanne, Switzerland, 2000; p. 15. [Google Scholar]
- Poncin-Epaillard, F.; Zorzi, W.; El Moualij, B.; Heinen, E.; Legeay, G.; Legeais, V.; Rouault, E. Nouveaux Supports pour l’Immuno-Détection d’Eléments et en Particulier pour le Dosage des Protéines Prions; Applications au Stockage, ou à la Capture Sélective de Molécules d’Intérêt. a) Résultats Complémentaires Dosage de la Protéine Prion. Patent WO 2006/105622, 2006. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mille, C.; Debarnot, D.; Zorzi, W.; Moualij, B.E.; Quadrio, I.; Perret-Liaudet, A.; Coudreuse, A.; Legeay, G.; Poncin-Epaillard, F. A New Approach for Detection Improvement of the Creutzfeldt-Jakob Disorder through a Specific Surface Chemistry Applied onto Titration Well. Biosensors 2012, 2, 433-447. https://doi.org/10.3390/bios2040433
Mille C, Debarnot D, Zorzi W, Moualij BE, Quadrio I, Perret-Liaudet A, Coudreuse A, Legeay G, Poncin-Epaillard F. A New Approach for Detection Improvement of the Creutzfeldt-Jakob Disorder through a Specific Surface Chemistry Applied onto Titration Well. Biosensors. 2012; 2(4):433-447. https://doi.org/10.3390/bios2040433
Chicago/Turabian StyleMille, Caroline, Dominique Debarnot, Willy Zorzi, Benaissa El Moualij, Isabelle Quadrio, Armand Perret-Liaudet, Arnaud Coudreuse, Gilbert Legeay, and Fabienne Poncin-Epaillard. 2012. "A New Approach for Detection Improvement of the Creutzfeldt-Jakob Disorder through a Specific Surface Chemistry Applied onto Titration Well" Biosensors 2, no. 4: 433-447. https://doi.org/10.3390/bios2040433
APA StyleMille, C., Debarnot, D., Zorzi, W., Moualij, B. E., Quadrio, I., Perret-Liaudet, A., Coudreuse, A., Legeay, G., & Poncin-Epaillard, F. (2012). A New Approach for Detection Improvement of the Creutzfeldt-Jakob Disorder through a Specific Surface Chemistry Applied onto Titration Well. Biosensors, 2(4), 433-447. https://doi.org/10.3390/bios2040433