Non-Interventional and High-Precision Temperature Measurement Biochips for Long-Term Monitoring the Temperature Fluctuations of Individual Cells
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chip Fabrication
2.2. Molecular Modification of the Surfaces of the Chips
2.3. Cell Culture and Temperature Monitoring
3. Results and Discussions
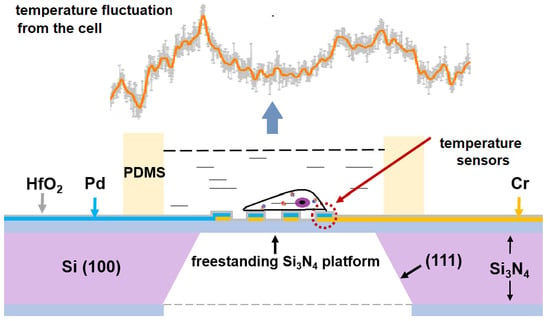
3.1. Design and Properties of the New Biochip
3.1.1. The Temperature-Measurement Chip and Control System
3.1.2. Improving Cell Compatibility by Molecular Modification of the Chip Surface
3.1.3. High Thermal Sensitivity
3.1.4. Low Thermal Noise
3.2. Preliminary Applications in Temperature-Measurements of Individual Cells
3.2.1. Temperature Fluctuations from the Basic Metabolism of Hela Cells
3.2.2. Temperature Drop of Hela Cells Subjected to a Lethal Fluid
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jung, J.; Zeng, H.; Horng, T. Metabolism as a guiding force for immunity. Nat. Cell Biol. 2019, 21, 85–93. [Google Scholar] [CrossRef]
- Martano, G.; Borroni, E.M.; Lopci, E.; Cattaneo, M.G.; Mattioli, M.; Bachi, A.; Decimo, I.; Bifari, F. Metabolism of Stem and Progenitor Cells: Proper Methods to Answer Specific Questions. Front. Molec. Neurosci. 2019, 12, 17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Urbauer, E.; Rath, E.; Haller, D. Mitochondrial Metabolism in the Intestinal Stem Cell Niche-Sensing and Signaling in Health and Disease. Front. Cell. Dev. Biol. 2021, 8, 14. [Google Scholar] [CrossRef] [PubMed]
- Weinan, H.E.; Yan, S.; Ning, G. Cellular temperature measurement and its application prospect in drug development. J. Pharm. Res. 2019. [Google Scholar] [CrossRef]
- Rodenfels, J.; Neugebauer, K.M.; Howard, J. Heat Oscillations Driven by the Embryonic Cell Cycle Reveal the Energetic Costs of Signaling. Dev. Cell. 2019, 48, 646–658. [Google Scholar] [CrossRef] [Green Version]
- Yoo, T.; Lim, K.; Sultan, M.T.; Lee, J.S.; Park, J.; Ju, H.W.; Park, C.; Jang, M. The real-time monitoring of drug reaction in HeLa cancer cell using temperature/impedance integrated biosensors. Sens. Actuators B-Chem. 2019, 291, 17–24. [Google Scholar] [CrossRef]
- Li, C.; Yan, S.; He, W.; Yang, S.; Sun, J.; Gu, N. Real-Time Temperature Measurements of HMEC-1 Cells During Inflammation Production and Repair Detected by Wireless Thermometry. IEEE Trans. Biomed. Eng. 2019, 66, 1898–1904. [Google Scholar] [CrossRef]
- Zhou, J.J.; del Rosal, B.; Jaque, D.; Uchiyama, S.; Jin, D.Y. Advances and challenges for fluorescence nanothermometry. Nat. Methods 2020, 17, 967–980. [Google Scholar] [CrossRef] [PubMed]
- Chung, C.W.; Schierle, G.S.K. Intracellular Thermometry at the Micro-/Nanoscale and its Potential Application to Study Protein Aggregation Related to Neurodegenerative Diseases. ChemBioChem 2021, 22, 1546–1558. [Google Scholar] [CrossRef]
- Liu, Z.; Lavis, L.; Betzig, E. Imaging Live-Cell Dynamics and Structure at the Single-Molecule Level. Mol. Cell. 2015, 58, 644–659. [Google Scholar] [CrossRef] [Green Version]
- Sydor, A.M.; Czymmek, K.J.; Puchner, E.M.; Mennella, V. Super-Resolution Microscopy: From Single Molecules to Supramolecular Assemblies. Trends Cell Biol. 2015, 25, 730–748. [Google Scholar] [CrossRef] [Green Version]
- Kiyonaka, S.; Sakaguchi, R.; Hamachi, I.; Morii, T.; Yoshizaki, T.; Mori, Y. Validating subcellular thermal changes revealed by fluorescent thermosensors. Nat. Methods 2015, 12, 801–802. [Google Scholar] [CrossRef] [PubMed]
- Baffou, G.; Rigneault, H.; Marguet, D.; Jullien, L. “Validating subcellular thermal changes revealed by fluorescent thermosensors” and “The 10(5) gap issue between calculation and measurement in single-cell thermometry” Reply. Nat. Methods 2015, 12, 803. [Google Scholar] [CrossRef]
- Savchuk, O.; Carvajal Marti, J.J.; Cascales, C.; Haro-Gonzalez, P.; Sanz-Rodriguez, F.; Aguilo, M.; Diaz, F. Bifunctional Tm3+,Yb3+:GdVO4@SiO2 Core-Shell Nanoparticles in HeLa Cells: Upconversion Luminescence Nanothermometry in the First Biological Window and Biolabelling in the Visible. Nanomaterials 2020, 10, 993. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, T.; Fukuda, N.; Uchiyama, S.; Inada, N. A Cell-Permeable Fluorescent Polymeric Thermometer for Intracellular Temperature Mapping in Mammalian Cell Lines. PLoS ONE 2015, 10, e0117677. [Google Scholar] [CrossRef] [Green Version]
- Nakano, M.; Arai, Y.; Kotera, I.; Okabe, K.; Kamei, Y.; Nagai, T. Genetically encoded ratiometric fluorescent thermometer with wide range and rapid response. PLoS ONE 2017, 12, e0172344. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chretien, D.; Benit, P.; Ha, H.H.; Keipert, S.; El-Khoury, R.; Chang, Y.T.; Jastroch, M.; Jacobs, H.T.; Rustin, P.; Rak, M. Mitochondria are physiologically maintained at close to 50 °C. PLoS Biol. 2018, 16, e2003992. [Google Scholar] [CrossRef] [Green Version]
- Homma, M.; Takei, Y.; Murata, A.; Inoue, T.; Takeoka, S. A ratiometric fluorescent molecular probe for visualization of mitochondrial temperature in living cells. Chem. Commun. 2015, 51, 6194–6197. [Google Scholar] [CrossRef]
- Bennaim, A.; Casadei, D. Modern Thermodynamics; World Scientific: Hackensack, NJ, USA, 2017. [Google Scholar]
- Stephan, K. Thermodynamik. Grundlagen und Technische Anwendungen; Springer: Berlin/Heidelberg, Germany, 2005. [Google Scholar]
- Tian, W.J.; Wang, C.L.; Wang, J.Q.; Chen, Q.H.; Sun, J.F.; Li, C.; Wang, X.; Gu, N. A high precision apparatus for intracellular thermal response at single-cell level. Nanotechnology 2015, 26, 9. [Google Scholar] [CrossRef]
- Rajagopal, M.C.; Brown, J.W.; Gelda, D.; Valavala, K.V.; Wang, H.; Llano, D.A.; Gillette, R.; Sinha, S. Transient heat release during induced mitochondrial proton uncoupling. Commun. Biol. 2019, 2, 279. [Google Scholar] [CrossRef]
- Yang, F.; Li, G.; Yang, J.; Wang, Z.; Han, D.; Zheng, F.; Xu, S. Measurement of local temperature increments induced by cultured HepG2 cells with micro-thermocouples in a thermally stabilized system. Sci. Rep. 2017, 7, 1721. [Google Scholar] [CrossRef] [Green Version]
- Xu, J.J.; Lei, Z.J.; Guo, J.K.; Huang, J.; Wang, W.; Reibetanz, U.; Xu, S.Y. Trapping and Driving Individual Charged Micro-particles in Fluid with an Electrostatic Device. Nano-Micro Lett. 2016, 8, 270–281. [Google Scholar] [CrossRef] [Green Version]
- Han, D.; Xu, J.; Wang, Z.; Yang, N.; Li, X.; Qian, Y.; Li, G.; Dai, R.; Xu, S. Penetrating effect of high-intensity infrared laser pulses through body tissue. RSC Adv. 2018, 8, 32344–32357. [Google Scholar] [CrossRef] [Green Version]
- Wang, Z.H.; Yang, F.; Han, D.H.; Li, G.; Xu, J.J.; Xu, S.Y. Alternative method to fabricate microdevices on a freestanding Si3N4 window. J. Vac. Sci. Technol. B 2017, 35, 6. [Google Scholar] [CrossRef]
- Bai, S.Y.; Tang, Z.A.; Huang, Z.X.; Yu, J. Thermal Characterization of Si3N4 Thin Films Using Transient Thermoreflectance Technique. IEEE Trans. Ind. Electron. 2009, 56, 3238–3243. [Google Scholar] [CrossRef]
- Alam, M.T.; Manoharan, M.P.; Haque, M.A.; Muratore, C.; Voevodin, A. Influence of strain on thermal conductivity of silicon nitride thin films. J. Micromech. Microeng. 2012, 22, 8. [Google Scholar] [CrossRef] [Green Version]
- Sze, S.M.; Lee, M.K. Semiconductor Devices: Physics and Technology, Desktop Edition, 3rd ed.; John Wiley & Sons: New York, NY, USA, 2012. [Google Scholar]
- Strnad, J.; Vengar, A. Stefan’s measurement of the thermal conductivity of air. Eur. J. Phys. 1984, 5, 9. [Google Scholar] [CrossRef]
- Yamada, M.; Akashi, K.; Ooka, R.; Miyado, K.; Akutsu, H. Mitochondrial Genetic Drift after Nuclear Transfer in Oocytes. Int. J. Mol. Sci. 2020, 21, 5880. [Google Scholar] [CrossRef]
- Wang, T.Q.; Yang, W.X. Nuclear regulation of mitochondrial functions during oocyte development. Biocell 2020, 44, 469–478. [Google Scholar] [CrossRef]
- Li, H.Z.; Slone, J.; Huang, T.S. The role of mitochondrial-related nuclear genes in age-related common disease. Mitochondrion 2020, 53, 38–47. [Google Scholar] [CrossRef]
- Suzuki, M.; Tseeb, V.; Oyama, K.; Ishiwata, S. Microscopic detection of thermogenesis in a single HeLa cell. Biophys. J. 2007, 92, L46–L48. [Google Scholar] [CrossRef] [Green Version]
- Ghasemi, F.; Shafiee, M.; Banikazemi, Z.; Pourhanifeh, M.H.; Khanbabaei, H.; Shamshirian, A.; Moghadam, S.A.; ArefNezhad, R.; Sahebkar, A.; Avan, A.; et al. Curcumin inhibits NF-kB and Wnt/beta-catenin pathways in cervical cancer cells. Pathol. Res. Pract. 2019, 215, 6. [Google Scholar] [CrossRef] [PubMed]
- Su, Z.Y.; Yang, H.; Zhao, M.; Wang, Y.L.; Deng, G.Y.; Chen, R.X. MicroRNA-92a Promotes Cell Proliferation in Cervical Cancer via Inhibiting p21 Expression and Promoting Cell Cycle Progression. Oncol. Res. 2017, 25, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Yamamura, M.; Hayatsu, H.; Miyamae, T. Heat production as a cell cycle monitoring parameter. Biochem. Biophys. Res. Commun. 1986, 140, 414–418. [Google Scholar] [CrossRef]
- Okabe, K.; Inada, N.; Gota, C.; Harada, Y.; Funatsu, T.; Uchiyama, S. Intracellular temperature mapping with a fluorescent polymeric thermometer and fluorescence lifetime imaging microscopy. Nat. Commun. 2012, 3, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ogino, Y.; Liang, R.W.; Mendonca, D.B.S.; Mendonca, G.; Nagasawa, M.; Koyano, K.; Cooper, L.F. RhoA-Mediated Functions in C3H10T1/2 Osteoprogenitors Are Substrate Topography Dependent. J. Cell. Physiol. 2016, 231, 568–575. [Google Scholar] [CrossRef] [Green Version]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, D.; Xu, J.; Wang, H.; Wang, Z.; Yang, N.; Yang, F.; Shen, Q.; Xu, S. Non-Interventional and High-Precision Temperature Measurement Biochips for Long-Term Monitoring the Temperature Fluctuations of Individual Cells. Biosensors 2021, 11, 454. https://doi.org/10.3390/bios11110454
Han D, Xu J, Wang H, Wang Z, Yang N, Yang F, Shen Q, Xu S. Non-Interventional and High-Precision Temperature Measurement Biochips for Long-Term Monitoring the Temperature Fluctuations of Individual Cells. Biosensors. 2021; 11(11):454. https://doi.org/10.3390/bios11110454
Chicago/Turabian StyleHan, Danhong, Jingjing Xu, Han Wang, Zhenhai Wang, Nana Yang, Fan Yang, Qundong Shen, and Shengyong Xu. 2021. "Non-Interventional and High-Precision Temperature Measurement Biochips for Long-Term Monitoring the Temperature Fluctuations of Individual Cells" Biosensors 11, no. 11: 454. https://doi.org/10.3390/bios11110454