Determination of Alanine Aminotransferase with an Electrochemical Nano Ir-C Biosensor for the Screening of Liver Diseases
Abstract
:1. Introduction

2. Experimental Section
2.1. Materials and Reagents
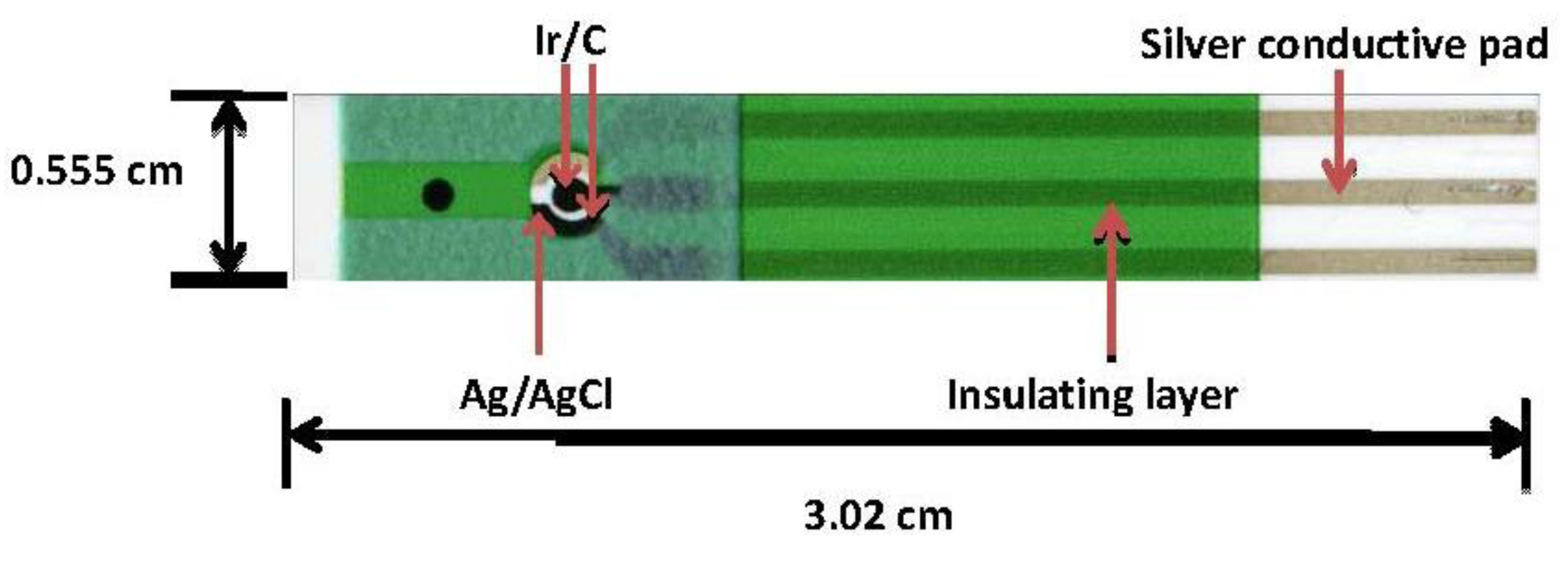
2.2. Thick-Film Screen-Printed Prototype
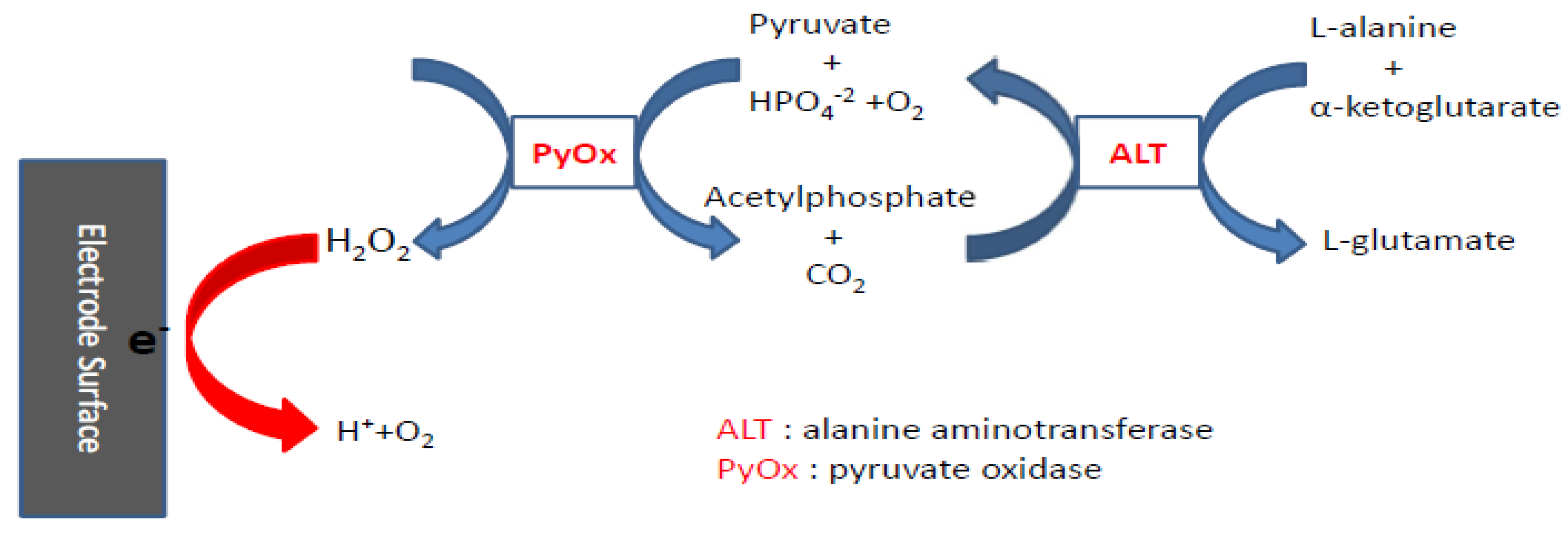
2.3. Experimental Testing Procedure
3. Results and Discussion
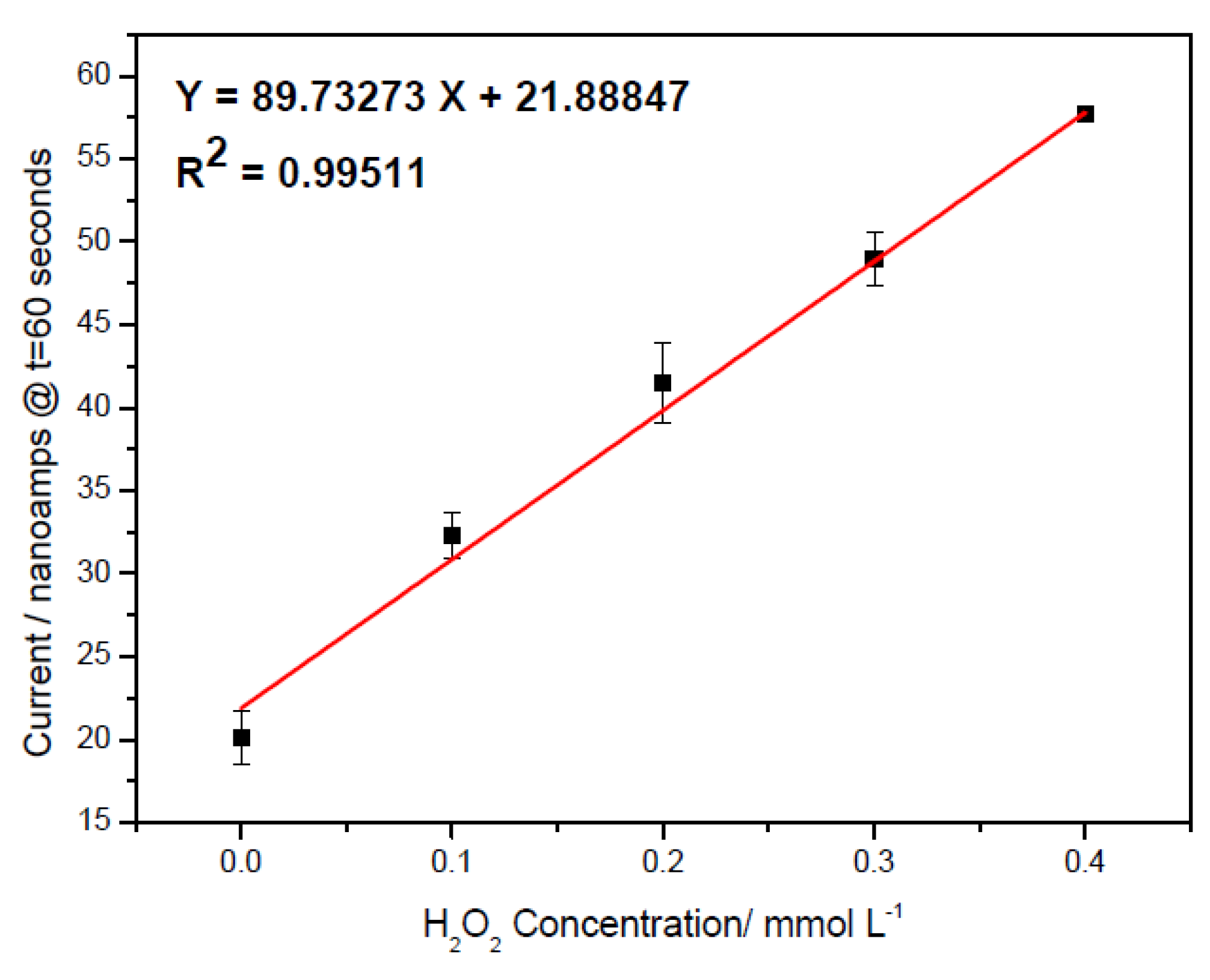
3.1. H2O2 Detection in Buffer Solution
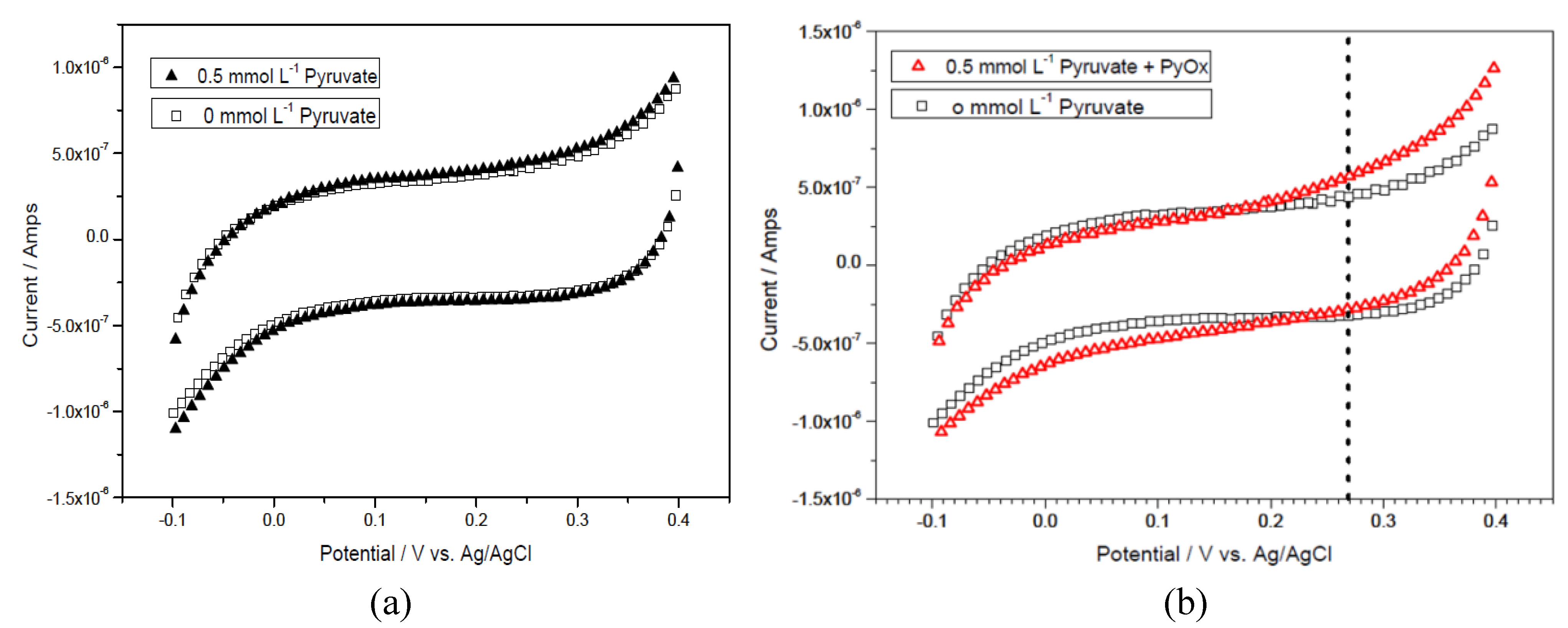
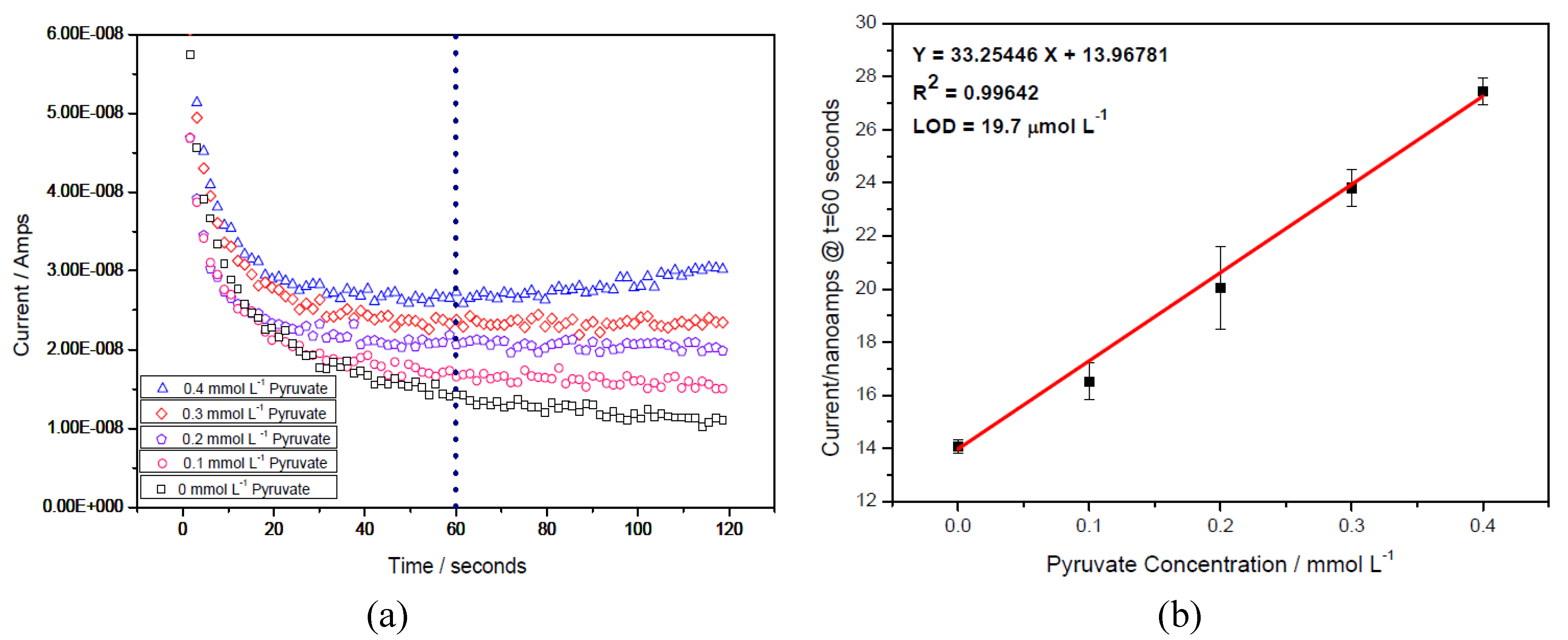
3.2. Determination of Pyruvate Concentration with Enzymatically Generated H2O2
3.3. Determination of Alanine Aminotransferase (ALT)
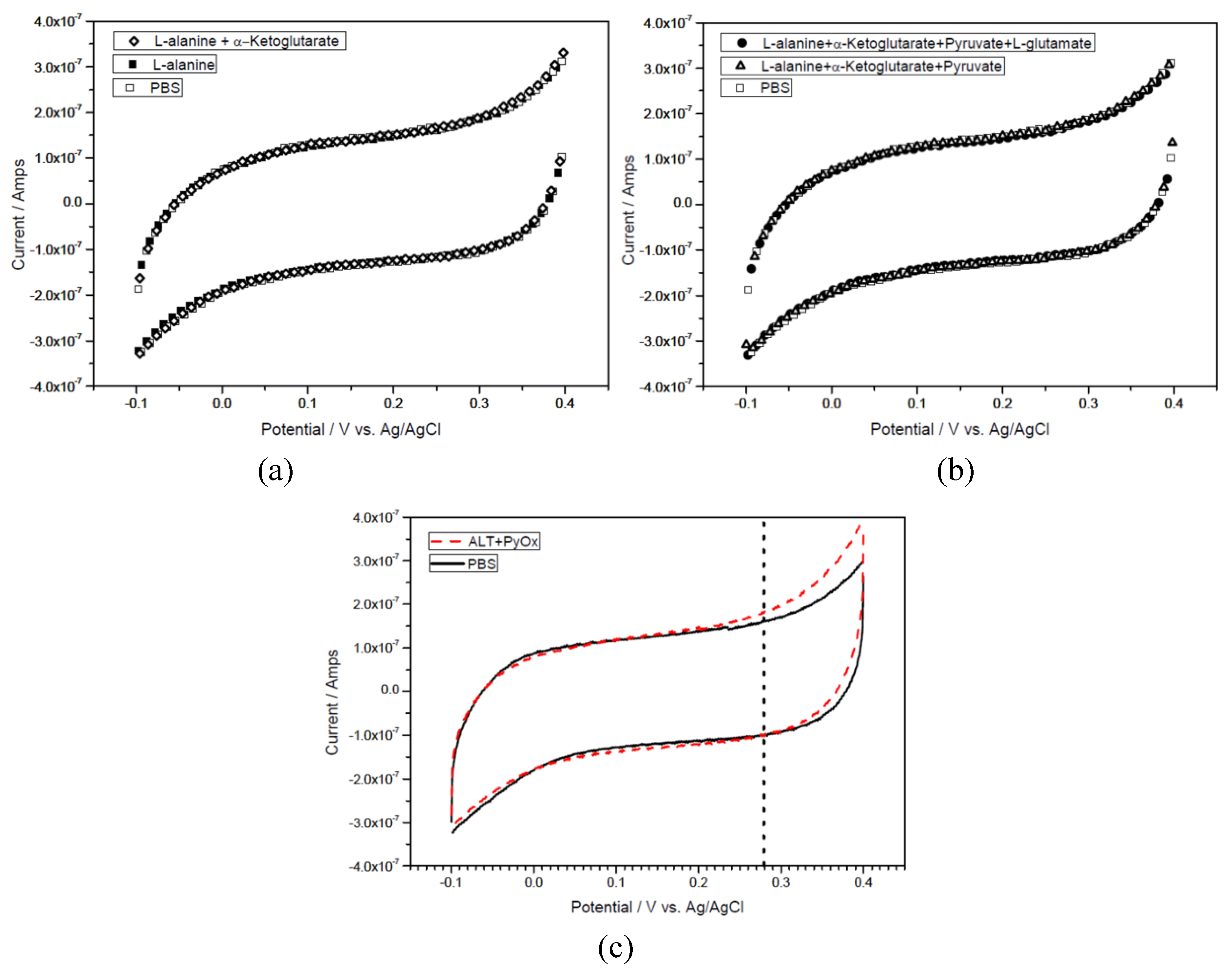
3.3.1. ALT Detection with Cyclic Voltammetry
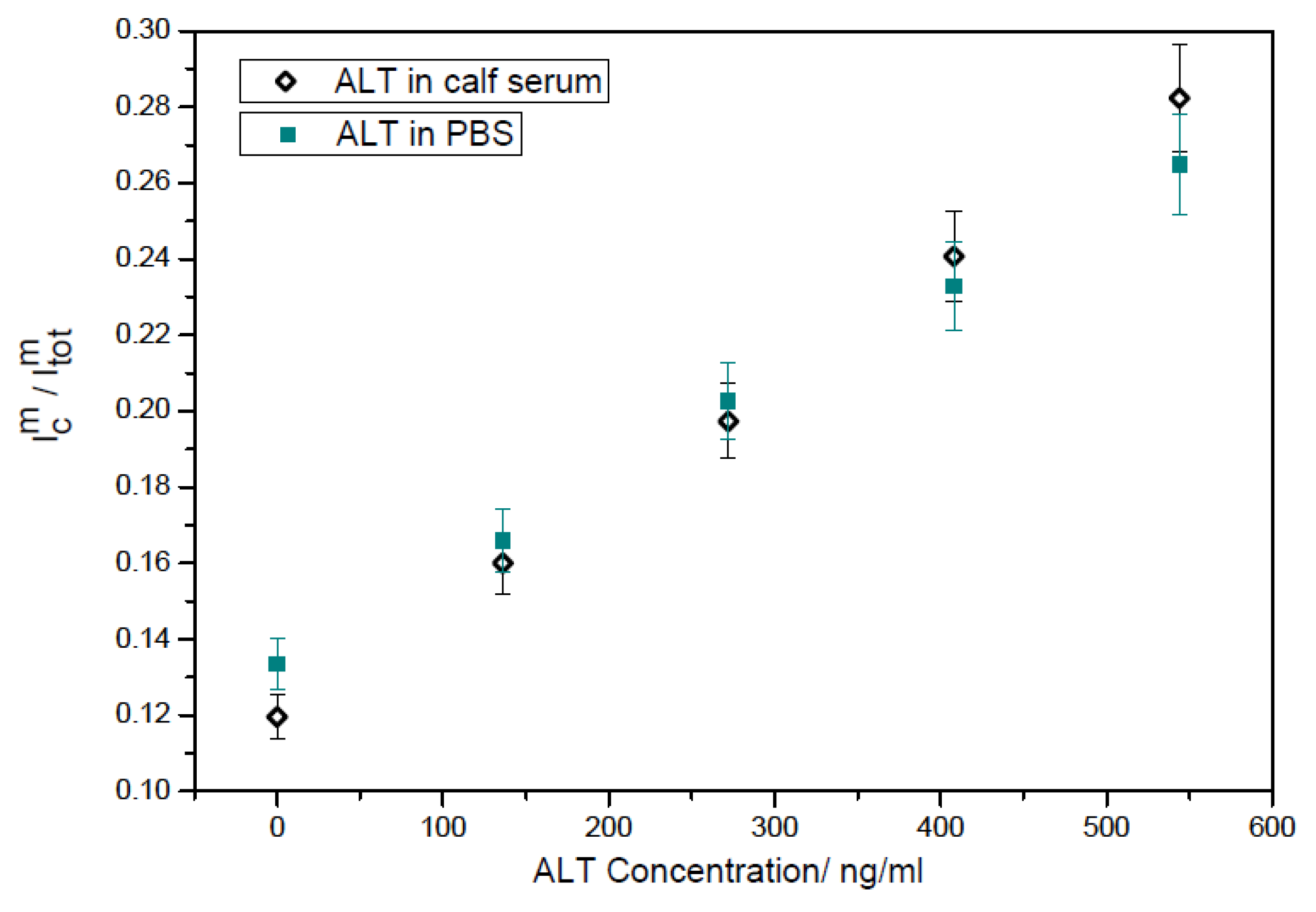
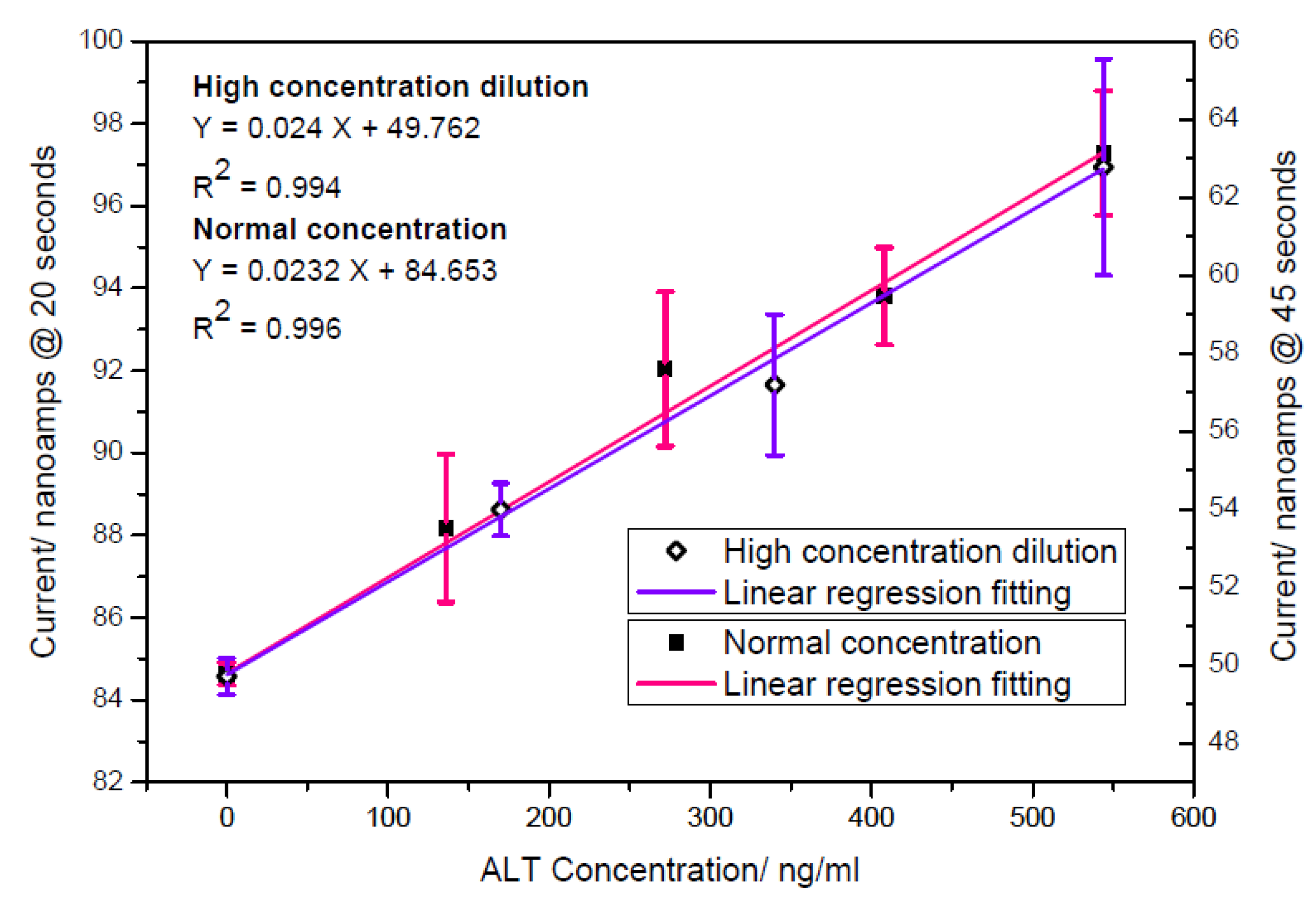
3.3.2. ALT Detection with Amperometric Method

4. Conclusions
Acknowledgments
References
- Dafour, D.R.; Lott, J.A.; Nolte, F.S.; Gretch, D.R.; Koff, R.S.; Seeff, L.B. Diagnosis and monitoring of hepatic injury. I. Performance characteristics of laboratory tests. Clin. Chem. 2000, 46, 2027–2049. [Google Scholar]
- Huang, X.J.; Choi, Y.K.; Im, H.S.; Oktay, Y.; Euisik, Y.; Kim, H.S. Aspartate aminotransferase (AST/GOT) and alanine aminotransferase (ALT/GPT) detection techniques. Sensors 2006, 6, 756–782. [Google Scholar] [CrossRef]
- Knight, J.A.; Hunter, D.T. Aspartic transaminase, alanine transaminase and lactate dehydrogenase determination by automated spectrophotometric analysis. J. Med. Lab Technol. 1968, 25, 106–111. [Google Scholar]
- Jamal, M.; Worsfold, O.; McCormac, T.; Dempsey, E. A stable and selective electrochemical biosensor for the liver enzyme alanine aminotransferase (ALT). Biosens. Bioelectron. 2009, 24, 2926–2930. [Google Scholar] [CrossRef]
- Song, M.J.; Yun, D.H.; Min, N.K.; Hong, S.I. Electrochemical biosensor array for liver diagnosis using silanization technique on nanoporous silicon electrode. J. Biosci. Bioeng. 2007, 103, 32–37. [Google Scholar] [CrossRef]
- Song, M.J.; Yun, D.H.; Hong, S.I. An electrochemical biosensor array for rapid detection of alanine aminotransferase and aspartate aminotransferase. Biosci. Biotechnol. Biochem. 2009, 73, 474–478. [Google Scholar] [CrossRef]
- Matos, R.C.; Augelli, M.A.; Lago, C.L.; Angnes, L. Flow injection analysis-amperometric determination of ascorbic and uric acids in urine using arrays of gold microelectrodes modified by electrodeposition of palladium. Anal. Chim. Acta 2000, 404, 151–157. [Google Scholar] [CrossRef]
- Chang, K.S.; Chang, C.K.; Chou, S.F.; Chen, C.Y. Sequential measurement of aminotransferase activities by amperometric biosensors. Biosens. Bioelectron. 2007, 22, 2914–2920. [Google Scholar] [CrossRef]
- Li, C.M.; Cha, C.S. Porous carbon composite/enzyme glucose microsensor. Front Biosci. 2004, 9, 3324–3330. [Google Scholar] [CrossRef]
- Xuan, G.S.; Oh, S.W.; Choi, E.Y. Development of an electrochemical immunosensor for alanine aminotransferase. Biosens. Bioelectron. 2003, 19, 365–371. [Google Scholar] [CrossRef]
- Guo, C.X.; Zheng, X.T.; Lu, Z.S.; Lou, X.W.; Li, C.M. Biointerface by cell growth on layered grapheme-artificial peroxidase-protein nanostructure for in situ quantitative molecular detection. Adv. Mater. 2010, 22, 5164–5167. [Google Scholar] [CrossRef]
- Guo, C.X.; Zheng, X.T.; Ng, S.R.; Lai, Y.; Lei, Y.; Li, C.M. In situ molecular detection of ischemic cells by enhanced protein direct electron transfer on a unique horseradish peroxidase-Au nanoparticles-polyaniline nanowires biofilm. Chem. Commun. 2011, 47, 2652–2654. [Google Scholar]
- Fang, L.; Wang, S.H.; Liu, C.C. An electrochemical biosensor of the ketone 3-[beta]-hydroxybutyrate for potential diabetic patient management. Sens. Actuat. B 2008, 2, 818–825. [Google Scholar] [CrossRef]
- Shen, J.; Dudik, L.; Liu, C.C. An iridium nanoparticles dispersed carbon based thick film electrochemical biosensor and its application for a single use, disposable glucose biosensor. Sens. Actuat. B 2007, 1, 106–113. [Google Scholar] [CrossRef]
- Bartling, B.; Li, L.; Liu, C.C. Determination of total bile acid levels using a thick-film screen-printed Ir/C sensor for the detection of liver disease. Analyst 2009, 134, 973–979. [Google Scholar] [CrossRef]
- Liao, W.Y.; Liu, C.C.; Wang, C. Detection of lipoprotein-associated phospholipase A2 using a nano-iridium particle catalyst-based biosensor. Sens. Actuat. B 2008, 2, 993–999. [Google Scholar] [CrossRef]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hsueh, C.-J.; Wang, J.H.; Dai, L.; Liu, C.-C. Determination of Alanine Aminotransferase with an Electrochemical Nano Ir-C Biosensor for the Screening of Liver Diseases. Biosensors 2011, 1, 107-117. https://doi.org/10.3390/bios1030107
Hsueh C-J, Wang JH, Dai L, Liu C-C. Determination of Alanine Aminotransferase with an Electrochemical Nano Ir-C Biosensor for the Screening of Liver Diseases. Biosensors. 2011; 1(3):107-117. https://doi.org/10.3390/bios1030107
Chicago/Turabian StyleHsueh, Chang-Jung, Joanne H. Wang, Liming Dai, and Chung-Chiun Liu. 2011. "Determination of Alanine Aminotransferase with an Electrochemical Nano Ir-C Biosensor for the Screening of Liver Diseases" Biosensors 1, no. 3: 107-117. https://doi.org/10.3390/bios1030107