1. Introduction
All the organic chemistry of terrestrial organisms is based on the L form of amino acids. Despite much effort devoted to elucidating how and why
l-amino acids were preferentially selected with respect to the
d-enantiomer no clear solution has yet been obtained [
1]. Some enantioselective mechanisms have been proposed however none of them deal with organic molecules in plausible prebiotic Earth environments [
2,
3,
4]. In the prebiotic chemistry research community, there is no unique, single, universal model of the prebiotic environmental conditions. There is no agreement as to what the exact conditions were regarding temperature, pH, red-ox potential, inorganic precursors, pressure (surface of liquid, gaseous phase, deep ocean) or even which were the first organic molecules that launched the auto-catalytic reactions. In practice, all plausible situations are explored experimentally because they may be representative of the present or past environmental conditions in the ancient Earth and also because they may be relevant to investigate the habitability of other outer Earth environments, such as Mars, Titan, Europa or even extra solar planets. Given that the prebiotic scenario has not yet been unequivocally identified, there is no agreement either as to what mechanism may have caused the original selection of one chiral handiness over the other. All scenarios are currently considered but to date no experiment has explored the plausible interaction of geomagnetic fields and primitive organic chiral molecules, such as amino acids, on the primordial ocean surface.
Simple symmetry arguments demonstrate that a static magnetic field cannot induce absolute enantioselection at thermodynamical equilibrium [
5,
6]. However, for far-from-equilibrium processes a magnetic field might induce enantioselection if the molecules are aligned since this constitutes a falsely chiral (
i.e., time-noninvariant enantiomorphous) arrangement [
5,
6]. According to this, a magnetic field alone might induce deracemization if the molecules are pre-aligned, as in a crystal or on the liquid-air surface and the process is far from equilibrium [
5]. For this research, we shall investigate an out-of-equilibrium process where amino-acid arrangement is possible: the crystallization, at the air-liquid interface, of a sodium chlorate solution with racemic aminoacid additives. This experimental observations illustrates how the geomagnetic field,
i.e., a falsely chiral influence, can induce deracemization and enantioselection in interaction with racemic amino-acid mixtures.
To investigate this effect, one needs a technique that is sensitive to tiny amounts of chiral hydrophobic amino acids located only at the upper molecular layers of a saline water solution. We propose to use the crystallization of sodium chlorate NaClO
3 (a process which has proven to be particularly sensitive to tiny amounts of hydrophobic chiral amino acids) as a probe for plausible amino acid enantiomeric interactions at this interface. NaClO
3 is an achiral molecule but when several molecules join into small crystal nuclei they crystallize in an ordered spiral-like structure which can be either levo (l) or dextro (d) rotatory. A saturated NaClO
3 solution left at room temperature in a wide vessel crystallizes into an almost racemic mixture of crystals,
i.e., on average there are no preferences for (l) or (d) crystals [
7]. The crystallization of NaClO
3 has shown to be a very convenient process to detect tiny chiral effects that take place within the water-air interface as well as to detect the chiral sign of amino acid impurities [
8,
9,
10,
11]. We shall test for biases in the water-air induced crystallization of NaClO
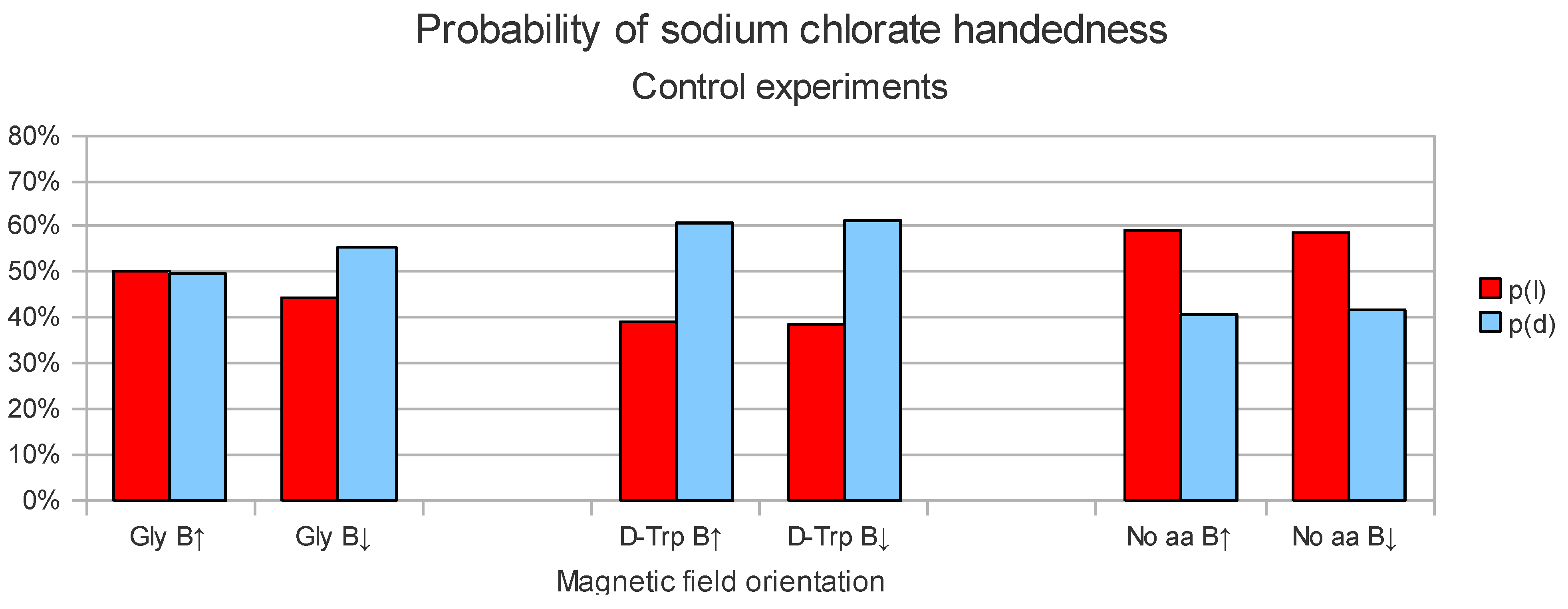
3 crystallized with racemic additives (of the order of a few ppms) of hydrophobic amino acids (which will tend to be at the water-air interface of the solution) and an upwards or downwards external magnetic field. Three additional sets of control experiments, one without additives, one with the achiral hydrophobic amino-acid Gly additive and one with only one handedness of chiral hydrophobic amino acid (
d-Trp), are investigated for comparison.
2. Experimental Section
In order to overcome the geomagnetic field at Madrid, where the experiments took place, and be able to control the direction of the local (downwards pointing) magnetic field we need to use an external magnetic field greater than 0.05 mT. We used a set of small Neodymium dipole magnets of about 5 mm height and 17 mm diameter. The magnetic field at the center has been measured and found to be 100 mT at a distance of 7 mm and 200 mT at 3 mm. This is the range of distances between the magnets and the water-air interface in our two different types of experimental setups. Which means that at the water-air interface which is right above the center of the magnets, where the maximal field is experienced, there is a magnetic field of 200 mT at most, i.e., roughly 4000 times greater than the actual maximal geomagnetic field.
All glass material was steamed for sterilization during 2 h. The experiments were run at room temperature. We used NaClO3 from Sigma-Aldrich S.A. (ACS quality) and ultrapure water (Millipore Milli-Q Q. Gard). The NaClO3 used has a 99.91% purity. The ferromagnetic metals Fe, Mn and Co are present as impurities in amounts of maximum 5 ppm each. These metallic impurities are unavoidable and constantly present as a background together with the salt ions and ambient impurities. For each crystallization process 50 mL of a NaClO3 aqueous solution (48.8 g NaClO3 in 100 g of dissolution, saturated 51.2%, 9.86 molal saturated at 30 °C) was prepared at 45 °C under vigorous stirring (500 rpm) together with a small amount (4 ppm for the aerosol experiments, and 20 ppm for the steady crystallization ones) of one or several amino acid forms, depending on the experiment. All the amino acids were obtained from Sigma-Aldrich. The hot solution was filtered, through a 0.2 μm-pore size sterile nylon.
To guarantee the independence of the observations with respect to the specific details of the experimental setup we propose two different techniques where the interface between the bulk liquid and the air is relevant. The solution described above is put to crystallize under two possible setups: an aerosol-liquid cycle (see
Figure 1-Left) or a steady crystallization (see
Figure 1-Right). For the aerosol-liquid setup, a round-bottom flask, with 5 mL of the liquid solution, was placed within a bath of the ultrasonic aerosol generator (a commercial, small ultrasound generator of 1'8 MHz, 30 W) [
10]. After 2 min, the ultrasonic device induces the formation of an optically thick cloud of aerosols generated from the water-air interface solution. There is a continuous recycling process, from the aerosol phase to the liquid bulk interface and back. Because of the intense evaporation in the aerosols, this is the first environment to reach supersaturation and crystal nucleation of NaClO
3 is initiated at the aerosol phase roughly after 2 h. This method induces in about 4 h the homochiral crystallization of NaClO
3 for the steady crystallization setup we have crystallized 1 mL of the solution described above in a small open crystal vessel, of about 2 cm diameter and 4 cm height under different experimental conditions. The first crystal nuclei are formed on the liquid pool surface which, due to the evaporation, is the first place to reach supersaturation. These crystals then grow further at the bottom of the vessel, reducing the supersaturation in NaClO
3 and preventing the formation of new crystals nuclei. After between one and three days there are observable crystals of few mm size within the remaining liquid solution.
Figure 1.
Water-air induced crystallization setups. (Left) Aerosol experimental setup. Here the flask has a magnet within the crystal vessel with the South pole facing upwards. (Right) Steady crystallization experimental setup, one vessel with magnetic field and one without.
Figure 1.
Water-air induced crystallization setups. (Left) Aerosol experimental setup. Here the flask has a magnet within the crystal vessel with the South pole facing upwards. (Right) Steady crystallization experimental setup, one vessel with magnetic field and one without.
Both crystallization setups share an essential feature: they initiate the crystallization with constituents of the solution at the interface between air and the liquid bulk and are very sensitive to water-air surface impurities [
10,
11]. As we will show later the final preferred handedness of NaClO
3 may be biased by chiral impurities (such as amino acids) located within this environment and is independent of the procedure used. Once this is confirmed and to run a larger number of experiments in parallel improving the statistics, the steady crystallization procedure will be the only one used.
The chirality of the crystals obtained under either crystallization setup was determined from their optical activity using a stereomicroscope and two linear polarizers [
12,
13,
14,
15]. See
Figure 2 for two examples of homochiral crystallization distributions. We count the experiments that lead to (l) homochiral experiments n
(l), and (d) homochiral experiments n
(d) for a set of experimental cases. If all (either one or many) the crystals in the vessel are levorotatory the result is marked as (l), if all are dextrorotatory the result is (d). The rest which have both (l) and (d) crystals in arbitrary rate are classified as (r) [
14,
15].
Figure 2.
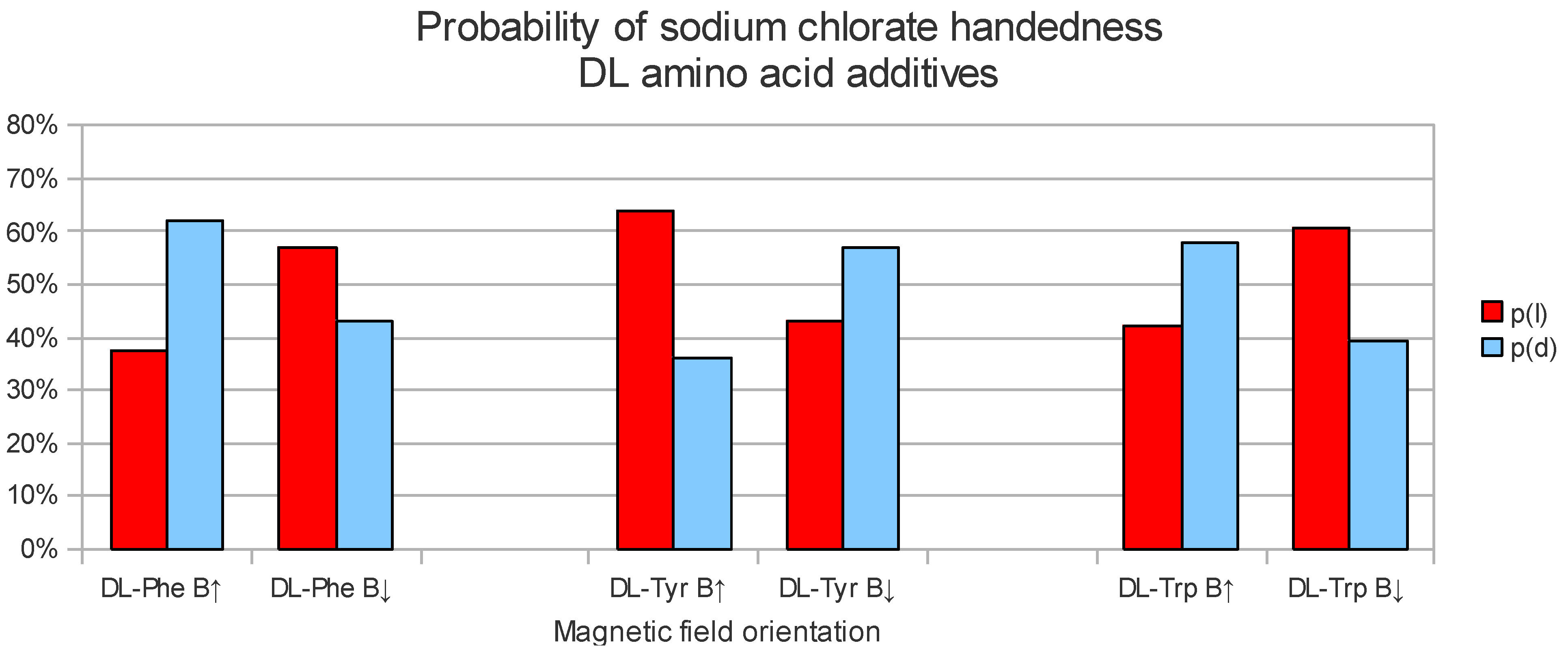
Examples of NaClO3 homochiral distributions of crystals obtained from an aerosol crystallization experiment (Left) and a steady crystallization experiments (Right). The biggest crystals are a few mm size. (Left) Dextro homochiral NaClO3 crystals (here seen as light crystals) obtained from the co-crystallization with 4 ppm of dl-Phe and an external upwards pointing magnetic field (B↑). (Right) Levo (here seen as dark crystals) homochiral crystals obtained from the co-crystallization with 20 ppm of dl-Phe and an external downwards pointing magnetic field (B↓).
Figure 2.
Examples of NaClO3 homochiral distributions of crystals obtained from an aerosol crystallization experiment (Left) and a steady crystallization experiments (Right). The biggest crystals are a few mm size. (Left) Dextro homochiral NaClO3 crystals (here seen as light crystals) obtained from the co-crystallization with 4 ppm of dl-Phe and an external upwards pointing magnetic field (B↑). (Right) Levo (here seen as dark crystals) homochiral crystals obtained from the co-crystallization with 20 ppm of dl-Phe and an external downwards pointing magnetic field (B↓).
In order to detect chiral effects at the interfaces, we first need to confirm that the handedness of NaClO
3 can be biased by the only presence of a chiral amino acid, the results are summarized in
Table 1. We performed an approximately equal number of experiments with each setup using solutions with
d- and
l-Phe,
d- and
l-Tyr. To guarantee the independence of the observations with respect to the specific details of the experimental setup (aerosol cycle
vs. steady crystal vessel), the observations of both crystallization setups were added. The biased is observed in both experimental setups equally, confirming that the water-air interface is the place where the final handedness is determined. Roughly 69% ± 4% of the experiments with homochiral amino acid additives lead to pure homochiral NaClO
3 crystallizations, the rest which have both (l) and (d) crystals are discarded in this analysis. In addition, experiments with
d- and
l-Trp have been performed however using only the steady crystallization process. In this later case, roughly 54% ± 12% of the experiments are homochiral. A control experiment (using only the steady crystallization process), has been run adding an achiral amino acid instead, Gly, see
Table 1. In this case roughly 55% ± 9% of the experiments are homochiral. Using the experimental data of
Table 1 we derive, with a confidence level of 95%, the true proportion
p of each handedness of NaClO
3 which varies depending upon the added amino acid. These probabilities are plot in
Figure 3.
Table 1.
Amino acid effect on the crystallizations of NaClO3 with amino acid impurities under water-air induced crystallization conditions. The addition of chiral hydrophobic amino acids favors a preferred handedness in the sodium chlorate crystallization, the achiral hydrophobic amino acid Gly does not induce a significant bias.
Table 1.
Amino acid effect on the crystallizations of NaClO3 with amino acid impurities under water-air induced crystallization conditions. The addition of chiral hydrophobic amino acids favors a preferred handedness in the sodium chlorate crystallization, the achiral hydrophobic amino acid Gly does not induce a significant bias.
| Chiral Amino Acid Impurity | n(l) | n(d) |
|---|
| (a) l-Phe | 31 | 41 |
| (b) d-Phe | 38 | 24 |
| (c) l-Tyr | 31 | 23 |
| (d) d-Tyr | 18 | 25 |
| (e) l-Trp | 69 | 73 |
| (f) d-Trp | 66 | 51 |
| (g) Gly | 90 | 82 |
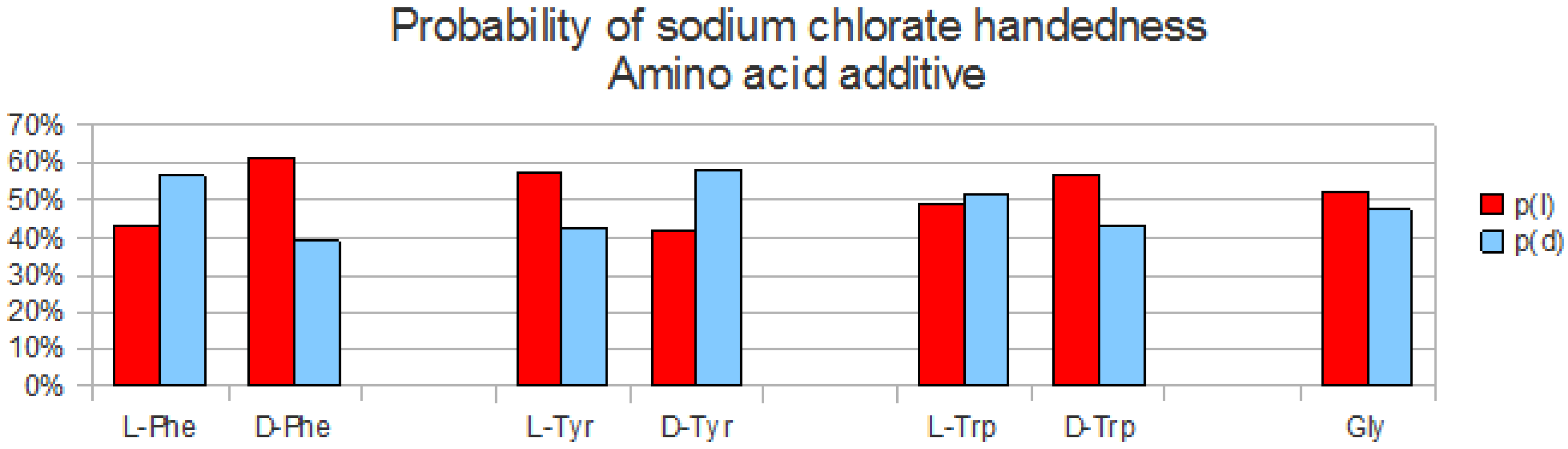
Figure 3.
True proportion or probability of sodium chlorate handedness, (l) vs. (d), as a function of added hydrophobic amino acid. Both handedness are equiprobable, up to the statistical error, in the case of Gly, whereas with chiral amino acids (l or d) Phe, Tyr and Trp additives, there is a significant bias in the proportion towards one or other handedness that depends on the chiral sign of the amino acid and on the amino acid type.
Figure 3.
True proportion or probability of sodium chlorate handedness, (l) vs. (d), as a function of added hydrophobic amino acid. Both handedness are equiprobable, up to the statistical error, in the case of Gly, whereas with chiral amino acids (l or d) Phe, Tyr and Trp additives, there is a significant bias in the proportion towards one or other handedness that depends on the chiral sign of the amino acid and on the amino acid type.
The crystallization of sodium chlorate is exposed to ambient contaminants and also to the unavoidable salt and additive impurities. To assess if the handedness of an amino acid additive alone drives a non-random biased output, we apply Fisher’s exact statistical test to pairs of experiments with opposing conditions. In each pair we shall evaluate if the variation of one single factor induces a significantly different output from one setup to the other, keeping in mind that all background effects (ambient, solute and solvant impurities) are common in both situations. By applying Fisher test to the results in
Table 1, we conclude that the handedness of NaClO
3 is biased by the presence of one or other enantiomer of the amino acid, with a level of confidence of 98.89% for Phe (rows a and b), with a level of confidence of 95.81% for Tyr (rows c and d) and with a level of confidence of 91.61% for Trp (rows e and f). In the case of Gly, the outcome is almost equally probable (52% ± 8% for (l) handedness) for one and other handedness showing that an achiral amino acid additive does not bias the crystallization of sodium chlorate. These tests confirm that these experimental setups can be used to detect the footprint of the organization of chiral hydrophobic amino acids at the water air interface.