Bridging the Fields of Solar Cell and Battery Research to Develop High-Performance Anodes for Photoelectrochemical Cells and Metal Ion Batteries
Abstract
:1. Introduction


2. The Challenge: A Systematic Study of Functionalized, Alloyed TiO2 Anodes
2.1. Project Scope
2.2. Specific Aims
- 1)
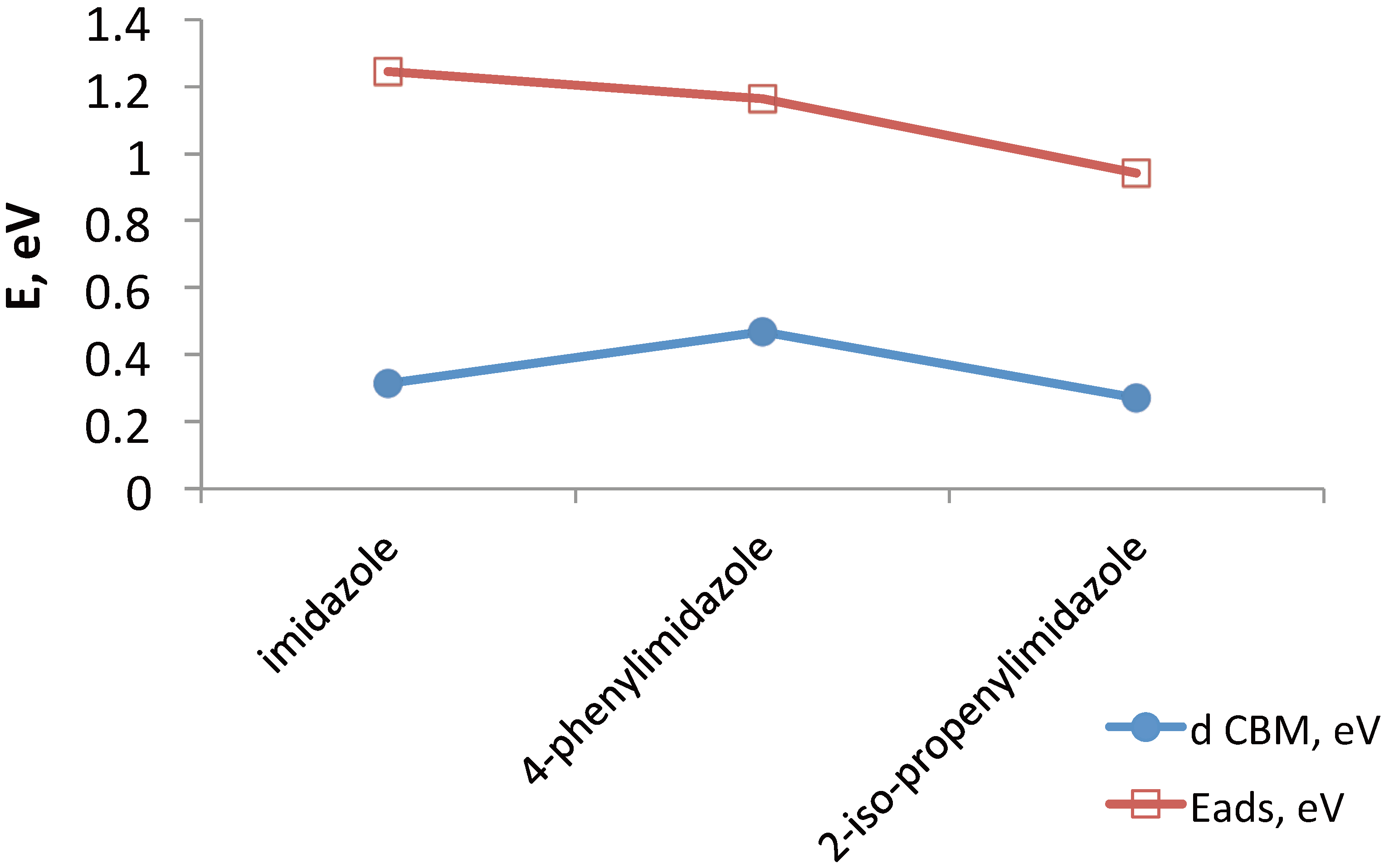
- How do molecular co-adsorbents affect the electronic structure of the dye-semiconductor interface and, consequently, electron injection into TiO2 and recombination of injected electrons with electrolyte species? We need to understand the trends in the effects of molecular co-adsorbents (specifically on dye adsorption configuration and energy and on energy level matching) depending on their molecular structure, including the influence of specific functional groups, such as nitrogen containing heterocycles. This knowledge will help design co-adsorbent-dye combinations enhancing the performance of mesoscopic solar cells.
- 2)
- Do Li or Na counterions insert into and de-insert from the TiO2 anode under the photoelectrochemical cell’s operating conditions? Specifically, does the metal ion concentration in TiO2 affect significantly the electronic structure, conduction, electron injection (short circuit current), diffusion and recombination (open circuit voltage)?
- 3)
- Can the interaction with the metal ion change the phase of the nanostructured anode material? If so, is this a reversible change? This could have a profound effect on the durability of mesoscopic solar cells.
- 4)
- How does this interaction depend on the kind of ion (e.g., Li vs. Na) and the specific polymorph of TiO2? How does it depend on the nanoparticle morphology and the surface indices of the facets approached by the ion? This includes a comparative study of anode structures (phases, facets) used in photoelectrochemical cells and those used as prospective anodes in Li or Na ion batteries. This knowledge will enable the design of anodes with desired insertion (for metal ion batteries), absorption and conduction band (for DSSC) properties.
- 5)
- To what extent is it possible to decouple the design of co-adsorbents and counterions from the design of dyes? There are conflicting reports in the literature about this [32].
3. Detailed Proposal
3.1. Detailed Justification
3.1.1. Importance of Developing Functionalized TiO2 Anodes for Mesoscopic Solar Cells
3.1.2. Ion Insertion into a TiO2 Anode: A Common Yet Little Studied Phenomenon in Photoelectrochemical Solar Cells and Batteries
3.2. Theoretical and Computational Analysis
3.2.1. The Effect of Molecular Co-Adsorbates on the Electronic Properties of the Anode
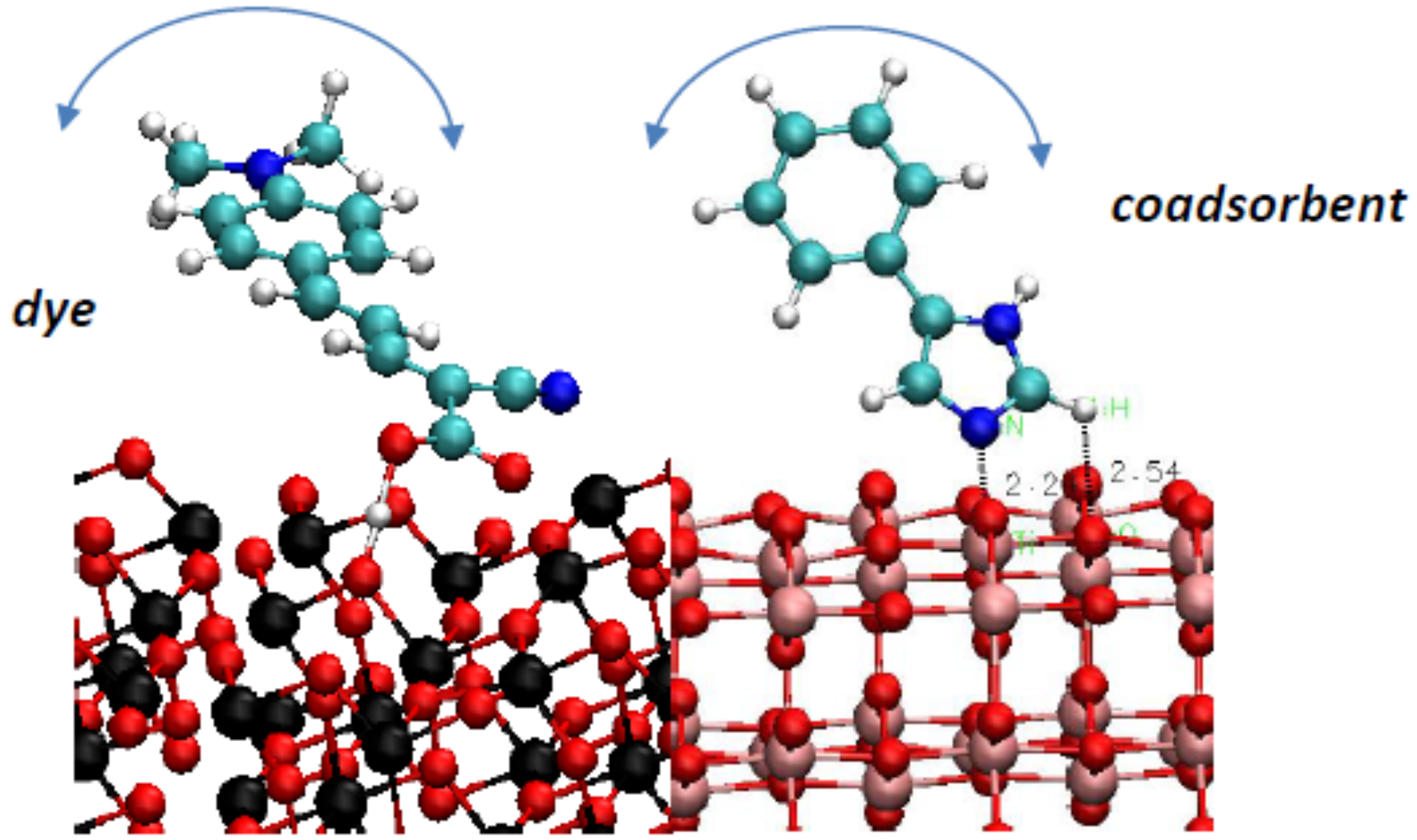
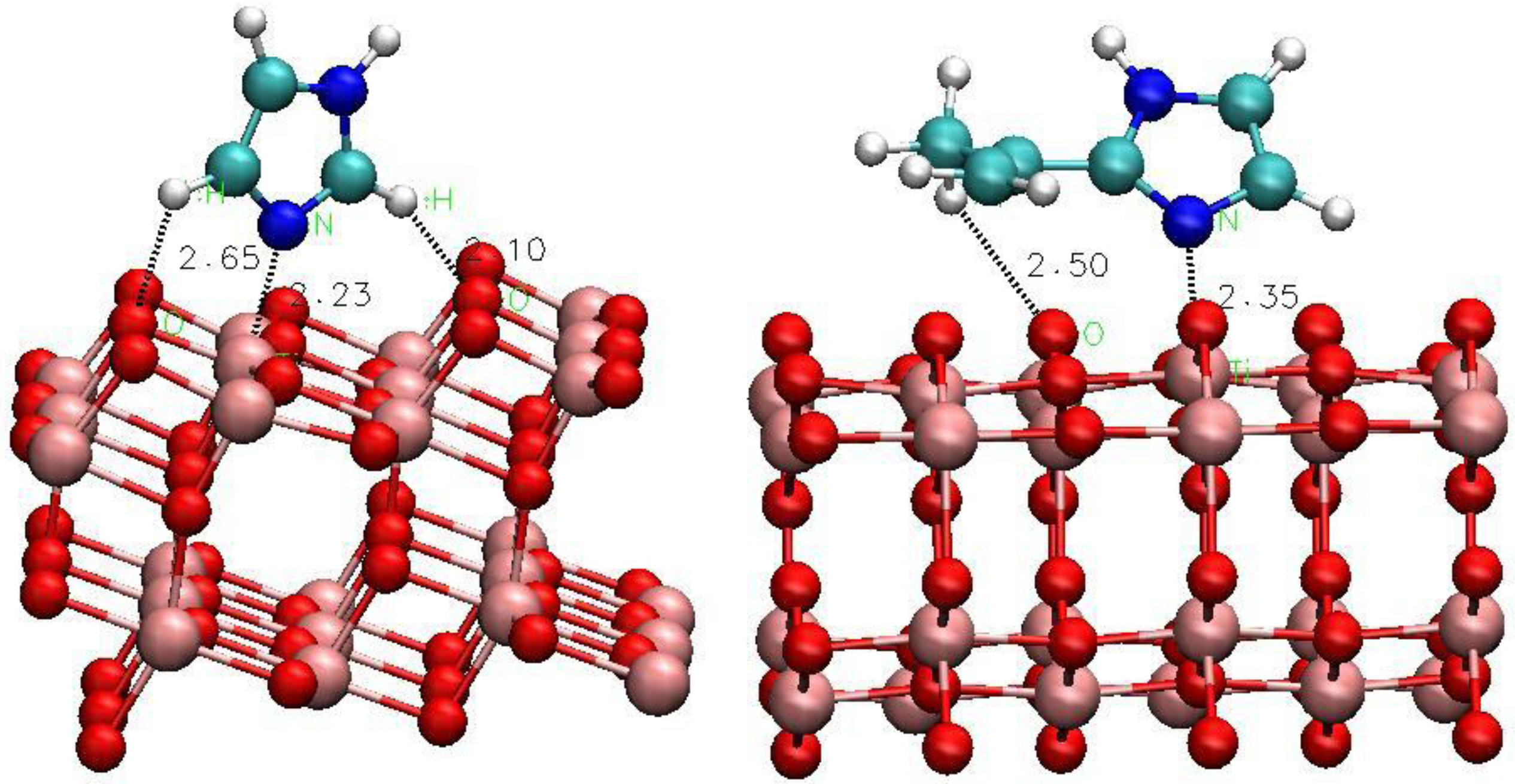
3.2.2. The Effect of Metal Ion Insertion on the Electronic Properties of the Anode and of Co-Adsorbents on Insertion
3.3. Interaction between Theory/Computation and Experiment
- -
- Investigation of electrochemical insertion of Li- and Na-ions into TiO2- anatase, rutile and bronze phases (standard battery characterization measurements of capacity-voltage curves at different rates) upon anode exposure to electrolytes with different Li/Na salt concentrations.
- -
- Estimation of metal ion content based on the storage capacity obtained.
- -
- Estimation of diffusion coefficients of Li and Na-ions in these structures, using GITT (galvanostatic intermittent titration technique), CV (cyclic voltammetry) and impedance studies.
- -
- Evaluation of electronic conductivity on prelithiated TiO2 films.
- -
- Experiments on photoelectrochemical cells with chemically pre-lithiated TiO2 anodes to investigate their performance.
- -
- Measurements of the effect on electron injection and recombination rates in dye-sensitized cells of different co-adsorbent-dyes pairs (using, for example, spectroscopy and impedance techniques [98]). This, together with calculations, will be used to build structure-property relations. These relations will facilitate computational screening of co-adsorbents used to achieve a desired effect on the electronic structure of the dye-semiconductor interface, to facilitate injection and inhibit recombination.
- -
- Measurements of I-V curves under different illumination intensities, transient photocurrent and photovoltage and AC impedance of solar cells utilizing different co-adsorbent molecules.
3.4. Expected Outcomes
4. Conclusions
4.1. A Systematic Theoretical Study to Derive Structure-Property Relations
4.2. Inclusion of Effects Due to Nuclear Dynamics
4.3. Considering the Effect of Counterion Intercalation on the Anode
4.4. Impact of Nanosizing
4.5. A Theoretical Study Tailored to Practice
Acknowledgments
References and Notes
- Barbour, E.; Wilson, I.A.G.; Bryden, I.G.; McGregor, P.G.; Mulheran, P.A.; Hall, P.J. Towards an objective method to compare energy storage technologies: Development and validation of a model to determine the upper boundary of revenue available from electrical price arbitrage. Energy Environ. Sci. 2012, 5, 5425–5436. [Google Scholar] [CrossRef]
- Hagfeldt, A.; Boschloo, G.; Sun, L.; Kloo, L.; Pettersson, H. Dye-sensitized solar cells. Chem. Rev. 2010, 110, 6595–6663. [Google Scholar] [CrossRef]
- Wei, D. Dye sensitized solar cells. Int. J. Mol. Sci. 2010, 11, 1103–1113. [Google Scholar] [CrossRef]
- Manzhos, S.; Jono, R.; Yamashita, K.; Fujisawa, J.-I.; Nagata, M.; Segawa, H. Study of interfacial charge transfer bands and electron recombination in the surface complexes of TCNE, TCNQ, and TCNAQ with TiO2. J. Phys. Chem. C 2011, 115, 21487–21493. [Google Scholar]
- Tao, C.S.; Jiang, J.; Tao, M. Natural resource limitations to terawatt-scale solar cells. Solar Energy Mater. Solar Cells 2011, 95, 3176–3180. [Google Scholar] [CrossRef]
- Branker, K.; Pathak, M.J.J.; Pearce, J.M. A review of solar photovoltaic levelized cost of electricity. Renew. Sust. Energ. Rev. 2011, 15, 4470–4482. [Google Scholar] [CrossRef]
- Graetzel, M. Photoelectrochemical cells. Nature 2001, 414, 338–344. [Google Scholar] [CrossRef]
- Boschloo, G.; Hagfeldt, A. Characteristics of the iodide/triiodide redox mediator in dye-sensitized solar cells. Acc. Chem. Res. 2009, 42, 1819–1826. [Google Scholar] [CrossRef]
- De Angelis, F.; Fantacci, S. Simulating dye-sensitized TiO2 Heterointerfaces in explicit solvent: Absorption spectra, energy levels, and dye desorption. J. Phys. Chem. Lett. 2011, 2, 813–817. [Google Scholar] [CrossRef]
- Aurbach, D.; Lu, Z.; Schechter, A.; Gofer, Y.; Gizbar, H.; Turgeman, R.; Cohen, Y.; Moshkovich, M.; Levi, E. Prototype systems for rechargeable magnesium batteries. Nature 2000, 407, 724–727. [Google Scholar] [CrossRef]
- Tran, T.T.; Obrovac, M.N. Alloy negative electrodes for high energy density metal-ion cells. J. Electrochem. Soc. 2011, 158, A1411–A1416. [Google Scholar] [CrossRef]
- Chevrier, V.L.; Ceder, G. Challenges for Na-ion negative rlectrodes. J. Electrochem. Soc. 2011, 158, A1011–A1014. [Google Scholar] [CrossRef]
- Kim, S.W.; Seo, D.H.; Ma, X.H.; Ceder, G.; Kang, K. Electrode materials for rechargeable sodium-ion batteries: Potential alternatives to current lithium-ion batteries. Adv. Energy Mater. 2012, 2, 710–721. [Google Scholar] [CrossRef]
- Hayashi, M.; Arai, H.; Ohtsuka, H.; Sakurai, Y. Electrochemical characteristics of calcium in organic electrolyte solutions and vanadium oxides as calcium hosts. J. Power Sources 2003, 119–121, 617–620. [Google Scholar]
- Palomares, V.; Serras, P.; Villaluenga, I.; Hueso, K.B.; Gonzalez, J.C.; Rojo, T. Na-ion batteries, recent advances and present challenges to become low cost energy storage systems. Energy Environ. Sci. 2012, 5, 5884–5901. [Google Scholar] [CrossRef]
- Liu, S.; Hu, J.J.; Yan, N.F.; Pan, G.L.; Li, G.R.; Gao, X.P. Aluminum storage behavior of anatase TiO2 nanotube arrays in aqueous solution for aluminum ion batteries. Energy Environ. Sci. 2012, 5, 9743–9746. [Google Scholar] [CrossRef]
- Shukla, A.K.; Kumar, T.P. Lithium economy: Will it get the electric traction? J. Phys. Chem. Lett. 2013, 4, 551–555. [Google Scholar] [CrossRef]
- Tarascon, J.-M. Is lithium the new gold? Nat. Chem. 2010, 2, 510. [Google Scholar] [CrossRef]
- Xu, K.; von Cresce, A. Li+-solvation/desolvation dictates interphasial processes on graphitic anode in Li ion cells. J. Mater. Res. 2012, 27, 2327–2341. [Google Scholar] [CrossRef]
- Leung, K. First-principles modeling of the initial stages of organic solvent decomposition on LixMn2O4(100) surfaces. J. Phys. Chem. C 2012, 116, 9852–9861. [Google Scholar] [CrossRef]
- Leung, K. Electronic structure modeling of electrochemical reactions at electrode/electrolyte interfaces in lithium ion batteries. J. Phys. Chem. C 2013, 117, 1539–1547. [Google Scholar] [CrossRef]
- Vatamanu, J.; Borodin, O.; Smith, G.D. Molecular dynamics simulation studies of the structure of a mixed carbonate/LiPF6 electrolyte near graphite surface as a function of electrode potential. J. Phys. Chem. C 2012, 116, 1114–1121. [Google Scholar] [CrossRef]
- Muldoon, J.; Bucur, C.B.; Oliver, A.G.; Sugimoto, T.; Matsui, M.; Kim, H.S.; Allred, G.D.; Zajicek, J.; Kotani, Y. Electrolyte roadblocks to a magnesium rechargeable battery. Energy Environ. Sci. 2012, 5, 5941–5950. [Google Scholar] [CrossRef]
- Yella, A.; Lee, H.W.; Tsao, H.N.; Yi, C.; Chandiran, A.K.; Nazeeruddin, M.K.; Diau, E.W.G.; Yeh, C.Y.; Zakeeruddin, S.M.; Grätzel, M. Porphyrin-sensitized solar cells with cobalt (II/III)-based electrolyte exceed 12% efficiency. Science 2011, 334, 629–634. [Google Scholar] [CrossRef]
- Auvinen, S.; Alatalo, M.; Haario, H.; Jalava, J.-P.; Lamminmäki, R.-J. Size and shape dependence of the electronic and spectral properties in TiO2 nanoparticles. J. Phys. Chem. C 2011, 115, 8484–8493. [Google Scholar]
- Spadavecchia, F.; Cappelletti, G.; Ardizzone, S.; Ceotto, M.; Falciola, L. Electronic structure of pure and N-doped TiO2 nanocrystals by electrochemical experiments and first principles calculations. J. Phys. Chem. C 2011, 115, 6381–6391. [Google Scholar] [CrossRef]
- Han, L.; Islam, A.; Chen, H.; Malapaka, C.; Chiranjeevi, B.; Zhang, S.; Yang, X.; Yanagida, M. High-efficiency dye-sensitized solar cell with a novel co-adsorbent. Energy Environ. Sci. 2012, 5, 6057–6060. [Google Scholar] [CrossRef]
- Lon, H.; Zhou, D.; Zhang, M.; Peng, C.; Uchida, S.; Wang, P. Probing dye-correlated interplay of energetics and kinetics in mesoscopic titania solar cells with 4-tert-butylpyridine. J. Phys. Chem. C 2011, 115, 14408–14414. [Google Scholar] [CrossRef]
- Ren, X.; Feng, Q.; Zhou, G.; Huang, C.-H.; Wang, Z.-S. Effect of cations in coadsorbate on charge recombination and conduction band edge movement in dye-sensitized solar cells. J. Phys. Chem. C 2010, 114, 7190–7195. [Google Scholar] [CrossRef]
- Koops, S.E.; O’Regan, B.C.; Barnes, P.R.; Durrant, J.R. Parameters influencing the efficiency of electron injection in dye-sensitized solar cells. J. Am. Chem. Soc. 2009, 131, 4808–4818. [Google Scholar]
- Wang, H.; Peter, L.M. Influence of electrolyte cations on electron transport and electron transfer in dye-sensitized solar cells. J. Phys. Chem. C 2012, 116, 10468–10475. [Google Scholar] [CrossRef] [Green Version]
- Shi, Y.S.; Wang, Y.H.; Zhang, M.; Dong, X.D. Influences of cation charge density on the photovoltaic performance of dye-sensitized solar cells: Lithium, sodium, potassium, and dimethylimidazolium. Phys. Chem. Chem. Phys. 2011, 13, 14590–14597. [Google Scholar] [CrossRef]
- Saravanan, K.; Ananthanarayanan, K.; Balaya, P. Mesoporous TiO2 with high packing density for superior lithium storage. Energy Environ. Sci. 2010, 3, 939–948. [Google Scholar] [CrossRef]
- Dalton, A.S.; Belak, A.A.; van der Ven, A. Thermodynamics of Lithium in TiO2(B) from first principles. Chem. Mater. 2012, 24, 1568–1574. [Google Scholar] [CrossRef]
- Kavan, L.; Kalbáč, M.; Zukalová, M.; Exnar, I.; Lorenzen, V.; Nesper, R.; Graetzel, M. Lithium storage in nanostructured TiO2 made by hydrothermal growth. Chem. Mater. 2004, 16, 477–485. [Google Scholar] [CrossRef]
- Wagemaker, M.; Borghols, W.J.H.; Mulder, F.M. Large impact of particle size on insertion reactions. A case for anatase LixTiO2. J. Am. Chem. Soc. 2007, 129, 4323–4327. [Google Scholar] [CrossRef]
- Yang, Z.; Choi, D.; Kerisit, S.; Rosso, K.M.; Wang, D.; Zhang, J.; Graff, G.; Liu, J. Nanostructures and lithium electrochemical reactivity of lithium titanites and titanium oxides: A review. J. Power Sources 2009, 192, 588–598. [Google Scholar] [CrossRef]
- Hu, Y.S.; Kienle, L.; Guo, Y.G.; Maier, J. High lithium electroactivity of nanometer-sized rutile TiO2. Adv. Mater. 2006, 18, 1421–1426. [Google Scholar] [CrossRef]
- Shin, J.-Y.; Joo, J.H.; Samuelis, D.; Maier, J. Oxygen-deficient TiO2-δ nanoparticles via hydrogen reduction for high rate capability lithium batteries. Chem. Mater. 2012, 24, 543–551. [Google Scholar] [CrossRef]
- Goodenough, J.B.; Kim, Y. Challenges for rechargeable Li batteries. Chem. Mater. 2010, 22, 587–603. [Google Scholar] [CrossRef]
- Zhang, L.; Zhang, Z.; Redfern, P.C.; Curtiss, L.A.; Amine, K. Molecular engineering towards safer lithium-ion batteries: A highly stable and compatible redox shuttle for overcharge protection. Energy Environ. Sci. 2012, 5, 8204–8207. [Google Scholar] [CrossRef]
- Nakayama, M.; Kotobuki, M.; Munakata, H.; Nogami, M.; Kanamura, K. First-principles density functional calculation of electrochemical stability of fast Li ion conducting garnet-type oxides. Phys. Chem. Chem. Phys. 2012, 14, 10008–10014. [Google Scholar] [CrossRef]
- Jalem, R.; Yamamoto, Y.; Shiiba, H.; Nakayama, M.; Munakata, H.; Kasuga, T.; Kanamura, K. Concerted migration mechanism in the Li ion dynamics of garnet-type Li7La3Zr2O12. Chem. Mater. 2013, 25, 425–430. [Google Scholar] [CrossRef]
- Yamagata, M.; Matsui, Y.; Sugimoto, T.; Kikuta, M.; Higashizaki, T.; Kono, M.; Ishikawa, M. High-performance graphite negative electrode in a bis(fluorosulfonyl)imide-based ionic liquid. J. Power Sources 2013, 227, 60–64. [Google Scholar] [CrossRef]
- Ryan, K.R.; Trahey, L.; Ingram, B.J.; Burrell, A.K. Limited stability of ether-based solvents in lithium–oxygen batteries. J. Phys. Chem. C 2012, 116, 19724–19728. [Google Scholar] [CrossRef]
- Fehse, M.; Fischer, F.; Tessier, C.; Stievano, L.; Monconduit, L. Tailoring of phase composition and morphology of TiO2-based electrode materials for lithium-ion batteries. J. Power Sources 2013, 231, 23–28. [Google Scholar] [CrossRef]
- Yu, J.; Sushko, M.L.; Kerisit, S.; Rosso, K.M.; Liu, J. Kinetic Monte Carlo study of ambipolar lithium ion and electron-polaron diffusion into nanostructured TiO2. J. Phys. Chem. Lett. 2012, 3, 2076–2081. [Google Scholar] [CrossRef]
- Ren, Y.; Zhang, J.; Liu, Y.; Li, H.; Wei, H.; Li, B.; Wang, X. Synthesis and superior anode performances of TiO2-carbon-rGO composites in lithium-ion batteries. Appl. Mater. Interfaces 2012, 4, 4776–4780. [Google Scholar] [CrossRef]
- Nishizawa, H.; Aoki, Y. The crystallization of anatase and the conversion to bronze-type TiO2 under hydrothermal conditions. J. Solid. State Chem. 1985, 56, 158–165. [Google Scholar] [CrossRef]
- Kopidakis, N.; Benkstein, K.D.; van de Lagemaat, J.; Frank, A.J. Transport-limited recombination of photocarriers in dye-sensitized nanocrystalline TiO2 solar cells. J. Phys. Chem. B 2003, 107, 11307–11315. [Google Scholar]
- Sun, Z.; Zhang, R.-K.; Xie, H.-H.; Wang, H.; Liang, M.; Xue, S. Nonideal charge recombination and conduction band edge shifts in dye-sensitized solar cells based on adsorbent doped poly(ethylene oxide) electrolytes. J. Phys. Chem. C 2013, 117, 4364–4373. [Google Scholar] [CrossRef]
- Kim, J.-Y.; Kim, J.Y.; Lee, D.-K.; Kim, B.; Kim, H.; Koe, M.J. Importance of 4-tert-butylpyridine in electrolyte for dye-sensitized solar cells employing SnO2 electrode. J. Phys. Chem. C 2012, 116, 22759–22766. [Google Scholar] [CrossRef]
- Asaduzzaman, A.M.; Schreckenbach, G. Computational studies on the interactions among redox couples, additives and TiO2: Implications for dye-sensitized solar cells. Phys. Chem. Chem. Phys. 2010, 12, 14609–14618. [Google Scholar] [CrossRef]
- Manzhos, S.; Segawa, H.; Yamashita, K. The effect of ligand substitution and water co-adsorption on the adsorption dynamics and energy level matching of amino-phenyl acid dyes on TiO2. Phys. Chem. Chem. Phys. 2012, 14, 1749–1755. [Google Scholar] [CrossRef]
- Manzhos, S.; Segawa, H.; Yamashita, K. Effect of nuclear vibrations, temperature, and orientation on injection and recombination conditions in amino-phenyl acid dyes on TiO2. Proc. SPIE 2012, 8438, 843814. [Google Scholar]
- Manzhos, S.; Segawa, H.; Yamashita, K. Effect of nuclear vibrations, temperature, co-adsorbed water, and dye orientation on light absorption, charge injection and recombination conditions in organic dyes on TiO2. Phys. Chem. Chem. Phys. 2013, 15, 1141–1147. [Google Scholar] [CrossRef]
- Haque, S.A.; Handa, S.; Peter, K.; Palomares, E.; Thelakkat, M.; Durrant, J.R. Supermolecular control of charge transfer in dye-sensitized nanocrystalline TiO2 films: Towards a quantitative structure–function relationship. Angew. Chem. Int. Ed. 2005, 44, 5740–5744. [Google Scholar] [CrossRef]
- Clifford, J.N.; Palomares, E.; Nazeeruddin, M.K.; Grätzel, M.; Nelson, J.; Li, X.; Long, N.J.; Durrant, J.R. Molecular control of recombination dynamics in dye-sensitized nanocrystalline TiO2 films: Free energy vs distance dependence. J. Am. Chem. Soc. 2004, 126, 5225–5233. [Google Scholar] [CrossRef]
- Ceder, G.; Hautier, G.; Jain, A.; Ong, S.P. Recharging lithium battery research with first-principles methods. MRS Bull. 2011, 36, 185–191. [Google Scholar]
- Malyi, O.I.; Tan, T.L.; Manzhos, S. A comparative computational study of structures, diffusion, and dopant interactions between Li and Na insertion into Si. Appl. Phys. Express 2013, 6, 027301. [Google Scholar] [CrossRef]
- Malyi, O.I.; Tan, T.L.; Manzhos, S. In search of high performance anode materials for Mg batteries: Computational studies of Mg in Ge, Si, and Sn. J. Power Sources 2013, 233, 341–345. [Google Scholar] [CrossRef]
- Wan, W.H.; Zhang, Q.F.; Cui, Y.; Wang, E.G. First principles study of lithium insertion in bulk silicon. J. Phys. Condens. Matter 2010, 22, 415501–415509. [Google Scholar] [CrossRef]
- Cui, Z.; Gao, F.; Cui, Z.; Qu, J. A second nearest neighbor embedded atom method interatomic potential for Li-Si alloys. J. Power Sources 2012, 207, 150–159. [Google Scholar] [CrossRef]
- Miao, L.; Wu, J.; Jiang, J.; Liang, P. First-principles study on the synergetic mechanism of SnO2 and graphene as a lithium ion battery anode. J. Phys. Chem. C 2013, 117, 23–27. [Google Scholar] [CrossRef]
- Kaghazchi, P. Theoretical studies of Li incorporation into Si(111). J. Phys. Condens. Matter 2013, 25, 095008. [Google Scholar] [CrossRef]
- Kaghazchi, P. Theoretical studies of lithium incorporation into α-Sn(100). J. Chem. Phys. 2013, 138, 054706. [Google Scholar] [CrossRef]
- Peng, B.; Cheng, F.; Tao, Z.; Chen, J. Lithium transport at silicon thin film: Barrier for high-rate capability anode. J. Chem. Phys. 2010, 133, 034701. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhang, W.; Wan, W.; Cui, Y.; Wang, E. Lithium insertion in silicon nanowires: An ab initio study. Nano Lett. 2010, 10, 3243–3249. [Google Scholar] [CrossRef]
- Jung, S.C.; Han, Y.-K. Facet-dependent lithium intercalation into Si crystals: Si(100) vs. Si(111). Phys. Chem. Chem. Phys. 2011, 13, 21282–21287. [Google Scholar] [CrossRef]
- Leung, K.; Qi, Y.; Zavadil, K.R.; Jung, Y.S.; Dillon, A.C.; Cavanagh, A.S.; Lee, S.-H.; George, S.M. Using atomic layer deposition to hinder solvent decomposition in lithium ion batteries: First-principles modeling and experimental studies. J. Am. Chem. Soc. 2011, 133, 14741–14754. [Google Scholar]
- Shao, N.; Sun, X.-G.; Dai, S.; Jiang, D. Oxidation potentials of functionalized sulfone solvents for high-voltage Li-ion batteries: A computational study. J. Phys. Chem. B 2012, 116, 3235–3238. [Google Scholar] [CrossRef]
- Kim, S.-P.; van Duin, A.C.T.; Shenoy, V.B. Effect of electrolytes on the structure and evolution of the solid electrolyte interphase (SEI) in Li-ion batteries: A molecular dynamics study. J. Power Sources 2011, 196, 8590–8597. [Google Scholar] [CrossRef]
- Ganesh, P.; Kent, P.R.C.; Jiang, D. Solid–electrolyte interphase formation and electrolyte reduction at Li-ion battery graphite anodes: Insights from first-principles molecular dynamics. J. Phys. Chem. C 2012, 116, 24476–24481. [Google Scholar] [CrossRef]
- Malyi, O.I.; Tan, T.L.; Manzhos, S. A computational study of the insertion of Li, Na, and Mg atoms into Si(111) nanosheets. Nano Energy 2013. [Google Scholar] [CrossRef]
- Carvalho, A.; Rayson, M.J.; Briddon, P.R.; Manzhos, S. Effect of the adsorption of ethylene carbonate on Si surfaces on the Li intercalation behavior. (Submitted).
- Kusama, H.; Orita, H.; Sugihara, H. TiO2 band shift by nitrogen-containing heterocycles in dye-sensitized solar cells: A periodic density functional study. Langmuir 2008, 24, 4411–4419. [Google Scholar] [CrossRef]
- Kawata, K.; Goto, T.; Yoshizaki, H. Additives for dye-sensitized solar cells. February 2013. Available online: http://www.sumobrain.com/patents/WO2013026563.html (accessed on 18 June 2013).
- Scheers, J.; Johansson, P.; Szczeciński, P.; Wieczorek, W.; Armand, M.; Jacobsson, P. Benzimidazole and imidazole lithium salts for battery electrolytes. J. Power Sources 2010, 195, 6081–6087. [Google Scholar] [CrossRef]
- Ichimura, A.S.; Mack, B.M.; Usmani, S.M.; Mars, D.G. Direct synthesis of anatase films with ~100% (001) facets and [001] preferred orientation. Chem. Mater. 2012, 24, 2324–2329. [Google Scholar] [CrossRef]
- Werner, J.H.; Zapf-Gottwick, R.; Koch, M.; Fischer, K. Toxic substances in photovoltaic modules. In Proceedings of the 21st International Photovoltaic Science and Engineering Conference, Fukuoka, Japan, 28 November–2 December 2011.
- O’Regan, B.; Grätzel, M. A low-cost, high-efficiency solar cell based on dye-sensitized colloidal TiO2 films. Nature 1991, 353, 737–740. [Google Scholar] [CrossRef]
- Zeng, W.; Cao, Y.; Bai, Y.; Wang, Y.; Shi, Y.; Zhang, M.; Wang, F.; Pan, C.; Wang, P. Efficient dye-sensitized solar cells with an organic photosensitizer featuring orderly conjugated ethylenedioxythiophene and dithienosilole blocks. Chem. Mater. 2010, 22, 1915–1925. [Google Scholar] [CrossRef]
- Yum, J.-H.; Baranoff, E.; Kessler, F.; Moehl, T.; Ahmad, S.; Bessho, T.; Marchioro, A.; Ghadiri, E.; Moser, J.-E.; Yi, C.; et al. A cobalt complex redox shuttle for dye-sensitized solar cells with high open-circuit potentials. Nat. Commun. 2012, 3, 631–638. [Google Scholar] [CrossRef]
- Shin, J.-Y.; Samuelis, D.; Maier, J. Sustained lithium-storage performance of hierarchical, nanoporous anatase TiO2 at high rates: Emphasis on interfacial storage phenomena. Adv. Funct. Mater. 2011, 21, 3464–3472. [Google Scholar] [CrossRef]
- Armstrong, A.R.; Arrouvel, C.; Gentili, V.; Parker, S.C.; Islam, M.S.; Bruce, P.G. Lithium coordination sites in LixTiO2(B): A structural and computational study. Chem. Mater. 2010, 22, 6426–6432. [Google Scholar] [CrossRef]
- Shen, L.; Zhang, X.; Li, H.; Yuan, C.; Cao, G. Design and tailoring of a three-dimensional TiO2-graphene-carbon nanotube nanocomposite for fast lithium storage. J. Phys. Chem. Lett. 2011, 2, 3096–3101. [Google Scholar] [CrossRef]
- Kang, J.; Wei, S.H.; Zhu, K.; Kim, Y.H. First-principles theory of electrochemical capacitance of nanostructured materials: Dipole-assisted subsurface intercalation of lithium in pseudocapacitive TiO2 anatase nanosheets. J. Phys. Chem. C 2011, 115, 4909–4915. [Google Scholar] [CrossRef]
- Zakharova, G.C.; Jähne, C.; Popa, A.; Täschner, Ch.; Gemming, Th.; Leonhardt, A.; Büchner, B.; Klingeler, R. Anatase nanotubes as an electrode material for lithium-ion batteries. J. Phys. Chem. C 2012, 116, 8714–8720. [Google Scholar] [CrossRef]
- Zhu, K.; Wang, Q.; Kim, J.-H.; Pesaran, A.A.; Frank, J.A. Pseudocapacitive lithium-ion storage in oriented anatase TiO2 nanotube arrays. J. Phys. Chem. C 2012, 116, 11895–11899. [Google Scholar] [CrossRef]
- Wagemaker, M.; Mulder, F.M. Properties and promises of nanosized insertion materials for Li-ion batteries. Acc. Chem. Res. 2012. [Google Scholar] [CrossRef]
- Manzhos, S.; Segawa, H.; Yamashita, K. Effect of isotopic substitution on elementary processes in dye-sensitized solar cells: Deuterated amino-phenyl acid dyes on TiO2. Computation 2013, 1, 1–15. [Google Scholar] [CrossRef]
- Miyasaka, T. Toward printable sensitized mesoscopic solar cells: Light-harvesting management with thin TiO2 films. J. Phys. Chem. Lett. 2011, 2, 262–269. [Google Scholar] [CrossRef]
- Kusama, H.; Sugihara, H.; Sayama, K. Effect of cations on the interactions of Ru dye and iodides in dye-sensitized solar cells: A density functional theory study. J. Phys. Chem. C 2011, 115, 2544–2552. [Google Scholar] [CrossRef]
- Li, R.; Liu, J.; Cai, N.; Zhang, M.; Wang, P. Synchronously reduced surface states, charge recombination, and light absorption length for high-performance organic dye-sensitized solar cells. J. Phys. Chem. B 2010, 114, 4461–4464. [Google Scholar] [CrossRef]
- Yang, H.; Huang, S.; Huang, X.; Fan, F.F.; Liang, W.T.; Liu, X.H.; Chen, L.Q.; Huang, J.Y.; Li, J.; Zhu, T.; et al. Orientation-dependent interfacial mobility governs the anisotropic swelling in lithiated silicon nanowires. Nano Lett. 2012, 12, 1953–1958. [Google Scholar] [CrossRef]
- Mizuno, Y.; Okubo, M.; Hosono, E.; Kudo, T.; Zhou, H.; Oh-ishi, K. Suppressed activation energy for interfacial charge transfer of a Prussian blue analog thin film electrode with hydrated ions (Li+, Na+, and Mg2+). J. Phys. Chem. C 2013, 117, 10877–10882. [Google Scholar] [CrossRef]
- Tatsumi, H.; Sasahara, A.; Tomitori, M. Adsorption of propylene carbonate molecules on a TiO2(110) surface. J. Phys. Chem. C 2013, 117, 10410–10416. [Google Scholar] [CrossRef]
- Listorti, A.; Creager, C.; Sommeling, P.; Kroon, J.; Palomares, E.; Fornelli, A.; Breen, B.; Barnes, P.R.F.; Durrant, J.R.; Law, C.; et al. The mechanism behind the beneficial effect of light soaking on injection efficiency and photocurrent in dye sensitized solar cells. Energy Environ. Sci. 2011, 4, 3494–3501. [Google Scholar] [CrossRef]
- Duncan, W.R.; Prezhdo, O.V. Theoretical studies of photoinduced electron transfer in dye-sensitized TiO2. Annu. Rev. Phys. Chem. 2007, 58, 143–184. [Google Scholar] [CrossRef]
- Hibino, M.; Abe, K.; Mochizuki, M.; Miyayama, M. Amorphous titanium dioxide electrode for high-rate discharge and charge. J. Power Sources 2004, 126, 139–143. [Google Scholar] [CrossRef]
- Armstrong, A.R.; Armstrong, G.; Canales, J.; Bruce, P.G. TiO2-B nanowires as negative electrodes for rechargeable lithium batteries. J. Power Sources 2005, 146, 501–506. [Google Scholar] [CrossRef]
- Wang, Q.; Wen, Z.H.; Li, J.H. Solvent-controlled synthesis and electrochemical lithium storage of one-dimensional TiO2 nanostructures. Inorg. Chem. 2006, 45, 6944–6949. [Google Scholar] [CrossRef]
- Kavan, L.; Rathousky, J.; Gratzell, M.; Shklover, V.; Zukal, A. Surfactant-templated TiO2 (anatase): Characteristic features of lithium insertion electrochemistry in organized nanostructures. J. Phys. Chem. B 2000, 104, 12012–12020. [Google Scholar] [CrossRef]
- Wagemaker, M.; Lutzenkirchen-Hecht, D.; van Well, A.A.; Frahm, R. Atomic and electronic bulk versus surface structure: Lithium intercalation in anatase TiO2. J. Phys. Chem. B 2004, 108, 12456–12464. [Google Scholar] [CrossRef]
- Li, J.R.; Tang, Z.L.; Zhang, Z.T. Preparation and novel lithium intercalation properties of titanium oxide nanotubes. Electrochem. Solid-State Lett. 2005, 8, A316–A319. [Google Scholar] [CrossRef]
- Subramanian, V.; Karki, A.; Gnanasekar, K.I.; Eddy, F.P.; Rambabu, B. Nanocrystalline TiO2 (anatase) for Li-ion batteries. J. Power Sources 2006, 159, 186–192. [Google Scholar] [CrossRef]
- Borghols, W.J.H.; Wagemaker, M.; Lafont, U.; Keller, E.M.; Mulder, F.M. Impact of nanosizing on lithiated rutile TiO2. Chem. Mater. 2008, 20, 2949–2955. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Manzhos, S.; Giorgi, G. Bridging the Fields of Solar Cell and Battery Research to Develop High-Performance Anodes for Photoelectrochemical Cells and Metal Ion Batteries. Challenges 2013, 4, 116-135. https://doi.org/10.3390/challe4010116
Manzhos S, Giorgi G. Bridging the Fields of Solar Cell and Battery Research to Develop High-Performance Anodes for Photoelectrochemical Cells and Metal Ion Batteries. Challenges. 2013; 4(1):116-135. https://doi.org/10.3390/challe4010116
Chicago/Turabian StyleManzhos, Sergei, and Giacomo Giorgi. 2013. "Bridging the Fields of Solar Cell and Battery Research to Develop High-Performance Anodes for Photoelectrochemical Cells and Metal Ion Batteries" Challenges 4, no. 1: 116-135. https://doi.org/10.3390/challe4010116




