S-Nitrosoglutathione Reverts Dietary Sucrose-Induced Insulin Resistance
Abstract
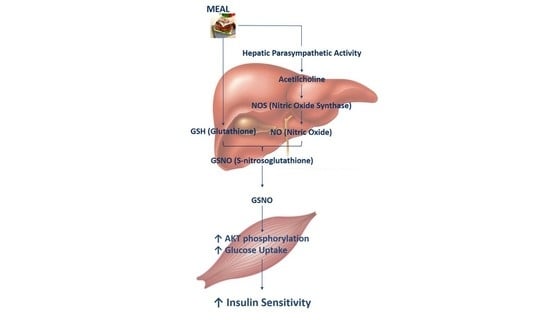
:1. Introduction
2. Materials and Methods
2.1. Presurgical Protocols
2.2. Surgical Protocol
2.3. Rapid Insulin Sensitivity Test (RIST)
2.4. Determination of Serum Levels of Glucose, Insulin, and c-peptide
2.5. Meal Tolerance Test
2.6. Measurement of Hepatic Nitric Oxide Synthase (NOS) Gene Expression
2.7. Measurement of Hepatic Nitric Oxide Synthase (NOS) Activity
2.8. Nitric Oxide Assessment
2.9. Hepatic Glutathione Measurement
2.10. Cell Culture for and L6 Myotubes
2.11. Glucose Uptake
2.12. Immunoblotting Analysis
2.13. Experimental Protocols
2.14. Statistical Analyses
3. Results
3.1. High Sucrose Intake Promotes Body Weight Gain with Unchanged Fasting Glycemia
3.2. Insulin Sensitivity is Impaired in High-Sucrose Diet Fed Animals
3.3. Hepatic Nitric Oxide Expression/Activity and Nitrate Levels, as well as Hepatic Glutathione, are Altered in High-Sucrose Feeding
3.4. Intravenous Administration of S-Nitrosoglutathione Ameliorates Sucrose Induced Insulin Resistance
3.5. S-Nitrosogluathione Promotes Glucose Uptake in L6 Myotubes
3.6. S-Nitrosoglutathione Promote Glucose Uptake by Activating the Insulin Signaling Pathway
4. Discussion
4.1. Body Weight, Water/Food Intake, and Biochemical Parameters
4.2. Weeks of Sucrose-Enriched Diet Leads to Impaired Insulin Secretion and Clearance in the Fed State
4.3. Glutathione and NO Levels are Impaired after 4 Weeks of Sucrose Diet
4.4. S-Nitrothiols are Sufficient to Overcome Insulin Resistance
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zheng, Y.; Ley, S.H.; Hu, F.B. Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat. Rev. Endocrinol. 2017, 14, 88–98. [Google Scholar] [CrossRef]
- Kolb, H.; Martin, S. Environmental/lifestyle factors in the pathogenesis and prevention of type 2 diabetes. BMC Med. 2017, 15, 131. [Google Scholar] [CrossRef] [PubMed]
- Kirchner, H.; Osler, M.E.; Krook, A.; Zierath, J.R. Epigenetic flexibility in metabolic regulation: Disease cause and prevention? Trends Cell Biol. 2013, 23, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Malik, V.S.; Popkin, B.; Bray, G.A.; Després, J.-P.; Willett, W.C.; Hu, F.B. Sugar-Sweetened Beverages and Risk of Metabolic Syndrome and Type 2 Diabetes. Diabetes Care 2010, 33, 2477–2483. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schaefer, E.J.; Gleason, J.A.; Dansinger, M.L. Dietary Fructose and Glucose Differentially Affect Lipid and Glucose Homeostasis. J. Nutr. 2009, 139, 1257S–1262S. [Google Scholar] [CrossRef] [Green Version]
- Melanson, K.J.; Angelopoulos, T.J.; Nguyen, V.; Zukley, L.; Lowndes, J.; Rippe, J.M.; Ishihara, J.; Inoue, M.; Iwasaki, M.; Sasazuki, S.; et al. High-fructose corn syrup, energy intake, and appetite regulation. Am. J. Clin. Nutr. 2008, 88, 1738S–1744S. [Google Scholar] [CrossRef] [Green Version]
- Sumiyoshi, M.; Sakanaka, M.; Kimura, Y. Chronic Intake of High-Fat and High-Sucrose Diets Differentially Affects Glucose Intolerance in Mice. J. Nutr. 2006, 136, 582–587. [Google Scholar] [CrossRef] [Green Version]
- Hannou, S.A.; Haslam, D.E.; McKeown, N.M.; Herman, M.A. Fructose metabolism and metabolic disease. J. Clin. Investig. 2018, 128, 545–555. [Google Scholar] [CrossRef]
- DiNicolantonio, J.J.; O’Keefe, J.H.; Lucan, S.C. Added fructose: A principal driver of type 2 diabetes mellitus and its consequences. Mayo Clin. Proc. 2015, 90, 372–381. [Google Scholar] [CrossRef] [Green Version]
- Miller, A.; Adeli, K. Dietary fructose and the metabolic syndrome. Curr. Opin. Gastroenterol. 2008, 24, 204–209. [Google Scholar] [CrossRef] [Green Version]
- Her, T.K.; Lagakos, W.S.; Brown, M.R.; Le Brasseur, N.K.; Rakshit, K.; Aleksey, V. Matveyenko., Dietary carbohydrates modulate metabolic and beta-cell adaptation to high-fat diet-induced obesity. Am. J. Physiol. Endocrinol. Metab. 2020, 318, E856–E865. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.C.; Marques, C.; Martins, F.O.; Viegas, I.; Tavares, L.; Macedo, M.P.; Jones, J.G. Determining contributions of exogenous glucose and fructose to de novo fatty acid and glycerol synthesis in liver and adipose tissue. Metab. Eng. 2019, 56, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Jarak, I.; Barosa, C.; Martins, F.O.; Silva, J.C.P.; Santos, C.; Belew, G.D.; Rito, J.; Viegas, I.; Teixeira, J.; Oliveira, P.J.; et al. Sources of hepatic glycogen synthesis in mice fed with glucose or fructose as the sole dietary carbohydrate. Magn. Reson. Med. 2018, 81, 639–644. [Google Scholar] [CrossRef] [PubMed]
- Samuel, V.T. Fructose induced lipogenesis: From sugar to fat to insulin resistance. Trends Endocrinol. Metab. 2011, 22, 60–65. [Google Scholar] [CrossRef]
- Sánchez-Lozada, L.G.; Mu, W.; Roncal, C.; Sautin, Y.Y.; Abdelmalek, M.; Reungjui, S.; Le, M.; Nakagawa, T.; Lan, H.-Y.; Yu, X.; et al. Comparison of free fructose and glucose to sucrose in the ability to cause fatty liver. Eur. J. Nutr. 2009, 49, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Ribeiro, R.T.; Lautt, W.W.; Legare, D.J.; Macedo, M.P. Insulin resistance induced by sucrose feeding in rats is due to an impairment of the hepatic parasympathetic nerves. Diabetologia 2005, 48, 976–983. [Google Scholar] [CrossRef]
- Folli, F.; Corradi, D.; Fanti, P.; Davalli, A.; Paez, A.; Giaccari, A.; Perego, C.; Muscogiuri, G. The role of oxidative stress in the pathogenesis of type 2 diabetes mellitus micro- and macrovascular complications: Avenues for a mechanistic-based therapeutic approach. Curr. Diabetes Rev. 2011, 7, 313–324. [Google Scholar] [CrossRef]
- Du, D.; Shi, Y.-H.; Le, G. Oxidative stress induced by high-glucose diet in liver of C57BL/6J mice and its underlying mechanism. Mol. Biol. Rep. 2010, 37, 3833–3839. [Google Scholar] [CrossRef]
- Francini, F.; Castro, M.C.; Schinella, G.; García, M.E.; Maiztegui, B.; Raschia, M.A.; Gagliardino, J.J.; Massa, M.L. Changes induced by a fructose-rich diet on hepatic metabolism and the antioxidant system. Life Sci. 2010, 86, 965–971. [Google Scholar] [CrossRef]
- Blouet, C.; Mariotti, F.; Mathe, V.; Tome, D.; Huneau, J.-F. Nitric Oxide Bioavailability and Not Production Is First Altered During the Onset of Insulin Resistance in Sucrose-Fed Rats. Exp. Biol. Med. 2007, 232, 1458–1464. [Google Scholar] [CrossRef]
- Guarino, M.P.; Correia, N.C.; Lautt, W.W.; Macedo, M.P. Insulin sensitivity is mediated by the activation of the ACh/NO/cGMP pathway in rat liver. Am. J. Physiol. Liver Physiol. 2004, 287, G527–G532. [Google Scholar] [CrossRef]
- Guarino, M.P.; Macedo, M.P. Co-administration of glutathione and nitric oxide enhances insulin sensitivity in Wistar rats. Br. J. Pharmacol. 2006, 147, 959–965. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guarino, M.P.; Afonso, R.A.; Raimundo, N.; Raposo, J.F.; Macedo, M.P. Hepatic glutathione and nitric oxide are critical for hepatic insulin-sensitizing substance action. Am. J. Physiol. Liver Physiol. 2003, 284, G588–G594. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mayer, B.; Pfeiffer, S.; Schrammel, A.; Koesling, D.; Schmidt, K.; Brunner, F. A New Pathway of Nitric Oxide/Cyclic GMP Signaling InvolvingS-Nitrosoglutathione. J. Biol. Chem. 1998, 273, 3264–3270. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schrammel, A. S-nitrosation of glutathione by nitric oxide, peroxynitrite, and •NO/O2•−. Free Radic. Biol. Med. 2003, 34, 1078–1088. [Google Scholar] [CrossRef]
- Benhar, M.; Forrester, M.T.; Stamler, J.S. Protein denitrosylation: Enzymatic mechanisms and cellular functions. Nat. Rev. Mol. Cell Biol. 2009, 10, 721–732. [Google Scholar] [CrossRef]
- Khan, M.; Dhammu, T.S.; Qiao, F.; Kumar, P.; Singh, A.K.; Singh, I. S-Nitrosoglutathione Mimics the Beneficial Activity of Endothelial Nitric Oxide Synthase-Derived Nitric Oxide in a Mouse Model of Stroke. J. Stroke Cerebrovasc. Dis. 2019, 28, 104470. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.; Shunmugavel, A.; Dhammu, T.S.; Khan, H.; Singh, I.; Singh, A.K. Combined treatment with GSNO and CAPE accelerates functional recovery via additive antioxidant activities in a mouse model of TBI. J. Neurosci. Res. 2018, 96, 1900–1913. [Google Scholar] [CrossRef]
- Stamler, J.S.; Lamas, S.; Fang, F.C. Nitrosylation. the prototypic redox-based signaling mechanism. Cell 2001, 106, 675–683. [Google Scholar] [CrossRef] [Green Version]
- Lima, B.; Forrester, M.T.; Hess, U.T.; Stamler, J.S. S-nitrosylation in cardiovascular signaling. Circ. Res. 2010, 106, 633–646. [Google Scholar] [CrossRef] [Green Version]
- Foster, M.W.; McMahon, T.J.; Stamler, J.S. S-nitrosylation in health and disease. Trends Mol. Med. 2003, 9, 160–168. [Google Scholar] [CrossRef]
- Fernandes, A.B.; Guarino, M.P.; Macedo, M.P. Understanding the in-vivo relevance of S-nitrosothiols in insulin action. Can. J. Physiol. Pharmacol. 2012, 90, 887–894. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, T.D.C.; Jiang, L.Q.; Zierath, J.R.; Nunes, M.T. L-Arginine enhances glucose and lipid metabolism in rat L6 myotubes via the NO/ c-GMP pathway. Metabolism 2013, 62, 79–89. [Google Scholar] [CrossRef]
- Lira, V.A.; Soltow, Q.A.; Long, J.H.D.; Betters, J.L.; Sellman, J.E.; Criswell, D.S. Nitric oxide increases GLUT4 expression and regulates AMPK signaling in skeletal muscle. Am. J. Physiol. Metab. 2007, 293, E1062–E1068. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lautt, W.W.; Wang, X.; Sadri, P.; Legare, D.J.; Macedo, M.P. Rapid insulin sensitivity test (RIST). Can. J. Physiol. Pharmacol. 1998, 76, 1080–1086. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C.(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Bredt, D.S.; Snyder, S.H. Nitric oxide: A physiologic messenger molecule. Annu. Rev. Biochem. 1994, 63, 175–195. [Google Scholar] [CrossRef]
- Harlow, E.; Lane, D.P. Bradford Assay. CSH Protoc. 2006, 2006. [Google Scholar] [CrossRef]
- Afonso, R.A.; Patarrão, R.S.; Macedo, M.P.; Carmo, M.M. Carvedilol Action Is Dependent on Endogenous Production of Nitric Oxide. Am. J. Hypertens. 2006, 19, 419–425. [Google Scholar] [CrossRef] [Green Version]
- Ignarro, L.J.; Fukuto, J.M.; Griscavage, J.M.; Rogers, N.E.; Byrns, R.E. Oxidation of nitric oxide in aqueous solution to nitrite but not nitrate: Comparison with enzymatically formed nitric oxide from L-arginine. Proc. Natl. Acad. Sci. USA 1993, 90, 8103–8107. [Google Scholar] [CrossRef] [Green Version]
- Moshage, H.; Kok, B.; Huizenga, J.R.; Jansen, P.L. Nitrite and nitrate determinations in plasma: A critical evaluation. Clin. Chem. 1995, 41, 892–896. [Google Scholar] [CrossRef] [PubMed]
- Wennmalm, A.; Benthin, G.; Petersson, A.-S. Dependence of the metabolism of nitric oxide (NO) in healthy human whole blood on the oxygenation of its red cell haemoglobin. Br. J. Pharmacol. 1992, 106, 507–508. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, F.; Troncy, E.; Francœur, M.; Vinet, B.; Vinay, P.; Czaika, G.; Blaise, G. Effects of reducing reagents and temperature on conversion of nitrite and nitrate to nitric oxide and detection of NO by chemiluminescence. Clin. Chem. 1997, 43, 657–662. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Onyango, I.G.; Khan, S.M. Oxidative stress, mitochondrial dysfunction, and stress signaling in Alzheimer’s disease. Curr. Alzheimer Res. 2006, 3, 339–349. [Google Scholar] [CrossRef] [PubMed]
- Huppertz, C.; Fischer, B.M.; Kim, Y.-B.; Kotani, K.; Vidal-Puig, A.; Slieker, L.J.; Sloop, K.W.; Lowell, B.B.; Kahn, B.B. Uncoupling Protein 3 (UCP3) Stimulates Glucose Uptake in Muscle Cells through a Phosphoinositide 3-Kinase-dependent Mechanism. J. Biol. Chem. 2001, 276, 12520–12529. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Furukawa, N.; Ongusaha, P.; Jahng, W.J.; Araki, K.; Choi, C.S.; Kim, H.-J.; Lee, Y.H.; Kaibuchi, K.; Kahn, B.B.; Masuzaki, H.; et al. Role of Rho-kinase in regulation of insulin action and glucose homeostasis. Cell Metab. 2005, 2, 119–129. [Google Scholar] [CrossRef] [Green Version]
- Kim, Y.-B.; Shulman, G.I.; Kahn, B.B. Fatty acid infusion selectively impairs insulin action on Akt1 and protein kinase C lambda/zeta but not on glycogen synthase kinase. J. Biol. Chem. 2002, 277, 32915–32922. [Google Scholar] [CrossRef] [Green Version]
- Afonso, R.A.; Gaspar, J.M.; Lamarão, I.; Lautt, W.W.; Macedo, M.P.; Information, P.E.K.F.C. Postprandial insulin action relies on meal composition and hepatic parasympathetics: Dependency on glucose and amino acids. J. Nutr. Biochem. 2016, 27, 70–78. [Google Scholar] [CrossRef]
- De Koning, L.; Malik, V.S.; Rimm, E.B.; Willett, W.C.; Hu, F.B. Sugar-sweetened and artificially sweetened beverage consumption and risk of type 2 diabetes in men. Am. J. Clin. Nutr. 2011, 93, 1321–1327. [Google Scholar] [CrossRef] [Green Version]
- Ludwig, D.; Peterson, K.; Gortmaker, S. Relation between consumption of sugar-sweetened drinks and childhood obesity: A prospective, observational analysis. Lancet 2001, 357, 505–508. [Google Scholar] [CrossRef]
- Malik, V.S.; Schulze, M.B.; Hu, F.B. Intake of sugar-sweetened beverages and weight gain: A systematic review. Am. J. Clin. Nutr. 2006, 84, 274–288. [Google Scholar] [CrossRef] [PubMed]
- Tordoff, M.G.; Alleva, A.M. Effect of drinking soda sweetened with aspartame or high-fructose corn syrup on food intake and body weight. Am. J. Clin. Nutr. 1990, 51, 963–969. [Google Scholar] [CrossRef]
- Sacramento, J.F.; Martins, F.O.; Rodrigues, T.; Matafome, P.; Ribeiro, M.J.; Olea, E.; Conde, S.V. A2 Adenosine Receptors Mediate Whole-Body Insulin Sensitivity in a Prediabetes Animal Model: Primary Effects on Skeletal Muscle. Front. Endocrinol. 2020, 11, 262. [Google Scholar] [CrossRef] [PubMed]
- Pinyo, J.; Hira, T.; Hara, H. Continuous feeding of a combined high-fat and high-sucrose diet, rather than an individual high-fat or high-sucrose diet, rapidly enhances the glucagon-like peptide-1 secretory response to meal ingestion in diet-induced obese rats. Nutritition 2019, 62, 122–130. [Google Scholar] [CrossRef] [PubMed]
- Martins, F.O.; Delgado, T.C.; Viegas, J.; Gaspar, J.M.; Scott, D.K.; O’Doherty, R.M.; Macedo, M.P.; Jones, J.G. Mechanisms by which the thiazolidinedione troglitazone protects against sucrose-induced hepatic fat accumulation and hyperinsulinaemia. Br. J. Pharmacol. 2015, 173, 267–278. [Google Scholar] [CrossRef] [Green Version]
- Abbasi, F.; Tern, P.J.W.; Reaven, G.M. Plasma glucose concentration 60 min post oral glucose load and risk of type 2 diabetes and cardiovascular disease: Pathophysiological implications. Diabetes Vasc. Dis. Res. 2019, 16, 337–343. [Google Scholar] [CrossRef]
- Tessari, P.; Cecchet, D.; Cosma, A.; Vettore, M.; Coracina, A.; Millioni, R.; Iori, E.; Puricelli, L.; Avogaro, A.; Vedovato, M. Nitric Oxide Synthesis Is Reduced in Subjects with Type 2 Diabetes and Nephropathy. Diabetes 2010, 59, 2152–2159. [Google Scholar] [CrossRef] [Green Version]
- Natali, A.; Ribeiro, R.T.; Baldi, S.; Tulipani, A.; Rossi, M.; Venturi, E.; Mari, A.; Macedo, M.P.; Ferrannini, E. Systemic inhibition of nitric oxide synthesis in non-diabetic individuals produces a significant deterioration in glucose tolerance by increasing insulin clearance and inhibiting insulin secretion. Diabetologia 2013, 56, 1183–1191. [Google Scholar] [CrossRef]
- Bahadoran, Z.; Mirmiran, P.; Ghasemi, A. Role of Nitric Oxide in Insulin Secretion and Glucose Metabolism. Trends Endocrinol. Metab. 2019, 31, 118–130. [Google Scholar] [CrossRef]
- Cruz, E.M.S.; De Morais, J.M.B.; Da Rosa, C.V.D.; Simões, M.D.S.; Comar, J.; Chuffa, L.G.D.A.; Seiva, F.R.F. Long-term sucrose solution consumption causes metabolic alterations and affects hepatic oxidative stress in Wistar rats. Biol. Open 2020, 9. [Google Scholar] [CrossRef] [Green Version]
- Andrich, D.E.; Melbouci, L.; Ou, Y.; Auclair, N.; Mercier, J.; Grenier, J.-C.; Lira, F.S.; Barreiro, L.B.; Danialou, G.; Comtois, A.-S.; et al. A Short-Term High-Fat Diet Alters Glutathione Levels and IL-6 Gene Expression in Oxidative Skeletal Muscles of Young Rats. Front. Physiol. 2019, 10, 372. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anavi, S.; Tirosh, O. iNOS as a metabolic enzyme under stress conditions. Free Radic. Biol. Med. 2020, 146, 16–35. [Google Scholar] [CrossRef]
- Carvalho-Filho, M.A.; Ueno, M.; Hirabara, S.M.; Seabra, A.B.; Carvalheira, J.B.; De Oliveira, M.G.; Velloso, L.A.; Curi, R.; Saad, M. S-Nitrosation of the Insulin Receptor, Insulin Receptor Substrate 1, and Protein Kinase B/Akt: A Novel Mechanism of Insulin Resistance. Diabetes 2005, 54, 959–967. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McGrowder, D.; Ragoobirsingh, D.; Brown, P. Acute effects of exogenous nitric oxide on glucose uptake in skeletal muscle of normoglycaemic and diabetic rats. Med. Sci. Monit. 2005, 12, BR28–BR35. [Google Scholar] [PubMed]





© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sousa-Lima, I.; Fernandes, A.B.; Patarrão, R.S.; Kim, Y.-B.; Macedo, M.P. S-Nitrosoglutathione Reverts Dietary Sucrose-Induced Insulin Resistance. Antioxidants 2020, 9, 870. https://doi.org/10.3390/antiox9090870
Sousa-Lima I, Fernandes AB, Patarrão RS, Kim Y-B, Macedo MP. S-Nitrosoglutathione Reverts Dietary Sucrose-Induced Insulin Resistance. Antioxidants. 2020; 9(9):870. https://doi.org/10.3390/antiox9090870
Chicago/Turabian StyleSousa-Lima, Inês, Ana B. Fernandes, Rita S. Patarrão, Young-Bum Kim, and M. Paula Macedo. 2020. "S-Nitrosoglutathione Reverts Dietary Sucrose-Induced Insulin Resistance" Antioxidants 9, no. 9: 870. https://doi.org/10.3390/antiox9090870