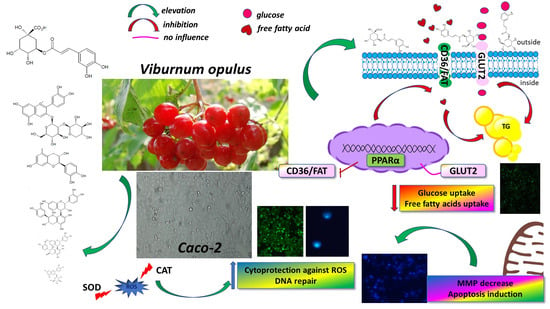
Viburnum opulus Fruit Phenolic Compounds as Cytoprotective Agents Able to Decrease Free Fatty Acids and Glucose Uptake by Caco-2 Cells
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Preparation of Viburnum Opulus Extracts
2.3. Determination of Total Phenolics Content and Phenolic Compounds Profile
2.4. Antioxidant Capacity Assays
2.5. Cell Culture and Exposure Conditions
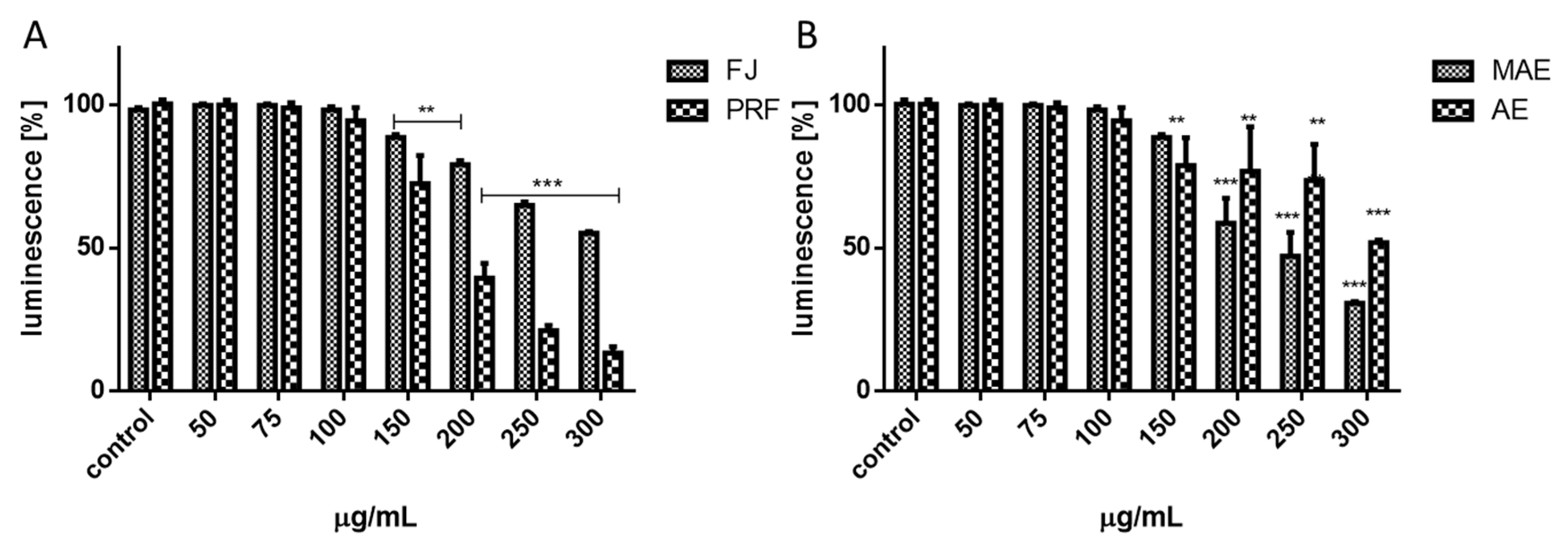
2.6. Cell Viability
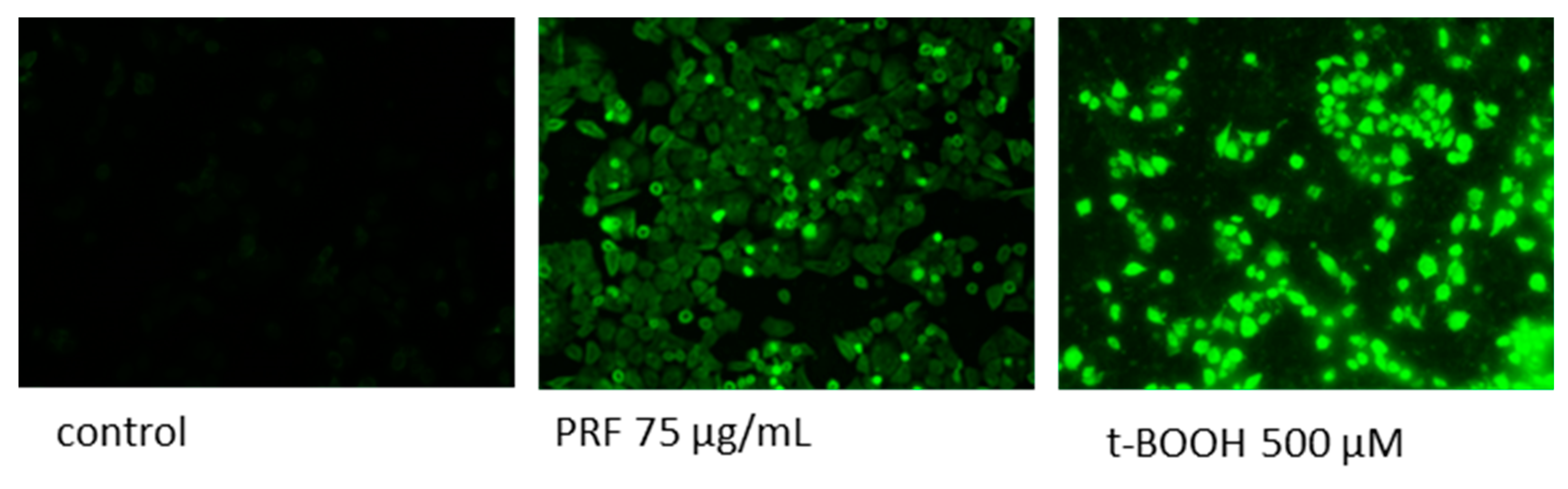
2.7. Detection of Intracellular Reactive Oxygen Species (ROS) Generation
2.8. Measurement of ATP Production
2.9. Measurement of Mitochondrial Membrane Potential (MMP)
2.10. Phosphatidylserine Externalization, Membrane Permeabilization and Apoptosis Induction
2.11. Glucose Uptake
2.12. Nile Red Staining
2.13. Fatty Acid Uptake
2.14. DNA Damage and Repair
2.15. Gene Expression Analysis
2.16. Statistical Analysis
3. Results and Discussion
3.1. Phenolic Compounds Profile and Content
3.2. Antioxidant Capacity of V. opulus Extracts
3.3. Assessment of Viburnum Opulus Phenolics on Cellular Viability
3.4. Viburnum opulus as a Chemopreventive Agent
3.5. Effect of V. opulus Extracts on Intracellular ATP Level, Intracellular Oxidative Stress and Apoptosis Induction
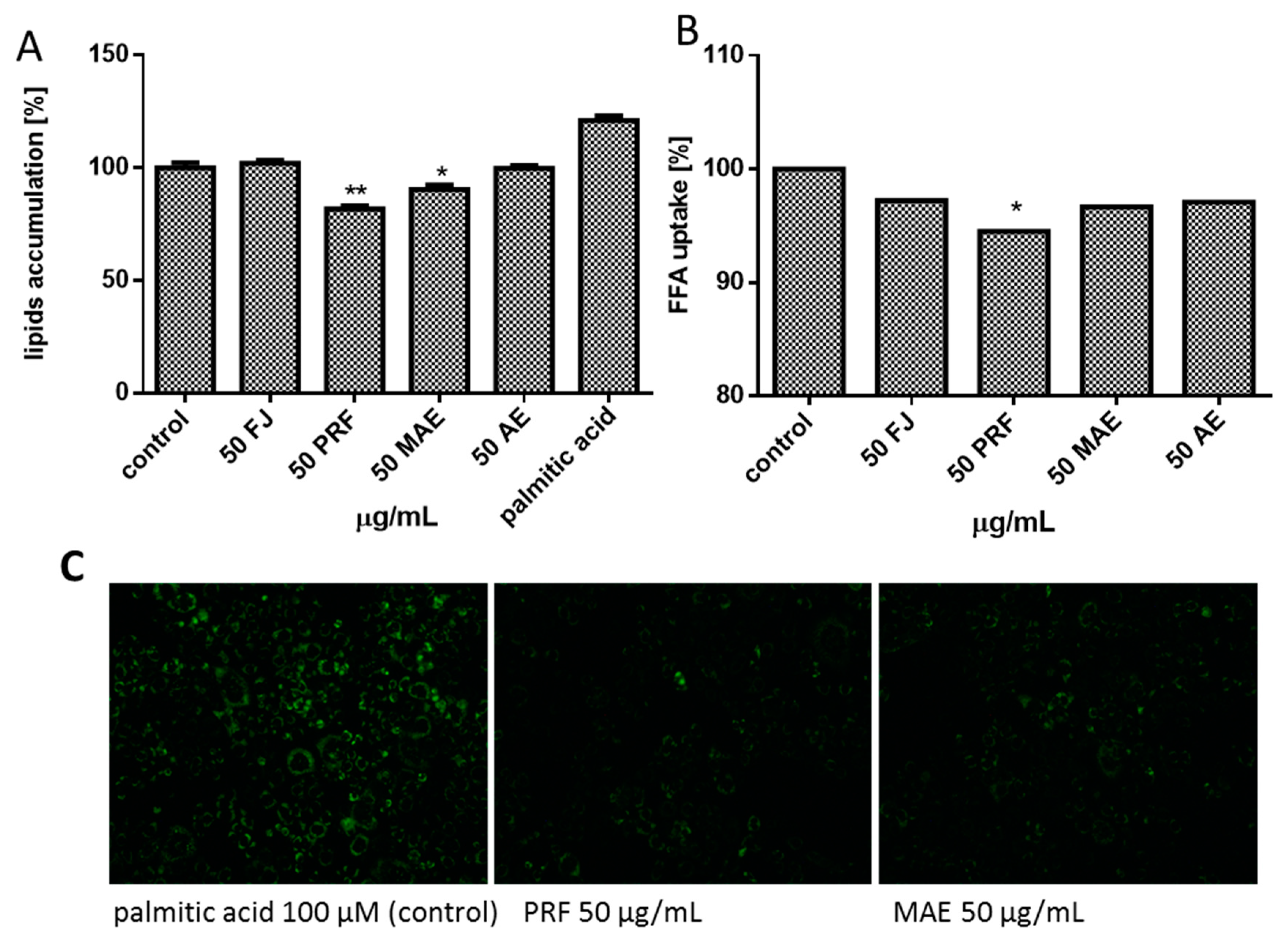
3.6. Viburnum opulus Phenolics as Modulators of Fatty Acids Uptake
3.7. Viburnum opulus Phenolic Compounds as Modulators of Glucose Uptake
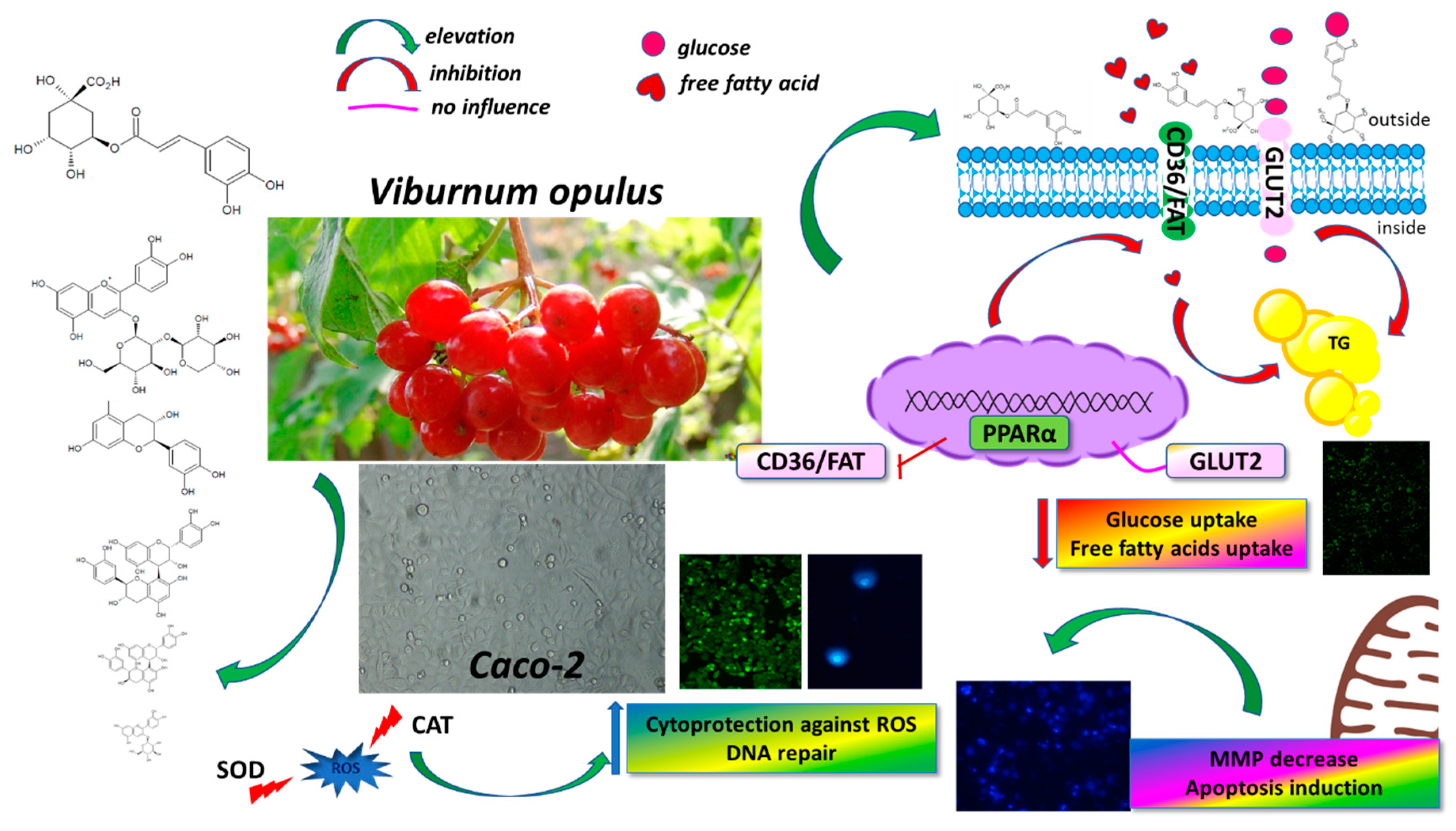
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Cory, H.; Passarelli, S.; Szeto, J.; Tamez, M.; Mattei, J. The Role of Polyphenols in Human Health and Food Systems: A Mini-Review. Front. Nutr. 2018, 5, 87. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eken, A.; Yücel, O.; İpek, İ.; Ayşe, B.; Endİrlİk, B.Ü. An Investigation on Protective Effect of Viburnum opulus L. Fruit Extract Against Ischemia/Reperfusion-Induced Oxidative Stress after lung transplantation in rats. Kafkas Üniversitesi Veteriner Fakültesi Dergisi 2017, 23, 437–444. [Google Scholar]
- Perova, I.B.; Zhogova, A.A.; Cherkashin, A.V.; Éller, K.I.; Ramenskaya, G.V. Biologically Active Substances From European Guelder Berry Fruits. Pharm. Chem. J. 2014, 48, 332–339. [Google Scholar] [CrossRef]
- Moldovan, B.; David, L.; Chişbora, C.; Cimpoiu, C. Degradation Kinetics of Anthocyanins from European Cranberrybush (Viburnum opulus L.) Fruit Extracts. Effects of Temperature, pH and Storage Solvent. Molecules 2012, 17, 11655–11666. [Google Scholar] [CrossRef] [PubMed]
- Kızılay, O.N.; Ülker, F.; Çelik, V.; Özdemir, T.; Çakmak, Ö.; Can, E. The evaluation of the effectiveness of Gilaburu (Viburnum opulus L.) extract in the medical expulsive treatment of distal ureteral stones The evaluation of the effectiveness of Gilaburu (Viburnum opulus L.) extract in the medical expulsive treatment of distal ureteral stones. Turk. J. Urol. 2019. [Google Scholar] [CrossRef]
- Ceylan, D.; Aksoy, A.; Ertekin, T.; Yay, A.H.; Nisari, M.; Karatoprak, G.Ş.; Ülger, H. The effects of gilaburu (Viburnum opulus) juice on experimentally induced Ehrlich ascites tumor in mice. J. Cancer Res. Ther. 2018, 14, 310–320. [Google Scholar]
- Kozlowska, W.; Wagner, C.; Moore, E.M.; Matkowski, A. Botanical Provenance of Traditional Medicines From Carpathian Mountains at the Ukrainian-Polish Border. Front. Pharmacol. 2018, 9, 295. [Google Scholar] [CrossRef]
- Zakłos-Szyda, M.; Majewska, I.; Redzynia, M.; Koziołkiewicz, M. Antidiabetic Effect of Polyphenolic Extracts from Selected Edible Plants as α-Amylase, α-Glucosidase and PTP1B Inhibitors, and β Pancreatic Cells Cytoprotective Agents—A Comparative Study. Curr. Top. Med. Chem. 2015, 15, 2431–2444. [Google Scholar] [CrossRef]
- Mihaylova, D.; Popova, A.; Alexieva, I.; Krastanov, A.; Lante, A. Polyphenols as Suitable Control for Obesity and Diabetes. Open Biotech. J. 2018, 12, 219–228. [Google Scholar] [CrossRef]
- Farrell, T.L.; Ellam, S.L.; Forrelli, T.; Williamson, G. Attenuation of glucose transport across Caco-2 cell monolayers by a polyphenol-rich herbal extract: Interactions with SGLT1 and GLUT2 transporters. Biofactors 2013, 39, 448–456. [Google Scholar] [CrossRef]
- Kwon, O.; Eck, P.; Chen, S.; Corpe, C.P.; Lee, J.H.; Kruhlak, M.; Levine, M. Inhibition of the intestinal glucose transporter GLUT2 by flavonoids. FASEB J. 2007, 21, 366–377. [Google Scholar] [CrossRef]
- Schreck, K.; Melzig, M.F. Intestinal Saturated Long-Chain Fatty Acid, Glucose and Fructose Transporters and Their Inhibition by Natural Plant Extracts in Caco-2 Cells. Molecules 2018, 23, 2544. [Google Scholar] [CrossRef]
- Manzano, S.; Williamson, G. Polyphenols and phenolic acids from strawberry and apple decrease glucose uptake and transport by human intestinal Caco-2 cells. Mol. Nutr. Food Res. 2010, 54, 1773–1780. [Google Scholar] [CrossRef]
- Stremmel, W.; Staffer, S.; Wannhoff, A.; Pathil, A. The overall fatty acid absorption controlled by basolateral chylomicron excretion under regulation of p-JNK1. Biochim. Biophys. Acta-Mol. Cell Biol. Lipids 2017, 1862, 917–928. [Google Scholar] [CrossRef]
- Podsędek, M.; Majewska, A.; Redzynia, I.; Sosnowska, M.; Koziołkiewicz, D. In vitro inhibitory effect on digestive enzymes and antioxidant potential of commonly consumed fruits. J. Agric. Food Chem. 2014, 62, 4610–4617. [Google Scholar] [CrossRef]
- Budryn, G.; Zakłos-Szyda, M.; Zaczyńska, D.; Żyżelewicz, D.; Grzelczyk, J.; Zduńczyk, Z.; Juśkiewicz, J. Green and roasted coffee extracts as antioxidants in βTC3 cells with induced oxidative stress and lipid accumulation inhibitors in 3T3L1 cells, and their bioactivity in rats fed high fat diet. Eur. Food Res. Technol. 2017, 243, 1323–1334. [Google Scholar] [CrossRef]
- Zakłos-Szyda, M.; Pawlik, N. Japanese quince (Chaenomeles japonica L.) fruit polyphenolic extract modulates carbohydrate metabolism in HepG2 cells via AMP-activated protein kinase. Acta Biochim. Pol. 2018, 65, 67–78. [Google Scholar] [CrossRef]
- Hogan, A.M.; Swaminathan, V.; Pallegar, N.K.; Christian, S.L. Nile Red and 2-NBDG Are Incompatible for the Simultaneous Detection of Lipid and Glucose Accumulation. Int. J. Spectrosc. 2016, 2016, 5215086. [Google Scholar] [CrossRef]
- Nowak, A.; Klewicki, R.; Lipin, L. Ellagitannins from Rubus idaeus L. Exert Geno—And Cytotoxic Effects against Human Colon Adenocarcinoma Cell Line Caco-2. J. Agric. Food Chem. 2017, 65, 2947–2955. [Google Scholar] [CrossRef]
- Peng, B.J.; Zhu, Q.; Zhong, Y.L.; Xu, S.H.; Wang, Z. Chlorogenic Acid Maintains Glucose Homeostasis through Modulating the Expression of SGLT-1, GLUT-2, and PLG in Different Intestinal Segments of Sprague-Dawley Rats Fed a High-Fat Diet. Biomed. Environ. Sci. 2015, 28, 894–903. [Google Scholar]
- Velioglu, Y.S.; Ekici, L.; Poyrazoglu, E.S. Original article Phenolic composition of European cranberrybush (Viburnum opulus L.) berries and astringency removal of its commercial juice. Int. J. Food Sci. Technol. 2006, 9205, 1011–1015. [Google Scholar] [CrossRef]
- Karaçelik, P.; Küçük, A.A.; Iskefiyeli, M.; Aydemir, Z.; de Smet, S.; Miserez, S.; Sandra, B. Antioxidant components of Viburnum opulus L. determined by on-line HPLC–UV–ABTS radical scavenging and LC–UV–ESI-MS methods. Food Chem. 2015, 175, 106–114. [Google Scholar] [CrossRef]
- Kraujalyte, V.; Rimantas, P.; Pukalskas, A.; Laima, C. Antioxidant properties and polyphenolic compositions of fruits from different European cranberrybush (Viburnum opulus L.) genotypes. Food Chem. 2013, 141, 3695–3702. [Google Scholar] [CrossRef]
- De Freitas, V.; Faria, A.; Pestana, D.; Azevedo, J.; Azevedo, I.; Mateus, N. Absorption of anthocyanins through intestinal epithelial cells—Putative involvement of GLUT2. Mol. Nutr. Food Res. 2009, 53, 1430–1437. [Google Scholar]
- Hajiaghaalipour, F.; Khalilpourfarshbafi, M.; Arya, A. Modulation of Glucose Transporter Protein by Dietary Flavonoids in Type 2 Diabetes Mellitus. Int. J. Biol. Sci. 2015, 11, 508. [Google Scholar] [CrossRef]
- Catarino, T.A.; Gonc, P. The effect of oxidative stress upon the intestinal epithelial uptake of butyrate. Eur. J. Pharmacol. 2013, 699, 88–100. [Google Scholar]
- Margina, D.; Gradinaru, D.; Manda, G.; Neagoe, I.; Ilie, M. Membranar effects exerted in vitro by polyphenols—Quercetin, epigallocatechin gallate and curcumin—On HUVEC and Jurkat cells, relevant for diabetes mellitus. Food Chem. Toxicol. 2013, 61, 86–93. [Google Scholar] [CrossRef]
- Amararathna, M.; Johnston, M.R.; Rupasinghe, H.P.V. Plant Polyphenols as Chemopreventive Agents for Lung Cancer. Int. J. Mol. Sci. 2016, 17, 1352. [Google Scholar] [CrossRef]
- Goszcz, K.; Duthie, G.G.; Stewart, D.; Leslie, S.J.; Megson, I.L. Bioactive polyphenols and cardiovascular disease: Chemical antagonists, pharmacological agents or xenobiotics that drive an adaptive response? Br. J. Pharmacol. 2017, 174, 1209–1225. [Google Scholar] [CrossRef]
- Liang, N.; Dupuis, J.H.; Yada, R.Y.; Kitts, D.D. Chlorogenic acid isomers directly interact with Keap 1-Nrf2 signaling in Caco-2 cells. Mol. Cell. Biochem. 2019, 457, 105–118. [Google Scholar] [CrossRef] [Green Version]
- Bonarska-kujawa, H.P.D. Effect of chlorogenic acid on the phase transition in phospholipid and phospholipid/cholesterol membranes. J. Thermal Anal. Calorim. 2014, 118, 943–950. [Google Scholar] [Green Version]
- Liang, N.; Kitts, D.D. Chlorogenic Acid (CGA) Isomers Alleviate Interleukin 8 (IL-8) Production in Caco-2 Cells by Decreasing Phosphorylation of p38 and Increasing Cell Integrity. Int. J. Mol. Sci. 2018, 19, 3837. [Google Scholar] [CrossRef]
- Wang, J.; Li, J.; Liu, J.; Xu, M.; Tong, X.; Wang, J. Chlorogenic acid prevents isoproterenol-induced DNA damage in vascular smooth muscle cells. Mol. Med. Rep. 2016, 14, 4063–4068. [Google Scholar] [CrossRef]
- Ekbatan, S.S.; Li, X.; Ghorbani, M.; Azadi, B. Chlorogenic Acid and Its Microbial Metabolites Exert Anti-Proliferative Effects, S-Phase Cell-Cycle Arrest and Apoptosis in Human Colon Cancer Caco-2 Cells. Int. J. Mol. Sci. 2018, 19, 723. [Google Scholar] [CrossRef]
- Cheah, K.Y.; Howarth, G.S.; Bindon, K.A.; Kennedy, J.A.; Bastian, S.E.P. Low molecular weight procyanidins from grape seeds enhance the impact of 5-Fluorouracil chemotherapy on Caco-2 human colon cancer cells. PLoS ONE 2014, 9, e98921. [Google Scholar] [CrossRef]
- Gorlach, S.; Wagner, W.; Podsędek, A.; Szewczyk, K.; Koziołkiewicz, M.; Dastych, J. Procyanidins from Japanese Quince (Chaenomeles japonica) fruit induce apoptosis in human colon cancer Caco-2 cells in a degree of polymerization-dependent manner. Nutr. Cancer 2011, 63, 1348–1360. [Google Scholar] [CrossRef]
- Rodríguez-ramiro, I.; Ramos, S.; Bravo, L.; Goya, L.; Martín, M.Á. Procyanidin B2 and a cocoa polyphenolic extract inhibit acrylamide-induced apoptosis in human Caco-2 cells by preventing oxidative stress and activation of JNK pathway. J. Nutr. Biochem. 2011, 22, 1186–1194. [Google Scholar] [CrossRef] [Green Version]
- Pandey, M.K.; Gupta, S.C.; Nabavizadeh, A.; Aggarwal, B.B. Seminars in Cancer Biology Regulation of cell signaling pathways by dietary agents for cancer prevention and treatment. Semin. Cancer Biol. 2017, 46, 158–181. [Google Scholar] [CrossRef]
- Dhuriya, Y.K.; Sharma, D. Necroptosis: A regulated inflammatory mode of cell death. J. Neuroinflamm. 2018, 15, 199. [Google Scholar] [CrossRef]
- Lin, W.; Tongyi, S. Role of Bax/Bcl-2 family members in green tea polyphenol induced necroptosis of p53-deficient Hep3B cells. Tumor Biol. 2014, 35, 8065–8075. [Google Scholar] [CrossRef]
- Fam, T.; Klymchenko, A.; Collot, M. Recent Advances in Fluorescent Probes for Lipid Droplets. Materials 2018, 11, 1768. [Google Scholar] [CrossRef]
- Rohm, B.; Riedel, A.; Ley, J.P.; Widder, S. Function activation and increase acetyl-coenzyme A. Food Funct. 2015, 6, 173–185. [Google Scholar]
- Glatz, J.F.C.; Luiken, J.J.F.P. Biochimie From fat to FAT (CD36/SR-B2): Understanding the regulation of cellular fatty acid uptake. Biochimie 2017, 136, 21–26. [Google Scholar] [CrossRef]
- Schneider, H.; Staudacher, S.; Poppelreuther, M.; Stremmel, W.; Ehehalt, R.; Füllekrug, J. Protein mediated fatty acid uptake: Synergy between CD36/FAT-facilitated transport and acyl-CoA synthetase-driven metabolism. Arch. Biochem. Biophys. 2014, 546, 8–18. [Google Scholar] [CrossRef]
- Perona, J.S. Biochimica et Biophysica Acta Membrane lipid alterations in the metabolic syndrome and the role of dietary oils. BBA-Biomembr. 2017, 1859, 1690–1703. [Google Scholar] [CrossRef]
- Sancheza, M.B.; Miranda-Pereza, E.; Verjanb, J.C.G.; Barreraa, M.D.F.; Perez-Ramosc, J.; Alarcon-Aguilaret, F.J. Potential of the chlorogenic acid as multitarget agent: Insulin-secretagogue and PPAR α/γ dual agonist. Biomed. Pharmacother. 2017, 94, 169–175. [Google Scholar] [CrossRef]
- Pang, Y.; Zhu, Q.; Kang, J.; Liu, M.; Wang, Z. Chlorogenic Acid Functions as a Novel Agonist of PPAR γ 2 during the Differentiation of Mouse 3T3-L1 Preadipocytes. BioMed Res. Int. 2018. [Google Scholar] [CrossRef]
- Vrbacky, M. Succinimidyl oleate, established inhibitor of CD36/FAT translocase inhibits complex III of mitochondrial respiratory chain. Biochem. Biophys. Res. Commun. 2010, 391, 1348–1351. [Google Scholar]
- Abumrad, N.A.; Cifarelli, V. Intestinal CD36 and Other Key Proteins of Lipid Utilization: Role in Absorption and Gut Homeostasis. Compr. Physiol. 2018, 8, 493–507. [Google Scholar]
- Zhang, D.; Zhang, R.; Liu, Y.; Sun, X.; Yin, Z.; Li, H. CD36 gene variants is associated with type 2 diabetes mellitus through the interaction of obesity in rural Chinese adults. Gene 2018, 659, 155–159. [Google Scholar] [CrossRef]
- Hemmersbach, S.; Brauer, S.S.; Hu, S.; Galla, H.; Humpf, H. Transepithelial Permeability Studies of Flavan-3-ol-C-glucosides and Procyanidin Dimers and Trimers across the Caco-2 Cell Monolayer. J. Agric. Food Chem. 2013, 61, 7932–7940. [Google Scholar] [CrossRef]
- Zou, T.; Feng, D.; Song, G.; Li, H.; Tang, H.; Ling, W. The Role of Sodium-Dependent Glucose Transporter 1 and Glucose Transporter 2 in the Absorption of Cyanidin-3-O-β-Glucoside in Caco-2 Cells. Nutrients 2014, 6, 4165–4177. [Google Scholar] [CrossRef]
- Hresko, R.C.; Kraft, T.E.; Quigley, A.; Carpenter, E.P.; Hruz, P.W. Mammalian Glucose Transporter Activity Is Dependent upon Anionic and Conical Phospholipids. J. Biol. Chem. 2016, 291, 17271–17282. [Google Scholar] [CrossRef] [Green Version]
- De, C.; Palacio, J.R.; Martínez, P.; Morros, A. Biochimica et Biophysica Acta Effect of oxidative stress on plasma membrane fluidity of THP-1 induced macrophages. BBA-Biomembr. 2013, 1828, 357–364. [Google Scholar]
- Phan, H.T.T.; Yoda, T.; Chahal, B.; Morita, M.; Takagi, M.; Vestergaard, C. Biochimica et Biophysica Acta Structure-dependent interactions of polyphenols with a biomimetic membrane system. BBA-Biomembr. 2014, 1838, 2670–2677. [Google Scholar] [CrossRef]
- Tsuchiya, H. Membrane Interactions of Phytochemicals as Their Molecular Mechanism Applicable to the Discovery of Drug Leads from Plants. Molecules 2015, 20, 18923–18966. [Google Scholar] [CrossRef]
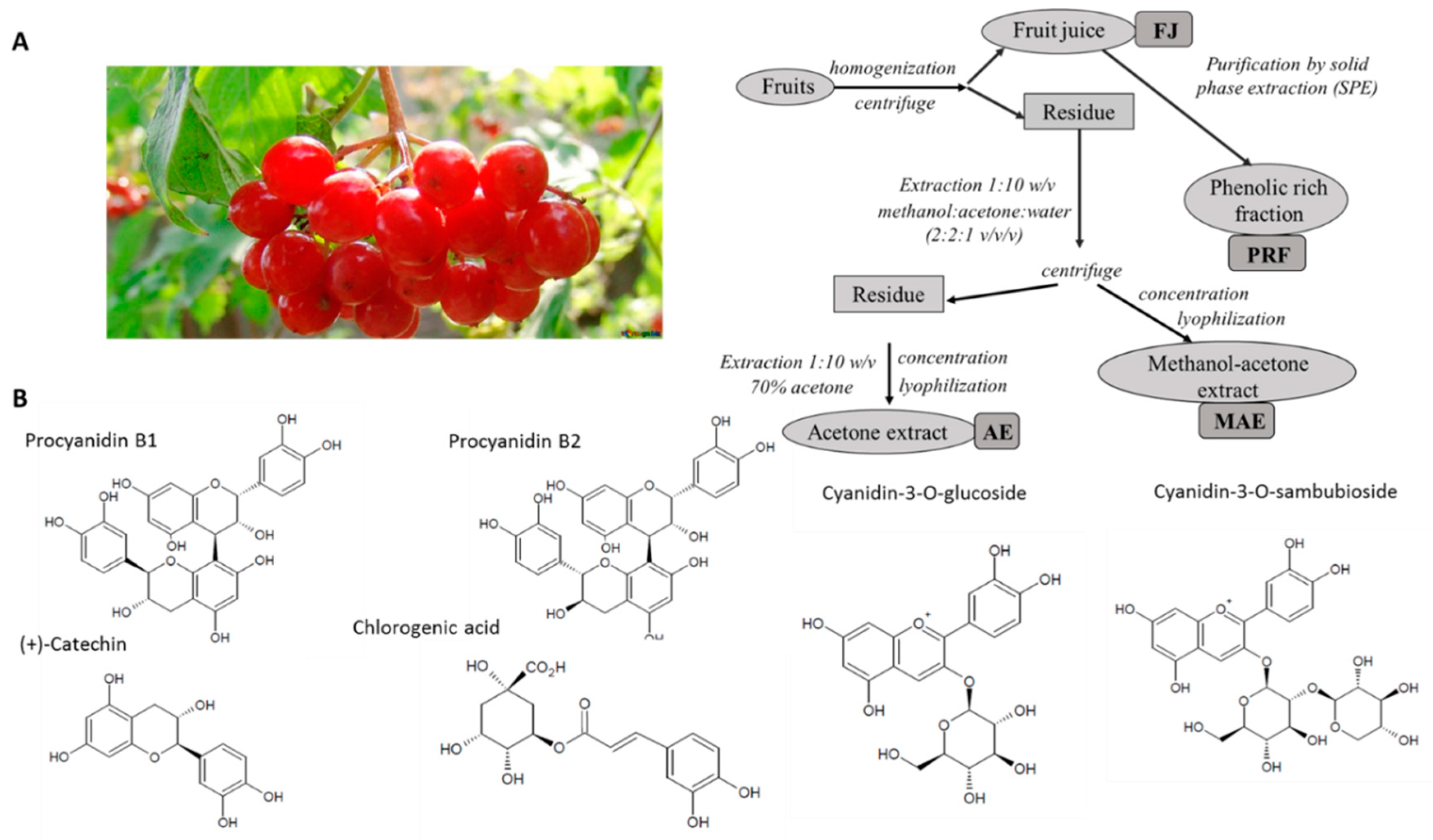








| Hydroxybenzoic Acid (HBA) | Rt [min] | λmax [nm] | FJ | MAE | AE | PRF |
|---|---|---|---|---|---|---|
| mg/g of Juice | mg/g of Extract | |||||
| Gallic acid | 1.92 | 282.7 | n.d. | 0.21 ± 0.00 a | 0.40 ± 0.00 b | n.d. |
| Flavanols | 1.21 ± 0.01 | 22.84 ± 0.05 | 15.46 ± 0.22 | 100.74 ± 0.10 | ||
| Procyanidin B1 | 4.50 | 278.7 | 0.31 ± 0.00 b | 3.28 ± 0.03 c | 2.46 ± 0.01 a | 29.76 ± 0.05 d |
| (+)-Catechin | 5.19 | 278.7 | 0.68 ± 0.01 b | 9.71 ± 0.01 c | 5.70 ± 0.00 a | 53.18 ± 0.03 d |
| Procyanidin B2 | 5.98 | 278.7 | 0.22 ± 0.00 a | 5.44 ± 0.02 c | 3.77 ± 0.03 b | 17.79 ± 0.02 d |
| (−)-Epicatechin | 6.85 | 277.7 | n.d. | 4.42 ± 0.02 b | 3.53 ± 0.18 a | n.d. |
| Hydroxycinnamic Acids (HCA) | 7.97 ± 0.02 | 96.37 ± 0.14 | 84.27 ± 0.26 | 645.85 ± 2.00 | ||
| Neochlorogenic acid | 3.94 | 323.7 | n.d. | 0.16 ± 0.00 b | 0.15 ± 0.00 a | 0,36 ± 0.00 c |
| Chlorogenic acid | 5.55 | 325.7 | 7.97 ± 0.02 a | 96.22 ± 0.14 c | 84.31 ± 0.26 b | 645.49 ± 2.00 |
| Flavonols | 0.01 ± 0.00 | 0.54 ± 0.00 | 0.48 ± 0.00 | 2.35 ± 0.00 | ||
| Rutin | 9.66 | 353.7 | 0.01 ± 0.00 b | n.d. | 0.12 ± 0.00 a | 1.09 ± 0.00 c |
| Isorhamnetin | 9.93 | 353.7 | 0.002 ± 0.000 a | n.d. | n.d. | 0.12 ± 0.00 b |
| Isorhamnetin 3-O-rutinoside | 11.20 | 353.7 | 0.01 ± 0.00 a | 0.26 ± 0.00c | 0.18 ± 0.00 b | 0.84 ± 0.00 d |
| Quercetin | 11.55 | 352.7 | n.d. | 0.28 ± 0.00 b | 0.17 ± 0.00 a | 0.30 ± 0.00 c |
| Anthocyanins | 1.12 ± 0.00 | 2.07 ± 0.01 | 1.45 ± 0.00 | 78.06 ± 0.68 | ||
| Cyanidin-3-O-sambubioside | 6.83 | 516.7 | 0.85 ± 0.00 | 0.74 ± 0.00 | 0.56 ± 0.00 | 55.78 ± 0.69 |
| Cyanidin-3-O-glucoside | 7.03 | 515.7 | 0.20 ± 0.01 | 0.86 ± 0.00 | 0.58 ± 0.00 | 17.08 ± 0.01 |
| Cyanidin-3-O-rutinoside | 7.36 | 518.7 | 0.06 ± 0.00 | 0.47 ± 0.00 | 0.31 ± 0.00 | 5.21 ± 0.01 |
| Phenolics Total | 10.32 ± 0.03 | 121.83 ± 0.19 | 101.67 ± 0.41 | 827.00 ± 1.68 | ||
| Sample | Total Phenolics | Antioxidant Capacity | |
|---|---|---|---|
| ABTS | ORAC | ||
| mg GAE/g | µmol TE/g | ||
| Methanol-Acetone Extract (MAE) | 143.77 ± 4.0 b | 1230.81 ± 26.1 b | 2388.58 ± 150.5c |
| Acetone Extract (AE) | 137.80 ± 5.6 b | 1227.73 ± 32.0 b | 2092.65 ± 48.3 b |
| Phenolic Rich Fraction (PRF) | 361.52 ± 13.3 c | 2619.59 ± 123.1c | 7810.29 ± 342.3d |
| Fresh Juice (FJ) | 5.98 ± 0.3 a | 38.36 ± 2.7 a | 103.50 ± 6.4 a |
| V. opulus Preparation | IC0 [µg/mL] | IC50 [µg/mL] | Cells Viability [%] 1 | |
| t-BOOH 500 μM | 79.50 ± 3.85 | |||
| FJ | 75.0 | 450.0 | 95.64 ± 8.78 | |
| PRF | 50.0 | 250.0 | 96.12 ± 10.12 | |
| MAE | 50.0 | 275.0 | 91.70 ± 8.31 | |
| AE | 100 | 400.0 | 90.50 ± 3.02 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zakłos-Szyda, M.; Pawlik, N.; Polka, D.; Nowak, A.; Koziołkiewicz, M.; Podsędek, A. Viburnum opulus Fruit Phenolic Compounds as Cytoprotective Agents Able to Decrease Free Fatty Acids and Glucose Uptake by Caco-2 Cells. Antioxidants 2019, 8, 262. https://doi.org/10.3390/antiox8080262
Zakłos-Szyda M, Pawlik N, Polka D, Nowak A, Koziołkiewicz M, Podsędek A. Viburnum opulus Fruit Phenolic Compounds as Cytoprotective Agents Able to Decrease Free Fatty Acids and Glucose Uptake by Caco-2 Cells. Antioxidants. 2019; 8(8):262. https://doi.org/10.3390/antiox8080262
Chicago/Turabian StyleZakłos-Szyda, Małgorzata, Nina Pawlik, Dominika Polka, Adriana Nowak, Maria Koziołkiewicz, and Anna Podsędek. 2019. "Viburnum opulus Fruit Phenolic Compounds as Cytoprotective Agents Able to Decrease Free Fatty Acids and Glucose Uptake by Caco-2 Cells" Antioxidants 8, no. 8: 262. https://doi.org/10.3390/antiox8080262