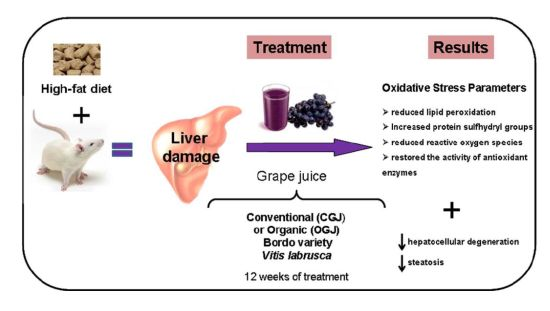
Hepatoprotective and Antioxidant Potential of Organic and Conventional Grape Juices in Rats Fed a High-Fat Diet
Abstract
:1. Introduction
2. Experimental Section
2.1. Chemicals
2.2. Diets
2.3. Grape Juices
2.4. Animals
2.5. Treatment
2.6. Weight Changes
2.7. Oxidative Stress Measurements
2.7.1. Tissue Preparation
2.7.2. Thiobarbituric Acid-Reactive Substances (TBARS) Measurement
2.8. Carbonyl Assay
2.9. Sulfhydryl Assay
2.10. Determination of Antioxidant Enzyme Activities
2.11. 2′,7′-Dihydrodichlorofluorescein Oxidation Assay
2.12. Protein Determination
2.13. Histopathological Analysis of the Liver Tissues
2.14. Statistical Analysis
3. Results
3.1. Effect of Treatment with HFD and Grape Juices on Animal Body Composition
| Parameters | Control | HFD | HFD + CGJ | HFD + OGJ |
|---|---|---|---|---|
| Weight (g) | 329.2 ± 16.04 | 315.4 ± 14.62 | 279.4 ± 4.95 * | 274.3 ± 9.04 * |
| Weight gain (g) | 274.4 ± 16.39 | 260.2 ± 14.44 | 230.6 ± 3.78 | 225.1 ± 9.1 * |
| Liver weight (g) | 9.2 ± 0.64 | 9.0 ± 0.47 | 8.6 ± 0.2 | 8.7 ± 0.4 |
| Body weight (g)/Liver weight (g) | 36.0 ± 2.92 | 34.9 ± 3.54 | 32.5 ± 2.42 | 30.6 ± 2.50 * |
3.2. Effect of Treatment with HFD and Grape Juices on Oxidative Stress Parameters
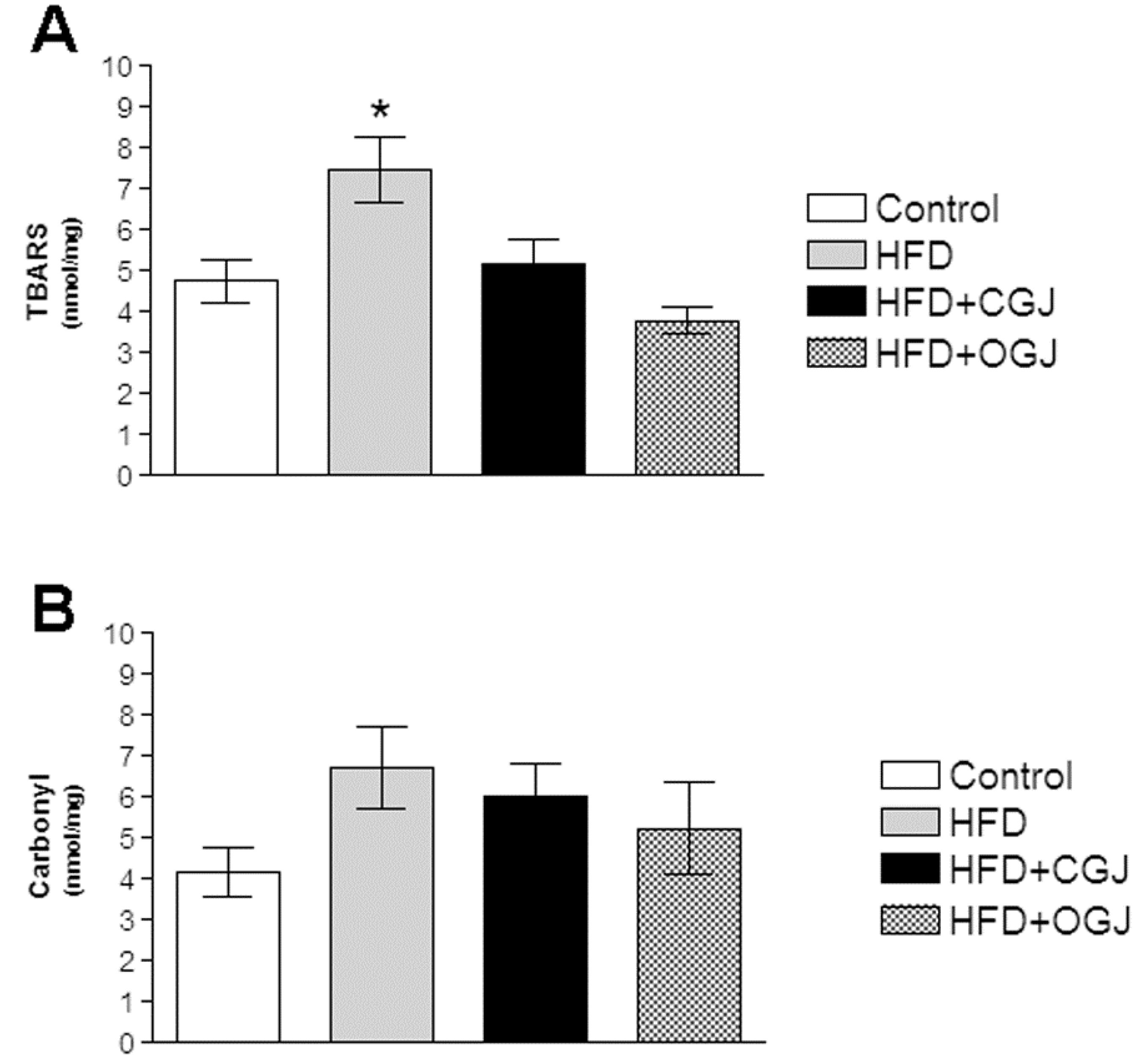


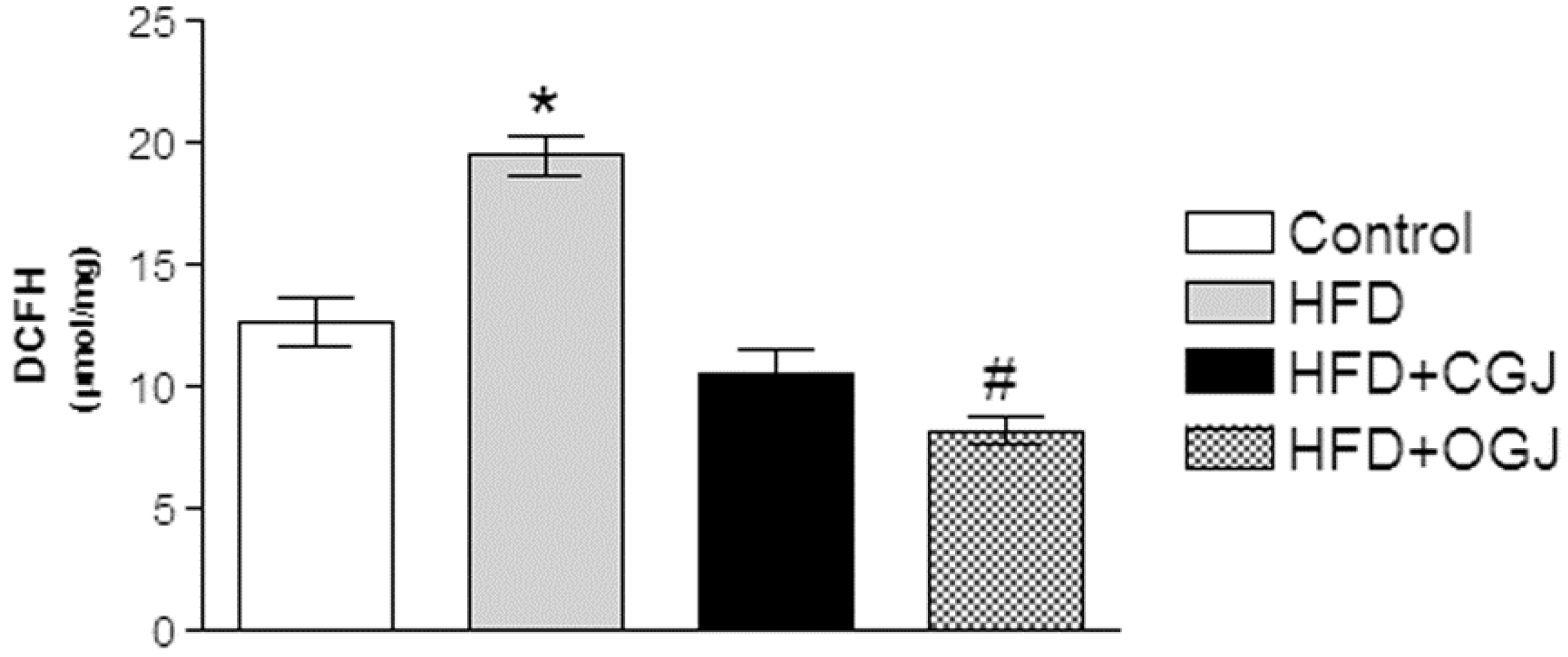
3.3. Effect of Treatment with HFD and Grape Juices on Cytoarchitecture and Histochemistry of Liver

4. Discussion
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- WHO—World Health Organization. Obesity and Overweight. Available online: http://ww.who.int/mediancenter/factsheets/fs311/en/index.html (accessed on 10 April 2013).
- Yang, X.; Telama, R.; Viikari, J.; Raitakari, O.T. Risk of obesity in relation to physical activity tracking from youth to adulthood. Med. Sci. Sports Exerc. 2006, 38, 919–925. [Google Scholar] [CrossRef]
- Yamamoto-Kimura, L.; Posadas-Romero, C.; Posadas-Sánchez, R.; Zamora-González, J.; Cardoso-Saldaña, G.; Méndez Ramírez, I. Prevalence and interrelations of cardiovascular risk factors in urban and rural Mexican adolescents. J. Adolesc. Health 2006, 38, 591–598. [Google Scholar] [CrossRef]
- Lomonaco, R.; Chen, J.; Cusi, K. An Endocrine Perspective of Nonalcoholic Fatty Liver Disease (NAFLD). Ther. Adv. Endocrinol. Metab. 2011, 2, 211–225. [Google Scholar] [CrossRef]
- Banerji, M.A.; Buckley, M.C.; Chaiken, R.L.; Gordon, D.; Lebovitz, H.E.; Kral, J.G. Liver fat, serum triglycerides and visceral adipose tissue in insulin-sensitive and insulin-resistant black men with NIDDM. Int. J. Obes. Relat. Metab. Disord. 1995, 19, 846–850. [Google Scholar] [PubMed]
- Oh, H.Y.; Shin, S.K.; Heo, H.S.; Ahn, J.S.; Kwon, E.Y.; Park, J.H.; Cho, Y.Y.; Park, H.J.; Lee, M.K.; Kim, E.J.; et al. Time-dependent network analysis reveals molecular targets underlying the development of diet-induced obesity and non-alcoholic steatohepatitis. Genes Nutr. 2012, 8, 301–316. [Google Scholar] [PubMed]
- Kabir, M.; Catalano, K.J.; Ananthnarayan, S.; Kim, S.P.; van Citters, G.W.; Dea, M.K.; Bergman, R.N. Molecular evidence supporting the portal theory: A causative link between visceral adiposity and hepatic insulin resistance. Am. J. Physiol. Endocrinol. Metab. 2005, 288, 454–461. [Google Scholar] [CrossRef]
- Ravel, A.P.; Dave, K.R.; Pérez-Pinzón, M.A. Resveratrol mimics ischemic preconditioning in the brain. J. Cereb. Blood Flow Metab. 2006, 26, 1141–1147. [Google Scholar] [PubMed]
- Zern, T.L.; Fernandez, M.L. Cardioprotective effect of dietary of polyphenols. J. Nutr. 2005, 135, 2291–2294. [Google Scholar] [PubMed]
- Silver, H.J.; Dietrich, M.S.; Niswender, K.D. Effects of grapefruit, grapefruit juice and water preloads on energy balance, weight loss, body composition, and cardiometabolic risk in free-living obese adults. Nutr. Metab. 2011, 8. [Google Scholar] [CrossRef]
- Boqué, N.; Campión, J.; de la Iglesia, R.; de la Garza, A.L.; Milagro, F.I.; Roman, B.S.; Bañuelos, O.; Martínez, J.Á. Screening of polyphenolic plant extracts for anti-obesity properties in Wistar rats. J. Sci. Food Agric. 2013, 93, 1226–1232. [Google Scholar] [CrossRef]
- Park, H.J.; Jung, U.J.; Lee, M.K.; Cho, S.J.; Jung, H.K.; Hong, J.H.; Park, Y.B.; Kim, S.R.; Shim, S.; Jung, J.; Choi, M.S. Modulation of lipid metabolism by polyphenol-rich grape skin extract improves liver steatosis and adiposity in high fat fed mice. Mol. Nutr. Food Res. 2013, 57, 360–364. [Google Scholar] [CrossRef]
- Lau, F.; Shukitt, H.; Joseph, J. The beneficial effects of fruit polyphenols on brain aging. Neurobiol. Aging 2005, 1, 128–132. [Google Scholar]
- Venturini, C.D.; Merlo, S.; Souto, A.A.; Fernandes, M.C.; Gomez, R.; Rhoden, C.R. Resveratrol and red wine function as antioxidants in the central nervous system without cellular proliferative effects during experimental diabetes. Oxid. Med. Cell. Longev. 2011, 3, 434–441. [Google Scholar]
- Yadav, M.; Jain, S.; Bhardwaj, A.; Nagpal, R.; Puniya, M.; Tomar, R.; Singh, V.; Parkash, O.; Prasad, G.B.; Marotta, F.; Yadav, H. Biological and medicinal properties of grapes and their bioactive constituents. J. Med. Food 2009, 12, 473–484. [Google Scholar] [CrossRef]
- Dani, C.; Pasquali, M.A.B.; Oliveira, M.R.; Umezu, F.M.; Salvador, M.; Henriques, J.A.P.; Moreira, J.C.F. Intake of purple grape juice as a hepatoprotective agent in Wistar rats. J. Med. Food 2008, 11, 127–132. [Google Scholar] [CrossRef]
- Osman, H.E.; Maalej, N.; Shanmuganayagam, D.; Folts, J.D. Grape juice but not orange or grapefruit juice inhibits platelet activity in dogs and monkeys (Macaca fasciularis). J. Nutr. 1998, 128, 2307–2312. [Google Scholar] [PubMed]
- Day, A.P.; Kemp, H.J.; Bolton, C.; Hartog, M.; Stansbie, D. Effect of concentrated red grape juice consumption on serum antioxidant capacity and low-density lipoprotein oxidation. Ann. Nutr. Metab. 1997, 41, 353–357. [Google Scholar] [CrossRef]
- Dani, C.; Oliboni, L.; Umezu, F.; Salvador, M.; Moreira, J.C.; Fonseca, J.A. Antioxidant and Antigenotoxic Activities of Purple Grape Juice Organic and Conventional in Adult Rats. J. Med. Food 2009, 12, 1111–1118. [Google Scholar] [CrossRef]
- Rodrigues, A.D.; Scheffel, T.B.; Scola, G.; Santos, M.T.; Fank, B.; de Freitas, S.C.; Dani, C.; Vanderlinde, R.; Henriques, J.A.; Coitinho, A.S.; et al. Neuroprotective and anticonvulsant effects of organic and conventional purple grape juices on seizures in Wistar rats induced by pentylenetetrazole. Neurochem. Int. 2012, 60, 799–805. [Google Scholar] [CrossRef]
- Dani, C.; Oliboni, L.S.; Vanderlinde, R.; Bonatto, D.; Salvador, M.; Henriques, J.A.P. Phenolic content and antioxidant activities of white and purple juices manufactured with organically- or conventionally-produced grapes. Food Chem. Toxicol. 2007, 45, 2574–2580. [Google Scholar] [CrossRef]
- Aquilano, K.; Baldelli, S.; Rotilio, G.; Ciriolo, M.R. Role of nitric oxide synthases in Parkinson’s disease: A review on the antioxidant and anti-inflammatory activity of polyphenols. Neurochem. Res. 2008, 33, 2416–2426. [Google Scholar] [CrossRef]
- Markus, M.A.; Morris, B.J. Resveratrol in prevention and treatment of common clinical conditions of aging. Clin. Interv. Aging 2008, 3, 331–339. [Google Scholar] [PubMed]
- Bose, M.; Lambert, J.D.; Ju, J.; Reuhl, K.R.; Shapses, S.A.; Yag, C.S. The major green tea polyphenol, (−)-epigallocatechin-3-gallate, inhibits obesity, metabolic syndrome, and fatty liver disease in high-fat-fed mice. J. Nutr. 2008, 138, 1677–1683. [Google Scholar] [PubMed]
- Yang, D.J.; Chang, Y.Y.; Hsu, C.L.; Liu, C.W.; Lin, Y.L.; Lin, Y.H.; Liu, K.C.; Chen, Y.C. Antiobesity and hypolipidemic effects of polyphenol-rich longan (Dimocarpus longans Lour.) flower water extract in hypercaloric-dietary rats. J. Agric. Food Chem. 2010, 58, 2020–2027. [Google Scholar] [CrossRef] [PubMed]
- Beltrán-Debón, R.; Rull, A.; Rodríguez-Sanabria, F.; Iswaldi, I.; Herranz-López, M.; Aragonès, G.; Camps, J.; Alonso-Villaverde, C.; Menéndez, J.A.; Micol, V.; et al. Continuous administration of polyphenols from aqueous rooibos (Aspalathus linearis) extract ameliorates dietary-induced metabolic disturbances in hyperlipidemic mice. Phytomedicine 2011, 18, 414–424. [Google Scholar] [CrossRef]
- Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 1979, 95, 351–358. [Google Scholar] [CrossRef]
- Reznick, A.Z.; Packer, L. Methods. Carbonyl assay for determination of oxidatively modified proteins. Methods Enzymol. 1994, 233, 357–363. [Google Scholar] [CrossRef]
- Aksenov, M.Y.; Markesberry, W.R. Change in thiol content and expression of glutathione redox system gene in the hippocampus and cerebellum in Alzheimer’s disease. Neurosci. Lett. 2001, 302, 141–145. [Google Scholar] [CrossRef]
- Bannister, J.V.; Calabrese, L. Assays for SOD. Method Biochem. Anal. 1987, 32, 279–312. [Google Scholar] [CrossRef]
- Aebi, H. Catalase in vitro. Methods Enzymol. 1984, 105, 121–126. [Google Scholar] [CrossRef]
- Flohe, L.; Gunzler, W.A. Assays of glutathione peroxidase. Methods Enzymol. 1984, 105, 114–121. [Google Scholar] [CrossRef]
- LeBel, C.P.; Ischiropoulos, H.; Bondy, S.C. Evaluation of the probe 2′,7′-dichlorofluorescin as an indicator of reactive oxygen species formation and oxidative stress. Chem. Res. Toxicol. 1992, 5, 227–231. [Google Scholar] [CrossRef]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Radall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951, 193, 265–267. [Google Scholar] [PubMed]
- Normann, C.A.B.M.; Moreira, J.C.F.; Cardoso, V.V. Micronuclei in red blood cells of armored catfish Hypostomus plecotomus exposed to potassium dichromate. AJB 2008, 7, 893–896. [Google Scholar]
- Jen, K.L.C.; Buison, A.; Pellizzon, M.; Ordiz, F., Jr.; Ana, L.S.; Brown, J. Differential Effects of Fatty Acids and Exercise on Body Weight Regulation and Metabolism in Female Wistar Rats. Exp. Biol. Med. 2003, 228, 843–849. [Google Scholar]
- Feoli, A.M.; Roehrig, C.; Rotta, L.N.; Kruger, A.H.; Souza, K.B.; Kessler, A.M.; Renz, S.V.; Brusque, A.M.; Souza, D.O.; Perry, M.L. Serum and liver lipids in rats and chicks fed with diets containing different oils. Nutrition 2003, 19, 789–793. [Google Scholar] [CrossRef]
- Perichart-Perera, O.; Balas-Nakash, M.; Rodríguez-Cano, A.; Muñoz-Manrique, C.; Monge-Urrea, A.; Vadillo-Ortega, F. Correlates of Dietary Energy Sources with Cardiovascular Disease Risk Markers in Mexican School—Age Children. J. Am. Diet. Assoc. 2010, 110, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Papaharalambus, C.A.; Griendling, K.K. Basic Mechanisms of Oxidative Stress and Reactive Oxygen Species in Cardiovascular Injury. Trends Cardiovasc. Med. 2007, 17, 48–54. [Google Scholar] [CrossRef]
- Melega, S.; Canistro, D.; Nicola, G.R.; Lazzeri, L.; Sapone, A.; Paolini, M. Protective effect of Tuscan black cabbage sprout extract against serum lipid increase and perturbations of liver antioxidant and detoxifying enzymes in rats fed a high-fat diet. Br. J. Nutr. 2013, 110, 988–997. [Google Scholar] [CrossRef]
- Mcdonald, R.B. Influence of dietary sucrose on biological aging. Am. J. Clin. Nutr. 1995, 62, 284–292. [Google Scholar]
- Halliwell, B.; Gutteridge, J.M.C. Measurement of reactive species. In Free Radicals in Biology and Medicine, 4th ed.; Oxford University Press: New York, NY, USA, 2007; pp. 268–340. [Google Scholar]
- Ferreira, A.L.A.; Matsubara, L.S. Radicais livres: Conceitos, doenças relacionadas, sistema de defesa e estresse oxidativo. Rev. Assoc. Med. Bras. 1997, 43, 61–68. [Google Scholar] [PubMed]
- Park, K.Y.; Park, E.; Kim, J.S.; Kang, M.H. Daily grape juice consumption reduces oxidative DNA damage and plasma free radical levels in healthy Koreans. Mutat. Res. 2003, 529, 77–86. [Google Scholar] [CrossRef]
- Robb, E.L.; Winkelmolen, L.; Visanji, N.; Brotchie, J.; Stuart, J.A. Dietary resveratrol administration increases MnSod expression and activity in mouse brain. Biochem. Biophys. Res. Commun. 2008, 372, 254–259. [Google Scholar] [CrossRef]
- Sinha, K.; Chaudhary, G.; Gupta, Y.K. Protective effect of resveratrol against oxidative stress in middle cerebral artery occlusion model of stroke in rats. Life Sci. 2002, 71, 655–665. [Google Scholar] [CrossRef]
- Han, Y.S.; Zheng, W.H.; Bastianetto, S.; Chabot, J.G.; Quirion, R. Neuroprotective effects of resveratrol against β-amyloid-induced neurotoxicity in rat hippocampal neurons: Involvement of protein kinase C. Brit. J. Pharmacol. 2004, 141, 997–1005. [Google Scholar] [CrossRef]
- Vingtdeux, V.; Werringloer, U.D.; Zhao, H.; Davies, P.; Marambaud, P. Therapeutic potential of resveratrol in Alzheimer’s disease. BMC Neurosci. 2008, 9. [Google Scholar] [CrossRef]
- Xia, N.; Daiber, A.; Habermeier, A.; Closs, E.I.; Thum, T.; Spanier, G.; Lu, Q.; Oelze, M.; Torzewki, M.; Lackner, K.J.; et al. Resveratrol reverses endothelial nitric-oxide synthase uncoupling in apolipoprotein E knockout mice. J. Pharmacol. Exp. Ther. 2010, 335, 149–154. [Google Scholar] [CrossRef]
- Lakshmi, B.V.; Sudhakar, M.; Aparna, M. Protective potential of Black grapes against lead induced oxidative stress in rats. Environ. Toxicol. Pharmacol. 2013, 35, 361–368. [Google Scholar] [CrossRef]
- Scola, G.; Scheffel, T.; Gambato, G.; Freitas, S.; Dani, C.; Funchal, C.; Gomez, R.; Coitinho, A.; Salvador, M. Flavan-3-ol compounds prevent pentylenetetrazol-induced oxidative damage in rats without producing mutations and genotoxicity. Neurosci. Lett. 2013, 534, 145–149. [Google Scholar] [CrossRef]
- Sharma, S.K.; Suman Vasudeva, N. Hepatoprotective activity of Vitis vinifera root extract against carbon tetrachloride-induced liver damage in rats. Acta Pol. Pharm. 2012, 69, 933–937. [Google Scholar] [PubMed]
- Chang, C.C.; Chang, C.Y.; Huang, J.P.; Hung, L.M. Effect of resveratrol on oxidative and inflammatory stress in liver and spleen of streptozotocin-induced type 1 diabetic rats. J. Physiol. 2012, 55, 192–201. [Google Scholar]
- Gourineni, V.; Shay, N.F.; Chung, S.; Sandhu, A.K.; Gu, L. Muscadine grape (Vitis rotundifolia) and wine phytochemicals prevented obesity-associated metabolic complications in C57BL/6J mice. J. Agric. Food Chem. 2012, 60, 7674–7681. [Google Scholar] [CrossRef]
- Nagao, K.; Jinnouchi, T.; Kai, S.; Yanagita, T. Effect of dietary resveratrol on the metabolic profile of nutrients in obese OLETF rats. Lipids Health Dis. 2013, 12. [Google Scholar] [CrossRef]
- Lagouge, M.; Argmann, C.; Gerhart-Hines, Z.; Meziane, H.; Lerin, C.; Daussin, F.; Messadeq, N.; Milne, J.; Lambert, P.; Elliott, P.; et al. Resveratrol improves mitochondrial function and protects against metabolic disease by activating SIRT1 and PGC-1α. Cell 2006, 127, 1109–1122. [Google Scholar] [CrossRef]
- Du, Z.; Yang, Y.; Hu, Y.; Sun, Y.; Zhang, S.; Peng, W.; Zhong, Y.; Huang, X.; Kong, W. A long-term high-fat diet increases oxidative stress, mitochondrial damage and apoptosis in the inner ear of D-galactose-induced aging rats. Hear. Res. 2012, 287, 15–24. [Google Scholar] [CrossRef]
- Fachinetto, R.; Burger, M.E.; Wagner, C.; Wondeacek, D.C.; Brito, V.B.; Nogueira, C.W.; Ferreira, J.; Rocha, J.B.T. High fat diet increases the incidence of orofacial dyskinesia and oxidative stress in specific brain regions of rats. Pharmacol. Biochem. Behav. 2005, 81, 585–592. [Google Scholar] [CrossRef]
- Ribeiro, M.C.P.; Barbosa, N.B.V.; Almeida, T.M.; Parcianello, L.M.; Perottoni, J.; de Ávila, D.S.; Rocha, J.B.T. High-fat diet and hydrochlorothiazide increase oxidative stress in brain of rats. Cell Biochem. Funct. 2009, 27, 473–478. [Google Scholar] [CrossRef]
- Muthulakshmi, S.; Saravanan, R. Efficacy of azelaic acid on hepatic key enzymes of carbohydrate metabolism in high fat diet induced type 2 diabetic mice. Biochimie 2013, 95, 1239–1244. [Google Scholar] [CrossRef]
- Rodrigues, A.D.; Scheffel, T.B.; Scola, G.; Dos Santos, M.T.; Fank, B.; Dani, C.; Vanderlinde, R.; Henriques, J.A.; Coitinho, A.S.; Salvador, M. Purple grape juices prevent pentylenetetrazol-induced oxidative damage in the liver and serum of Wistar rats. Nutr. Res. 2013, 33, 120–125. [Google Scholar] [CrossRef]
- Dani, C.; Pasquali, M.A.B.; Oliveira, M.R.; Umezu, F.M.; Salvador, M.; Henriques, J.A.P.; Moreira, J.C.F. Protective effects of purple grape juice on carbon tetrachloride-induced oxidative stress in brains of adult Wistar rats. J. Med. Food 2008, 11, 55–61. [Google Scholar] [CrossRef]
- Bayram, I.; Erten, R.; Bayram, Y.; Bulut, G.; Ozbek, H. The hepatoprotective effects of dihydromyrcenol and geranyl formate in an experimental model of acute hepatic injury induced by the use of carbon tetrachloride. Turk. J. Gastroenterol. 2011, 22, 594–601. [Google Scholar] [PubMed]
- Prabu, S.M.; Shagirtha, K.; Renugadevi, J. Naringenin in combination with vitamins C and E potentially protects oxidative stress-mediated hepatic injury in cadmium-intoxicated rats. J. Nutr. Sci. Vitaminol. 2011, 57, 177–185. [Google Scholar] [CrossRef]
- El-Beshbishy, H.A.; Mohamadin, A.M.; Nagy, A.A.; Abdel-Naim, A.B.; Indian, J. Amelioration of tamoxifen-induced liver injury in rats by grape seed extract, black seed extract and curcumin. Exp. Biol. 2010, 48, 280–288. [Google Scholar]
- Lee, E.S.; Shin, M.O.; Yoon, S.; Moon, J.O. Resveratrol inhibits dimethylnitrosamine-induced hepatic fibrosis in rats. Arch. Pharm. Res. 2010, 33, 925–932. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Buchner, I.; Medeiros, N.; Lacerda, D.D.S.; Normann, C.A.B.M.; Gemelli, T.; Rigon, P.; Wannmacher, C.M.D.; Henriques, J.A.P.; Dani, C.; Funchal, C. Hepatoprotective and Antioxidant Potential of Organic and Conventional Grape Juices in Rats Fed a High-Fat Diet. Antioxidants 2014, 3, 323-338. https://doi.org/10.3390/antiox3020323
Buchner I, Medeiros N, Lacerda DDS, Normann CABM, Gemelli T, Rigon P, Wannmacher CMD, Henriques JAP, Dani C, Funchal C. Hepatoprotective and Antioxidant Potential of Organic and Conventional Grape Juices in Rats Fed a High-Fat Diet. Antioxidants. 2014; 3(2):323-338. https://doi.org/10.3390/antiox3020323
Chicago/Turabian StyleBuchner, Iselde, Niara Medeiros, Denise Dos Santos Lacerda, Carlos Augusto B. M. Normann, Tanise Gemelli, Paula Rigon, Clovis Milton Duval Wannmacher, João Antônio Pegas Henriques, Caroline Dani, and Cláudia Funchal. 2014. "Hepatoprotective and Antioxidant Potential of Organic and Conventional Grape Juices in Rats Fed a High-Fat Diet" Antioxidants 3, no. 2: 323-338. https://doi.org/10.3390/antiox3020323