Antioxidant and Free Radical Scavenging Capacity of Seed and Shell Essential Oils Extracted from Abrus precatorius (L)
Abstract
:1. Introduction
2. Experimental Section
2.1. Chemicals Used
2.2. Plant Material and Extraction of Essential Oils
2.3. DPPH Assay
2.4. ABTS Assay
2.5. Lipid Peroxides Radical Scavenging Capacity
2.6. Nitric Oxide Radical Scavenging Capacity
2.7. Statistical Analyses
3. Results and Discussion
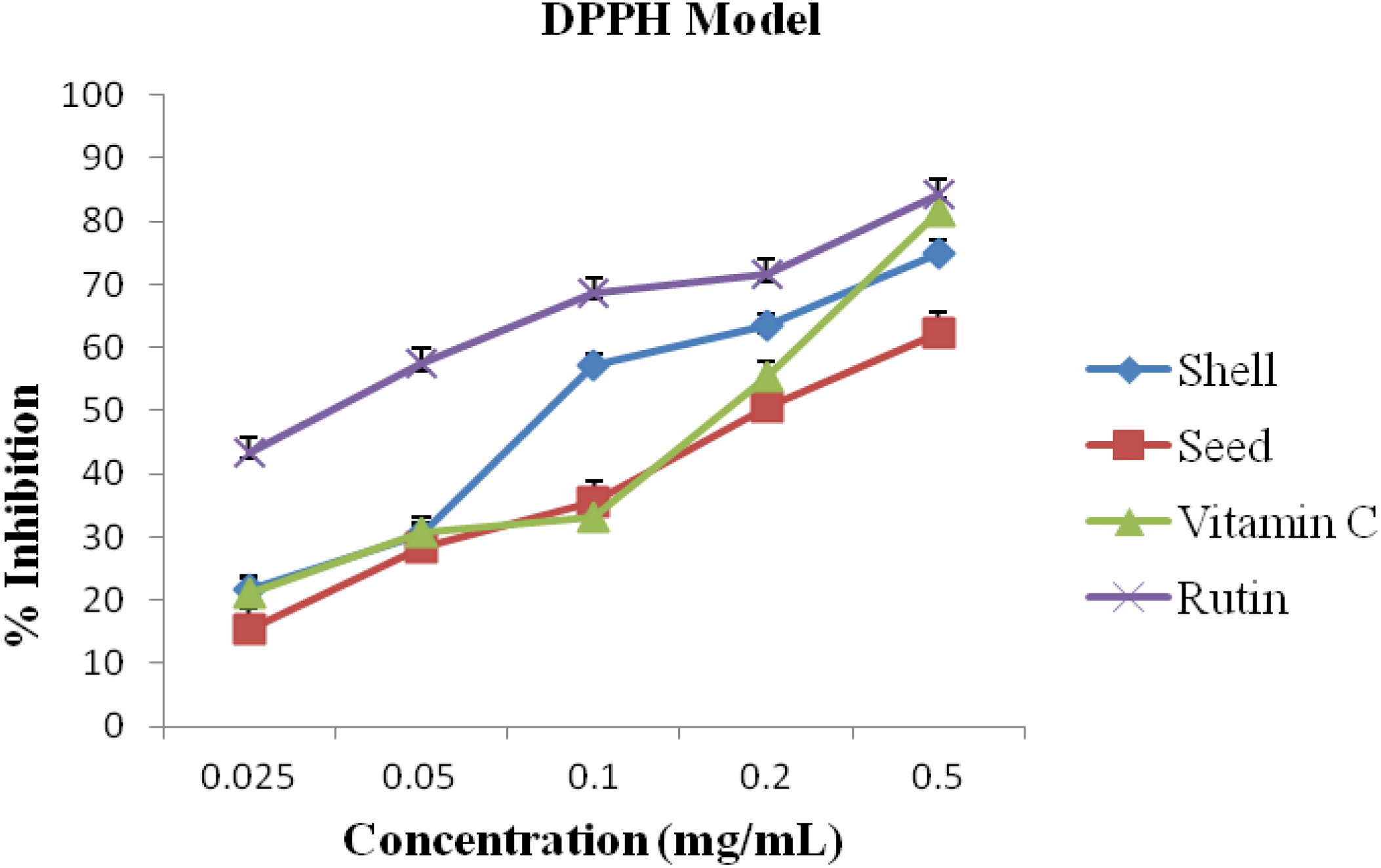
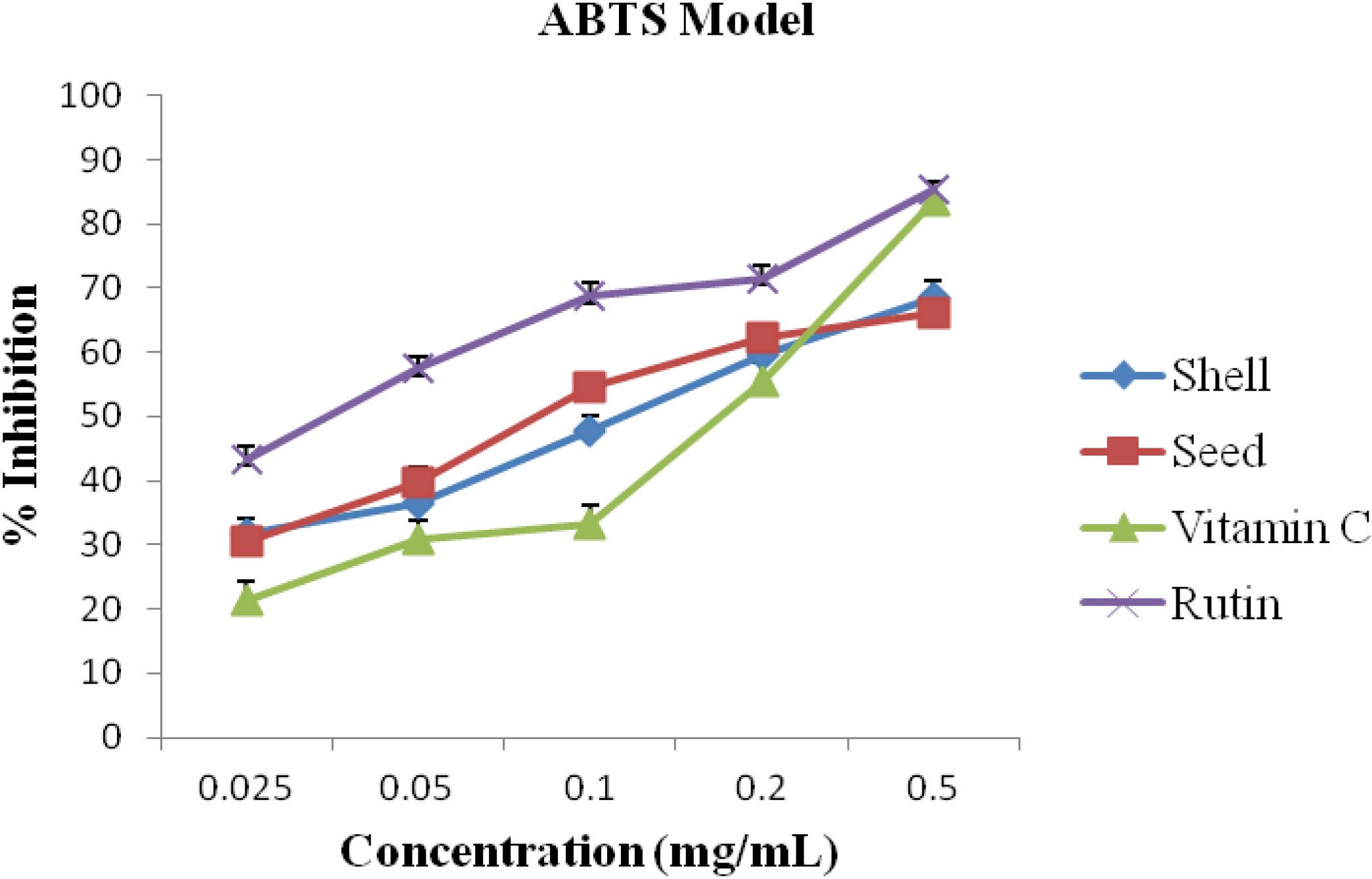
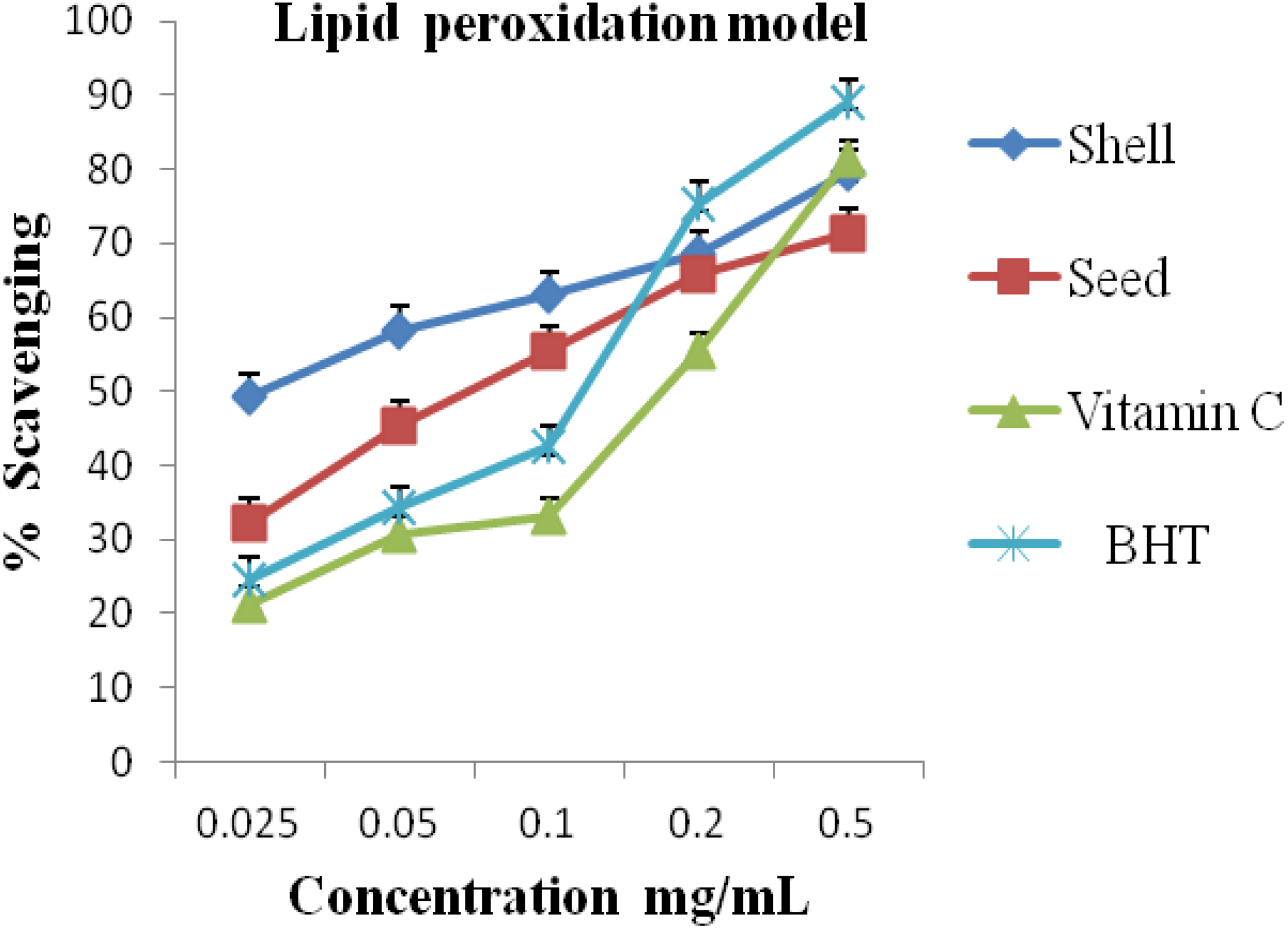
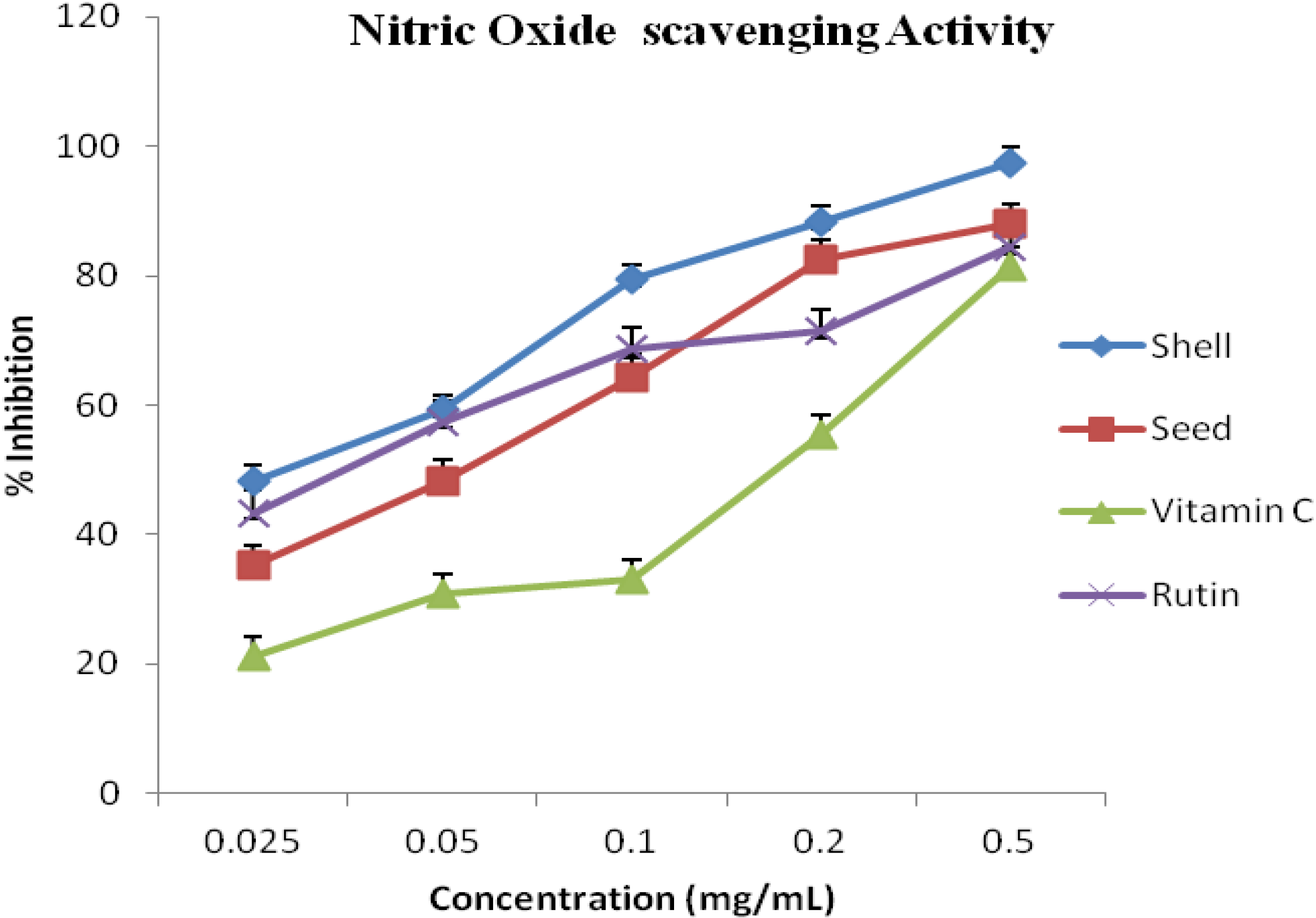
| S/N | Activity | A. precatorius | Standard/Commercial Antioxidants
(Positive Controls) | |||
|---|---|---|---|---|---|---|
| Seed Oil (IC50) | Shell Oil (IC50) | Vitamin C (IC50) | Rutin (IC50) | BHT b (IC50) | ||
| 1 | DPPH• | 5.0 3 ± 0.24 | 3.03 ± 0.11 | 1.50 ± 0.01 | 0.50 ± 0.04 | ND |
| 2 | ABTS+• | 2.95 ± 0.31 | 3.07 ± 0.22 | 0.10 ± 0.04 | 0.10 ± 0.01 | ND |
| 3 | LP• | 1.92 ± 2.10 | 1.42 ± 0.40 | 1.83 ± 0.33 | ND | 0.83 ± 0.40 |
| 4 | NO• | 2.10 ± 0.40 | 1.20 ± 0.20 | 1.20 ± 0.20 | 3.40 ± 0.01 | ND |
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Saikat, S.; Chakraborty, R.; Sridhar, C.Y.; Reddy, S.R.; Biplab, D. Free radicals, antioxidants, diseases and phytomedicine: Current status and future prospect. Int. J. Pharm. Sci. Rev. Res. 2010, 3, 91–100. [Google Scholar]
- Sachdev, S.; Davies, K. Production, detection, and adaptive responses to free radicals in exercise. Free Radic. Biol. Med. 2008, 44, 215–223. [Google Scholar] [CrossRef]
- Mahmood, R.M.; Soheila, M.; Saeid, A. Radical scavenging and reducing power of Salvia mirzayanii subfractions. Molecules 2008, 13, 2804–2813. [Google Scholar] [CrossRef]
- Mimica-Dukic, N.; Dušan, B.; Slavenko, M.; Dragana, V.G.; Branka, O.D. Essential oil of Myrtus communis L. as a potential antioxidant and antimutagenic agents. Molecules 2010, 15, 2759–2770. [Google Scholar] [CrossRef]
- Paul, S. Trachyspermum ammi (L.). Fruit essential oil influencing on membrane permeability and surface characteristics in inhibiting food-borne pathogens. Food Control 2011, 22, 725–731. [Google Scholar] [CrossRef]
- Morten, H.; Mygind, T.; Rikke, L. Essential oils in food preservation: Mode of action, synergies, and interactions with food matrix components. Front. Microbiol. 2012, 3, 12. [Google Scholar] [PubMed]
- Millogo-Kone, M.; Lompo, F.; Nacoulma, O. Evaluation of flavonoids, total phenolic contents of P. biglobosa and free radical scavenging and antimicrobial activities. Res. J. Med. Sci. 2009, 3, 70–74. [Google Scholar]
- Brenes, A.; Roura, E. Essential oils in poultry nutrition: Main effects and modes of action. Anim. Feed Sci. Technol. 2010, 158, 1–14. [Google Scholar] [CrossRef]
- Nweze, E.I.; Okafor, J.I. Activities of a wide range of medicinal plants and essential oil against Scedospaorium isolates. Am. Eurasian J. Sci. Res. 2010, 5, 161–169. [Google Scholar]
- Tuttolomondo, T.; Virga, G.; Curcuruto, G.; la Bella, S.; Leto, C.; Licata, M.; Napoli, E.; Pasquale, A.; Saija, A.; Siracusa, L.; et al. Biomolecular characterization of wild sicilian oregano: Phytochemical screening of essential oils and extracts, and evaluation of their antioxidant activities. Chem. Biodivers. 2013, 10, 411–433. [Google Scholar] [CrossRef]
- Mansoub, N.; Myandoab, P. Research opinion in animal. Vet. Sci. 2012, 2, 7–9. [Google Scholar]
- Wang, W.; Wu, N.; Zu, G.; Fu, Y. Antioxidant activity of Rosmarinus officinalis (L), essential oil compared to its main components. Food Chem. 2008, 108, 1019–1022. [Google Scholar] [CrossRef]
- Miller, E.R.; Pastor-Barriuso, R.; Dalal, D.; Riemersma, R.A.; Appel, L.J.; Guallar, E. Vitamin E supplementation may increase all-cause mortality. Ann. Intern. Med. 2005, 1, 37–46. [Google Scholar]
- Jesudoss, V.A.; Jayaraman, J.; Madhavan, S. d-limonene alleviates insulin resistance and oxidative stress in rat. Eur. J. Nutr. 2012, 51, 57–68. [Google Scholar] [CrossRef]
- Horvathova, E.; Katarina, K.; Srancikova, A.; Hunakova, L.; Galova, E.; Sevcovicova, A.; Slamenova, D. Borneol administration protects primary rat hepatocytes against exogenous oxidative DNA damage. Mutagenesis 2012, 27, 581–588. [Google Scholar] [CrossRef]
- Bidinotto, L.T.; Costa, C.A.; Salvadori, D.M.; Costa, M.; Rodrigues, M.A.; Barbisan, L.F. Effects lemongrass oil on DNA damage and carcinogenesis in female mice. J. Appl. Toxicol. 2011, 31, 536–544. [Google Scholar] [CrossRef]
- Kaurinovic, B.; Vlaisavljevic, S.; Popovic, M.; Vastag, D.; Djurendic-Brenesel, M. Antioxidant properties of Marrubium peregrinum essential oil. Molecules 2010, 15, 5943–5955. [Google Scholar] [CrossRef]
- Avlessi, F.; Alitonou, G.A.; Sohounhloue, D.K.; Bessiere, J.M.; Menut, C. Chemical and Biological evaluation of leaf essential oil of Commiphora africana from Benin. J. Essent. Oil Res. 2005, 17, 569. [Google Scholar] [CrossRef]
- John, D.B.; Benjamin, P.J.; Rathna, K.; Herin, D.S. Abrus precatorius L.: A medicinal plant with potential as antibacterial agent. J. Pharm. Res. 2012, 5, 1207–1209. [Google Scholar]
- Pal, R.S.; Ariharasivakumar, G.; Girhepunje, K.; Upadhyay, A. In vitro antioxidant activity of phenolic and flavonoids compounds extracted from seeds of Abrus precatorius. Int. J. Pharm. Pharm. Sci. 2009, 1, 136–140. [Google Scholar]
- Okoh, S.O.; Asekun, O.T.; Familoni, O.B. The essential oils and chemical composition of leaf, seed and shell of Abrus precatorius from Nigeria. In Proceedings of the 4th Annual Research Conference and Fair, University of Lagos, Lagos, Nigeria, 8 January 2009; University of Lagos Press: Lagos, Nigeria, 2008. [Google Scholar]
- Okoh, S.O.; Asekun, O.T.; Familoni, O.B.; Afolayan, A.J. Composition and antioxidant activities of leaf and root volatile oils of Morida lucida. J. Nat. Prod. Commun. 2011, 6, 1537–1541. [Google Scholar]
- Witayapan, N.; Sombat, C.; Siriporn, O. Antioxidant and antimicrobial activities of Hyptis suaveolens essential oil. Sci. Pharm. 2007, 75, 35–46. [Google Scholar] [CrossRef]
- Badmus, J.A.; Odunola, O.A.; Obuotor, E. Phytochemical and in vitro antioxidant potentials of Holarrhena floribunda leaf. Afr. J. Biotechnol. 2010, 9, 340–346. [Google Scholar]
- Makhija, I.K.; Aswatha Ram, H.N.; Shreedhara, C.S.; Vijay Kumar, S.; Devkar, R. In vitro antioxidant studies of Sitopaladi Churna, a polyherbal Ayurvedic formulation. Free Radic. Antioxid. 2011, 1, 37–41. [Google Scholar] [CrossRef]
- Guerrini, A.; Sacchetti, G.; Rossi, D.; Paganetto, G.; Muzzoli, M.; Andreotti, E. Bioactivities of Piper aduncum L. and Piper obliquum Ruiz & Pavon (Piperaceae) essential oils from Eastern Ecuador. Environ. Toxicol. Pharmacol. 2009, 27, 39–48. [Google Scholar] [CrossRef]
- Valko, M.; Leibfritz, D.; Moncol, J.; Mazur, M.; Telser, J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol. 2007, 39, 44–84. [Google Scholar] [CrossRef]
- Sathyavathi, A.; Suchetha, N.; Vijay, R.; Ullal, D.; Praveen, A. Status of Phosphodiesterase, Nirtic oxide and Arginase levels in hypo and hyperthyroidism. Int. J. Res. Pharm. Biomed. Sci. 2012, 3, 541–544. [Google Scholar]
- Fini, M.A.; Johnson, R.J.; Stenmark, K.R.; Wright, R.M. Hypertension, nitrate-nitrite, and xanthine oxidoreductase catalyzed nitric oxide generation: Pros and Cons. Hypertension 2013, 62, e9. [Google Scholar] [CrossRef]
- Luh, S.; Wong, S.; El-shimi, E. Effect of processing on some chemical constituents of Pistachio nuts. J. Food Qual. 2007, 5, 33–41. [Google Scholar]
- Foti, M.C.; Ingold, K.U. Unexpected superoxide dismutase antioxidant activity of Ferric chloride in acetonitrile. J. Org. Chem. 2009, 68, 9162–9125. [Google Scholar] [CrossRef]
- Peana, A.; Marzocco, S.; Popolo, A. Linalool inhibits in vitro NO formation: Probable involvement in the antinociceptive activity of this monoterpene compound. Life Sci. 2006, 78, 719–723. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Okoh, S.O.; Asekun, O.T.; Familoni, O.B.; Afolayan, A.J. Antioxidant and Free Radical Scavenging Capacity of Seed and Shell Essential Oils Extracted from Abrus precatorius (L). Antioxidants 2014, 3, 278-287. https://doi.org/10.3390/antiox3020278
Okoh SO, Asekun OT, Familoni OB, Afolayan AJ. Antioxidant and Free Radical Scavenging Capacity of Seed and Shell Essential Oils Extracted from Abrus precatorius (L). Antioxidants. 2014; 3(2):278-287. https://doi.org/10.3390/antiox3020278
Chicago/Turabian StyleOkoh, Sunday O., Olayinka T. Asekun, Oluwole B. Familoni, and Anthony J. Afolayan. 2014. "Antioxidant and Free Radical Scavenging Capacity of Seed and Shell Essential Oils Extracted from Abrus precatorius (L)" Antioxidants 3, no. 2: 278-287. https://doi.org/10.3390/antiox3020278
APA StyleOkoh, S. O., Asekun, O. T., Familoni, O. B., & Afolayan, A. J. (2014). Antioxidant and Free Radical Scavenging Capacity of Seed and Shell Essential Oils Extracted from Abrus precatorius (L). Antioxidants, 3(2), 278-287. https://doi.org/10.3390/antiox3020278





