Effectiveness of α-, γ- and δ-Tocopherol in a CLA-Rich Oil
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials and Samples
2.2. Oxidation Procedures
2.3. Analytical Methods
2.3.1. Determination of Tocopherols
2.3.2. Quantitation of Triacylglycerol Dimers and Higher Oligomers by High-Performance Size-Exclusion Chromatography (HPSEC)
2.3.3. Determination of the Peroxide Value (PV)
2.3.4. Analysis of Fatty Acid Composition
2.4. Statistical Analysis
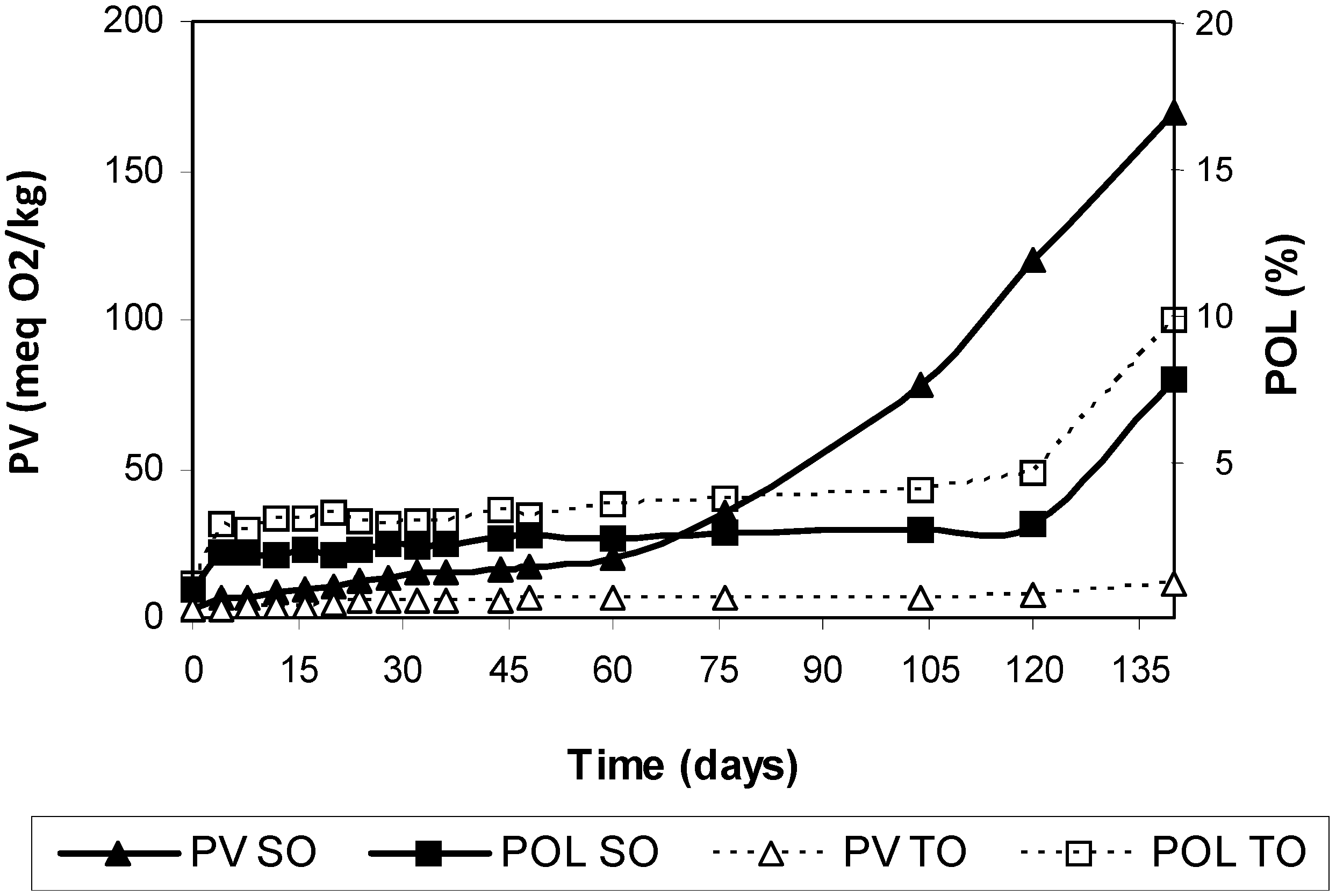
3. Results and Discussion
| Parameter | Safflower Oil | Tonalin Oil |
|---|---|---|
| Fatty acid composition (%) | ||
| 16:0 | 7.2 ± 0.2 | 2.4 ± 0.1 |
| 18:0 | 2.6 ± 0.1 | 2.6 ± 0.1 |
| 18:1 | 13.7 ± 0.5 | 14.2 ± 0.6 |
| 18:2 9c, 12c | 74.7 ± 0.7 | 0.5 ± 0.1 |
| 18:2 9c, 11t (CLA) | 38.2 ± 0.7 | |
| 18:2 10t, 12c (CLA) | 38.6 ± 0.7 | |
| Others | 1.8 ± 0.1 | 3.5 ± 0.2 |
| Tocopherols (mg/kg) | ||
| α | 266 ± 13 | 28 ± 2 |
| γ | 324 ± 17 | |
| δ | 215 ± 13 | |
| Peroxide value (meq O2/kg) | 2.9 ± 0.2 | 2.5 ± 0.3 |
| Oil Stability Index (h) | 5.8 ± 0.4 | 5.7 ± 0.3 |
| Polymers (%) | 1.0 ± 0.3 | 1.1 ± 0.4 |
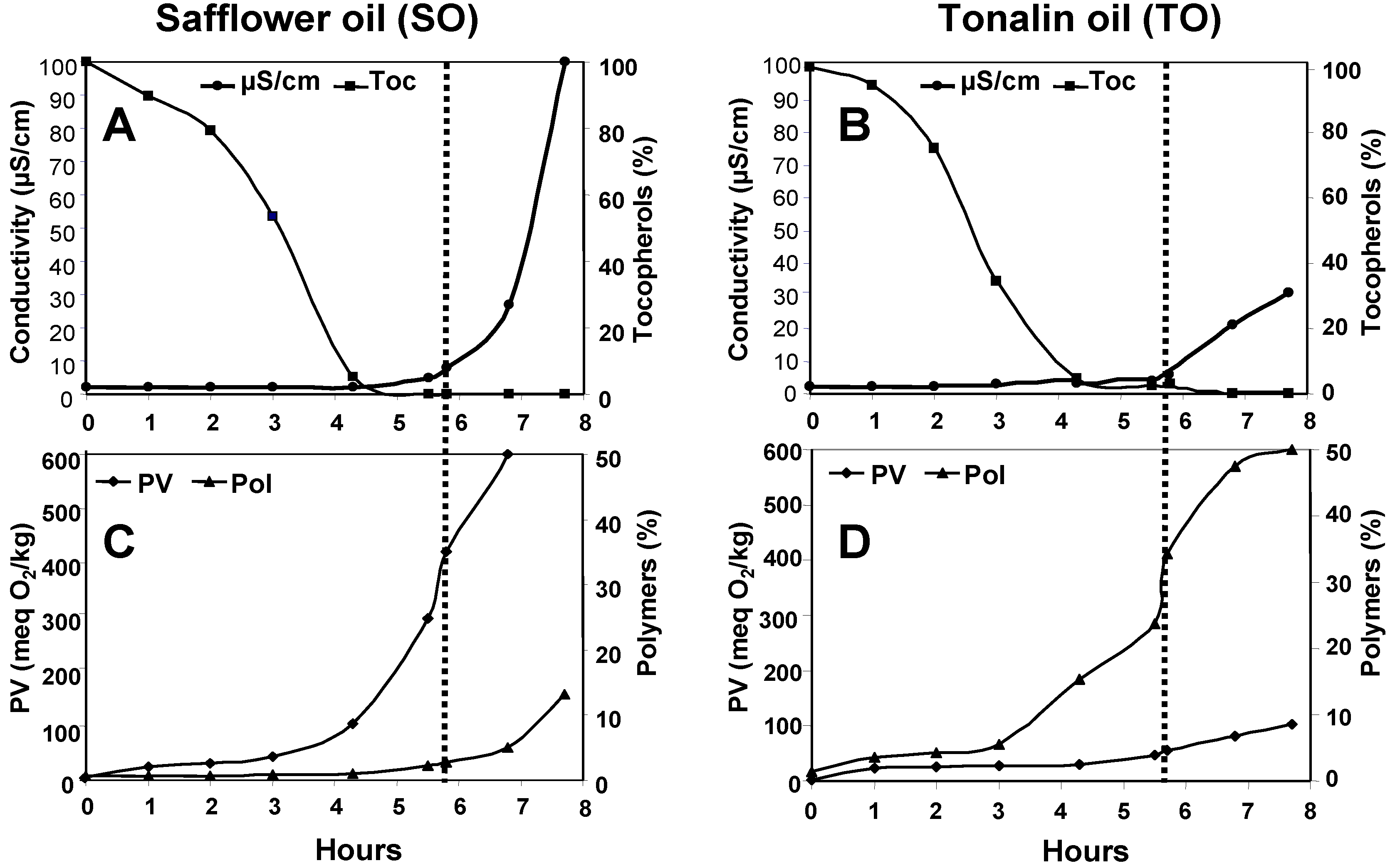
| Days | Dimers (%) | Oligomers (%) | Total Polymers (%) | Peroxide Value (meq O2/kg) | Tocopherols (mg/kg) |
|---|---|---|---|---|---|
| 0 | 1.0 | 0.0 | 1.0 | 2.9 | 266 |
| 4 | 2.0 | 0.2 | 2.2 | 6.9 | 192 |
| 8 | 1.9 | 0.2 | 2.2 | 6.3 | 179 |
| 12 | 1.9 | 0.2 | 2.1 | 8.4 | 174 |
| 16 | 2.1 | 0.2 | 2.3 | 9.9 | 150 |
| 20 | 1.9 | 0.2 | 2.1 | 10.0 | 139 |
| 24 | 2.0 | 0.3 | 2.3 | 12.8 | 139 |
| 28 | 2.1 | 0.4 | 2.5 | 13.3 | 118 |
| 32 | 2.1 | 0.3 | 2.4 | 15.0 | 100 |
| 36 | 2.1 | 0.3 | 2.4 | 15.7 | 80 |
| 44 | 2.3 | 0.4 | 2.7 | 16.2 | 42 |
| 48 | 2.3 | 0.4 | 2.8 | 17.6 | 40 |
| 60 | 2.3 | 0.4 | 2.7 | 20.3 | 33 |
| 76 | 2.4 | 0.4 | 2.8 | 35.3 | 29 |
| 104 | 2.5 | 0.4 | 2.9 | 77.9 | 18 |
| 120 | 2.7 | 0.5 | 3.2 | 120.0 | 11 |
| 140 | 5.9 | 2.1 | 8.0 | 170.0 | 0 |
| Days | Dimers (%) | Oligomers (%) | Total Polymers (%) | Peroxide Value (meq O2/kg) | Tocopherols (mg/kg) |
|---|---|---|---|---|---|
| 0 | 0.0 | 1.1 | 1.1 | 2.5 | 568 |
| 4 | 0.2 | 2.9 | 3.2 | 2.9 | 532 |
| 8 | 0.2 | 2.7 | 2.9 | 3.3 | 525 |
| 12 | 0.2 | 3.1 | 3.4 | 4.0 | 480 |
| 16 | 0.2 | 3.1 | 3.4 | 4.0 | 445 |
| 20 | 0.3 | 3.3 | 3.6 | 4.9 | 446 |
| 24 | 0.2 | 3.0 | 3.3 | 5.3 | 446 |
| 28 | 0.2 | 2.9 | 3.1 | 5.7 | 442 |
| 32 | 0.2 | 3.1 | 3.3 | 5.6 | 429 |
| 36 | 0.2 | 3.0 | 3.2 | 5.9 | 377 |
| 44 | 0.2 | 3.4 | 3.7 | 5.8 | 358 |
| 48 | 0.2 | 3.3 | 3.4 | 6.3 | 357 |
| 60 | 0.2 | 3.6 | 3.8 | 6.5 | 322 |
| 76 | 0.2 | 3.8 | 4.0 | 6.9 | 275 |
| 104 | 0.2 | 4.1 | 4.3 | 7.1 | 180 |
| 120 | 1.6 | 3.3 | 4.9 | 7.8 | 107 |
| 140 | 1.8 | 7.2 | 10.0 | 11.0 | 0 |
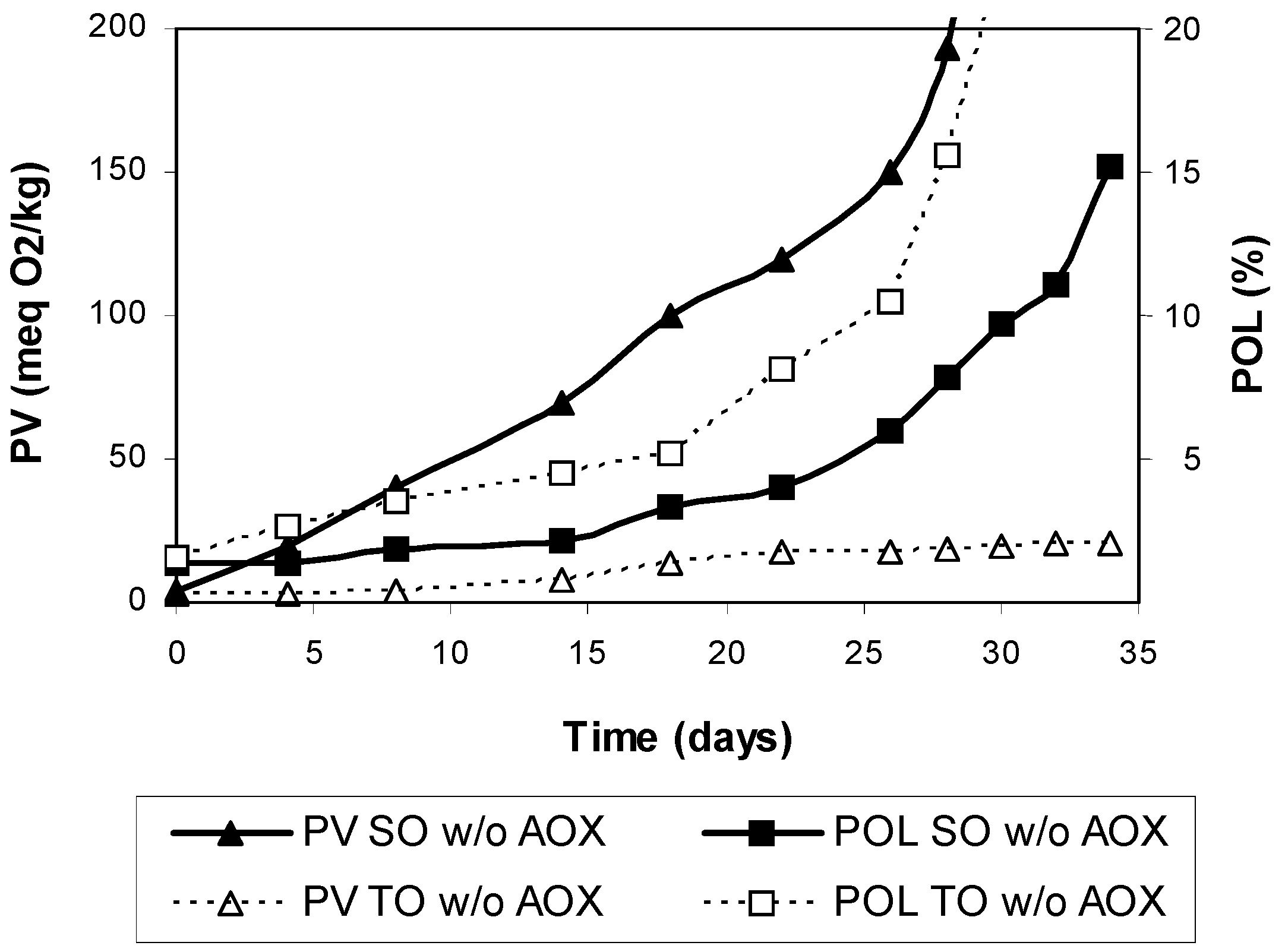
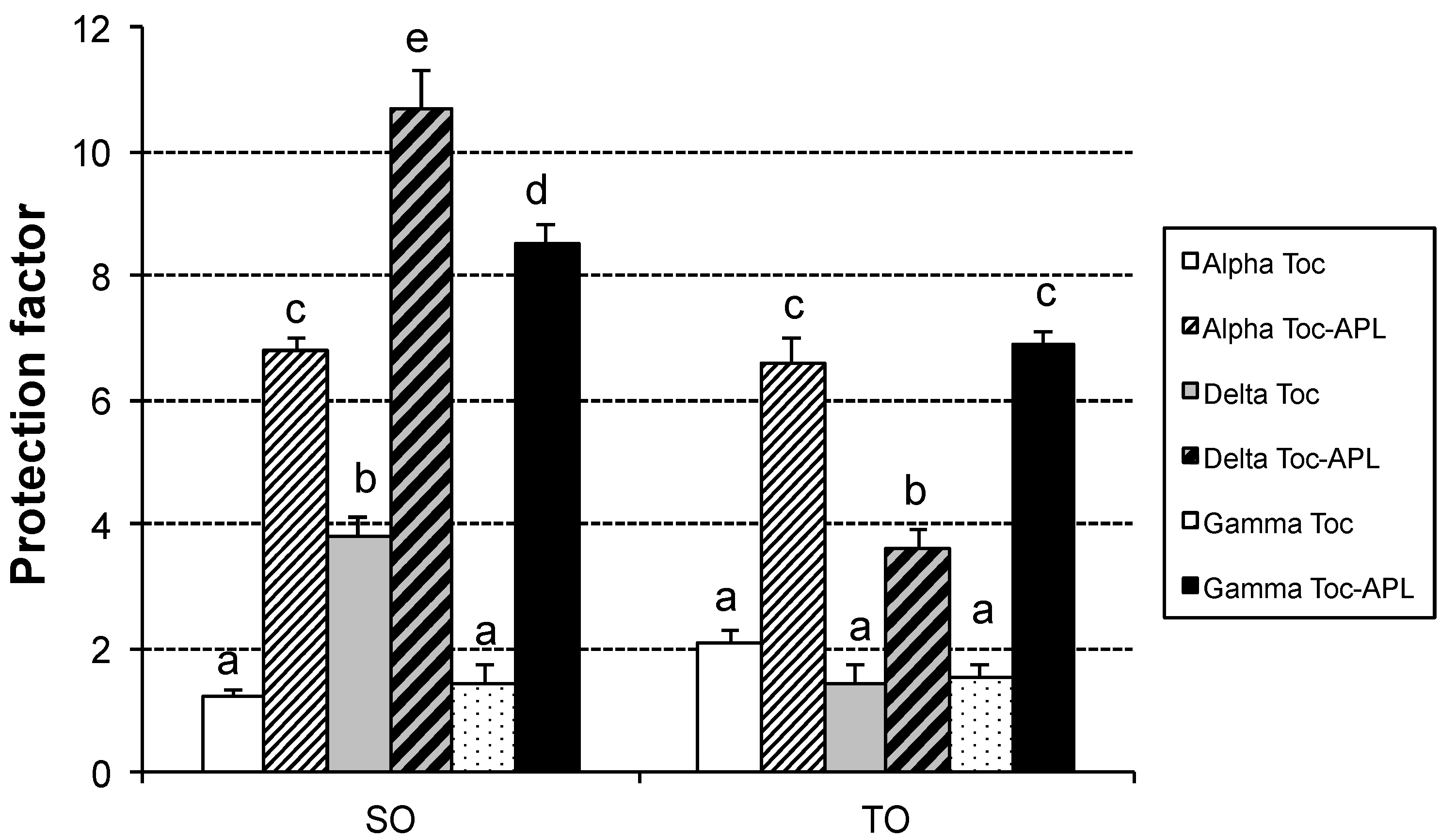
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Pariza, M.W.; Park, Y.; Cook, M.E. The biologically active isomers of conjugated linoleic acid. Prog. Lipid Res. 2001, 40, 283–298. [Google Scholar] [CrossRef]
- Saebo, A. Commercial Synthesis of Conjugated Linoleate. In Advances in CLA Research; AOCS Press: Champaign, IL, USA, 2003; Volume 2, pp. 71–81. [Google Scholar]
- Gaullier, J.M.; Halse, J.; Høye, K.; Kristiansen, K.; Fagertun, H.; Vik, H.; Gudmundsen, O. Supplementation with conjugated linoleic acid for 24 months is well tolerated by and reduces body fat mass in healthy, overweight humans. J. Nutr. 2005, 135, 778–784. [Google Scholar] [PubMed]
- Park, Y.; Pariza, M.W. Mechanisms of body fat modulation by conjugated linoleic acid (CLA). Food Res. Int. 2007, 40, 311–323. [Google Scholar] [CrossRef]
- García-Martínez, M.C.; Márquez-Ruiz, G. Lipid oxidation in functional dairy products. Curr. Nutr. Food Sci. 2009, 5, 209–216. [Google Scholar] [CrossRef]
- Dobarganes, M.C.; Márquez-Ruiz, G. Oxidized fats in foods. Curr. Opin. Clin. Nutr. Metab. Care 2003, 6, 157–163. [Google Scholar] [CrossRef]
- Brimberg, U.I.; Kamal-Eldin, A. On the kinetics of the autoxidation of fats: Substrates with conjugated double bonds. Eur. J. Lipid Sci. Technol. 2003, 105, 17–22. [Google Scholar] [CrossRef]
- Eulitz, K.; Yurawecz, M.P.; Ku, Y. The Oxidation of Conjugated Linoleic Acid. In Advances in CLA Research; AOCS Press: Champaign, IL, USA, 1999; Volume 1, pp. 55–63. [Google Scholar]
- Hämäläinen, T.I.; Sundberg, S.; Mäkinen, M.; Kaltia, S.; Hase, T.; Hopia, A. Hydroperoxide formation during autoxidation of conjugated linoleic acid methyl ester. Eur. J. Lipid Sci.Technol. 2001, 103, 588–593. [Google Scholar] [CrossRef]
- Hämäläinen, T.I.; Sundberg, S.; Hase, T.; Hopia, A. Stereochemistry of the hydroperoxides formed during autoxidation of CLA methyl ester in the presence of α-tocopherol. Lipids 2002, 37, 533–540. [Google Scholar] [CrossRef]
- Luna, P.; de la Fuente, M.A.; Salvador, D.; Márquez-Ruiz, G. Differences in oxidation kinetics between conjugated and non-conjugated methyl linoleate. Lipids 2007, 42, 1085–1092. [Google Scholar] [CrossRef]
- Yurawecz, M.P.; Delmonte, P.; Vogel, T.; Kramer, J.K.G. Oxidation of Conjugated Linoleic Acid: Initiators and Simultaneous Reactions: Theory and Practice. In Advances in CLA Research; AOCS Press: Champaign, IL, USA, 2003; Volume 2, pp. 56–70. [Google Scholar]
- Yurawecz, M.P.; Sehat, N.; Mossoba, M.M.; Roach, J.A.G.; Ku, Y. Oxidation Products of Conjugated Linoleic Acid and Furan Fatty Acids. In New Techniques and Applications in Lipid Analysis; AOCS Press: Champaign, IL, USA, 2007; pp. 183–215. [Google Scholar]
- García-Martínez, M.C.; Márquez-Ruiz, G.; Fontecha, J.; Gordon, M.H. Volatile oxidation compounds in a conjugated linoleic acid-rich oil. Food Chem. 2009, 113, 926–931. [Google Scholar] [CrossRef]
- Suzuki, R.; Abe, M.; Miyashita, K. Comparative study of the autoxidation of TAG containing conjugated and nonconjugated C18 PUFA. J. Am. Oil Chem. Soc. 2004, 81, 563–569. [Google Scholar] [CrossRef]
- Tsuzuki, T.; Igarashi, M.; Iwata, T.; Yamauchi-Sato, Y.; Yamamoto, T.; Ogita, K.; Suzuki, T.; Miyazawa, T. Oxidation rate of conjugated linoleic acid and conjugated linolenic acid is slowed by triacylglycerol esterification and α-tocopherol. Lipids 2004, 39, 475–480. [Google Scholar] [CrossRef]
- Ko, S.-N.; Kim, C.-J.; Kim, C.-T.; Kim, Y.; Kim, I.-H. Effects of tocopherols and tocotrienols on the inhibition of autoxidation of conjugated linoleic acid. Eur. J. Lipid Technol. 2010, 112, 496–501. [Google Scholar] [CrossRef]
- Lee, J.; Lee, S.-M.; Kim, I.-H.; Jeong, J.-H.; Rhee, C.; Lee, K.-W. Oxidative instability of CLA concentrate and its avoidance with antioxidants. J. Am. Oil Chem. Soc. 2003, 80, 807–810. [Google Scholar] [CrossRef]
- Yoshida, H.; Kondo, I.; Kajimoto, G. Participation of free fatty acids in the oxidation of purified soybean oil during microwave-heating. J. Am. Oil Chem. Soc. 1992, 69, 1136–1140. [Google Scholar] [CrossRef]
- AOCS. Official Methods and Recommended Practices of the American Oil Chemists’ Society, 4th ed.; AOCS Press: Champaign, IL, USA, 1996. [Google Scholar]
- IUPAC. Standard Methods for the Analysis of Oils, Fats and Derivatives, 7th ed.; International Union of Pure and Applied Chemistry, Blackwell Scientific: Oxford, UK, 1992. [Google Scholar]
- Márquez-Ruiz, G.; Dobarganes, M.C. High-Performance Size-Exclusion Chromatography for Lipid Analysis in Organic Media. In Lipid Analysis and Lipidomics. New Techniques and Applications; AOCS Press: Champaign, IL, USA, 2007; pp. 205–238. [Google Scholar]
- Márquez-Ruiz, G.; Martín-Polvillo, M.; Velasco, J.; Dobarganes, M.C. Formation of oxidation compounds in sunflower and olive oils under oxidative stability index conditions. Eur. J. Lipid Sci. Technol. 2008, 110, 465–471. [Google Scholar] [CrossRef]
- Márquez-Ruiz, G.; Martín-Polvillo, M.; Dobarganes, M.C. Quantitation of oxidized triglyceride monomers and dimers as an useful measurement for early and advanced stages of oxidation. Grasas Aceites 1996, 47, 48–53. [Google Scholar] [CrossRef]
- Márquez-Ruiz, G.; Martín-Polvillo, M.; Dobarganes, M.C. Effect of temperature and addition of α-tocopherol on the oxidation of trilinolein model systems. Lipids 2003, 38, 233–240. [Google Scholar] [CrossRef]
- Martín-Polvillo, M.; Márquez-Ruiz, G.; Dobarganes, M.C. Oxidative stability of sucflower oils differeing in unsaturation degree during long term storage at room temperature. J. Am. Oil Chem. Soc. 2004, 81, 577–583. [Google Scholar] [CrossRef]
- Frankel, E.N. Lipid Oxidation; The Oily Press: Dundee, UK, 2005. [Google Scholar]
- Miller, A.B.; Claxton, E. Atmospheric oxidation of methyl and glycol esters of β-eleostearic acid. Ind. Eng. Chem. 1928, 20, 43–48. [Google Scholar] [CrossRef]
- Allen, R.R.; Jackson, A.; Kummerow, F.A. Factors which affect the stability of highly unsaturated fatty acids. Difference in the oxidation of conjugated and nonconjugated linoleic acid. J. Am. Oil Chem. Soc. 1949, 26, 395–399. [Google Scholar] [CrossRef]
- Jackson, A.H.; Kummerow, F.A. Factors which affect the stability of highly unsaturated fatty acids. II. The autoxidation of linoleic and alkali conjugated acid in the presence of metallic naphthenates. J. Am. Oil Chem. Soc. 1949, 26, 460–465. [Google Scholar] [CrossRef]
- Oyman, Z.O.; Ming, W.; van der Linde, R. Oxidation of drying oils containing non-conjugated and conjugated double bonds catalyzed by a cobalt catalyst. Prog. Org. Coat. 2005, 54, 198–204. [Google Scholar] [CrossRef]
- Fuster, M.D.; Lampi, A.-M.; Hopia, A.; Kamal-Eldin, A. Effects of α- and γ-tocopherols on the autoxidation of purified sunflower triacylglycerols. Lipids 1998, 33, 715–722. [Google Scholar] [CrossRef]
- Lampi, A.-M.; Kamal-Eldin, A. Effect of α- and γ-tocopherols on thermal polymerization of purified high-oleic sunflower triacylglycerols. J. Am. Oil Chem. Soc. 1998, 75, 1699–1703. [Google Scholar] [CrossRef]
- Réblová, Z. The effect of temperature on the antioxidant activity of tocopherols. Eur. J. Lipid Sci. Technol. 2006, 108, 858–863. [Google Scholar] [CrossRef]
- Braunrath, R.; Isnardy, B.; Solar, S.; Elmadfa, I. Influence of α-, γ- and δ-tocopherol on the radiation induced formation of peroxides in rapeseed oil triacylglycerols. Food Chem. 2009, 117, 349–351. [Google Scholar] [CrossRef]
- Kamal-Eldin, A.; Appleqvist, L.-A. The chemistry and antioxidant properties of tocopherols and tocotrienols. Lipids 1996, 31, 671–701. [Google Scholar] [CrossRef]
- Bartee, S.D.; Kim, H.J.; Min, D.B. Effects of antioxidants on the oxidative stability of oils containing arachidonic, docosaentaenoic and docosahexaenoic acids. J. Am. Oil Chem. Soc. 2007, 84, 363–368. [Google Scholar] [CrossRef]
- Drusch, S.; Grob, N.; Schwarz, K. Efficient stabilization of bulk fish oil rich in long-chain polyunsaturated fatty acids. Eur. J. Lipid Sci. Technol. 2008, 110, 351–359. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Márquez-Ruiz, G.; García-Martínez, M.D.C.; Holgado, F.; Velasco, J. Effectiveness of α-, γ- and δ-Tocopherol in a CLA-Rich Oil. Antioxidants 2014, 3, 176-188. https://doi.org/10.3390/antiox3010176
Márquez-Ruiz G, García-Martínez MDC, Holgado F, Velasco J. Effectiveness of α-, γ- and δ-Tocopherol in a CLA-Rich Oil. Antioxidants. 2014; 3(1):176-188. https://doi.org/10.3390/antiox3010176
Chicago/Turabian StyleMárquez-Ruiz, Gloria, María Del Carmen García-Martínez, Francisca Holgado, and Joaquín Velasco. 2014. "Effectiveness of α-, γ- and δ-Tocopherol in a CLA-Rich Oil" Antioxidants 3, no. 1: 176-188. https://doi.org/10.3390/antiox3010176




