Pitting Corrosion of Steel Induced by Al2O3 Inclusions
Abstract
:1. Introduction
2. Experimental Procedures
2.1. Preparation and Processing of Experimental Materials
2.2. Experimental Schemes
3. Results and Discussion
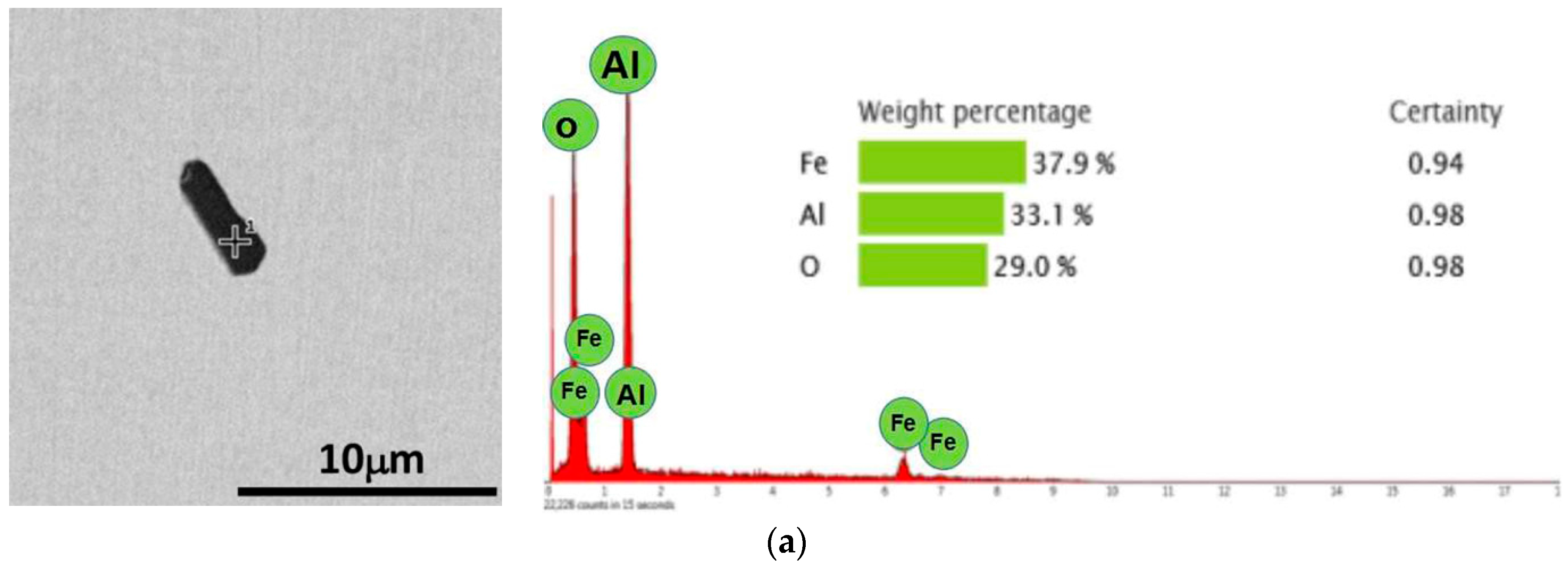
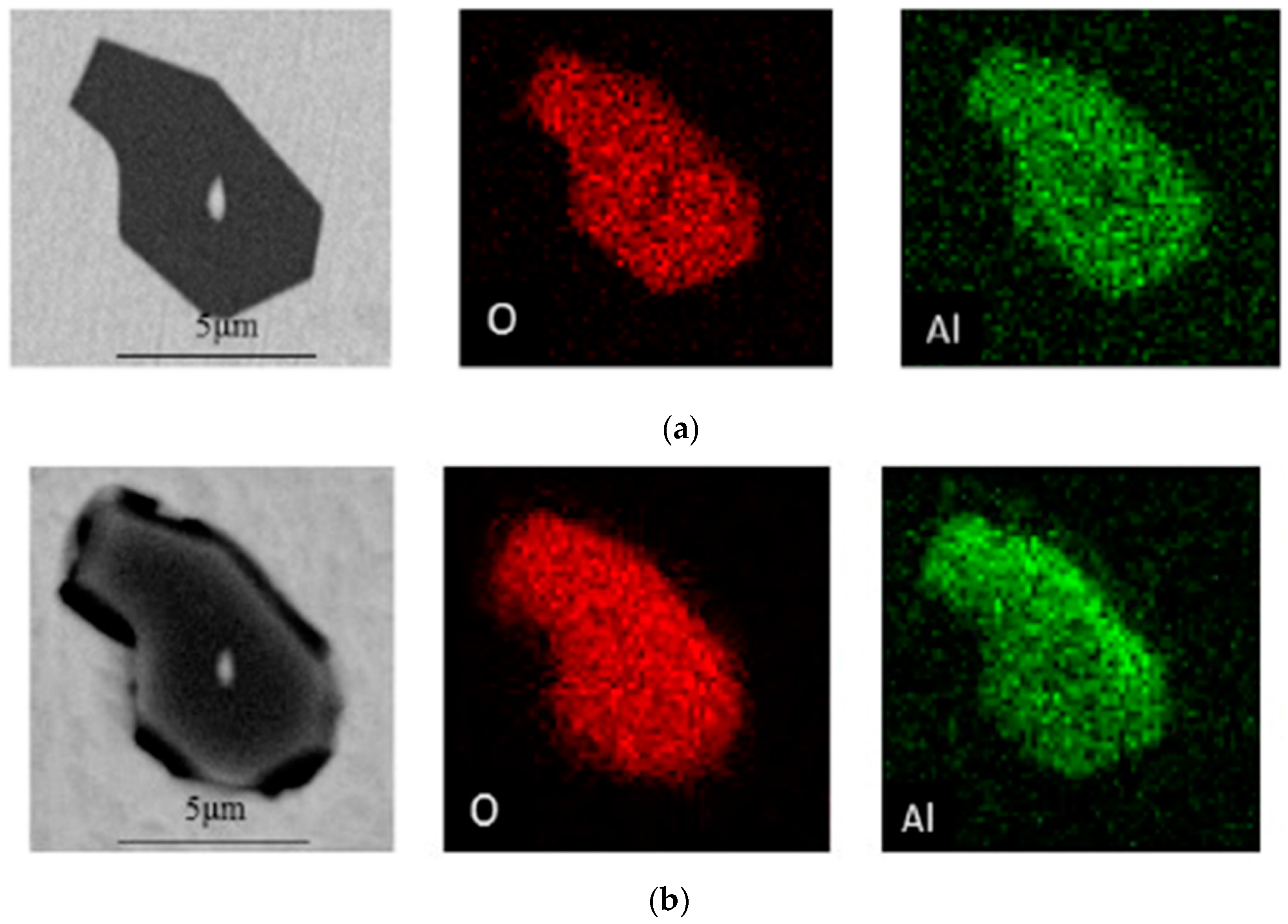
3.1. Observation and Control of Al2O3 Inclusions
3.2. Study on Corrosion Induced by Al2O3 Inclusions
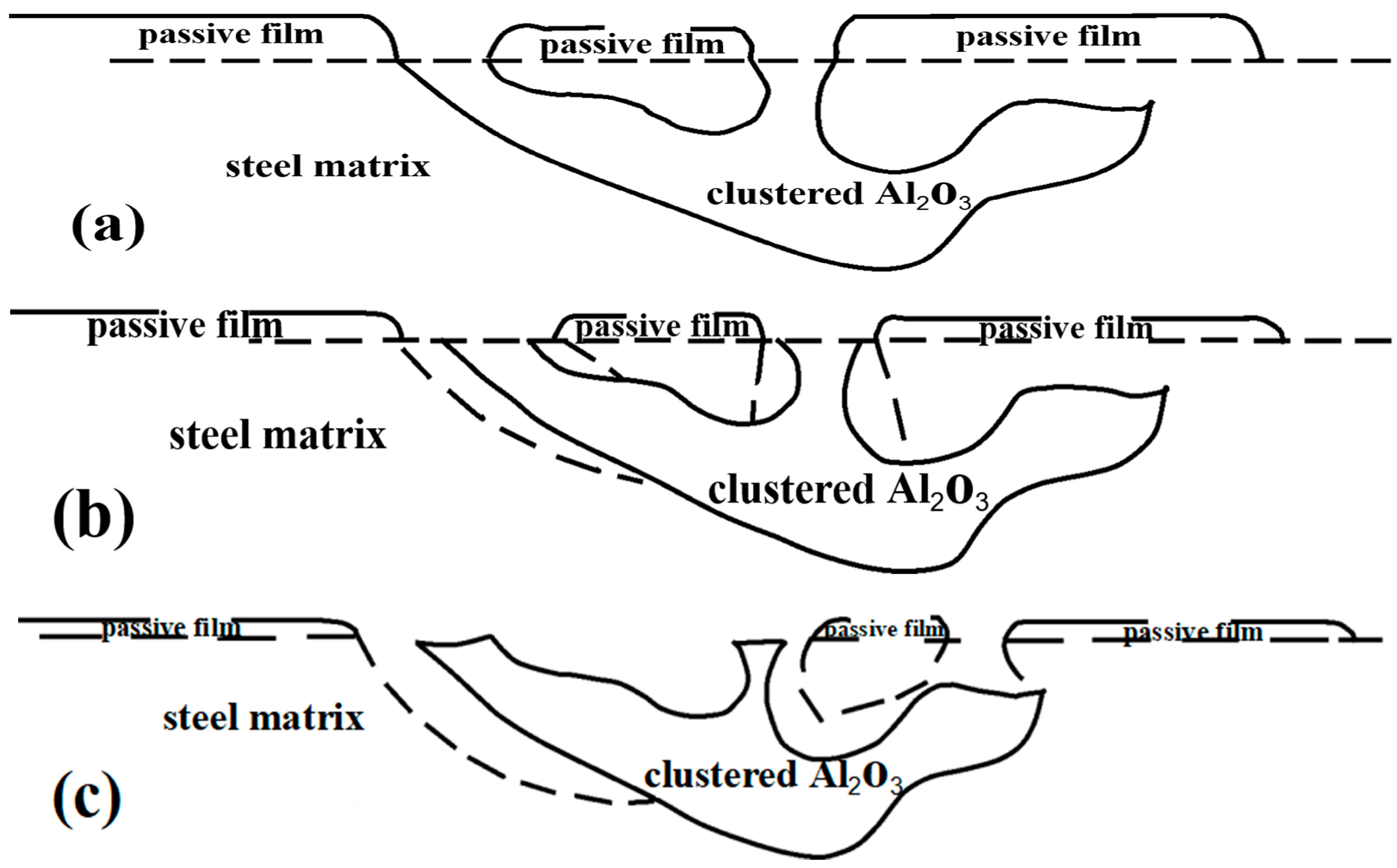
3.2.1. Effect of Clustered Al2O3 Inclusions on Corrosion
3.2.2. Effect of Lone Distributed Al2O3 Inclusions on Corrosion
3.2.3. Analysis of Corrosion Induced Mechanism of Al2O3 Inclusions
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ke, W. Progress in public inquiry concerning corrosion in Chinese industrial and natural environments. Corros. Prot. 2004, 25, 1–8. [Google Scholar]
- Frankel, G.S. Pitting corrosion of metals. J. Electrochem. Soc. 1998, 145, 2186. [Google Scholar] [CrossRef]
- Song, Y.W.; Shan, D.Y.; Han, E.H. Pitting corrosion of a rare earth Mg alloy GW93. J. Mater. Sci. Technol. 2017, 1, 1–7. [Google Scholar] [CrossRef]
- Williams, D.E.; Kilburn, M.R.; Cliff, J.; Waterhouse, G.I.N. Composition changes around sulfide inclusions in stainless steels, and implications for the initiation of pitting corrosion. Corros. Sci. 2010, 52, 3702–3716. [Google Scholar] [CrossRef]
- Zhang, H.; Chen, X.Q.; Chang, W.S. Effect of metallurgy factors on susceptibility to pitting initiation in steels. J. Chin. Soc. Corros. Prot. 2009, 29, 127–131. [Google Scholar]
- Caines, S.; Khan, F.; Shirokoff, J. Analysis of pitting corrosion on steel under insulation in marine environments. J. Loss Prev. Proc. 2013, 26, 1466–1483. [Google Scholar] [CrossRef]
- Uhlig, H.H. The cost of corrosion to the United States. Corrosion 1950, 6, 29–33. [Google Scholar] [CrossRef]
- Wang, J.M.; Chen, X.Q.; Li, G.M. Susceptibility of two types of low-alloy hull steels to pit initiation. J. Univ. Sci. Technol. B 2004, 11, 555–560. [Google Scholar]
- Park, I.-J.; Lee, S.-M.; Kang, M.; Lee, S.; Lee, Y.-K. Pitting corrosion behavior in advanced high strength steels. J. Alloys Compd. 2015, 619, 205–210. [Google Scholar] [CrossRef]
- Punckt, C.; Bölscher, M.; Rotermund, H.H.; Mikhailov, A.S.; Organ, L.; Budiansky, N.; Scully, J.R.; Hudson, J.L. Sudden onset of pitting corrosion on stainless steel as a critical phenomenon. Science 2004, 305, 1133–1136. [Google Scholar] [CrossRef] [PubMed]
- Szkzarska-Smialowska, Z. The effect of inclusions on the susceptibility of steels to pitting, stress corrosion cracking and hydrogen embrittlement. Mater. Corros. 1981, 32, 478–485. [Google Scholar] [CrossRef]
- Ha, H.Y.; Park, C.J.; Kwon, H.S. Effects of misch metal on the formation of non-metallic inclusions and the associated resistance to pitting corrosion in 25% Cr duplex stainless steels. Scr. Mater. 2006, 55, 991–994. [Google Scholar] [CrossRef]
- Schmuki, P.; Hildebrand, H.; Friedrich, A.; Virtanen, S. The composition of the boundary region of MnS inclusions in stainless steel and its relevance in triggering pitting corrosion. Corros. Sci. 2005, 47, 1239–1250. [Google Scholar] [CrossRef]
- Chiba, A.; Muto, I.; Sugawara, Y.; Hara, N. A microelectrochemical system for in situ high-resolution optical microscopy: morphological characteristics of pitting at MnS inclusion in stainless steel. J. Electrochem. Soc. 2013, 26, 1466–1483. [Google Scholar] [CrossRef]
- Zheng, S.Q.; Li, C.Y.; Qi, Y.M.; Chen, L.Q.; Chen, C.F. Mechanism of (Mg, Al, Ca)-oxide inclusion-induced pitting corrosion in 316L stainless steel exposed to sulfur environments containing chloride ion. Corros. Sci. 2013, 67, 20–31. [Google Scholar] [CrossRef]
- Muto, I.; Ito, D.; Hara, N. Microelectrochemical investigation on pit initiation at sulfide and oxide inclusions in type 304 stainless steel. J. Electrochem. Soc. 2009, 156, 55–61. [Google Scholar] [CrossRef]
- Choi, J.Y.; Kim, S.K.; Kang, Y.B.; Lee, H.G. Compositional evolution of oxide inclusions in austenitic stainless steel during continuous casting. Steel Res. Int. 2014, 86, 284–292. [Google Scholar] [CrossRef]
- Liu, C.S.; Yang, S.F.; Li, J.S.; Ni, H.W.; Zhang, X.L. The influence of FeO on the reaction between Fe–Al–Ca alloy and Al2O3–CaO–FeO oxide during heat treatment at 1473 K. Metals 2017, 7, 129. [Google Scholar] [CrossRef]
- Neerav, V.; Petrus, P.; Richard, F.; Michael, P.; Minna, L.; Scott, S. Transient inclusion evolution during modification of alumina inclusions by calcium in liquid steel: Part I. Background, experimental techniques and analysis methods. Metall. Mater. Trans. B 2011, 42, 711–719. [Google Scholar]
- Minna, L.; Lauri, H. Transformation of alumina inclusions by calcium treatment. Metall. Mater. Trans. B 2010, 41, 359–366. [Google Scholar]
- Yang, S.F.; Li, J.S.; Wang, Z.F.; Li, J.; Lin, L. Modification of MgO·Al2O3 spinel inclusions in Al-killed steel by Ca-treatment. Int. J. Min. Met. Mater. 2011, 18, 18–23. [Google Scholar] [CrossRef]
- Eklund, G. Initiation of pitting at sulfide inclusions in stainless steel. J. Electrochem. Soc. 1974, 121, 467–473. [Google Scholar] [CrossRef]
- JScully, R.; Budiansky, N.D.; Tiwary, Y.; Mikhailov, A.S.; Hudson, J.L. An alternate explanation for the abrupt current increase at the pitting potential. Corros. Sci. 2008, 50, 316–324. [Google Scholar] [CrossRef]
- Ryan, M.P.; Williams, D.E.; Chater, R.J.; Hutton, B.M.; McPhail, D.S. Why stainless steel corrodes. Nature 2002, 415, 770–774. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.M.; Zuo, Y.; Zhao, H. The current fluctuations and accumulated pitting damage of mild steel in NaNO2-NaCl solution. Appl. Surf. Sci. 2005, 243, 82–88. [Google Scholar] [CrossRef]
- Xu, C.C.; Zhang, X.B.; Li, Z.Q.; Liu, Y.P. Effect of cathodic polarization on chemical and electrochemical states inside occluded cells of localized corrosion of A3 steel. Corros. Sci. Prot. Technol. 2000, 12, 260–263. [Google Scholar]










| C | Si | Mn | P | S | Al | Ni | Cr | Cu | Ti |
|---|---|---|---|---|---|---|---|---|---|
| 0.003 | 0.03 | 0.05 | 0.010 | 0.006 | 0.03 | 0.03 | 0.02 | 0.03 | 0.02 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Q.; Yang, S.; Zhao, M.; Zhu, L.; Li, J. Pitting Corrosion of Steel Induced by Al2O3 Inclusions. Metals 2017, 7, 347. https://doi.org/10.3390/met7090347
Liu Q, Yang S, Zhao M, Zhu L, Li J. Pitting Corrosion of Steel Induced by Al2O3 Inclusions. Metals. 2017; 7(9):347. https://doi.org/10.3390/met7090347
Chicago/Turabian StyleLiu, Qing, Shufeng Yang, Mengjing Zhao, Libin Zhu, and Jingshe Li. 2017. "Pitting Corrosion of Steel Induced by Al2O3 Inclusions" Metals 7, no. 9: 347. https://doi.org/10.3390/met7090347




