A Critical Review of Mg-Based Hydrogen Storage Materials Processed by Equal Channel Angular Pressing
Abstract
:1. Introduction
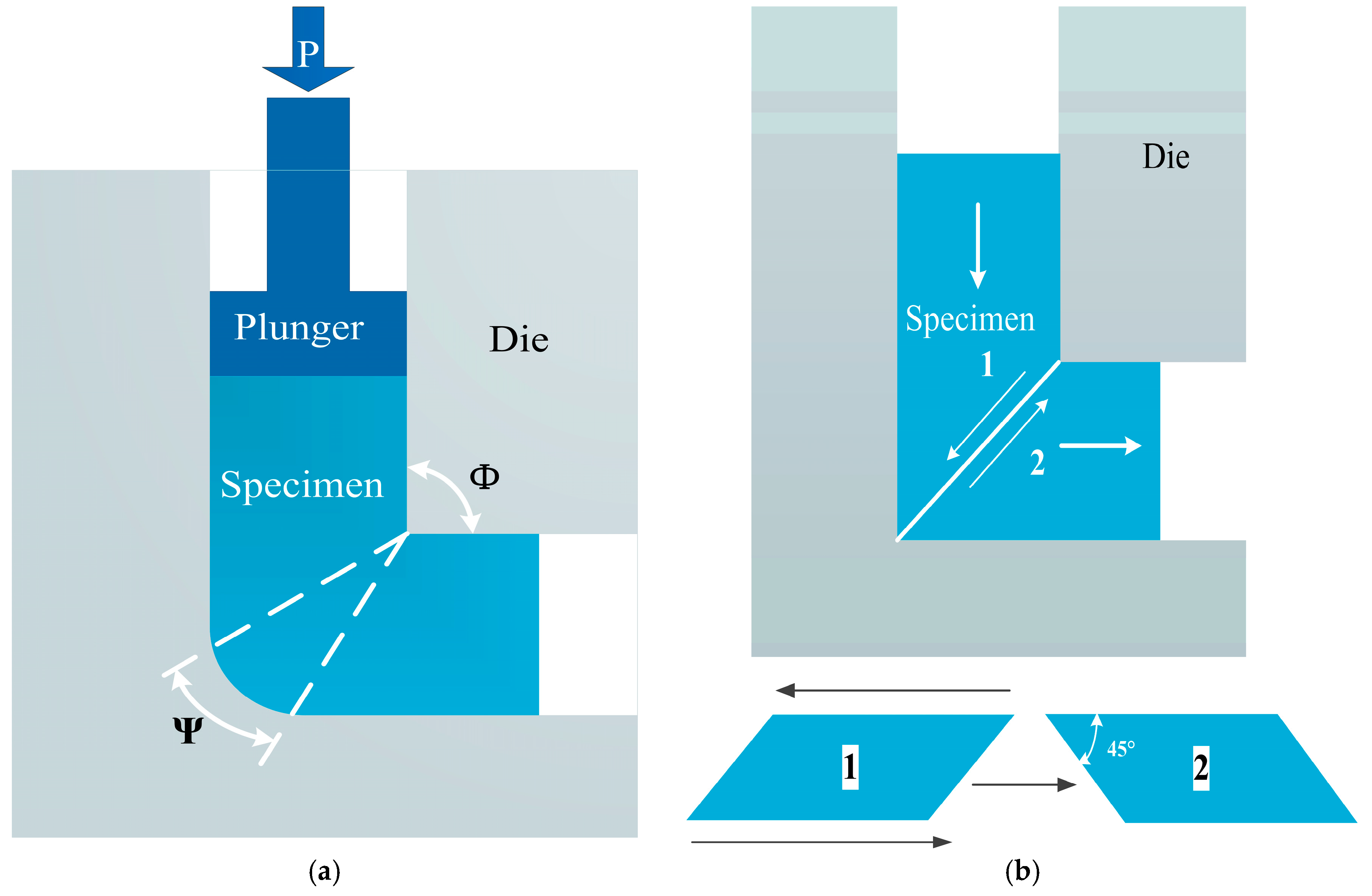
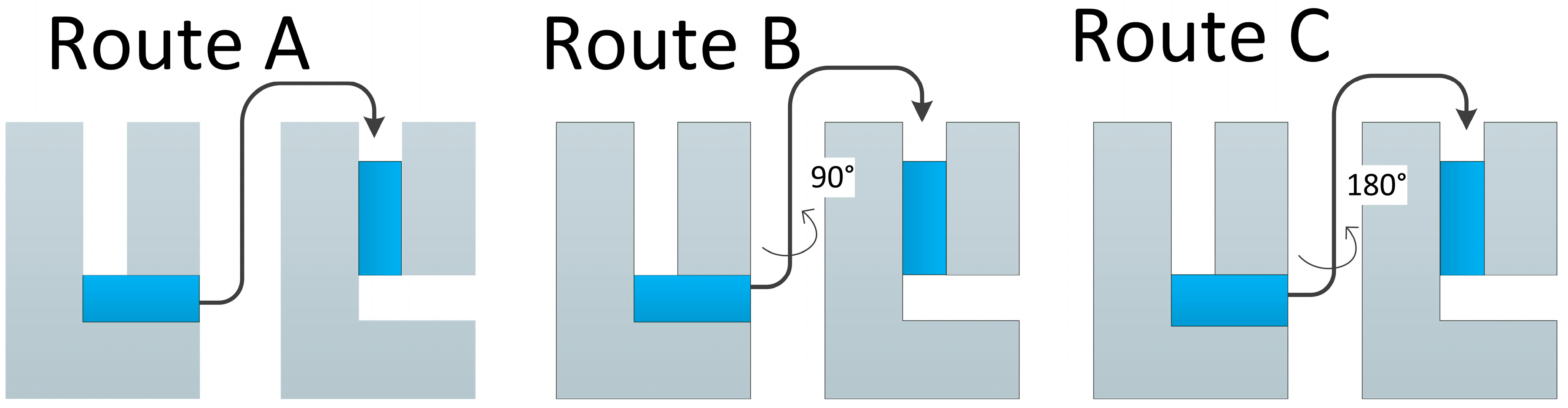
2. ECAP Technology
3. Effect of ECAP on the Hydrogen Storage Behavior of Mg-Based Metal Hydrides
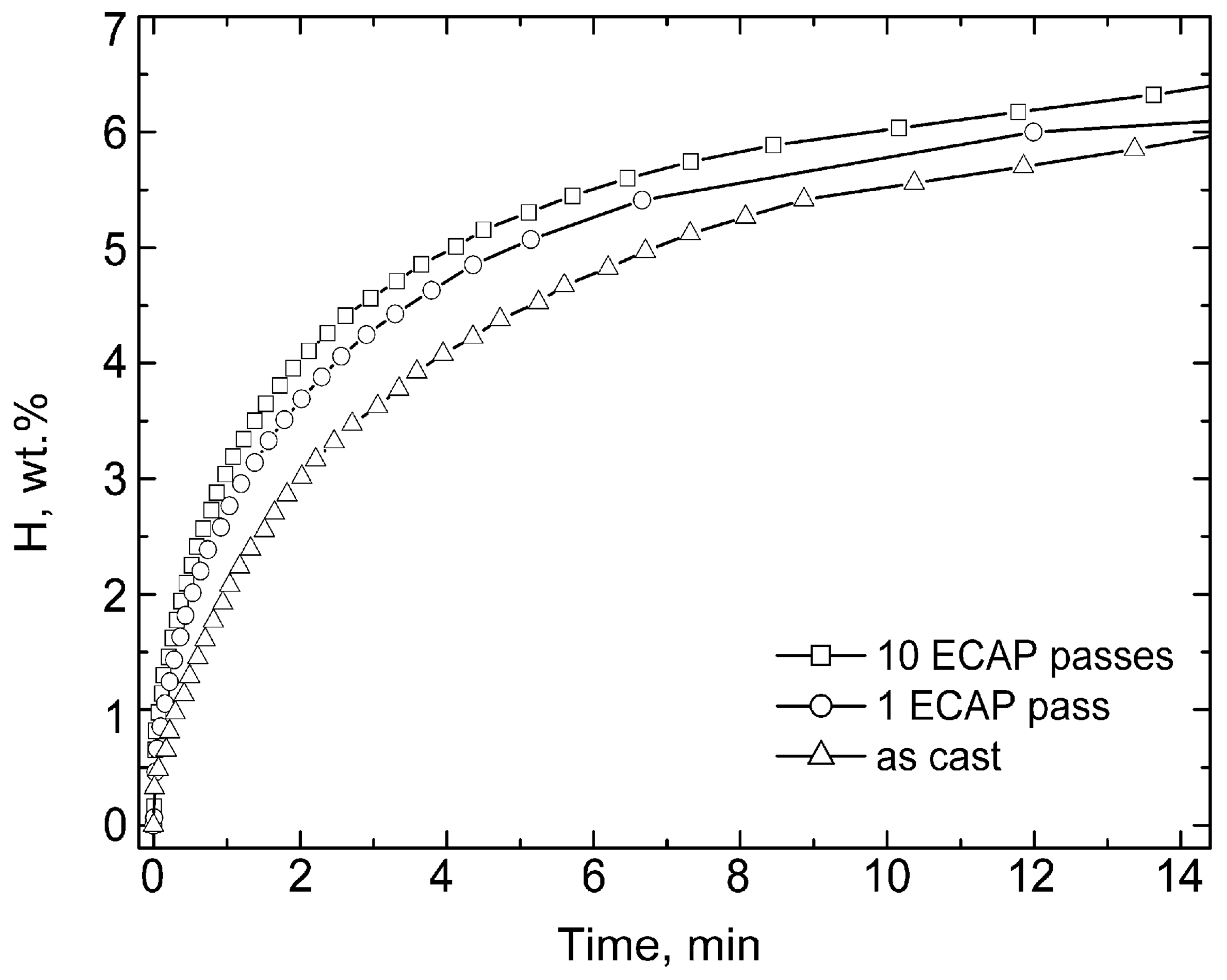
3.1. Dynamics
3.2. Capacity
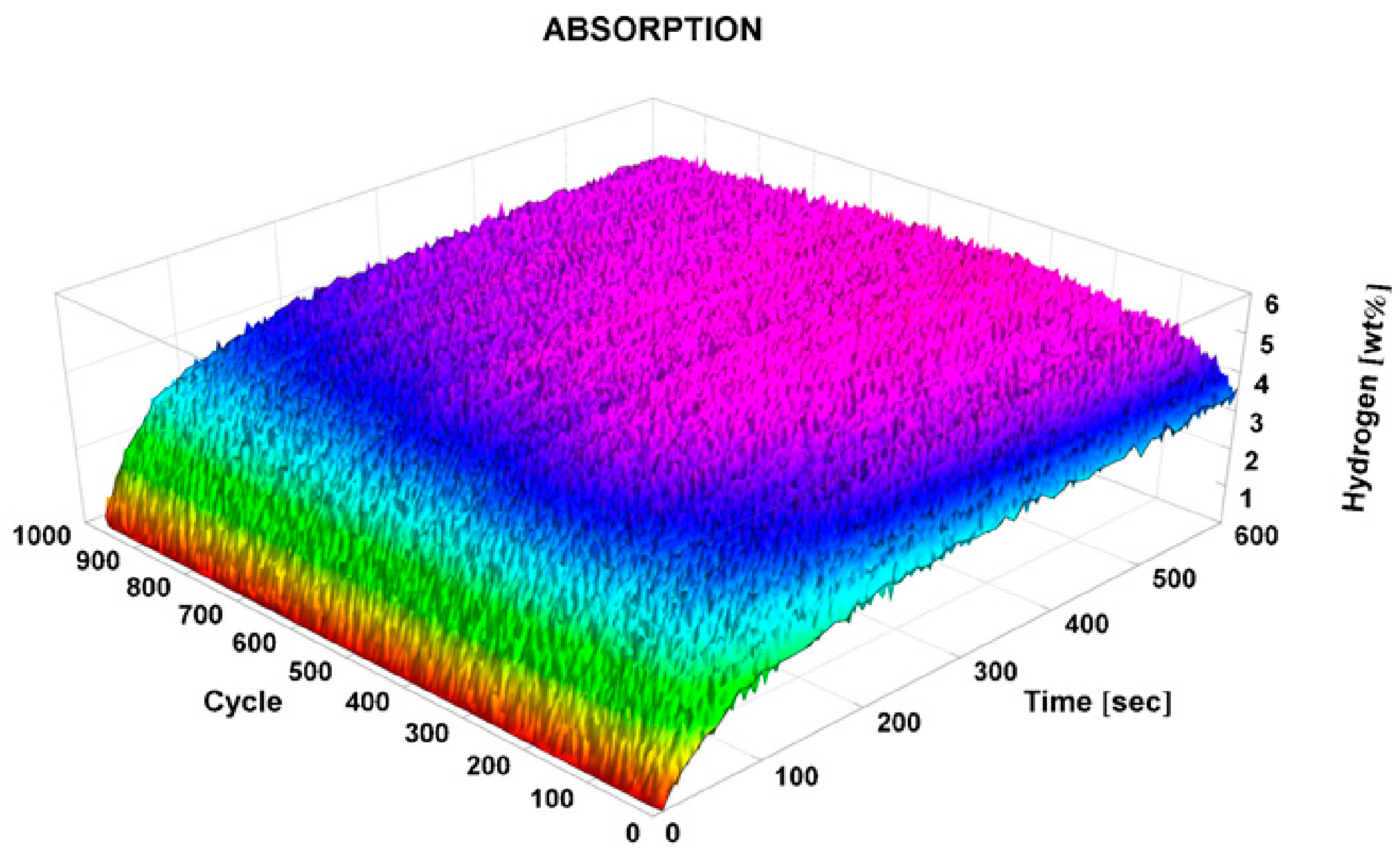
3.3. Cycling Stability
4. Analysis of the Factors Affecting Hydrogen Storage Properties
4.1. Grain Size and Crystallographic Defects
4.2. Catalysts
4.3. Grain Boundaries
4.4. Textures
5. Conclusions and Prospects
- Mechanism study. The diffusion path for H atoms from the surface to the bulk Mg alloys should be precisely investigated from the atomic scale. Special additives can be added into the bulk to systematically explore the interactions between additive elements and MgH2. Advanced theoretical calculations could be used in the research of the MgH2 hydrogenation/de-hydrogenation process.
- Microstructure optimization. The present microstructures of UFG or nanostructured Mg alloys need to be optimized on the basis of severe plastic deformation, in order to maximize the positive role for microstructure coordination acting on the hydrogen storage behavior of Mg hydrogen applications, as well as to explore the appropriate microstructures with the maximum hydrogen capacity, kinetic properties, and the longest cycling stability.
- Electrochemical hydrogen storage property study. Except for the gaseous hydrogen storage condition, UFG Mg alloys are also promising candidates for cathode materials; thus, more attention should be paid to the microstructure control introduced by ECAP to improve the electrochemical hydrogen storage property of UFG Mg alloys by means of microstructure coordination.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Rusman, N.A.A.; Dahari, M. A review on the current progress of metal hydrides material for solid-state hydrogen storage applications. Int. J. Hydrogen Energy 2016, 41, 12108–12126. [Google Scholar] [CrossRef]
- International Energy Agency. World Energy Outlook, Executive Summary; International Energy Agency: Paris, France, 2010. [Google Scholar]
- Skripnyuk, V.M.; Rabkin, E.; Estrin, Y.; Lapovok, R. Improving hydrogen storage properties of magnesium based alloys by equal channel angular pressing. Int. J. Hydrogen Energy 2009, 34, 6320–6324. [Google Scholar] [CrossRef]
- Wu, D.; Ouyang, L.; Wu, C.; Gu, Q.; Wang, H.; Liu, J.; Zhu, M. A Phase transition and hydrogen storage properties of Mg17Ba2 compound. J. Alloys Compd. 2017, 690, 519–522. [Google Scholar] [CrossRef]
- Sakintuna, B.; Lamari-Darkrim, F.; Hirscher, M. Metal hydride materials for solid hydrogen storage: A review. Int. J. Hydrogen Energy 2007, 32, 1121–1140. [Google Scholar] [CrossRef]
- Ellinger, F.H.; Holley, C.E.; McInteer, B.B.; Pavone, D. The preparation and some properties of magnesium hydride. J. Am. Chem. Soc. 1955, 77, 2647–2648. [Google Scholar] [CrossRef]
- Crivello, J.C.; Denys, R.V.; Dornheim, M.; Felderhoff, M.; Grant, D.M.; Huot, J.; Jensen, T.R.; De Jongh, P.; Latroche, M.; Walker, G.S.; et al. Mg-based compounds for hydrogen and energy storage. Appl. Phys. A 2016, 122, 85. [Google Scholar] [CrossRef]
- Kim, W.J.; Sa, Y.K. Micro-extrusion of ECAP processed magnesium alloy for production of high strength magnesium micro-gears. Scr. Mater. 2006, 54, 1391–1395. [Google Scholar] [CrossRef]
- Mostaed, E.; Vedani, M.; Hashempour, M.; Bestetti, M. Influence of ECAP process on mechanical and corrosion properties of pure Mg and ZK60 magnesium alloy for biodegradable stent applications. Biomatter 2014, 4, e28283. [Google Scholar] [CrossRef] [PubMed]
- Zeng, K.; Klassen, T.; Oelerich, W.; Bormann, R. Critical assessment and thermodynamic modeling of the Mg–H system. Int. J. Hydrogen Energy 1999, 24, 989–1004. [Google Scholar] [CrossRef]
- Skripnyuk, V.M.; Rabkin, E.; Estrin, Y.; Lapovok, R. The effect of ball milling and equal channel angular pressing on the hydrogen absorption/desorption properties of Mg–4.95 wt % Zn–0.71 wt % Zr (ZK60) alloy. Acta Mater. 2004, 52, 405–414. [Google Scholar] [CrossRef]
- Segal, V.M. Materials processing by simple shear. Mater. Sci. Eng. A 1995, 197, 157–164. [Google Scholar] [CrossRef]
- Valiev, R.Z.; Langdon, T.G. Principles of equal-channel angular pressing as a processing tool for grain refinement. Prog. Mater. Sci. 2006, 51, 881–981. [Google Scholar] [CrossRef]
- Iwahashi, Y.; Wang, J.; Horita, Z.; Nemoto, M.; Langdon, T.G. Principle of equal-channel angular pressing for the processing of ultra-fine grained materials. Scr. Mater. 1996, 35, 143–146. [Google Scholar] [CrossRef]
- Huot, J.; Skryabina, N.Y.; Fruchart, D. Application of severe plastic deformation techniques to magnesium for enhanced hydrogen sorption properties. Metals 2012, 2, 329–343. [Google Scholar] [CrossRef]
- Wieczorek, A.K.; Krystian, M.; Zehetbauer, M.J. SPD processed alloys as efficient vacancy-hydrogen systems. Solid State Phenom. 2006, 114, 177–182. [Google Scholar] [CrossRef]
- Furukawa, M.; Iwahashi, Y.; Horita, Z.; Nemoto, M.; Langdon, T.G. The shearing characteristics associated with equal-channel angular pressing. Mater. Sci. Eng. A 1998, 257, 328–332. [Google Scholar] [CrossRef]
- Song, D.; Ma, A.B.; Jiang, J.H.; Lin, P.H.; Yang, D.H.; Fan, J.F. Corrosion behaviour of bulk ultra-fine grained AZ91D magnesium alloy fabricated by equal-channel angular pressing. Corros. Sci. 2011, 53, 362–373. [Google Scholar] [CrossRef]
- Skripnyuk, V.; Buchman, E.; Rabkin, E.; Estrin, Y.; Popov, M.; Jorgensen, S. The effect of equal channel angular pressing on hydrogen storage properties of a eutectic Mg-Ni alloy. J. Alloys Compd. 2007, 436, 99–106. [Google Scholar] [CrossRef]
- Asselli, A.A.C.; Leiva, D.R.; Huot, J.; Kawasaki, M.; Langdon, T.G.; Botta, W.J. Effects of equal-channel angular pressing and accumulative roll-bonding on hydrogen storage properties of a commercial ZK60 magnesium alloy. Int. J. Hydrogen Energy 2015, 40, 16971–16976. [Google Scholar] [CrossRef]
- Soyama, J.; Floriano, R.; Leiva, D.R.; Guo, Y. Severely deformed ZK60 + 2.5% Mm alloy for hydrogen storage produced by two different processing routes. Int. J. Hydrogen Energy 2016, 41, 11284–11292. [Google Scholar] [CrossRef]
- Crivello, J.C.; Dam, B.; Denys, R.V.; Dornheim, M.; Grant, D.M.; Huot, J.; Jensen, T.R.; De Jongh, P.; Latroche, M.; Milanese, C.; et al. Review of magnesium hydride-based materials: Development and optimisation. Appl. Phys. A 2016, 122, 97. [Google Scholar] [CrossRef]
- Broom, D.P. The absorption and desorption properties of hydrogen storage material. In Hydrogen Storage Materials the Characterisation of Their Storage Properties, 1st ed.; China Machine Press: Beijing, China, 2013. (In Chinese) [Google Scholar]
- Krystian, M.; Zehetbauer, M.J.; Kropik, H.; Mingler, B.; Krexner, G. Hydrogen storage properties of bulk nanostructured ZK60 Mg alloy processed by equal channel angular pressing. J. Alloys Compd. 2011, 509, 449–455. [Google Scholar] [CrossRef]
- Bogdanović, B.; Hartwig, T.H.; Spliethoff, B. The development, testing and optimization of energy storage materials based on the MgH2-Mg system. Int. J. Hydrogen Energy 1993, 18, 575–589. [Google Scholar] [CrossRef]
- Liu, Z.; Lei, Z. Cyclic hydrogen storage properties of Mg milled with nickel nano-powders and MnO2. J. Alloys Compd. 2007, 443, 121–124. [Google Scholar] [CrossRef]
- Lei, Z.; Liu, Z.; Chen, Y. Cyclic hydrogen storage properties of Mg milled with nickel nano-powders and NiO. J. Alloys Compd. 2009, 470, 470–472. [Google Scholar] [CrossRef]
- Grill, A.; Horky, J.; Panigrahi, A.; Krexner, G.; Zehetbauer, M. Long-term hydrogen storage in Mg and ZK60 after severe plastic deformation. Int. J. Hydrogen Energy 2015, 40, 171–178. [Google Scholar] [CrossRef]
- Sato, K.; Yoshiie, T.; Satoh, Y.; Xu, Q.; Kuramoto, E.; Kiritani, M. Point defect production under high internal stress without dislocations in Ni and Cu. Radiat. Eff. Defects Soilds 2002, 157, 171–178. [Google Scholar] [CrossRef]
- Gammer, C.; Karnthaler, H.P.; Rentenberger, C. Reordering a deformation disordered intermetallic compound by antiphase boundary movement. J. Alloys Compd. 2017, 713, 148–155. [Google Scholar] [CrossRef]
- Wang, M.; Vo, N.Q.; Campion, M.; Nguyen, T.D. Forced atomic mixing during severe plastic deformation: Chemical interactions and kinetically driven segregation. Acta Mater. 2014, 66, 1–11. [Google Scholar] [CrossRef]
- Straumal, B.B.; Mazilkin, A.A.; Baretzky, B. Accelerated diffusion and phase transformations in co–cu alloys driven by the severe plastic deformation. Mater. Trans. 2012, 53, 63–71. [Google Scholar] [CrossRef]
- Sha, G.; Wang, Y.B.; Liao, X.Z. Influence of equal-channel angular pressing on precipitation in an Al-Zn-Mg-Cu alloy. Acta Mater. 2009, 57, 3123–3132. [Google Scholar] [CrossRef]
- Ma, A.; Jiang, J.; Saito, N.; Shigematsu, I.; Yuan, Y.; Yang, D.; Nishida, Y. Improving both strength and ductility of a Mg alloy through a large number of ECAP passes. Mater. Sci. Eng. A 2009, 513, 122–127. [Google Scholar] [CrossRef]
- Setman, D.; Schafler, E.; Korznikova, E.; Zehetbauer, M.J. The presence and nature of vacancy type defects in nanometals detained by severe plastic deformation. Mater. Sci. Eng. A 2008, 493, 116–122. [Google Scholar] [CrossRef]
- Viswanathan, V.; Laha, T.; Balani, K. Challenges and advances in nanocomposite processing techniques. Mater. Sci. Eng. R Rep. 2006, 54, 121–285. [Google Scholar] [CrossRef]
- Tran, N.E.; Lambrakos, S.G.; Imam, M.A. Analyses of hydrogen sorption kinetics and thermodynamics of magnesium-misch metal alloys. J. Alloys Compd. 2006, 407, 240–248. [Google Scholar] [CrossRef]
- Oelerich, W.; Klassen, T.; Bormann, R. Metal oxides as catalysts for improved hydrogen sorption in nanocrystalline Mg-based materials. J. Alloys Compd. 2001, 315, 237–242. [Google Scholar] [CrossRef]
- Coelho, R.S.; Pinto, H.; Requena, G.C. Casting in the Semi-Solid state of ZK60 magnesium alloy modified with rare earth addition. Adv. Mater. Res. 2014, 922, 694–699. [Google Scholar] [CrossRef]
- Vittori Antisari, M.; Aurora, A.; Mirabile Gattia, D.; Montone, A. On the nucleation step in the Mg-MgH2 phase transformation. Scr. Mater. 2009, 61, 1064–1067. [Google Scholar] [CrossRef]
- Montone, A.; Aurora, A.; Mirabile Gattia, D.; Vittori Antisari, M. On the barriers limiting the reaction kinetics between catalysed Mg and hydrogen. Scr. Mater. 2010, 63, 456–459. [Google Scholar] [CrossRef]
- Zaluski, L.; Zaluska, A.; Ström-Olsen, J.O. Nanocrystalline metal hydrides. J. Alloys Compd. 1997, 253, 70–79. [Google Scholar] [CrossRef]
- Hongo, T.; Edalati, K.; Arita, M. Significance of grain boundaries and stacking faults on hydrogen storage properties of Mg2Ni intermetallics processed by high-pressure torsion. Acta Mater. 2015, 92, 46–54. [Google Scholar] [CrossRef]
- Kusadome, Y.; Ikeda, K.; Nakamori, Y.; Orimo, S.; Horita, Z. Hydrogen storage capability of MgNi2 processed by high pressure torsion. Scr. Mater. 2007, 57, 751–753. [Google Scholar] [CrossRef]
- Edalati, K.; Yamamoto, A.; Horita, Z.; Ishihara, T. High-pressure torsion of pure magnesium: Evolution of mechanical properties, microstructures and hydrogen storage capacity with equivalent strain. Scr. Mater. 2011, 64, 880–883. [Google Scholar] [CrossRef]
- Mütschele, T.; Kirchheim, R. Segregation and diffusion of hydrogen in grain boundaries of palladium. Scr. Mater. 1987, 21, 135–140. [Google Scholar] [CrossRef]
- Mütschele, R.T.; Kirchheim, R. Hydrogen as a probe for the average thickness of a grain boundary. Scr. Mater. 1987, 21, 1101–1104. [Google Scholar] [CrossRef]
- Iwaoka, H.; Arita, M.; Horita, Z. Hydrogen diffusion in ultrafine-grained palladium: Roles of dislocations and grain boundaries. Acta Mater. 2016, 107, 168–177. [Google Scholar] [CrossRef]
- Oda, T. Thermodynamic model for grain boundary effects on hydrogen solubility, diffusivity and permeability in poly-crystalline tungsten. Fusion Eng. Des. 2016, 112, 102–116. [Google Scholar] [CrossRef]
- Hurley, C.; Martin, F.; Marchetti, L.; Chêne, J.; Blanc, C.; Andrieu, E. Role of grain boundaries in the diffusion of hydrogen in nickel base alloy 600: Study coupling thermal desorption mass spectroscopy with numerical simulation. Int. J. Hydrogen Energy 2016, 41, 17145–17153. [Google Scholar] [CrossRef]
- Wang, F.; Lai, W.; Li, R.; He, B.; Li, S. Fast hydrogen diffusion along the Σ7 grain boundary of α-Al2O3: A first-principles study. Int. J. Hydrogen Energy 2016, 41, 22214–22220. [Google Scholar] [CrossRef]
- Sun, L.; Jin, S.; Zhou, H.; Zhang, Y.; Lu, G. Dissolution and diffusion of hydrogen in a molybdenum grain boundary: A first-principles investigation. Comput. Mater. Sci. 2015, 102, 243–249. [Google Scholar] [CrossRef]
- Oudriss, A.; Creus, J.; Bouhattate, J.; Conforto, E.; Berziou, C.; Savall, C.; Feaugas, X. Grain size and grain-boundary effects on diffusion and trapping of hydrogen in pure nickel. Acta Mater. 2012, 60, 6814–6828. [Google Scholar] [CrossRef]
- Valiev, R.Z.; Islamgaliev, R.K.; Alexandrov, I.V. Bulk nanostructured materials from severe plastic deformation. Prog. Mater. Sci. 2000, 45, 103–189. [Google Scholar] [CrossRef]
- Jorge, A.M., Jr.; Prokofiev, E.; Ferreira De Lima, G.; Rauch, E.; Veron, M.; Botta, W.J.; Kawasaki, M.; Langdon, T.G. An investigation of hydrogen storage in a magnesium-based alloy processed by equal-channel angular pressing. Int. J. Hydrogen Energy 2013, 38, 8306–8312. [Google Scholar] [CrossRef]
- Lima, G.F.; Triques, M.R.M.; Kiminami, C.S.; Botta, W.J.; Jorge, A.M., Jr. Hydrogen storage properties of pure Mg after the combined processes of ECAP and cold-rolling. J. Alloys Compd. 2014, 586, 405–408. [Google Scholar] [CrossRef]
- Jacobson, N.; Tegner, B.; Schröder, E.; Hyldgaard, P.; Lundqvist, B.I. Hydrogen dynamics in magnesium and graphite. Comput. Mater. Sci. 2002, 24, 273–277. [Google Scholar] [CrossRef]
- Jorge, A.M., Jr.; Ferreira De Lima, G.; Martins Triques, M.R.; Botta, W.J.; Kiminami, C.S.; Nogueira, R.P.; Yavari, A.R.; Langdon, T.G. Correlation between hydrogen storage properties and textures induced in magnesium through ECAP and cold rolling. Int. J. Hydrogen Energy 2014, 39, 3810–3821. [Google Scholar] [CrossRef]
- Singh, S.; Eijt, S.W.H.; Zandbergen, M.W.; Legerstee, W.J.; Svetchnikov, V.L. Nanoscale structure and the hydrogenation of Pd-capped magnesium thin films prepared by plasma sputter and pulsed laser deposition. J. Alloys Compd. 2007, 441, 344–351. [Google Scholar] [CrossRef]

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, L.; Jiang, J.; Ma, A.; Li, Y.; Song, D. A Critical Review of Mg-Based Hydrogen Storage Materials Processed by Equal Channel Angular Pressing. Metals 2017, 7, 324. https://doi.org/10.3390/met7090324
Wang L, Jiang J, Ma A, Li Y, Song D. A Critical Review of Mg-Based Hydrogen Storage Materials Processed by Equal Channel Angular Pressing. Metals. 2017; 7(9):324. https://doi.org/10.3390/met7090324
Chicago/Turabian StyleWang, Lisha, Jinghua Jiang, Aibin Ma, Yuhua Li, and Dan Song. 2017. "A Critical Review of Mg-Based Hydrogen Storage Materials Processed by Equal Channel Angular Pressing" Metals 7, no. 9: 324. https://doi.org/10.3390/met7090324



