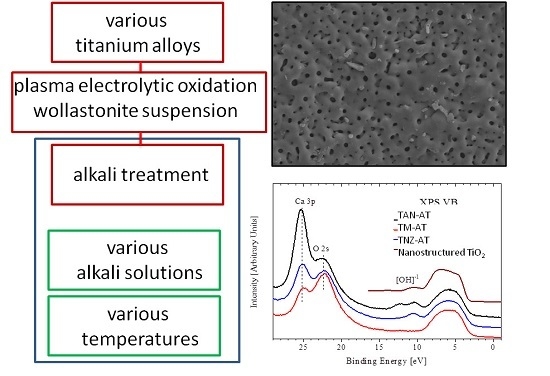
Influence of Alkali Treatment on Anodized Titanium Alloys in Wollastonite Suspension
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Procedures of Surface Treatment
2.2. Characterization of the Anodized Surfaces
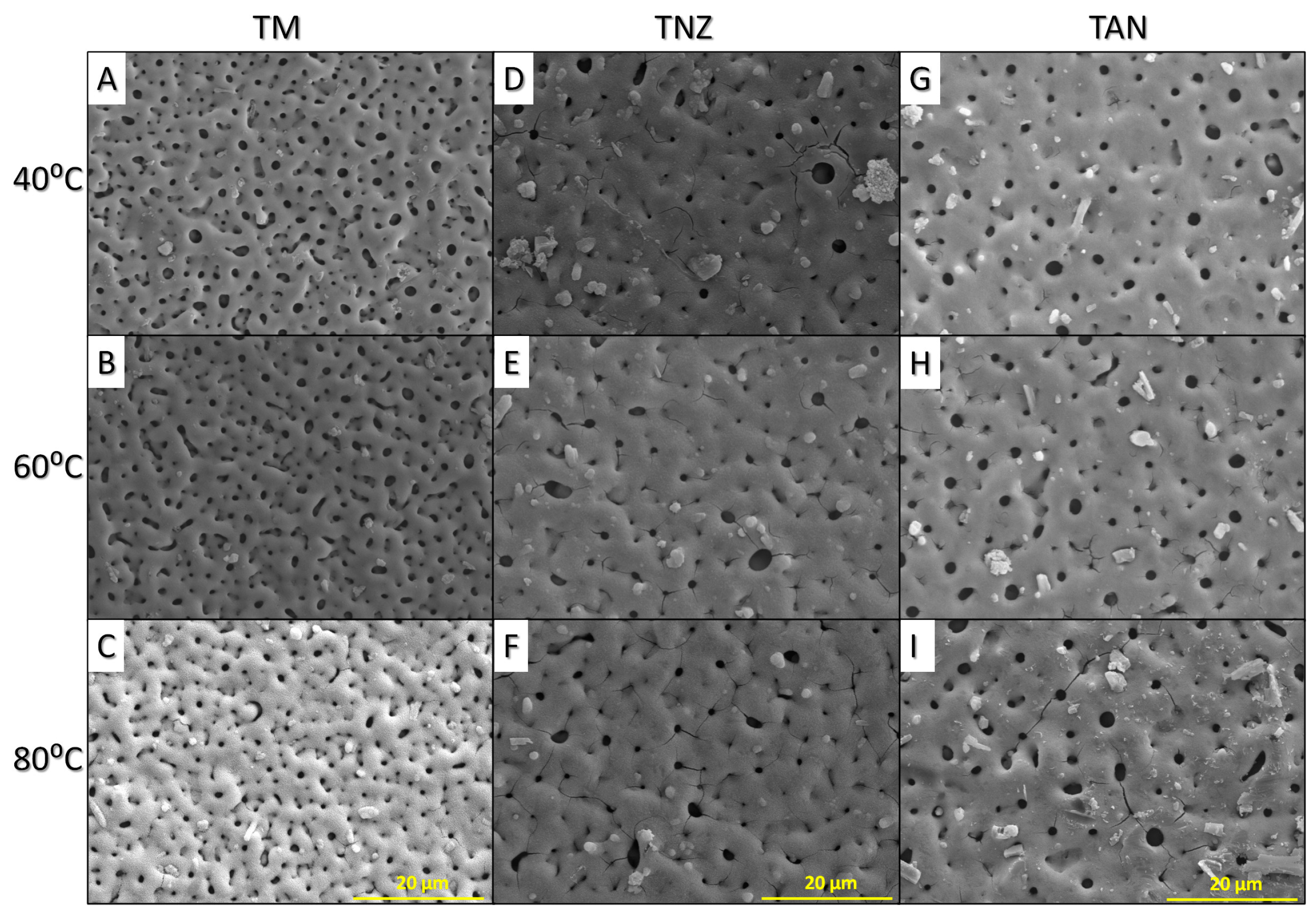
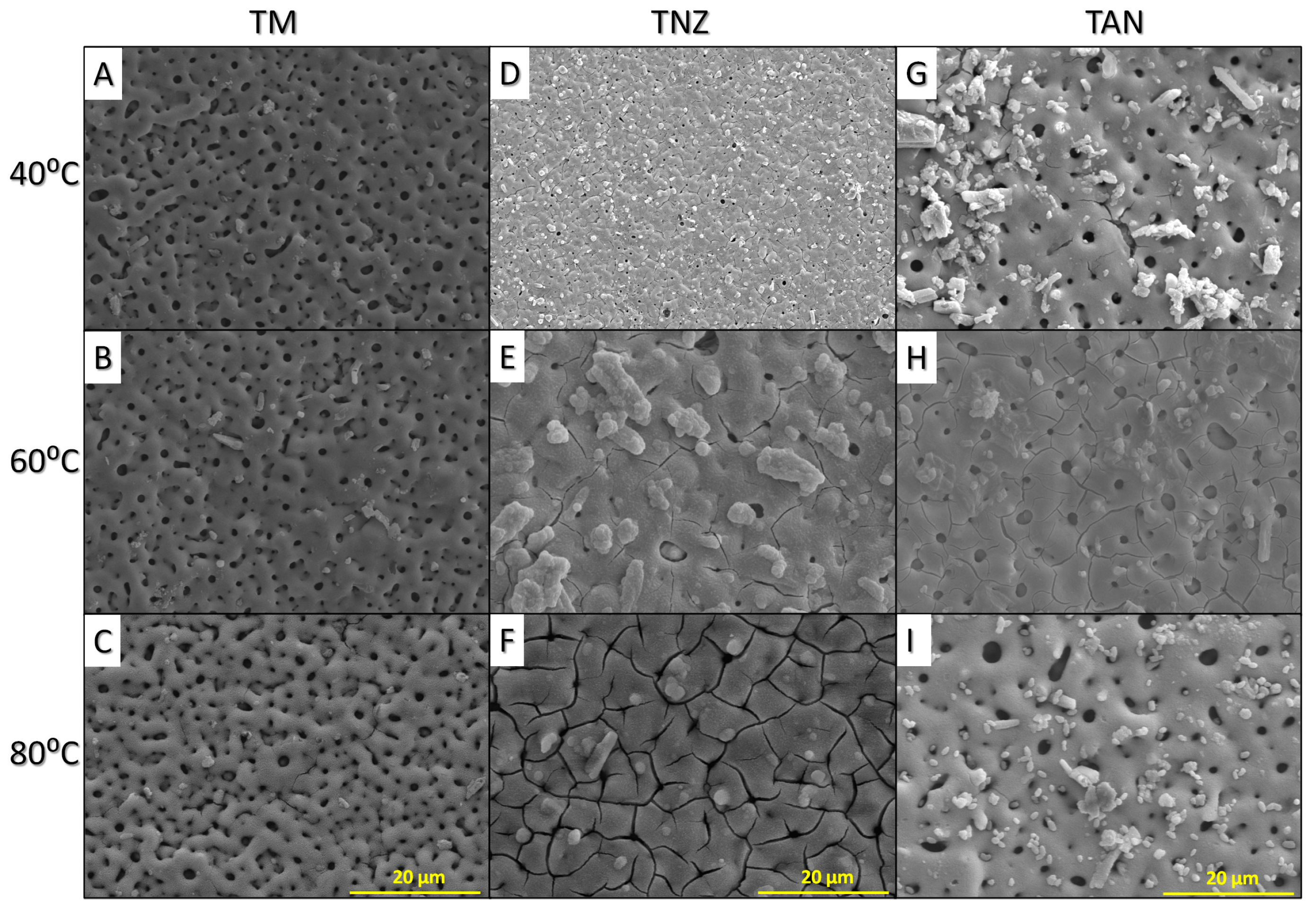
2.2.1. Microstructure, Surface Wettability and Roughness
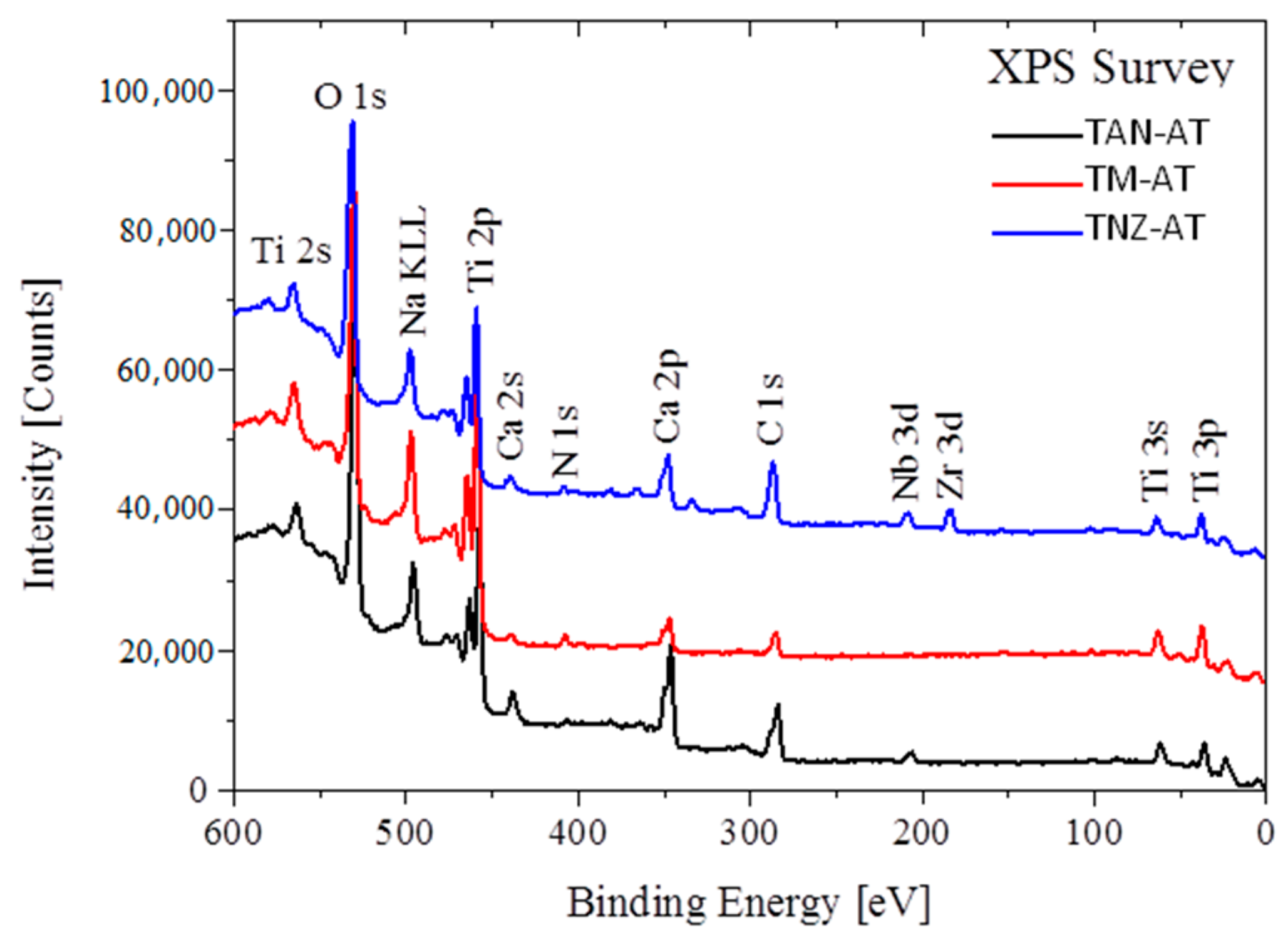
2.2.2. Chemical Composition
3. Results and Discussion
3.1. Microstructure of the Modified Titanium Alloy Surfaces
3.2. Wettability and Surface Roughness
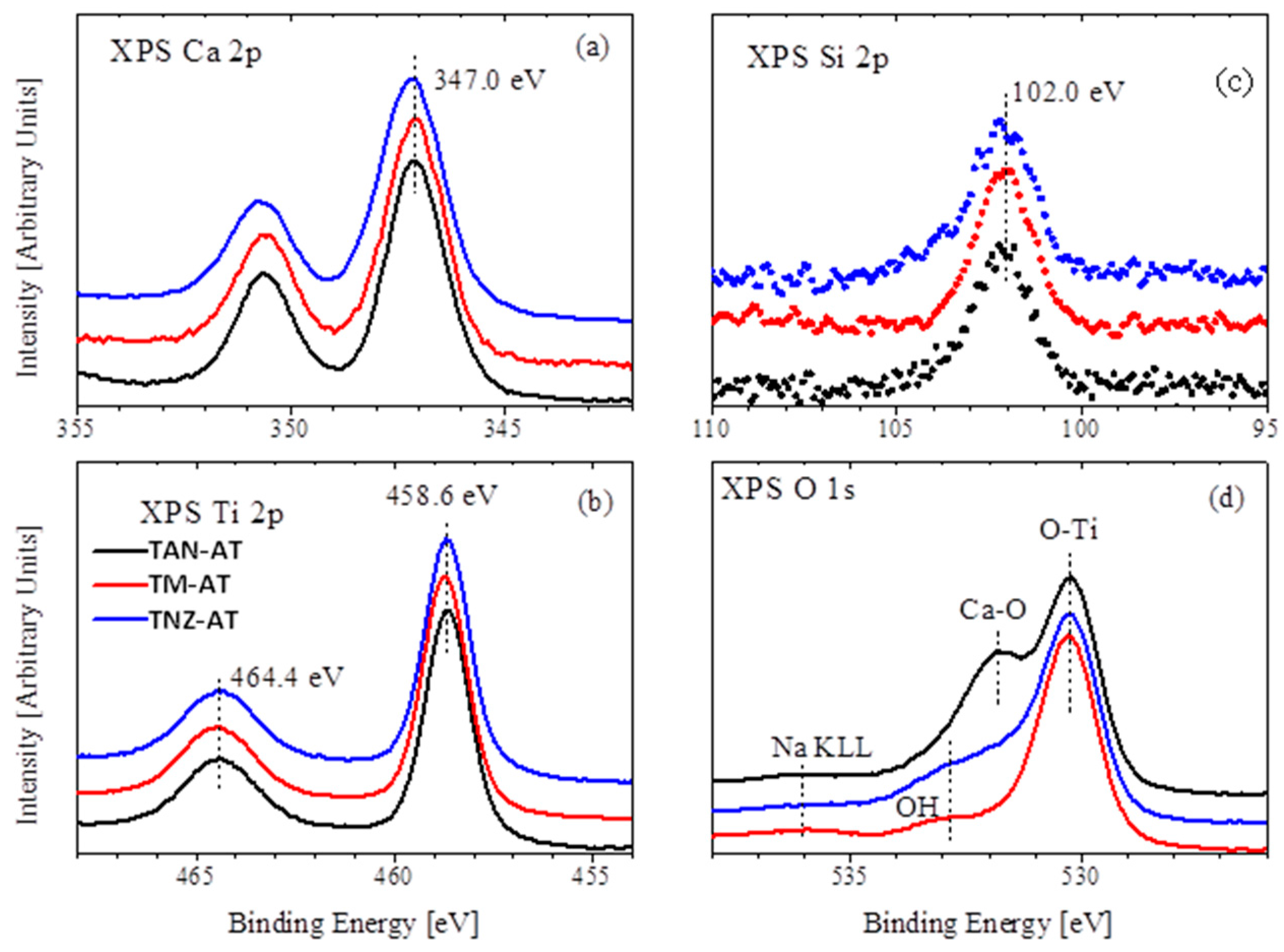
3.3. Chemical Composition Analysis
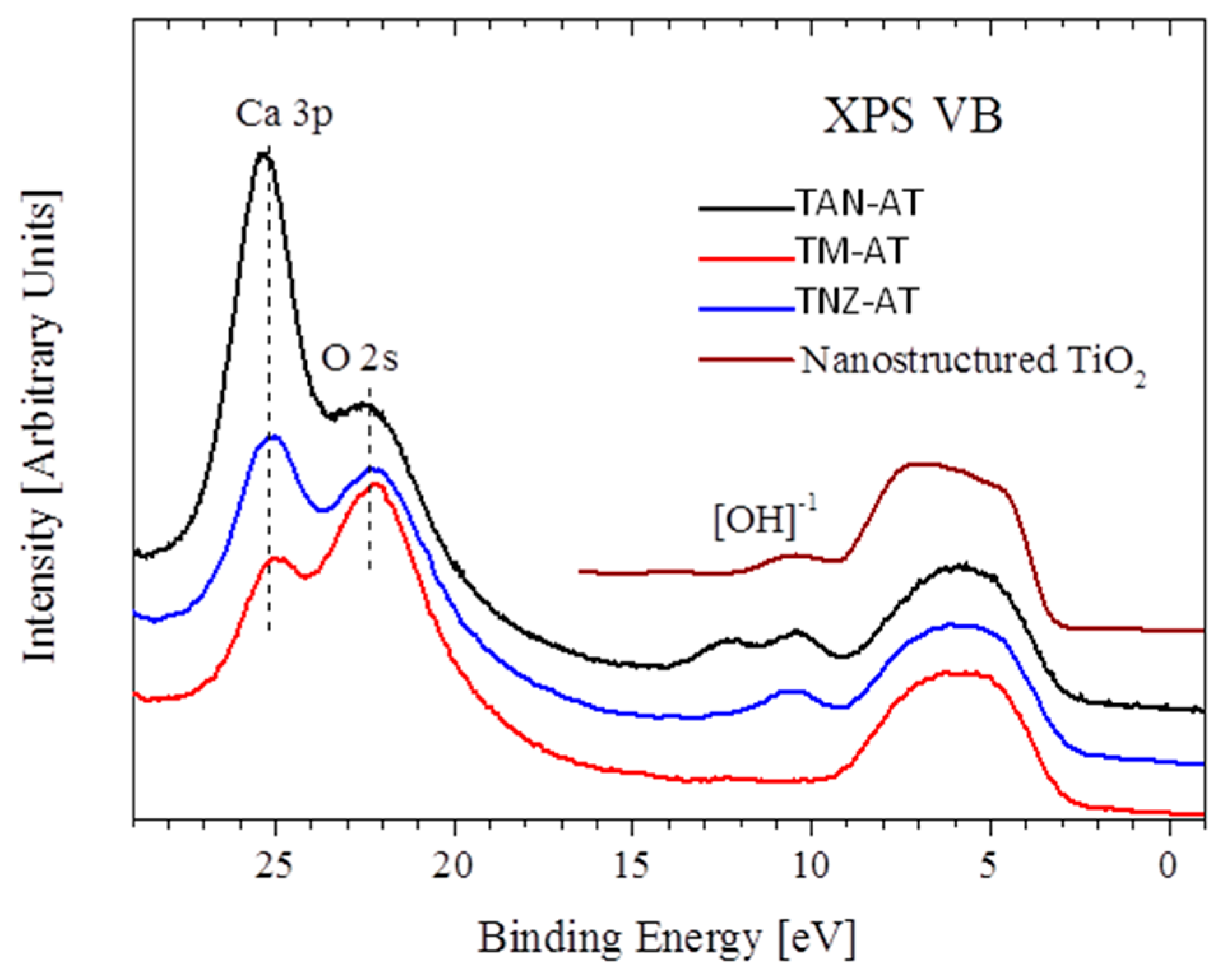
3.4. XPS Measurement
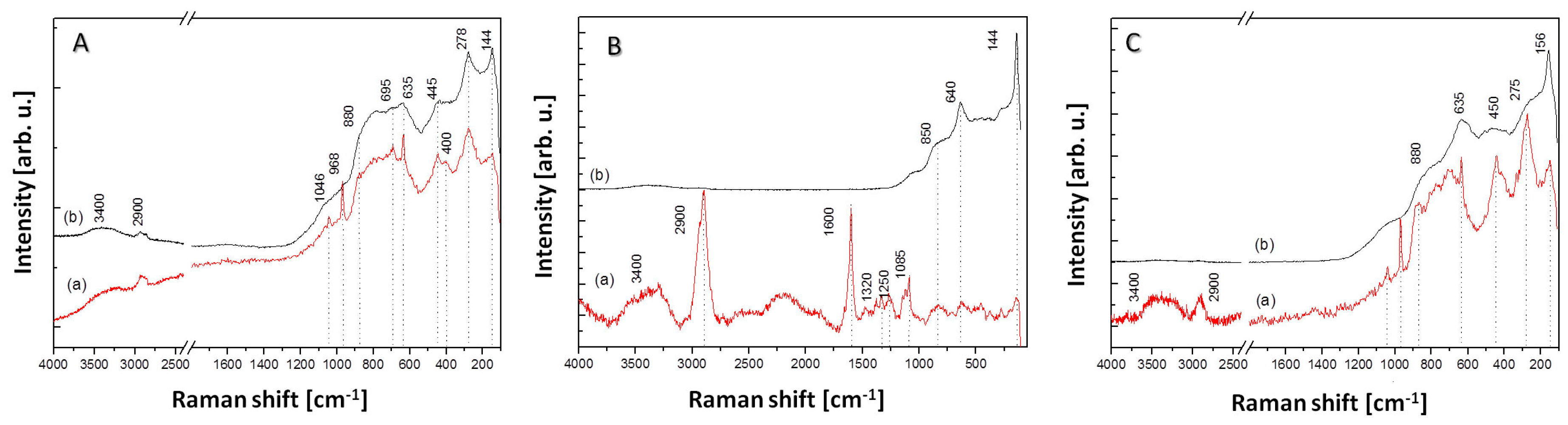
3.5. Raman Spectroscopy
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Prasad, S.; Ehrensberger, M. Biomaterial properties of titanium in dentistry. J. Oral Biosci. 2015, 57, 192–199. [Google Scholar] [CrossRef]
- Cordeiro, J.M.; Barão, V.A.R. Is there scientific evidence favoring the substitution of commercially pure titanium with titanium alloys for the manufacture of dental implants? Mater. Sci. Eng. C 2017, 71, 1201–1215. [Google Scholar] [CrossRef] [PubMed]
- Mahapatro, A. Bio-functional nano-coatings on metallic biomaterials. Mater. Sci. Eng. C 2015, 55, 227–251. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Thouas, G.A. Metallic implant biomaterials. Mater. Sci. Eng. R Rep. 2015, 87, 1–57. [Google Scholar] [CrossRef]
- Mani, G. Surface Properties and Characterization of Metallic Biomaterials. In Surface Coating and Modification of Metallic Biomaterials, 1st ed.; Wen, C., Ed.; Elsevier: Amsterdam, The Netherlands, 2016; Volume 3, pp. 61–77. [Google Scholar]
- Kaluđerović, M.R.; Schreckenbach, J.P.; Graf, H.L. Titanium dental implant surfaces obtained by anodic spark deposition—From the past to the future. Mater. Sci. Eng. C 2016, 69, 1429–1441. [Google Scholar] [CrossRef] [PubMed]
- Rokosz, K.; Hryniewicz, T.; Matỳsek, D.; Raaen, S.; Valíček, J.; Dudek, Ł.; Harničárová, M. SEM, EDS and XPS analysis of the coatings obtained on titanium after plasma electrolytic oxidation in electrolytes containing copper nitrate. Materials 2016, 9, 318. [Google Scholar] [CrossRef] [PubMed]
- Rokosz, K.; Hryniewicz, T.; Malorny, W. Characterization of coatings created on selected titanium alloys by plasma electrolytic oxidation. Adv. Mater. Sci. 2016, 6, 5–16. [Google Scholar] [CrossRef]
- Lee, B.H.; Kim, Y.D.; Shin, J.H.; Lee, K.H. Surface modification by alkali and heat treatments in titanium alloys. J. Biomed. Mater. Res. 2002, 61, 466–473. [Google Scholar] [CrossRef] [PubMed]
- Yavari, A.S.; Ahmadi, S.M.; Stok, J.; Wauthle, R.; Riemslag, A.C.; Janssen, M.; Schrooten, J.; Weinans, H.; Zadpoor, A.A. Effects of bio-functionalizing surface treatments on the mechanical behavior of open porous titanium biomaterials. J. Mech. Behav. Biomed. Mater. 2014, 36, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Yan, Z.; Wu, M.; Qin, H.; He, Y.; Yu, X.; Chen, L. ZrO2/TiO2 films prepared by plasma electrolytic oxidation and a post treatment. Surf. Coat. Technol. 2017, 309, 331–336. [Google Scholar] [CrossRef]
- Bai, Y.; Deng, Y.; Zheng, Y.; Li, Y.; Zhang, R.; Lv, Y.; Zhao, Q.; Wei, S. Characterization, corrosion behavior, cellular response and in vivo bone tissue compatibility of titanium–niobium alloy with low Young’s modulus. Mater. Sci. Eng. C 2016, 59, 565–576. [Google Scholar] [CrossRef] [PubMed]
- Correa, D.R.N.; Vicente, F.B.; Donato, T.A.G.; Arana-Chavez, V.E.; Buzalaf, M.A.R.; Grandini, C.R. The effect of the solute on the structure, selected mechanical properties, and biocompatibility of Ti–Zr system alloys for dental applications. Mater. Sci. Eng. C 2014, 34, 354–359. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Ramaswamy, Y.; Boughton, P.; Zreiqat, H. Improvement of mechanical and biological properties of porous CaSiO3 scaffolds by poly(d,l-lactic acid) modification. Acta Biomater. 2008, 4, 343–353. [Google Scholar] [CrossRef] [PubMed]
- Sadjadia, M.S.; Ebrahimia, H.R.; Meskinfam, M.; Zare, K. Silica enhanced formation of hydroxyapatite nanocrystals in simulated body fluid (SBF) at 37 °C. Mater. Chem. Phys. 2011, 130, 67–71. [Google Scholar] [CrossRef]
- Siriphannon, P.; Kameshima, Y.; Yasumori, A.; Okada, K.; Hayashi, S. Formation of hydroxyapatite on CaSiO3 powders in simulated body fluid. J. Eur. Ceram. Soc. 2002, 22, 511–520. [Google Scholar] [CrossRef]
- Liu, X.; Ding, C.; Chu, P.K. Mechanism of apatite formation on wollastonite coatings in simulated body fluid. Biomaterials 2004, 25, 1755–1761. [Google Scholar] [CrossRef] [PubMed]
- Kazek-Kęsik, A.; Dercz, G.; Kalemba, I.; Suchanek, K.; Kukharenko, A.I.; Korotin, D.M.; Michalska, J.; Krząkała, A.; Piotrowski, J.; Kurmaev, E.Z.; et al. Surface characterisation of Ti–15Mo alloy modified by a PEO process in various suspensions. Mater. Sci. Eng. C 2014, 39, 259–272. [Google Scholar] [CrossRef] [PubMed]
- Kazek-Kęsik, A.; Dercz, G.; Suchanek, K.; Kalemba-Rec, I.; Piotrowski, J.; Simka, W. Biofunctionalization of Ti–13Nb–13Zr alloy surface by plasma electrolytic oxidation. Part I. Surf. Coat. Technol. 2015, 276, 59–69. [Google Scholar] [CrossRef]
- ISO 4287:1999 Geometrical Product Specification (GPS)—Surface Texture: Profile Methods—Terms, Definitions and Surface Texture Parameters. Available online: https://www.iso.org/standard/10132.html (accessed on 18 August 2017).
- Wu, J.M.; Song, X.M.; Yan, M. Alkaline hydrothermal synthesis of homogeneous titania microspheres with urchin-like nanoarchitectures for dye effluent treatments. J. Hazard. Mater. 2011, 194, 338–344. [Google Scholar] [CrossRef] [PubMed]
- Kokubo, T.; Yamaguchi, S. Bioactive titanate layers formed on titanium and its alloys by simple chemical and heat treatments. Open Biomed. Eng. J. 2015, 9, 29–41. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; Yoo, D. Large-area sodium titanate nanorods formed on titanium surface via. NaOH alkali treatment. Arch. Metall. Mater. 2015, 60, 1371–1374. [Google Scholar] [CrossRef]
- Sainitya, R.; Sriram, M.; Kalyanaraman, V.; Dhivya, S.; Saravanan, S.; Vairamani, M.; Sastry, T.P.; Selvamurugan, N. Scaffolds containing chitosan/carboxymethyl cellulose/mesoporous wollastonite for bone tissue engineering. Int. J. Biol. Macromol. 2015, 80, 481–488. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Kim, Y.L.; Park, L.S.; Jin, G.C.; Bae, T.S.; Lee, M.H. Effect of alkali and heat treatments for bioactivity of TiO2 nanotubes. Appl. Surf. Sci. 2014, 321, 412–419. [Google Scholar] [CrossRef]
- Park, S.Y.; Jo, C.I.; Choe, H.C.; Brantley, W.A. Hydroxyapatite deposition on micropore-formed Ti-Ta-Nb alloys by plasma electrolytic oxidation for dental applications. Surf. Coat. Technol. 2016, 294, 15–20. [Google Scholar] [CrossRef]
- Zhao, G.L.; Xiao, L.; Zhong, B.; Wu, S.S.; Song, L.; Wen, G.W. Effect of alkali treatments on apatite formation of microarc-oxidized coating on titanium alloy surface. Trans. Nonferrous Met. Soc. China 2015, 25, 1151–1157. [Google Scholar] [CrossRef]
- Jalota, S.; Bhaduri, S.; Bhaduri, S.B. A protocol to develop crack-free biomimetic coatings on Ti6Al4V substrates. J. Mater. Res. 2007, 22, 1593–1600. [Google Scholar] [CrossRef]
- Baoe, L.; Jun, L.; Chunyong, L.; Haipeng, L.; Litong, G.; Shimin, L.; Hongshui, W. Surface roughness and hydrophilicity of titanium after anodic oxidation. Rare Met. Mater. Eng. 2016, 45, 858–862. [Google Scholar]
- Lampin, M.; Warocquier-Clérout, R.; Legris, C.; Degrange, M.; Sigot-Luizard, M. Correlation between substratum roughness and wettability, cell adhesion, and cell migration. J. Biomed. Mater. Res. 1997, 36, 99–108. [Google Scholar] [CrossRef]
- Gittens, R.A.; Scheideler, L.; Rupp, F.; Hyzy, S.L.; Geis-Gerstorfer, J.; Schwartz, Z.; Boyan, B.D. A review on the wettability of dental implant surfaces II: Biological and clinical aspects. Acta Biomater. 2014, 10, 2907–2918. [Google Scholar] [CrossRef] [PubMed]
- Song, W.H.; Jun, Y.K.; Han, Y.; Hong, S.H. Biomimetic apatite coatings on micro-arc oxidized titania. Biomaterials 2004, 25, 3341–3349. [Google Scholar] [CrossRef] [PubMed]
- Matykina, E.; Arrabal, R.; Skeldon, P.; Thompson, G.E. Transmission electron microscopy of coatings formed by plasma electrolytic oxidation of titanium. Acta Biomater. 2009, 5, 1356–1366. [Google Scholar] [CrossRef] [PubMed]
- Padial-Molina, M.; Galindo-Moreno, P.; Fernández-Barbero, J.E.; O’Valle, F.; Jódar-Reyes, A.B.; Ortega-Vinuesa, J.L.; Ramón-Torregrosa, P.J. Role of wettability and nanoroughness on interactions between osteoblast and modified silicon surfaces. Acta Biomater. 2011, 7, 771–778. [Google Scholar] [CrossRef] [PubMed]
- Martin, J.Y.; Schwartz, Z.; Hummert, T.W.; Schraub, T.M.; Simpson, J.; Lankford, J.; Dean, D.D.; Cochran, D.L.; Boyan, B.D. Effect of titanium surface roughness on proliferation, differentiation, and protein synthesis of human osteoblast-like cells (MG63). J. Biomed. Mater. Res. 1995, 29, 389–401. [Google Scholar] [CrossRef] [PubMed]
- Jokanovic, V.; Vilotijevic, M.; Jokanovic, B.; Jenko, M.; Anzel, I.; Stamenkovic, D.; Lazic, V.; Rudolf, R. Investigations of corrosion on the surface of titanium substrate caused by combined alkaline and heat treatment. Corros. Sci. 2014, 82, 180–190. [Google Scholar] [CrossRef]
- Zhang, W.; Du, K.; Yan, C.; Wang, F. Preparation and characterization of a novel Si-incorporated ceramic film on pure titanium by plasma electrolytic oxidation. Appl. Surf. Sci. 2008, 254, 5216–5223. [Google Scholar] [CrossRef]
- Wagner, C.D.; Riggs, W.M.; Davis, L.E.; Moulder, J.E.; Mulenger, G.E. Handbook of X-ray Photoelectron Spectroscopy, 1st ed.; Perkin Elmer Corp.: Eden Prairie, MN, USA, 1979. [Google Scholar]
- Fusi, M.; Maccallini, E.; Caruso, T.; Casari, C.S.; Li Bassi, A.; Bottani, C.E.; Rudolf, P.; Prince, K.C.; Agostino, R.G. Surface electronic and structural properties of nanostructured titanium oxide grown by pulsed laser deposition. Surf. Sci. 2011, 605, 333–340. [Google Scholar] [CrossRef]
- Thevenot, P.; Hu, W.; Tang, L. Surface chemistry influences implant biocompatibility. Curr. Top. Med. Chem. 2008, 8, 270–280. [Google Scholar] [PubMed]
- Nguyen-Phan, T.D.; Pham, V.H.; Kim, E.J.; Oh, E.S.; Hur, S.H.; Chung, J.S.; Lee, B.; Shin, E.W. Reduced graphene oxide–titanate hybrids: Morphologic evolution by alkali-solvothermal treatment and applications in water purification. Appl. Surf. Sci. 2012, 258, 4551–4557. [Google Scholar] [CrossRef]
- Nebe, J.B.; Müller, L.; Lüthen, F.; Ewald, A.; Bergemann, C.; Conforto, E.; Müller, F.A. Osteoblast response to biomimetically altered titanium surfaces. Acta Biomater. 2008, 4, 1985–1995. [Google Scholar] [CrossRef] [PubMed]
- De Aza, P.N.; Luklinska, Z.B.; Anseau, M.R.; Hector, M.; Guitiàn, F.; De Aza, S. Reactivity of a wollastonite–tricalcium phosphate bioeutectic ceramic in human parotid saliva. Biomaterials 2000, 21, 1735–1741. [Google Scholar] [CrossRef]
- Shao, H.; He, Y.; Fu, J.; He, D.; Yang, X.; Xie, J.; Yao, C.; Ye, J.; Xu, S.; Gou, Z. 3D printing magnesium-doped wollastonite/β-TCP bioceramics scaffolds with high strength and adjustable degradation. J. Eur. Ceram. Soc. 2016, 35, 1495–1503. [Google Scholar] [CrossRef]



| Titanium Alloy | Al | Nb | Zr | Ta | Mo | Fe | C | N | H | O | Ti |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Ti-6Al-7Nb (TAN) | 6.05 | 6.80 | - | 0.35 | - | 0.08 | 0.04 | 0.01 | 0.003 | 0.11 | balance |
| Ti-13Nb-13Zr (TNZ) | - | 13.30 | 13.00 | - | - | 0.08 | 0.05 | 0.0009 | 0.005 | 0.10 | |
| Ti-15Mo (TM) | - | - | - | - | 14.73 | 0.06 | 0.08 | 0.016 | 0.01 | 0.15 |
| Sample | Contact Angle/° | Surface Roughness, μm | ||
|---|---|---|---|---|
| Ra | Rq | Rz | ||
| TM | 47.6 ± 5.7 | 0.28 | 0.36 | 0.30 |
| TM-AT | 2.3 ± 0.7 | 1.08 | 2.19 | 309.06 |
| TNZ | 120 ± 5.0 | 0.51 | 0.63 | 9.77 |
| TNZ-AT | 21.8 ± 4.6 | 2.74 | 3.39 | 168.09 |
| TAN | 51.2 ± 4.7 | 0.30 | 0.48 | 1.48 |
| TAN-AT | 11.7 ± 2.7 | 2.37 | 2.91 | 92.40 |
| Sample | Concentration, at % | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Ti | Al | Nb/Mo | Ca | Na | Zr | C | O | Si | |
| TM-AT | 17.1 | - | 0.1 | 2.4 | 13.4 | - | 11.1 | 55.3 | 0.6 |
| TNZ-AT | 9.5 | - | 0.5 | 3.4 | 6.3 | 0.9 | 27.7 | 50.9 | 0.8 |
| TAN-AT | 9.8 | 0.3 | 0.3 | 5.6 | 7.5 | - | 24.6 | 51.4 | 0.5 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kazek-Kęsik, A.; Leśniak, K.; Zhidkov, I.S.; Korotin, D.M.; Kukharenko, A.I.; Cholakh, S.O.; Kalemba-Rec, I.; Suchanek, K.; Kurmaev, E.Z.; Simka, W. Influence of Alkali Treatment on Anodized Titanium Alloys in Wollastonite Suspension. Metals 2017, 7, 322. https://doi.org/10.3390/met7090322
Kazek-Kęsik A, Leśniak K, Zhidkov IS, Korotin DM, Kukharenko AI, Cholakh SO, Kalemba-Rec I, Suchanek K, Kurmaev EZ, Simka W. Influence of Alkali Treatment on Anodized Titanium Alloys in Wollastonite Suspension. Metals. 2017; 7(9):322. https://doi.org/10.3390/met7090322
Chicago/Turabian StyleKazek-Kęsik, Alicja, Katarzyna Leśniak, Ivan S. Zhidkov, Danila M. Korotin, Andrey I. Kukharenko, Seif O. Cholakh, Izabela Kalemba-Rec, Katarzyna Suchanek, Ernst Z. Kurmaev, and Wojciech Simka. 2017. "Influence of Alkali Treatment on Anodized Titanium Alloys in Wollastonite Suspension" Metals 7, no. 9: 322. https://doi.org/10.3390/met7090322