Microwave Pretreatment for Thiourea Leaching for Gold Concentrate
Abstract
:1. Introduction
2. Materials and Methods
2.1. Gold Concentrates
2.2. Pretreatment of Gold Concentrate Using Microwaves
2.3. Kinetic Au Leaching Experiments
3. Result and Discussion
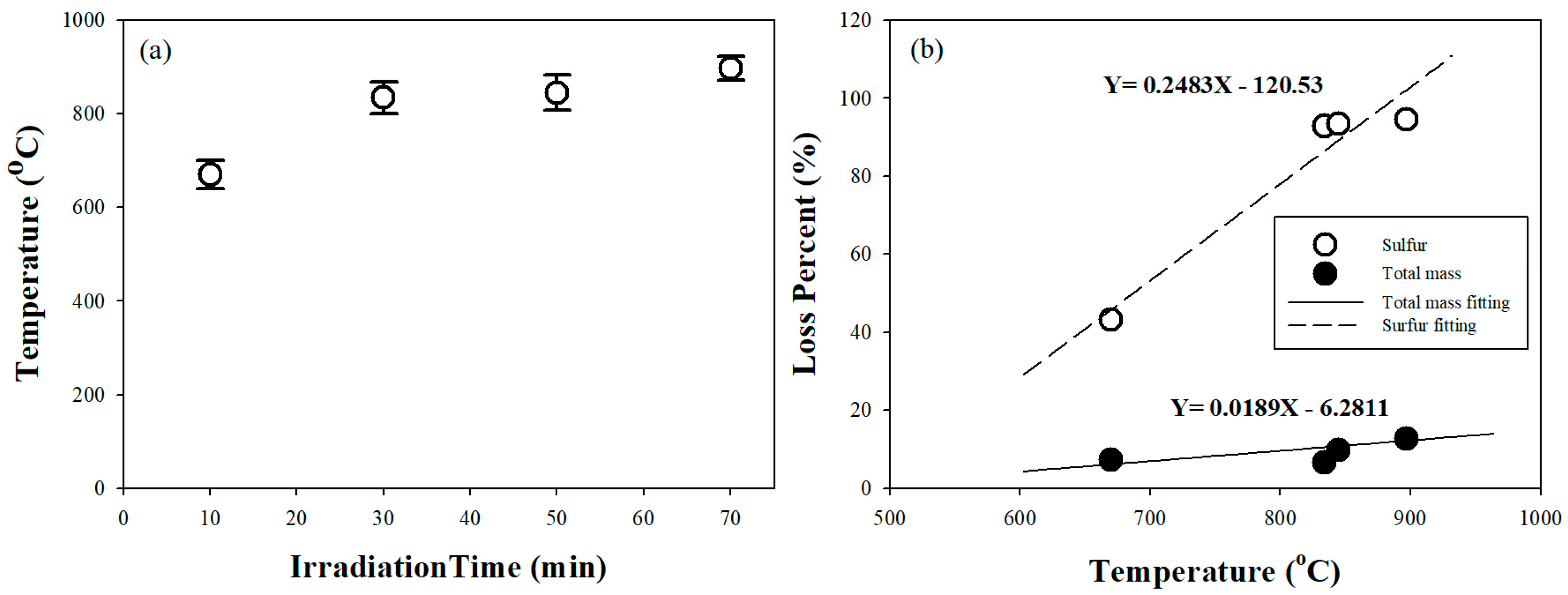
3.1. Effect of Microwave Irradiation on Gold Concentrate
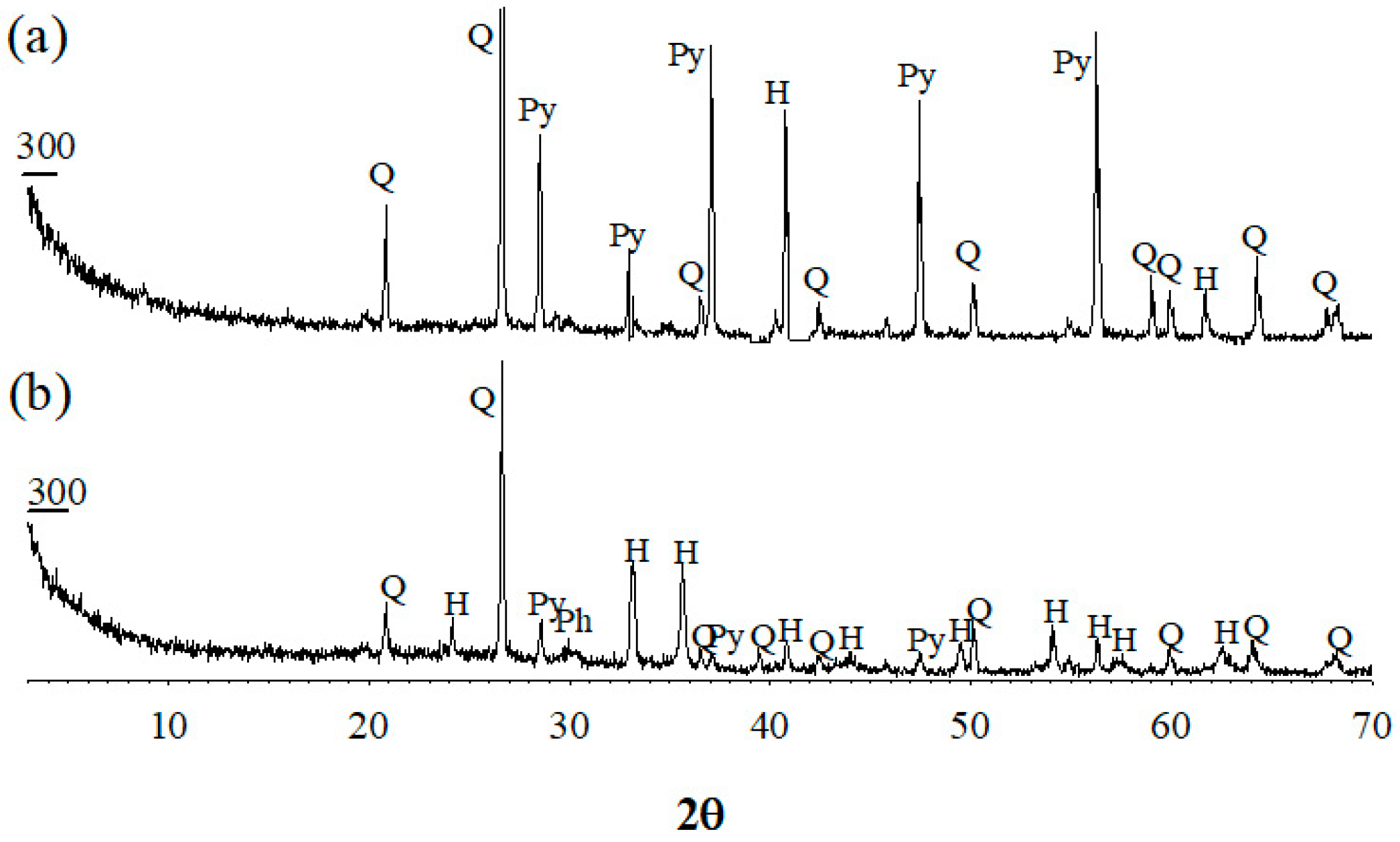
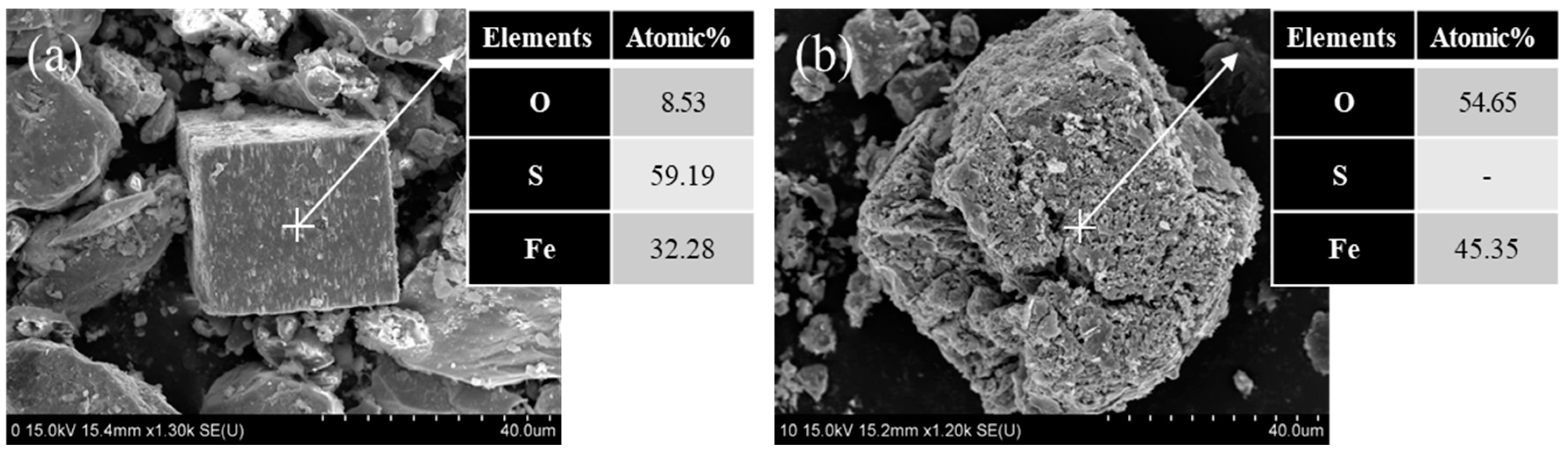
3.2. Mineralogical Change in Gold Concentrate by Microwave Irradiation
3.3. Gold Leaching
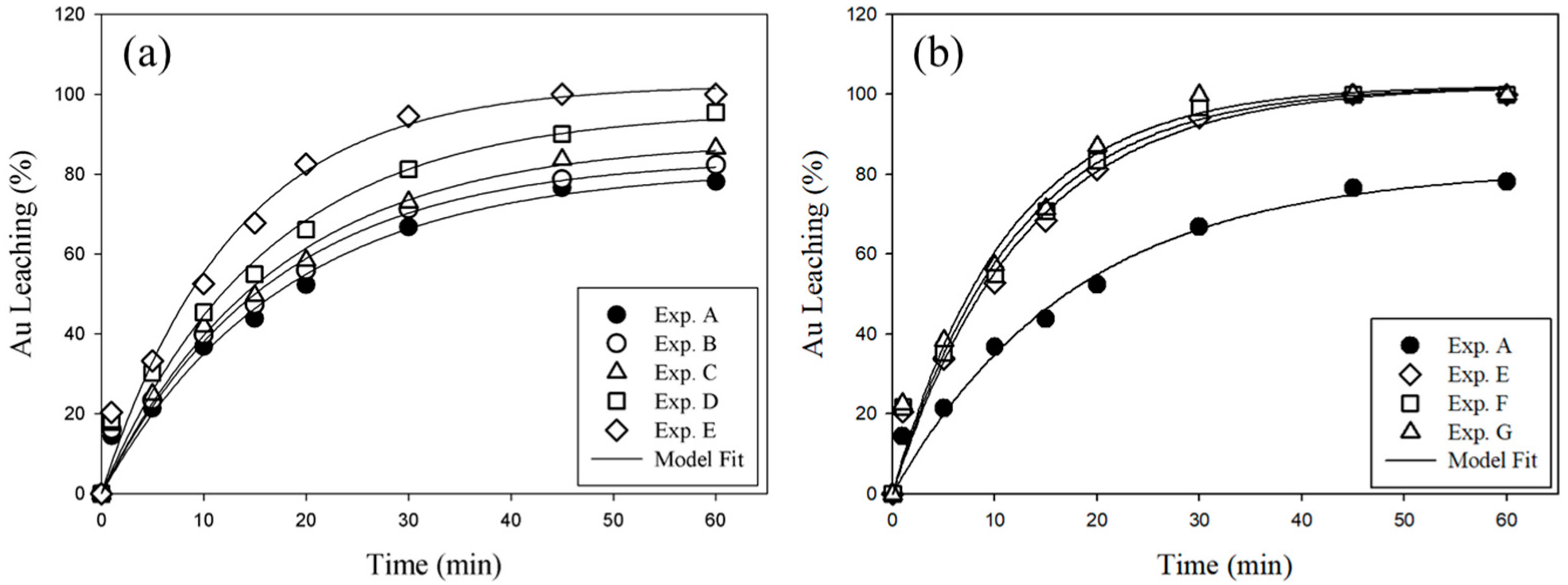
3.3.1. Effect of Microwave Irradiation on Au Leaching Efficiency
3.3.2. Effect of Thiourea Concentration on Au Leaching Efficiency
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Xu, B.; Yang, Y.; Li, Q.; Li, G.; Jiang, T. Fluidized roasting-stage leaching of a silver and gold bearing polymetallic sulfide concentrate. Hydrometallurgy 2014, 147, 79–82. [Google Scholar] [CrossRef]
- Örgül, S.; Atalay, Ü. Reaction chemistry of gold leaching in thiourea solution for a turkish gold ore. Hydrometallurgy 2002, 67, 71–77. [Google Scholar] [CrossRef]
- Aylmore, M.G.; Muir, D.M. Thermodynamic analysis of gold leaching by ammoniacal thiosulfate using Eh/pH and speciation diagrams. Miner. Metall. Process. 2001, 18, 221–227. [Google Scholar]
- Xu, B.; Yang, Y.; Li, Q.; Jiang, T.; Li, G. Stage leaching of a complex polymetallic sulfide concentrate: Focus on the extraction of Ag and Au. Hydrometallurgy 2016, 159, 87–94. [Google Scholar] [CrossRef]
- Filmer, A. The dissolution of gold from roasted pyrite concentrates. J. S. Afr. Inst. Min. Metall. 1982, 82, 90–94. [Google Scholar]
- Dunn, J.; Chamberlain, A. The recovery of gold from refractory arsenopyrite concentrates by pyrolysis-oxidation. Miner. Eng. 1997, 10, 919–928. [Google Scholar] [CrossRef]
- Boyabat, N.; Özer, A.; Bayrakceken, S.; Gülaboğlu, M. Thermal decomposition of pyrite in the nitrogen atmosphere. Fuel Process. Technol. 2004, 85, 179–188. [Google Scholar] [CrossRef]
- Ozkan, A.; Aydogan, S.; Akdemir, U. Bacterial leaching as a pre-treatment step for gold recovery from refractory ores. Fizykochem. Probl. Miner. 1998, 32, 173–181. [Google Scholar]
- Pickles, C. Microwaves in extractive metallurgy: Part 2—A review of applications. Miner. Eng. 2009, 22, 1112–1118. [Google Scholar] [CrossRef]
- Uslu, T.; Atalay, Ü. Microwave heating of coal for enhanced magnetic removal of pyrite. Fuel Process. Technol. 2004, 85, 21–29. [Google Scholar] [CrossRef]
- Huang, J.; Rowson, N. Heating characteristics and decomposition of pyrite and marcasite in a microwave field. Miner. Eng. 2001, 14, 1113–1117. [Google Scholar] [CrossRef]
- Ma, S.; Luo, W.; Mo, W.; Su, X.; Liu, P.; Yang, J. Removal of arsenic and sulfur from a refractory gold concentrate by microwave heating. Miner. Eng. 2010, 23, 61–63. [Google Scholar] [CrossRef]
- Amankwah, R.; Khan, A.; Pickles, C.; Yen, W. Improved grindability and gold liberation by microwave pretreatment of a free-milling gold ore. Miner. Process. Extr. Metall. 2005, 114, 30–36. [Google Scholar] [CrossRef]
- Kim, K.-J.; Ahn, H.-G. The effect of pore structure of zeolite on the adsorption of VOCs and their desorption properties by microwave heating. Microporous Mesoporous Mater. 2012, 152, 78–83. [Google Scholar] [CrossRef] [Green Version]
- Haque, K.E. Microwave energy for mineral treatment processes—A brief review. Int. J. Miner. Process. 1999, 57, 1–24. [Google Scholar] [CrossRef]
- Amankwah, R.; Ofori-Sarpong, G. Microwave heating of gold ores for enhanced grindability and cyanide amenability. Miner. Eng. 2011, 24, 541–544. [Google Scholar] [CrossRef]
- Amankwah, R.; Pickles, C. Microwave roasting of a carbonaceous sulphidic gold concentrate. Miner. Eng. 2009, 22, 1095–1101. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhao, Z.K. Solid acid and microwave-assisted hydrolysis of cellulose in ionic liquid. Carbohydr. Res. 2009, 344, 2069–2072. [Google Scholar] [CrossRef] [PubMed]
- Pert, E.; Carmel, Y.; Birnboim, A.; Olorunyolemi, T.; Gershon, D.; Calame, J.; Lloyd, I.K.; Wilson, O.C. Temperature measurements during microwave processing: The significance of thermocouple effects. J. Am. Ceram. Soc. 2001, 84, 1981–1986. [Google Scholar] [CrossRef]
- Menéndez, J.; Menéndez, E.; Garcia, A.; Parra, J.; Pis, J. Thermal treatment of active carbons: A comparison between microwave and electrical hating. J. Microw. Power Electromagn. Energy 1999, 34, 137–143. [Google Scholar] [CrossRef]
- Robinson, J. The extraction of gold from sulphidic concentrates by roasting and cyanidation. J. S. Afr. Inst. Min. Metall. 1988, 88, 117–130. [Google Scholar]
- Swash, P. A mineralogical investigation of refractory gold ores and their beneficiation, with special reference to arsenical ores. J. S. Afr. Inst. Min. Metall. 1988, 88, 173–180. [Google Scholar]
- Aylmore, M.G. Treatment of a refractory gold-copper sulfide concentrate by copper ammoniacal thiosulfate leaching. Miner. Eng. 2001, 14, 615–637. [Google Scholar] [CrossRef]
- Tanrıverdi, M.; Mordoğan, H.; İpekoğlu, Ü. Leaching of ovacık gold ore with cyanide, thiourea and thiosulphate. Miner. Eng. 2005, 18, 363–365. [Google Scholar] [CrossRef]
- McLaughlin, J.; Agar, G. Development and application of a first order rate equation for modelling the dissolution of gold in cyanide solution. Miner. Eng. 1991, 4, 1305–1314. [Google Scholar] [CrossRef]
- McGill, S.; Walkiewicz, J.W.; Clark, A. Microwave Heating of Chemicals and Minerals; US Department of the Interior, Bureau of Mines: Washington, DC, USA, 1995.
- Salsman, J.; Williamson, R.; Tolley, W.; Rice, D. Short-pulse microwave treatment of disseminated sulfide ores. Miner. Eng. 1996, 9, 43–54. [Google Scholar] [CrossRef]
- Uslu, T.; Atalay, Ü.; Arol, A. Effect of microwave heating on magnetic separation of pyrite. Colloids Surf. A Physicochem. Eng. Asp. 2003, 225, 161–167. [Google Scholar] [CrossRef]
- Ling, H. Study on waste liquid return process in bacterial oxidation of low-sulfur hard-handle gold concentrates. Mod. Min. 2013, 2, 009. [Google Scholar]
- Vaughan, D.J.; Craig, J.R. The crystal chemistry of iron-nickel thiospinels. Am. Mineral. 1985, 70, 1036–1043. [Google Scholar]
- Hwang, J.-Y.; Shi, S.; Xu, Z.; Huang, X. Oxygenated leaching of copper sulfide mineral under microwave-hydrothermal conditions. J. Miner. Mater. Charact. Eng. 2002, 1, 111. [Google Scholar] [CrossRef]
- Vorster, W.; Rowson, N.; Kingman, S. The effect of microwave radiation upon the processing of neves corvo copper ore. Int. J. Miner. Process. 2001, 63, 29–44. [Google Scholar] [CrossRef]
- Olubambi, P.; Potgieter, J.; Hwang, J.; Ndlovu, S. Influence of microwave heating on the processing and dissolution behaviour of low-grade complex sulphide ores. Hydrometallurgy 2007, 89, 127–135. [Google Scholar] [CrossRef]
- Wang, G.; Radziszewski, P.; Ouellet, J. Particle modeling simulation of thermal effects on ore breakage. Comput. Mater. Sci. 2008, 43, 892–901. [Google Scholar] [CrossRef]
- Wang, Y.; Djordjevic, N. Thermal stress FEM analysis of rock with microwave energy. Int. J. Miner. Process. 2014, 130, 74–81. [Google Scholar] [CrossRef] [Green Version]
- Beeby, J.P. The Recovery of a Valuable Species from an Ore. Google Patents WO 1,992,018,249 A1, 29 October 1992. [Google Scholar]
- Kingman, S.; Vorster, W.; Rowson, N. The influence of mineralogy on microwave assisted grinding. Miner. Eng. 2000, 13, 313–327. [Google Scholar] [CrossRef]
- Kingman, S.; Jackson, K.; Bradshaw, S.; Rowson, N.; Greenwood, R. An investigation into the influence of microwave treatment on mineral ore comminution. Powder Technol. 2004, 146, 176–184. [Google Scholar] [CrossRef]
| Experiment | Irradiation Time (min) | Lixiviant (mM) | ||
|---|---|---|---|---|
| Thiourea | Sodium Sulfate | Ferric Sulfate | ||
| A | - | 34.7 | 0.6 | 3.5 |
| B | 10 | 34.7 | ||
| C | 30 | 34.7 | ||
| D | 50 | 34.7 | ||
| E | 70 | 34.7 | ||
| F | 70 | 43.4 | ||
| G | 70 | 52.0 | ||
| Irradiation Time (min) | Elements (mg/kg) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Si | Fe | Cu | Zn | Pb | Co | S * | Ag | Au | |
| Raw | 10,460 | 40,560 | 290 | 140 | 4550 | 2550 | 20,140 | 1026 | 133 |
| 10 | 10,412 | 40,588 | 296 | 152 | 4566 | 2245 | 11,454 | 1022 | 139 |
| 30 | 10,388 | 41,356 | 291 | 144 | 4597 | 2188 | 1445 | 1012 | 150 |
| 50 | 10,401 | 41,599 | 284 | 143 | 4603 | 1384 | 1322 | 996 | 152 |
| 70 | 10,122 | 42,560 | 277 | 138 | 4612 | 1355 | 1120 | 999 | 159 |
| Experiment | (%) | k (min−1) | R2 |
|---|---|---|---|
| A | 81.42 | 0.0561 | 0.9804 |
| B | 84.06 | 0.0601 | 0.9780 |
| C | 88.45 | 0.0612 | 0.9757 |
| D | 96.02 | 0.0624 | 0.9768 |
| E | 100.00 | 0.0783 | 0.9830 |
| F | 100.00 | 0.0837 | 0.9805 |
| G | 100.00 | 0.0901 | 0.9773 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choi, N.-C.; Kim, B.-J.; Cho, K.; Lee, S.; Park, C.-Y. Microwave Pretreatment for Thiourea Leaching for Gold Concentrate. Metals 2017, 7, 404. https://doi.org/10.3390/met7100404
Choi N-C, Kim B-J, Cho K, Lee S, Park C-Y. Microwave Pretreatment for Thiourea Leaching for Gold Concentrate. Metals. 2017; 7(10):404. https://doi.org/10.3390/met7100404
Chicago/Turabian StyleChoi, Nag-Choul, Bong-Ju Kim, Kanghee Cho, Soonjae Lee, and Cheon-Young Park. 2017. "Microwave Pretreatment for Thiourea Leaching for Gold Concentrate" Metals 7, no. 10: 404. https://doi.org/10.3390/met7100404




