A Novel Research on Behavior of Zinc Ferrite Nanoparticles in Different Concentration of Poly(vinyl pyrrolidone) (PVP)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. Laboratory Determinations
3. Results and Discussion
3.1. Thermal Analyses of PVP
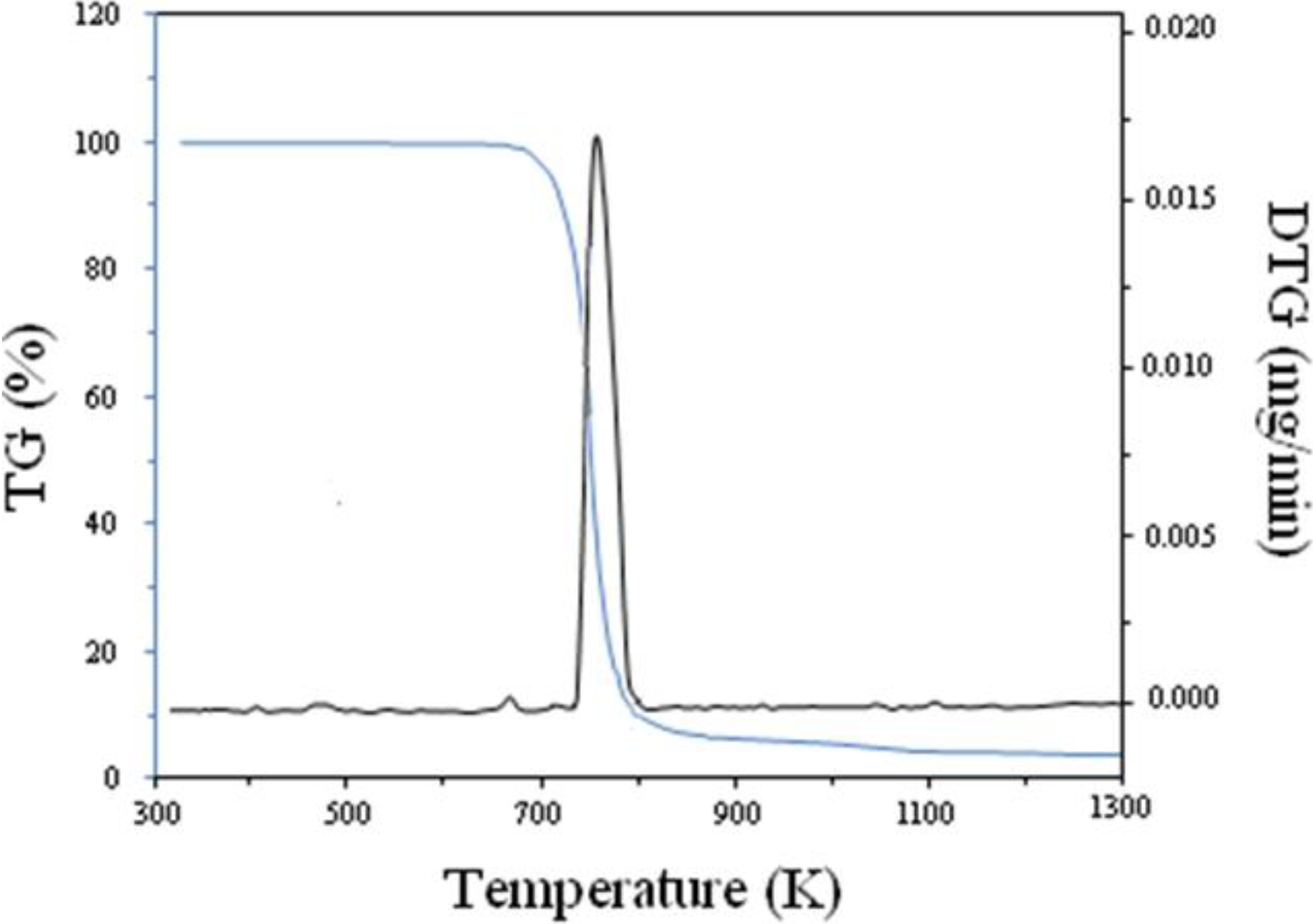
3.2. Mechanism of Formation

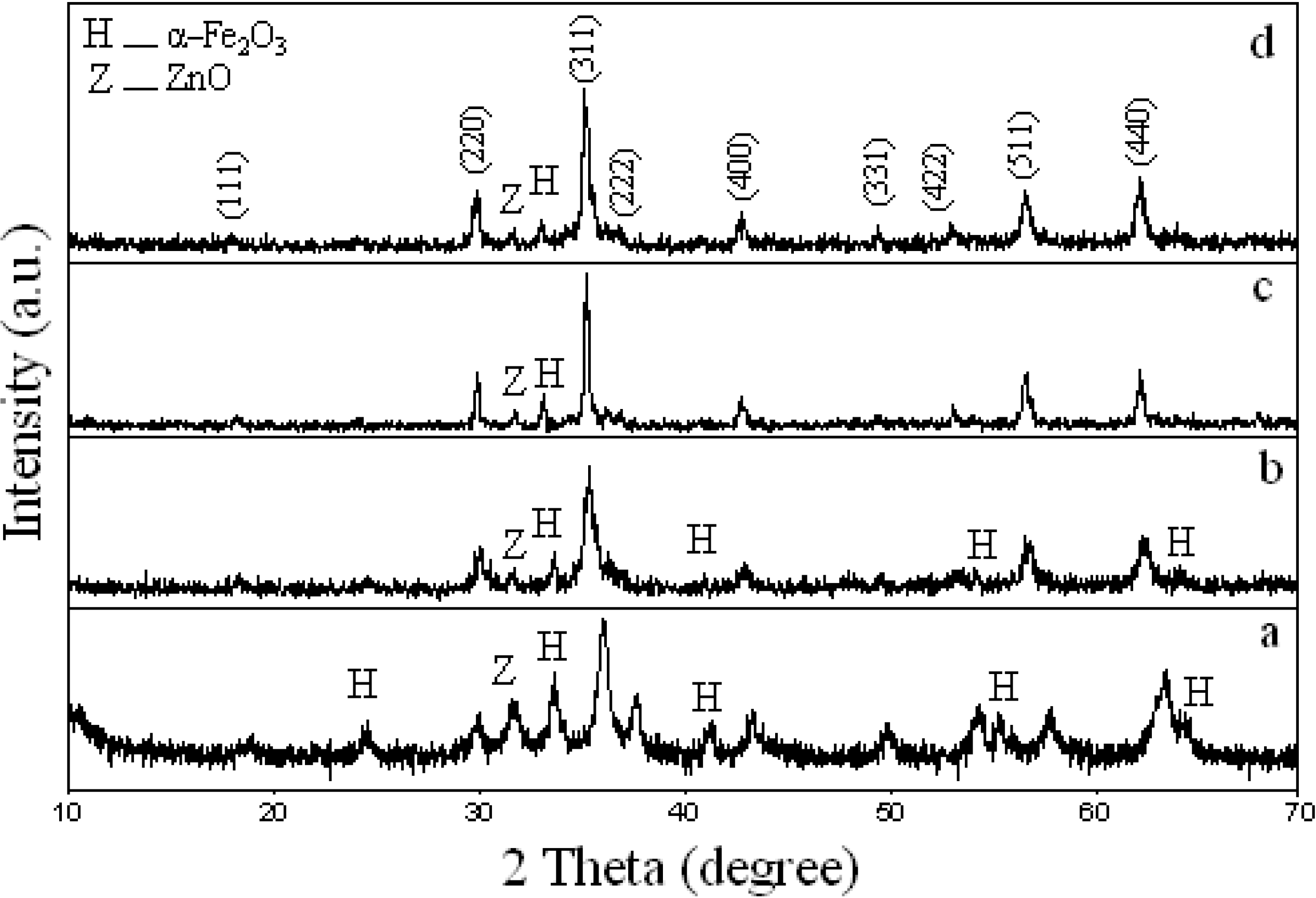
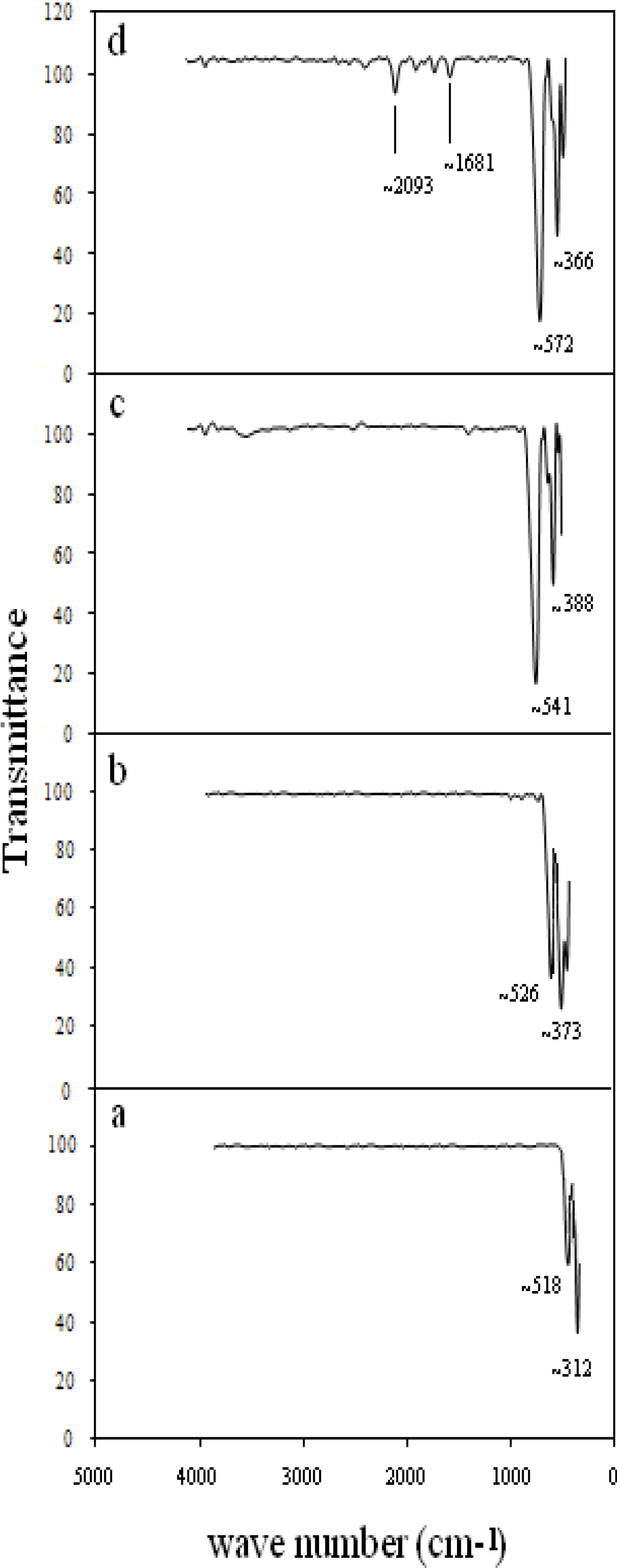
3.3. Effect of Calcination Temperature on Phase Composition, Morphology and Magnetic Properties
3.4. Effect of Concentration of PVP on Phase Composition, Morphology and Magnetic Properties
| ZnFe2O4 nanoparticles | Concentrations of PVP (g/L) | Percentage of hematite phase, vol.% | Percentage of zincite phase, vol.% | Percentage of spinel phase, vol.% | Particle size XRD (nm) | Particle size TEM (nm) |
|---|---|---|---|---|---|---|
| ZnFerrite 1 | 0 | 27.3 | 5.4 | 67.3 | - | - |
| ZnFerrite 2 | 15 | 4.8 | 0.8 | 94.4 | 29 | 28 ± 2 |
| ZnFerrite 3 | 40 | 1.7 | 0.5 | 97.8 | 33 | 31 ± 11 |
| ZnFerrite 4 | 55 | 1.6 | 0.5 | 97.9 | 35 | 34 ± 9 |


| ZnFe2O4 nanoparticles | Concentrations of PVP (g/L) | Saturation magnetization Ms (emu/g) |
|---|---|---|
| ZnFerrite 1 | 0 | 0.43 |
| ZnFerrite 2 | 15 | 1.1 |
| ZnFerrite 3 | 40 | 0.74 |
| ZnFerrite 4 | 55 | 0.60 |
4. Conclusions
Conflicts of Interest
References
- Rosensweig, R.E. Ferrohydrodynamics; Dover: New York, NY, USA, 1985. [Google Scholar]
- Tirosh, E.; Shemer, G.; Markovich, G. Optimizing cobalt ferrite nanocrystal synthesis using a magneto-optical probe. Chem. Mater. 2006, 18, 465–470. [Google Scholar] [CrossRef]
- Luders, U.; Barthelemy, A.; Bibes, M. NiFe2O4: A versatile spinel magterial brings new opportunities for Spintronics. Adv. Mater. 2006, 18, 1733–1736. [Google Scholar] [CrossRef]
- Pankhurst, Q.A.; Connolly, J.; Jones, S.K. Applications of magnetic nanoparticles in biomedicine. J. Phys. D 2003, 36, R167–R181. [Google Scholar] [CrossRef]
- Berry, C.C. Possible exploitation of magnetic nanoparticles-cell interaction for biomedical applications. J. Mater. Chem. 2005, 15, 543–547. [Google Scholar] [CrossRef]
- Selvan, R.K.; Kalaiselvi, N.; Augustin, C.O. SnO2 pinning: An approach to enhance the electrochemical properties of anodes for batteries. Electrochem. Solid State Lett. 2006, 9, A390–A394. [Google Scholar] [CrossRef]
- Atif, M.; Hasanian, S.K.; Nadeem, M. Magnetization of sol-gel prepared zinc ferrite nanoparticles: Effects of inversion and particle size. J. Sci. Commun. 2006, 138, 416–421. [Google Scholar]
- Jiang, J.Z.; Wynn, P.; Morup, S. Magnetic structure evolution in mechanically milled nanostructured ZnFeO2 particles. Nanostruct. Mater. 1999, 12, 737–740. [Google Scholar] [CrossRef]
- Shenoy, S.D.; Joy, P.A.; Anantharaman, M.R. Effect of mechanical milling on the structural, magnetic and dielectric properties of coprecipitated ultrafine zinc ferrite. J. Magn. Magn. Mater. 2004, 269, 217–226. [Google Scholar] [CrossRef]
- Hamdeh, H.H.; Ho, J.C.; Oliver, S.A.; Willey, R.J.; Oliveri, G.; Busca, G. Magnetic properties of partially-inverted zinc ferrite aerogel powders. J. Appl. Phys. 1997, 81, 1851–1858. [Google Scholar] [CrossRef]
- Yu, S.H.; Fujino, T.; Yoshimura, M. Hydrothermal synthesis of ZnFe2O4 ultrafine particles with high magnetization. J. Magn. Magn. Mater. 2003, 256, 420–427. [Google Scholar]
- Morrison, S.A.; Cahill, C.L.; Carpenter, E.E. Magnetic and structural properties of nickel zinc ferrite nanoparticles synthesized at room temperature. J. Appl. Phys. 2004, 95, 6392–6395. [Google Scholar] [CrossRef]
- Hochepied, J.P.; Bonville, J.F.; Pileni, M.P. Non stoichiometric zinc ferrite nancrystals: Syntheses and magnetic properties. J. Phys. Chem. 2000, 104, 905–912. [Google Scholar] [CrossRef]
- Rana, S.; Gallo, A.; Srivastava, R.S. On the suitability of nanocrystalline ferrites as a magnetic carrier for drug delivery: Functionalization, conjugation, and drug release behavior. Acta Biomater. 2007, 3, 233–242. [Google Scholar] [CrossRef]
- Naseri, M.G.; Saion, E.B.; Abbastabar, A.H.; Hashim, M.; Shaari, A.H. Simple preparation and characterization of nickel ferrite nanocrystals by a thermal treatment method. Powder Technol. 2011, 212, 80–88. [Google Scholar] [CrossRef]
- Naseri, M.G.; Saion, E.B.; Hashim, M.; Shaari, A.H.; Ahangar, H.A. Synthesis and characterization of zinc ferrite nanoparticles by a thermal treatment method. Solid State Commun. 2011, 151, 1031–1035. [Google Scholar] [CrossRef]
- Kumar, S.V.; Musturappa, T.E.; Prasannakumar, S.; Mahadevan, K.M.; Sherigara, B.S. N-Vinylpyrrolidone and ethoxyethyl methacrylate copolymer: Synthesis, characterization and reactivity ratios. J. Macromol. Sci. Part A 2007, 44, 1161–1169. [Google Scholar] [CrossRef]
- Silva, M.F.; Silva, C.A.; Fogo, F.C.; Pineda, E.A.G.; Hechenleitner, A.A.W. Thermal and FTIR study of polyvinylpyrrolidone/lignin blends. J. Ther. Anal. Calorim. 2005, 79, 367–370. [Google Scholar] [CrossRef]
- Lau, C.; Mi, Y. A study of blending and complexation of poly(acrylic acid)/poly(vinyl pyrrolidone). Polymer 2002, 43, 823–829. [Google Scholar] [CrossRef]
- Sivakumar, P.; Ramesh, R.; Ramanand, A. Synthesis and characterization of NiFe2O4 nanosheet via polymer assisted co-precipitation method. Mater. Lett. 2011, 65, 483–485. [Google Scholar] [CrossRef]
- Koebel, M.M.; Jones, L.C.; Somorjai, G.A. Preparation of size-tunable, highly monodisperse PVP-protected Pt-nanoparticles by seed-mediated growth. J. Nanopart. Res. 2008, 10, 1063–1069. [Google Scholar]
- Roosen, A.R.; Carter, W.C. Simulations of microstructural evolution: Anisotropic growth and coarsening. Phys. A 1998, 261, 232–247. [Google Scholar] [CrossRef]
- Ghosh, G.; Naskar, M.K.; Patra, A. Synthesis and characterization of PVP-encapsulated ZnS nanoparticles. Opt. Mater. 2006, 28, 1047–1053. [Google Scholar] [CrossRef]
- Shao, H.; Hi, Y.; Lee, H. Effect of PVP on the morphology of cobalt nanoparticles prepared by thermal decomposition of cobalt acetate. Curr. Appl. Phys. 2006, 6, e195–e197. [Google Scholar] [CrossRef]
- Huang, W.; Xu, G. Characterization of nano-Ag/PVP composites synthesized via ultra-violet irradiation. J. Coal Sci. Eng. 2010, 16, 188–192. [Google Scholar] [CrossRef]
- Tsuji, M.; Hashimoto, M.; Nishizawa, Y.; Tsuji, T. Synthesis of gold nanorods and nanowires by microwave-polyol method. Mater. Lett. 2004, 58, 2326–2330. [Google Scholar] [CrossRef]
- Chinnasamy, C.N.; Ponpandian, A.N.; Chattopadhyay, K.; Guerault, H.; Greneche, J.M. Magnetic properties of nanostructured ferrimagnetic zinc ferrite. J. Phys. 2000, 12, 7795–7805. [Google Scholar]
- Laokul, P.; Amornkitbamrung, V.; Seraphin, S. Characterization and magnetic properties of nanocrystalline CuFe2O4, NiFe2O4, ZnFe2O4 powders prepared by the Aloe vera extract solution. Curr. Appl. Phys. 2011, 11, 101–108. [Google Scholar] [CrossRef]
- Cullity, B.D. Elements of X-ray Diffraction; Addison-Wesley: London, UK, 1987. [Google Scholar]
- Sharma, D.R.; Mathur, R.; Vadera, S.R. Synthesis of nanocomposites of Ni–Zn ferrite in aniline formaldehyde copolymer and studies on their pyrolysis products. J. Alloys Compd. 2003, 358, 193–204. [Google Scholar] [CrossRef]
- Elsayed, A.H.; Mohy Eldin, M.S.; Elsyed, A.M. Synthesis and properties of polyaniline/ferrites nanocomposites. Int. J. Electrochem. Sci. 2011, 6, 206–221. [Google Scholar]
- Niasari, M.S.; Davar, F.; Mahmoudi, T. A simple route to synthesize nanocrystalline nickel ferrite (NiFe2O4) in the presence of octanoic acid as a surfactant. Polyhedron 2009, 28, 1455–1458. [Google Scholar] [CrossRef]
- Nyutu, E.K.; Canner, W.C.; Auerbach, S.M.; Chen, C.H.; Suib, S.L. Ultrasonic nozzle spray in situ mixing and microwave-assisted preparation of nanocrystalline spinel metal oxides: Nickel ferrite and zinc aluminate. J. Phys. Chem. C 2008, 112, 1407–1414. [Google Scholar]
- Mathew, D.S.; Juang, R.S. An overview of the structure and magnetism of spinel ferrite nanoparticles and their synthesis in microemulsions. Chem. Eng. J. 2007, 129, 51–65. [Google Scholar] [CrossRef]
- Li, F.S.; Wang, L.; Wang, J.B.; Zhou, Q.G.; Zhou, X.Z.; Kunkel, H.P.; Williams, G. Site preference of Fe in nanoparticles of ZnFe2O4. J. Magn. Magn. Mater. 2004, 268, 332–339. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kamari, H.M.; Naseri, M.G.; Saion, E.B. A Novel Research on Behavior of Zinc Ferrite Nanoparticles in Different Concentration of Poly(vinyl pyrrolidone) (PVP). Metals 2014, 4, 118-129. https://doi.org/10.3390/met4020118
Kamari HM, Naseri MG, Saion EB. A Novel Research on Behavior of Zinc Ferrite Nanoparticles in Different Concentration of Poly(vinyl pyrrolidone) (PVP). Metals. 2014; 4(2):118-129. https://doi.org/10.3390/met4020118
Chicago/Turabian StyleKamari, Halimah Mohamed, Mahmoud Goodarz Naseri, and Elias B. Saion. 2014. "A Novel Research on Behavior of Zinc Ferrite Nanoparticles in Different Concentration of Poly(vinyl pyrrolidone) (PVP)" Metals 4, no. 2: 118-129. https://doi.org/10.3390/met4020118




